Abstract
Phosphate sorption-desorption parameters like maximum phosphorus (P) adsorption capacity (P max), equilibrium phosphorus concentration (EPC), water desorbable P (α), potentially bioavailable P (β), and mobility index (α/β ratio) were determined in order to understand the sediment source-sink nature in Guaporé catchment in southern Brazil during summer and winter 2013 and 2014. The result showed a significant (p = 0.05) variation across sediment site or seasons and revealed the most sorption-desorption parameters (P max, α, β) with the increments following the order urban sediments > intensive agriculture under CT > intensive agriculture under NT > low agriculture sub-catchments (sub1 and sub2) > native forest. In the main river points, these parameters decreased along the river (P1 to P5). The results were more obvious in winter than the summer season. In contrast, the low values of λ and α/β ratio in the sediment from native forest and relatively less polluted catchment (sub1) during summer season show the quick P desorption when compared to specific Fe and Al oxides bound to stable P in intensive agriculture sediments. These findings clearly indicated that agricultural practices, sediment characteristics, and hydrological factors have a major impact on seasonal sediment P bioavailability and mobility. The urban untreated discharges may be a single major P source and, if it is not wisely managed, proves a major threat to water quality. These results have serious implications for the river ecosystem and will be of great importance to improve the environmental and economic performance of agricultural practices aiming to reduce soil-based P legacy to surface waters.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Eutrophication of fresh waters is mainly linked to upstream anthropogenic activities in the catchment like overfertilization and untreated urban and or industrial sewage discharge containing high amounts of dissolved nutrients, which pose a serious threat to the ecological environment (Sharpley et al. 2013; Withers et al. 2015). Non-point phosphate reaching to fresh water rivers and streams is more difficult to manage as it is bound to sediments and organic materials. However, due to land-use change, catchment geographical features like rainfall and runoff, seasonal pH and dissolved oxygen (DO) shifts, and sediment chemical composition, this sediment-bound P could be released into surrounding water by ion exchange (Withers and Jarvie 2008; Upreti et al. 2015). The sorption-desorption P balance considerably contributes to the final tropic level of water bodies (Jarvie et al. 2006; Zhang et al. 2014).
It is a fact that the P adsorption capacity to a larger extent is dependent on the sediment composition. Therefore, the most reactive sites for phosphate interaction are those minerals such as Fe and Al oxyhydroxides and surface hydroxyl groups that could be exchanged by phosphate anions on these sites of sediments. These variations are more obvious under highly weathered ferralsol under subtropical environments like most of the Southern Brazil. The high affinity of these soils for P adsorption has been reported in recent studies (Bortoluzzi et al. 2015; Tiecher et al. 2015; Fink et al. 2016). Their results have shown that these soils are predominant in Fe and Al sesquioxides, kaolinite clay, and 2:1 clay type with interlayer hydroxy-Al (Caner et al. 2014).
Furthermore, particle physical characteristics like size distribution are linked to the total surface area and must be considered while determining P retention. Results from the previous studies (Zhang and Huang 2007; Bortoluzzi et al. 2015) have shown that the P adsorption increases exponentially with decreasing particle size i.e., increasing clay fraction. Consequently, the sediment originating from these acidic soils facilitates the P sorption preferably via bidentate binding with active Fe and can contribute to as much as 50 to 70 % of total adsorbed P in sediment (Kim et al. 2011; Jan et al. 2015).
The determination of sediment-bound total phosphorus (TP) to predict either they act as source or sink in the river is ambiguous, as only some P is desorbed and become biologically available. For a better understanding of the sediment P mobilization, maximum phosphorus adsorption capacity (P max) and equilibrium phosphorus concentration (EPC) have been used to estimate river-sediment P dynamics. Several mathematical models have been developed and reported in literature (Lopez et al. 1996; Rheinheimer et al. 2000; Tu et al. 2002) for this purpose, but the superiority and wide adoption of nonlinear model of Langmuir have already been reported in many studies to describe the best P adsorption (House and Denison 2000; Bubba et al. 2003; Guardini et al. 2012). However, when the sediment has a considerable amount of native desorbable P (Q 0), it affects the Langmuir model validity to quantify accurately the P adsorption, as a fact that water desorbable P also participates in the adsorption equilibrium and, therefore, it must be accounted. To overcome this situation, several modifications in Langmuir model have been proposed depending on the sediment type (Zhu et al. 2011); however, the modification introduced by Koski-Vahala and Hartikainem (2001) is more practicable as it visually separates the P desorbed in distilled water (Q 0).
In freshwater river sediments, both total P (TP) and bioavailable P (BAP) contents also vary according to land-use heterogeneity and catchment characteristics and are not clear indicators of the eutrophic state of water. All P adsorbed into the sediment can be thermodynamically desorbed (Barrow 1983), yet, the determination of P adsorption capacity and EPC does not fully reveal the P release at the sediment-water interface. However, McKean and Warren (1996) proposed first-order kinetics equation which allows to estimate the instantaneous particulate bioavailable P (α), potential particulate bioavailable P or P buffering capacity (β), and desorption rate constant (λ) sediment-water interface and is used in many recent studies (Rheinheimer and Anghinoni 2001; Rheinheimer et al. 2008; Pellegrini et al. 2010; Bortoluzzi et al. 2013).
So far, few studies focusing on water and sediment-bound P transportation have been conducted in Southern Brazil and were limited to field/plot or small catchment scale to understand the land use (Pellegrini et al. 2010), rain event basis (Bortoluzzi et al. 2013), high-intensity cropland (Capoane et al. 2015), successive slurry application under no-till soil management (Guardini et al. 2012), and sediment source finger printing (Tiecher et al. 2015); yet, a comprehensive catchment scale temporal study integrating all these aspects is lacking.
The main objectives of current study were (a) to determine whether the surface sediments act as sink or source for river water, (b) to predict the particulate bioavailable P (algal bioavailable) and sediment P buffering capacity in response to the land use and management for each monitoring site, and (c) to assess and differentiate the overall seasonal variations in P adsorption-desorption kinetics based on geochemical composition in order to refine the upstream fertilization plan and soil management in the Guaporé catchment which is currently under highly anthropic and intensive agriculture region in Southern Brazil.
Materials and methods
Description of Guaporé catchment
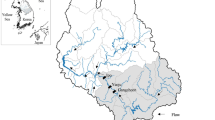
The study area Guaporé catchment is located at coordinates 28°54′41″S and 51°57′10″W in the south of the Brazil. The basin altitude varies from 40 to 800 m from south to north (Fig. 1). The climate of the region is humid subtropical climate with average annual rainfall from 1400 and 2000 mm and well distributed throughout the year, but high winter rain events are frequent from August to October months, and the average annual temperature is 17.4 °C. It has a total basin area of over 2000 km2 with areas under representative land uses for cropping pattern and fertilization management. The basin has high heterogeneity in soil geology and lithology, and many soil classes like Entisols, Luvisol, Cambisol, Oxisol, Ultisol, Chernosol are present due to specific pedo-climatic variations (Didoné et al. 2014). The region is characterized with intensive agriculture and high anthropic activities including rapid expansion in dairy and poultry industry over past 30 years. Therefore, surface water reservoirs are under threat of severe impairment due to high rainfall, sloppy landforms, high water erosion and sediment loads, and increased P discharges either from intensive agriculture lands or untreated urban sewage reaching to headwater streams (Didoné et al. 2014). It is a major source of drinking water for Marau town and a population of over 0.5 million residing in Guaporé region and have great importance for livestock, fisheries, and recreation in the region. Being a tributary of river Taquari, and a key contributor to the regional hydrographic of Guaíba Lake, the high sediment production and associated P drain to the Atlantic Ocean near Porto Alegre city, Rio Grande do Sul Brazil. The Guaporé catchment thus has a great importance both for regional and international water resource management.
Sediment samplings
To collect representative suspended sediment samples, a total of 11 monitoring stations, of which 6 were installed on sub-catchment scale and 5 monitoring stations on the main river, were selected after analyzing the land use and geographical location with the help of a digital elevation model originating in mapping mission of Earth relief–SRTM (Shuttle Radar Topography Mission) using the software ArcGIS 10 (ESRI 2011), and maps were developed. Suspended sediments were collected from summer (early February) and winter (late July) during 2013 and 2014 with the time-integrated suspended sediment samplers (Maier 2013). The samples were taken into acid-washed 20-L gallon and brought to soil and water chemistry laboratory of Federal University of Santa Maria, Brazil, and then oven-dried at 50 °C, gently disaggregated using a rubber head pestle and mortar, and sieved through a 63-μm mesh before chemical analysis. A set of original oven-dried sample was passed through 2-mm mesh and kept for particle size analysis.
Sediment characterization
Sediment (<63-μm) TP was determined with H2SO4 and H2O2 digestion in the presence of saturated MgCl2 (Olsen and Sommers 1982), while the organic P (OP) was estimated by ignition at 550 °C for 6 h and subsequently determined by colorimetry (Murphy and Riley 1962). The inorganic P (IP) was calculated as the difference of OP to TP. The total organic carbon (TOC) was analyzed by wet oxidation with K2Cr2O7 and H2SO4 following Walkley and Black method (1934). Amorphous Fe and Al were extracted with 0.2 mol L−1 ammonium oxalate at pH 3 in the dark (Fe ox and Alox), as described by Schwertmann (1964). The crystalline forms of Fe oxyhydroxides (Fe dcb ) and Al (Aldcb) were estimated in dithionite–citrate–bicarbonate (DCB) extraction as explained by Mehra and Jackson (1960). The particle size distribution was determined in 2-mm sediment by the laser particle size analyzer (Muggler et al. 1997) after oxidation of soil organic matter with H2O2 and subsequent dispersion with 6 % NaOH solution.
Phosphorus sorption study
To determine the P adsorption behavior, only summer sediments were selected, and 0.5 g of powdered suspended sediment (<63 μm) was weighed into 15 mL polypropylene centrifuge tube. Thereafter, 10 mL of 0.01 M L−1 CaCl2 solution containing 0, 2, 5, 10, 20, 50, 100, 200, 400, 800, 1200, and 1600 mg L−1 of equivalent P concentration from KH2PO4 was added, respectively. The samples were shaken for 16 h on an end-over-end shaker at 33 rpm at 25 °C, and subsequently the samples were centrifuged and P concentration was colorimetrically measured in the supernatant (Murphy and Riley 1962). The amount of adsorbed P was calculated as the difference of quantity added to P remaining in the solution at equilibrium. The sorption data was fitted to Langmuir equation with modification of the Koski-Vahala and Hartikainem (2001) and given as follow:
where
P max is the maximum P adsorption capacity (mg kg−1); km is the P concentration in the solution that allows half of the maximum P adsorption (mg L−1); P solution is the equilibrium P concentration in solution (mg L−1); and Q 0 is water desorbed P (mg L−1). The EPC was calculated by the expression:
Desorption kinetics study
Bioavailable particulate phosphorus in suspended sediments was estimated by a single extraction with anion exchange resin (AER) strips (AMI-7001S; Membranes International Inc., NJ) as described by Rheinheimer et al. (2000). Briefly, 0.5 g of powdered suspended sediment (<63-μm) and 10 mL of distilled water were taken in 15-mL falcon tube. Then 0.5 mol L−1 NaHCO3-saturated anion exchange resin strip was added (Gatiboni et al. 2005), and the tubes were shaken for 16 h on an end-over-end shaker at 33 rpm and 25 °C. The anion resin membrane was removed and washed with water before transferring into a 15-mL glass tube with 10 mL of 0.5 M L−1 HCl. After leaving for 2 h, the tubes were shaken on an oscillating agitator for further 30 min and P concentration was determined on an aliquot of the solution by Murphy and Riley (1962) method, and this P concentration is considered as bioavailable particulate P. To attain the cumulative amount of P sorbed, the successive extraction was carried out for 13 days in the same sediment samples until the P concentration in extract became constant and reached zero.
To estimate P desorption parameters, the first-order kinetic model proposed by the McKean and Warren (1996) in Eq. 3 was employed.
where
(Pdes) is amount of P desorbed; (β), potentially bioavailable particulate P; (α), instantaneous bioavailable particulate P; (λ), desorption rate constant, and (t), extraction time in minutes. The P mobility index was calculated as = (α × 100/β).
Statistics
One-way analysis of variance (ANOVA) followed by Scott-Knott test (p < 0.05) was conducted using Sisvar statistical software (version 5.3) to determine the significant differences between seasons and sampling stations (Ferreira 2011). The correlation coefficient was determined, and principal component analysis between a pair of means of P sorption-desorption parameters and sediment characteristics was also performed.
Results
Sediment characteristics
Under the acidic environments, the sediment P retention is closely related to and or influenced by metal oxides (Al and Fe) and clay contents. In this study, the seasonal variation in Fe and Al oxides is presented in Tables 2 and 3 while particle size distribution is shown in Fig. 2. The rank order of the Fe and Al oxides and most of chemical components concentration for the sediments generally followed a pattern as highly anthropic sediments (sub5) > intense agriculture under Ct (sub3) > agriculture under no tillage (NT) (sub4) > low agriculture sub-catchments (sub1, 2) > native forest. The main river sediments (P1 to P5) showed a descending order along river continuum for most of the chemical characteristics. Average across four seasons and 11 monitored locations, the sediment Feox ranged (4.4 to 7.4 g kg−1), Fedcb (33.7 to 53.6 g kg−1), Alox (1.1 to 2.2 g kg−1), Aldcb (2.8 to 5.9 g kg−1), TOC (18.2 to 33.7 g kg−1), and clay (43.3 to 137.7 g kg−1).
Individually, the sediment amorphous Fe (Feox) concentration collected in 2013 was higher during winter for agriculture sub-catchments (sub1 < sub2 < sub3 < sub4) and in the summer for main river sediment (P1 to P5); whereas, during 2014, most of the sediments from all monitoring stations showed higher Feox concentration in the summer. The relative increase in Feox content varied from 8.5 to 49.2 % summer and 37.8 to 113.5 % winter during 2013 and from 37.2 to 93 % summer and 59.5 to 110.8 % winter during 2014, over control (forest). The crystalline Fe (Fedcb) was higher in summer sediments than winter during both years, and higher concentration for highly anthropic followed by the both CT and NT intensive agriculture sediments was observed over the rest of the locations. The relative increase in Fedcb due to land use or seasonal changes ranged between 31 to 94.5 % and 16.5 to 30.9 % (summer and winter 2013) and between 22.4 to 71.1 % and 16.6 to 39.8 % (summer and winter 2014) over the reference forest site (Table 1).
The Alox did not showed much variation either for seasons or among sampling sites and differed between 1 and 2 g kg−1Alox for most of the sediments except highly anthropic (sub5) which showed as much as 3.37 g kg−1 Alox. However, Aldcb showed a significant difference, and a relative increase lies between 12.2 to 109 % and 20.9 to 88.1 % (summer and winter 2013) and 18.3 to 189 % and 7.6 to 96.6 % (summer and winter 2014) over the forest site (Table 1).
The TOC content was higher in most of winter sediments for both years, except a relatively higher TOC content in main river sediments during summer 2013. The relative increase in TOC content over the forest varied between 4.6 to 248.9 % in summer and 5.8 to 80.6 % in winter during 2013, and from 7.2 to 78.4 % and 10.6 to 308.9 % during summer and winter 2014, respectively (Tables 2 and 3).
The particle size distribution of different sediments collected over four seasons is presented in Fig. 2. The silt fraction appeared as the major proportion in the sediments, and it accounts for 50 to 70 % of the total while the clay showed a small contribution, accounting for 4 to 15 % of the total. The seasonal trend revealed that the high clay and sand proportion was recorded for intensive sub-catchment under CT (sub3) during winter period in both 2013 and 2014.
Sediment organic, inorganic, and total phosphorus fractions
The seasonal variations in OP, IP, and TP concentrations showed significant differences according to Scott-Knott test (p < 0.05) for 11 monitored sites and four seasons and are summarized in Tables 4 and 5. Along different land uses, the OP contents followed an order: intense agriculture under NT (sub4) > intense agriculture under CT (sub3) > highly anthropic sediments (sub5) > low agriculture sub-catchment (sub2) > native forest > sub1. The main river sediments (P1 to P5) showed descending trend along the river downstream except P4, which showed maximum values for OP over rest of the main river sediments. The relative increase in OP of disturbed sediment over native forest varied from 6.2 to 98.8 % in summer and 70.6 to 129.0 % in winter during the year 2013, while from 35.3 to 361.9 % in summer and 31.0 to 170.5 % in winter during 2014 was recorded.
The highest IP content was 1927.5 mg kg−1 in urban sub-catchment during summer 2013, while a minimum of 600.1 mg kg−1 was found in the native forest during winter in 2014 (Tables 2 and 3). A higher IP in sub-catchment with intensive agriculture under CT (sub3) was observed in the winter season in both years. The intensive agriculture sub-catchments (sub3 and sub4) showed relatively higher IP values during winter 2013 while higher IP contents were recorded in summer 2014. A similar trend for main river sediment (P1 to P5) was also observed. Across different sediment locations, the relative increase in IP contents varied between 24.2 to 193.5 % and 24.2 to 178.8 % (summer and winter 2013) and from 0.4 to 112.4 % to 15.8 to 99.8 % (summer and winter 2014) over the forest sediments (Tables 4 and 5).
A high TP during winter period was detected during both years and anthropogenic activity almost tripled the TP (2222.2 mg kg−1) in urban sub-catchment when compared to the reference forest sediment (864.2 mg kg−1) in winter 2013. The intensive agriculture site either under CT (sub3) or NT (sub4) also showed the higher 1642.0 and 1910.6 mg kg−1 during 2013, and 1415.5 and 1296.9 mg kg−1 TP during 2014 over the rest of sediments and the winter months retained higher TP in both years. The percent relative increase over forest site varied from 16.3 to 163.7 % (summer 2013), 12.4 to 157.7 % (winter 2013), 5.0 to 102.7 % (summer 2014), and from 18.8 to 98.9 % in winter 2014, respectively.
Phosphorus adsorption
The P adsorption behavior of suspended sediment was carried on sediments collected during the summer period for both 2013 and 2014. The time was selected since there are low flow conditions in the Guaporé River during this period, and it is also considered as an ecologically sensitive period. The land uses and seasonal changes showed a significant effect on the P sorption parameters (i.e., P max, km, Q 0 and EPC) according to Scott-Knott test (p < 0.05), and the results are presented in Table 6. Across the sediment origin, the highest values for sorption parameters were recorded for the intensive agriculture sub-catchment (sub3) under CT (P max = 4032 and 2342 mg kg−1), (km = 29.9 and 13.6 mg L−1), and (Q 0 = 2.4 and 2.2 mg L−1) during 2013 and 2014, respectively. The low agriculture catchment (sub2) also showed increasing trend for P sorption and P max (3258 and 2608 mg kg−1), km (27.40 and 15.68 mg L−1), and Q 0 (0.70 and 2.08 mg L−1) were recorded in both years. The main river sediments (P1 to P5) showed the second highest category for the P max, km, and Q 0, followed by the highly urban sub5, then by the NT (sub4), and least in sub1 and forest, respectively (Tables 4 and 5).
The sediments have finite P retention capacity and when saturated with P will reach the environmental limit, and the EPC estimation can differentiate this limit. The results indicated that in forest control sediments, the EPC was 2.73 and 3.01 μg L−1 during 2013 and 2014, respectively. The intensive agriculture sub-catchment under CT during both years (2013–2014) showed highest EPC (17.2 and 12.5 μg L−1), while low agriculture sub-catchment showed only high EPC (12.5 μg L−1) during 2014. Similarly, the sediments from highly punctual urban P source showed high equilibrium concentration (5.5 and 6.7 μg L−1) when compared to the forest. The sediments collected on the main river showed lower EPC concentration when compared to the highly impacted catchments. However, the intermediate P2 and P4 showed higher value over rest of the three points (Tables 4 and 5).
Phosphorus desorption kinetics
Results regarding seasonal P desorption kinetics from different land uses in Guaporé region are presented in Figs. 3, 4, 5, and 6. Individually, across the four seasons, the highest (96.3 mg kg−1) instantaneous bioavailable particulate P (α) was recorded for highly anthropic urban site during winter 2013, followed by the intensive agriculture site under CT with the (57.8 mg P kg−1) during winter 2014. While on the main river points from P1 to P5, a descending trend in α content was observed. The relative increase in α of disturbed sediment over native forest ranged from 19.6 to 175.3 % in summer and 8 to 235.5 % in winter during 2013, while it ranged from 53.2 to 165.8 % in summer and 15.1 to 123.2 % in winter during 2014 (Fig. 3).
Changes in the readily desorbable P (α, bioavailable P) of suspended sediment samples during summer-winter of 2013 and 2014 across 11 different monitoring sites in Guapore catchment, Southern Brazil. Lowercase letters on each bar show seasonal variation among different land use, while uppercase letters indicate annual variation within monitoring site and the values followed by the same letter are not significant at p < 0.05 according to the Scott-Knott test
Similarly, the potentially bioavailable P (β) exhibited varied response and showed an order in land use as highly anthropic sediments > intense agriculture under CT > intense agriculture under NT >low agriculture sub-catchments (sub1 and sub2) > native forest. The main river points (P1 to P5) showed relatively higher amount than agriculture treatments due to receiving highly diffused P inputs but followed the descending order along the river continuum (Fig. 4). The relative increase in potentially bioavailable P varied from 19.6 to 146 % in summer and 31.4 to 312.2 % in winter during the year 2013, and during 2014 from 39.6 to 148.3 % in summer and 31.3 to 110.9 % in winter. Among seasons, winter sediment considerably showed higher values than the summer at sub-catchment scale, while a less clear difference in the main river sediments was observed.
Changes in the potentially available P (β, potentially available P) of suspended sediment samples during summer-winter of 2013 and 2014 across 11 different monitoring sites in Guapore catchment, Southern Brazil. Lowercase letters on each bar show seasonal variation among different land use, while uppercase letters indicate annual variation within monitoring site and the values followed by the same letter are not significant at p < 0.05 according to the Scott-Knott test
The results of the desorption rate constant (λ) revealed that the highest values were recorded for downstream main river sediments (P3 and P4) during winter 2013. The less anthropic sediments (forest, sub1 and sub2) showed lower λ values when compared to the intense agriculture and highly anthropic sediments (Fig. 5). To estimate the sediment eutrophication hazard, the ratios between α and β have been used as a useful tool to describe the P mobility index. Averaging across seasons, among the monitored sites, the sediments from low anthropic sub-catchments have higher values for α/β ratios (forest = 26.4 %, sub1 = 29 % and sub2 = 25 %) than the highly anthropic sub5 (23.9 %). The sediments from the main river showed higher value of P mobility index than the native forest but they did not follow the upstream-downstream phenomena (Fig. 5).
Changes in the desorption rate constant (λ) of the first-order desorption reaction of suspended sediment samples during summer-winter of 2013 and 2014 across 11 different monitoring sites in Guapore catchment, Southern Brazil. Lowercase letters on each bar show seasonal variation among different land use, while uppercase letters indicate annual variation within monitoring site and the values followed by the same letter are not significant at p < 0.05 according to the Scott-Knott test
The cumulative P release revealed that the different sediments showed a clear desorption behavior for all four seasons during the 2013 and 2014 (Fig. 7). To attain the nearly complete removal of sediment-bound P from all sediments, 13 successive (192 h) AER extractions were performed to reach nearly a constant P concentration, although the sediment of low anthropic activity (forest, sub1, and sub2) attained this level between 7 to 10 extractions. Figure 7 indicates that in each season, the successive resin extraction resulted in maximum of cumulative P in highly anthropic urban sub5 sediments while lowest in the native forest sediments.
Principal component analysis and correlations
The principal component analysis (PCA) of P sorption-desorption parameters and sediment characteristics helped to identify the main factor that control the P transformations at the sediment-water interface. The sum of the first two factors accounted for 70.7 % of the variance of sediment data (Fig. 8). Factor 1 showed 53.1 % of total variance and was mainly related with Fedcb, Feox, α, β, IP, and Aldcb. As α and β are the fractions of IP and ultimately of TP, they are naturally correlated. Therefore, the important factor that correlates them under these environments is Fe/Al oxides. This indicates that the instantaneous bioavailable P and potentially bioavailable P concentration are strongly controlled by Fedcb and Feox. Factor 2 accounting for 17.6 % of total variability and EPC and P max was strongly correlated to clay and TOC contents. The PCA further revealed that at sub-catchment scale, the intense agriculture under NT (sub4), highly anthropic–urban (sub5), and P1 sediment showed the highest values for P sorption-desorption parameters, while intensive agriculture under CT (sub3) and main river P2 and P4 ranked in the second highest group.
The Pearson correlation revealed that many P sorption-desorption parameters and sediment characteristics have a significant correlation. The highest significant correlation coefficients between α and β with Fe and Al extracted by ammonium oxalate and dithionite–citrate–bicarbonate were recorded (r = 0.68 to 0.84). The P max was correlated with clay content (r = 0.65) and TOC content (r = 0.60), while EPC was only correlated with clay content (r = 0.67; Table 7).
Discussion
Variation in sediment phosphorus and chemical characteristics
Phosphorus mainly reaches the river system through domestic wastes and agricultural runoff; however, several biogeochemical factors and landscape hydrodynamics control this phenomenon. Firstly, the land use and managements play a key role. The study landscape showed a great variation in land use and soil management. The land cover variations are key contributor, and a shift from forest sub-catchment (0 % arable vs. 69 % forest) to intensive agriculture either under CT-sub3 (50 % arable vs. 37 % forest) or under NT-sub4 (71 % arable vs. 17 % forest) showed direct influence on the runoff and sediment delivery resulting in higher amount of TP, IP, and OP transport from these catchments. Similarly, the urban source (sub5) is directly receiving untreated city discharges into the river. Recent studies in the Guaporé catchment have shown high hydrological inundation, steep slope in CT sub-catchment, and high sediment production from agriculture catchments even under the NT management (Didoné et al. 2014; Minella et al. 2015). Similarly, Blanc et al. (2014) have shown a significant variation in the upstream and downstream river P concentration in urban Brazil. The landscape geology, geographical heterogeneity, and hydrological feature have a great control on the surface erosion and sediment transportation, and it can also be described that increasing area under NT can enhance the P export due to P saturation of top layer as recently reported by Sprague and Gronberg (2012) and Upreti et al. (2015).
Similarly, particle size distribution is also crucial while accounting for sediment-bound P. In our study, the sub-catchments under CT and steep slopes (sub2 and sub3) showed higher clay content than the NT (sub4) and urban catchment. The increased clay content in the CT catchments could be the result of tobacco cultivation during the winter months, and soil is thoroughly tilled and more vulnerable to water erosion and thus transports higher amounts of clay in the suspended colloids. Similar results were found by the Kröger et al. (2012) with changing land use and geography, while Zhang and Huang (2007) and Cooper et al. (2015) have reported that at even a small percentage (16–20) of the total weight, the clay fraction can account up to 70 % of the total surface area.
A complex overall situation with respect to defining P loss arises as a result of the size selective nature of soil erosion events. The loss of total P in the finer-eroded fraction would be greater relative to the original whole soil; however, this more easily transported material may support a lower solution P concentration compared to coarser-sized fractions (Scalenghe et al. 2007).
The mineralogical heterogeneity and geochemical composition, particularly Fe/Al content in the landscape, are other key features that regulate the form and fate of P along the river continuum. In the 1:1 type clays, specially kaolinite, the aluminum groups strongly attract proton that is generally protonated (Al-OH2 +), and it can be easily displaced by phosphate causing strongly bonded specific adsorption with and low equilibrium concentration (Chen et al. 1973). The P sorption-desorption parameters indicate that the iron hydroxides are the major binding agent of ion phosphate in these soils (Parfitt 1978; Sposito 2008).
Most soils of the study area are Oxisols/Ultisols, and these soils have a high content of Fe and Al oxides. The intensive site under CT (sub3) lies in Ultisols while that under NT and urban are located under Oxisols. Earlier studies in the region on many soils and sediments have shown similar geochemical behavior of P due to their high clay and Fe/Al contents (Bortoluzzi et al. 2013, 2015; Fink et al. 2014, 2016).
Another factor that might influence the P export is the successive surface application of manure and slurry under NT over prolonged time on small landholdings (sub4). This can saturate the surface charges and thus create a high risk to water quality (Sharpley 2003). Similarly, the highest OP concentration in NT sediment explains the evidence of soil OP in the top carbon stratified layer in the area. Many recent studies have proved that the fertilization and crop management in NT accelerate particulate bound P and its desorption from soils and suspended sediments (Kaiser et al. 2009; Borda et al. 2014; Couto et al. 2015; Fink et al. 2016). On the other hand, under CT, the freshly applied industrial P fertilizer had great risk to be lost during the tobacco cultivation. The impact is coincidently accelerated by the high rainfall during the same period (July to October) as observed in an earlier study (Pellegrini et al. 2010).
Phosphorus sorption
Land-based P enters to the river water mainly found as particulate P (PP) and often exceeds 80 % of the TP, as it can be stored in sediments via adsorption. Under acidic subtropical environments, the greater portion of PP is present as IP and occurs principally as Al-P or Fe-bound P and also as Ca-P in relatively small proportions. Many studies have documented that relationship (Némery and Garnier 2007; Withers and Jarvie 2008; Jing et al. 2012; Meng et al. 2015).
In this study, we used paired forest (natural) sediments as a reference for the comparison with anthropically impacted sediments. The relatively lower values of P max, km, Q 0, and EPC in forest sediments are attributed to the low clay content and low Al/Fe oxides and the fact that this area was never fertilized with external P and no anthropic activity. This leads to a very low release of P when these sediments reach to the river waters. A similar trend of P release in forest sediment was recorded by Pellegrini et al. (2010) and Zhu et al. (2011), and the latter authors found less seasonal variability in P release from forest sediments as they are nearly under balanced equilibrium.
Conversely, the sediments coming from intensively cultivated site under CT (sub3 = 50 % arable) located on steep slope landform and coupled with high rainfall generating very high erosion (Didoné et al. 2014) with a higher clay content, and higher amount of ammonium oxalate and or dithionite–citrate–bicarbonate extracted Al and Fe. Moreover, this site receives a high dose of industrial fertilizers and animal waste. Consequently, the sediments originated yet showed the higher capacity to absorb free P from the river water. Many authors have shown the high P retention capacity of the colloids under 1:1 type clays and where Al and Fe predominance (Bortoluzzi et al. 2013, 2015; Jan et al. 2015; Fink et al. 2016).
Similarly, the low agriculture catchment under CT (sub2) is located in the hilly area, and sediment predominantly comes from conventional cultivation areas (Tiecher et al. 2015). Due to the smaller area under cultivation in relation to sub3 (35 vs. 50 %), and the variation in forest coverage (50 % vs. 37 %), it justifies sediments’ intact nature with lower clay, low Alox, Feox and Aldcb, Fedcb that leads to lower P max, Q 0, and EPC (Tables 4 and 5). The sediments of the sub1, although with the same percent of the arable area as that cultivated in sub3 but high coverage with native vegetation, emphasize the importance of the distribution of arable and forest vegetation in the landscape ecology (Table 6). A similar result in earlier studies explaining the connectivity of P source and the river is critical and that upstream land use is influential and regulates the sediment P retention (Buck et al. 2004; Withers and Jarvie 2008; Pellegrini et al. 2010).
The sediment originated from intensive agriculture under NT (70 % arable and only 17 % forest) has low P max, very low km, lower Q 0 and especially much smaller value of EPC in comparison to under CT (50 % arable and 37 % forest).
This indicates the physical conservation of soil under NT (low erosion) but higher P loss risk due to increased fine sediment transportation from top phosphate saturated soil layer. A similar observation in recent studies has raised a serious question on the suitability of prolonged NT (>15 years) in relation to P release and water quality (Sharpley et al. 2013; Borda et al. 2014; Gatiboni et al. 2015).
Untreated domestic and city discharges are also a major source of P to rivers. In this study, we can compare two sub-catchments with the same proportion of arable area (70 %) and are under similar land use and soil management system (NT-soybean) (sub4 and sub5). The urban site receives untreated domestic sewage and agro-industrial discharges of the Marau city (45,000 inhabitants). This induces a high physico-chemical variation in sediments’ behavior and affect the sediment P dynamics over time. The adsorption results reported here reflect this effect, and many authors have shown urban P pollution in sediment with higher P max, km, Q 0, and EPC0 with lower clay content. Therefore, these sediments can easily act as a P source in the river system (Peterse et al. 2003; Jarvie et al. 2012; Blanc et al. 2014).
The overall particle size distribution indicated that the sediment from intensive agriculture and CT sub-catchment (sub3) with higher clay content showed higher values of modified Langmuir parameters i.e., P max, km, Q 0, and EPC0. Earlier studies under the similar soil and management system have shown a relationship between clay content and P sorption for high P max value (Agudelo et al. 2011; Bortoluzzi et al. 2013; Erikkison et al. 2015; Fink et al. 2016).
The main river sediments showed a great variability along the river continuum for P sorption. As the diffuse sources are invariable related to the land use, agriculture, animal production, there are several individual sources which define the P export potential of that all upstream drainage area. A better interpretation of these anthropogenic sources in relation to their runoff and sediment loss can help to differentiate their rate of P delivery and its composition (Minella et al. 2015; Tiecher et al. 2015). In this study, the P1 sediments receiving P input from the 70 % arable under NT and urban contributions showed high P max and showed a decreasing trend downstream except P4. This point of the river lies downstream too much of the conventional cultivation and receives the similar sediment as of two neighboring sub-catchment under CT (sub2 and sub3). Therefore, it was likely obvious that runoff from these CT sediments showed higher seasonal values for P max.
Phosphorus desorption
The significant variability in AER-extracted P in the different sediments is a useful tool to predict the instant bioavailable P and P buffering capacity and has been used in many soil and sediment P desorption studies (McKean and Warren 1996; Rheinheimer and Anghinoni 2001; Pellegrini et al. 2010; Su et al. 2014; Fink et al. 2016).
P-desorption-based indices require careful calibration for each desorption scenario. Torrent and Delgado (2001) proposed an index relatively insensitive to soil type, which makes it potentially useful in areas with high soil diversity. The low values of α and β in the forest and low anthropic sub1 were detected when compared to the intensive agriculture sub2 and sub3. This indicates that P is poorly retained in the forest and sub1 sediment than sub3 and sub4. This phenomenon can be contributed to the presence of high amount of binding partners of P in the form of amorphous and crystalline Fe or Al in intensively cultivated soils. This is also indicated by the higher correlation between α and Feox, Fedcb and Alox, Aldcb (r = 0.72, 0.84; 0.69, 0.74—Fig. 8, Table 6) and between β and Feox, Fedcb and Alox, Aldcb (r = 0.74, 0.83; 0.75, 0.78—Fig. 7, Table 6). The high potential of bioavailable P and cumulative desorbed P during winter months can be attributed to the high rainfall erosivity and increased sediment production during the tobacco cultivation (Pellegrini et al. 2010; Didoné et al. 2014).
The higher α and β in urban sediment (sub5) are directly related to the higher amount of untreated sewage and P discharges but are variable and are a combined function of both background agricultural loads and intensity of urban P export. This trend is very obvious in sub5 as indicated by the continuous higher β values (i.e., 396 vs. 96 mg kg−1) than in forest during winter 2013 as well as overall sub5 which showed highest P buffering capacity in all four seasons during 2013 and 2014 (Fig. 6). The inconsistent pattern of the main river sediment towards the α and β again shows the ambiguity and overlapping of several factors contributing to sediment and P transport. However, when compared to the forest, all five sediments showed a double increase for α and β values.
Variation in the mobility index (α × 100/β) of suspended sediment samples during summer-winter of 2013 and 2014 across 11 different monitoring sites in Guapore catchment, Southern Brazil. Lowercase letters on each bar show seasonal variation among different land use, while uppercase letters indicate annual variation within monitoring site and the values followed by the same letter are not significant at p < 0.05 according to the Scott-Knott test
These results imply that at sub-catchment scale, the intensive agriculture (sub3 and sub4) and urban (sub5) monitoring locations have specifically sediment-bound P which is stable and can be transported over longer distances in the landscape and or receiving water bodies as later evidenced by the consistent high values of α and β in the main river sediments along the river continuum than forest. The similar results for P desorption parameters were found in conditions (Tiessen et al. 1984; Agudelo et al. 2011).
The relatively higher desorption rate constant (λ) values in downstream sediment are indicative of higher organic carbon in these sediments with increasing drainage area and to some extent, also, higher surface runoff during winter months. However, during the summer season, relatively higher λ values in forest sediment than agriculture sediments are indicative that forest sediment can rapidly desorb P than the specifically bound P in agriculture sediment. Similarly, the higher values for the P mobility index (α / β ratios) both in the reference forest and relatively less disturbed (sub1) sediments than those of intensive agriculture (sub3 and sub4) and urban (sub5) clearly indicate that forest and low anthropic sediments easily desorb P than the sediments from the high agriculture and anthropic influence. Our results are in accordance with the Pellegrini et al. (2010), Guardini et al. (2012), and Fink et al. (2016) and authors observed a similar trend for P desorption parameters for low and high P conditions.
Conclusions
This intra-catchment comparison study has reflected the seasonal P sorption-desorption potential of sediments in relation to landscape controls using a range of sediment properties, catchment land use, and demographic characteristics to estimate P release potential and improve different upstream land use and management approaches. The seasonal and intra-catchment variation in TP and sediment-bound P release appeared as the combined effect of sediment mineralogical composition, landscape geographic features, and land use and management. However, the TP and IP showed higher correlation towards Feox concentration. Within the different monitored sites, the suspended sediment from intensive agriculture site under CT presented higher P storage (P max) which is closely associated with the high Al/Fe-P complexes. This leads to the lower EPC values indicating the high P release to the river waters. The desorption kinetics showed that urban (sub5) intensive agriculture (sub3 and sub4 including sub2) and P1 to P5 (main river) showed higher bioavailable P (α) and total desorbed P (β), which can pose a greater threat to stream waters by acting as P source during seasonal shifts and oxic-anoxic variations on sediment-water interface in the presence of high Fe, Al, and Mn ions. Finally, desorption rate (λ) and the P mobility index (α × 100/β) were highest in intensive agriculture sediment (sub3) as well as in highly anthropic (sub5), and main river sediment, though less pronounced variation was observed. The results clearly suggest that improvement in reduction of soil-based P is required to maintain water quality and implement the conservation practices to understand eutrophication process for wise hydrological source management.
References
Agudelo, S. C., Nelson, N. O., Barnes, P. L., Keane, T. D., & Pierzynski, G. M. (2011). Phosphorus adsorption and desorption potential of stream sediments. Journal of Environmental Quality, 40, 144–152.
Barrow, N. J. (1983). A mechanistic model for describing the sorption and desorption of phosphate by soil. Journal of Soil Science, 34, 733–750.
Blanc, L. R., Moreira, S., Gonçalves, A. M., Serafim, R., Abreu, S., Baroni, L., et al. (2014). Contamination in a Brazilian river: a risk of exposure to untreated effluents. Journal of Environmental Quality, 42, 1596–1601.
Borda, T., Celi, L., Bünemann, E. K., Oberson, A., Frossard, E., & Barberis, E. (2014). Fertilization strategies affect phosphorus forms and release from soils and suspended solids. Journal of Environmental Quality, 43, 1024–1031.
Bortoluzzi, E. C., dos Santos, D. R., Santanna, M. A., & Caner, L. (2013). Mineralogy and nutrient desorption of suspended sediments during a storm event. Journal of Soils and Sediments, 13, 1093–1105.
Bortoluzzi, E. C., Pérez, C. A. S., Ardisson, J. D., Tiecher, T., & Caner, L. (2015). Occurrence of iron and aluminum sesquioxides and their implications for the P sorption in subtropical soils. Applied Clay Science, 104, 196–204.
Bubba, D. M., Arias, C. A., & Brix, H. (2003). Phosphorus adsorption maximum of sands for use as media in subsurface flow constructed reed beds as measured by the Langmuir isotherm. Water Research, 37, 3390–3400.
Buck, O., Niyogi, D. K., & Townsend, C. R. (2004). Scale-dependence of land use effects on water quality of streams in agricultural catchments. Environmental Pollution, 130, 287–299.
Capoane, V., Tiecher, T., Schaefer, G. L., Ciotti, L. H., & Rheinheimer, D. S. (2015). Transferência de nitrogênio e fósforo para águas superficiais em uma bacia hidrográfica com agricultura e produção pecuária intensiva no Sul do Brasil (Transfer of nitrogen and phosphorus to surface water in a watershed with agriculture and intensive livestock production in Southern Brazil). Ciência Rural, 45, 647–650.
Chen, Y. S. R., Btuter, J. N., & Stumm, W. (1973). Kinetic study of phosphate reaction with aluminum oxide and kaolinite. Environmental Science and Technology, 7, 327–332.
Caner, L., Radtke, L. M., Vignol-lelarge, M. L., Inda, A. V., Bortoluzzi, E. C., & Mexias, A. S. (2014). Basalt and rhyo-dacite weathering and soil clay formation under subtropical climate in Southern Brazil Rio Grande do Sul State South America. Geoderma, 235-236, 100–112.
Cooper, R. J., Rawlins, B. G., Krueger, T., Lézé, B., Hiscock, K. M., & Pedentchouk, N. (2015). Contrasting controls on the phosphorus concentration of suspended particulate matter under base flow and storm event conditions in agricultural headwater streams. Science of the Total Environment, 533, 49–59.
Couto, R., Santos, M., Comin, J. J., Carlos, L., Martini, P., Colpo, L., et al. (2015). Environmental vulnerability and phosphorus fractions of areas with pig slurry applied to the soil. Journal of Environmental Quality, 44, 162–173.
Didoné, E. J., Gomes, J. P., Reichert, J. M., Merten, H., Dalbianco, L., Peixoto Barrros, C. A., & Ramon, R. (2014). Impact of no-tillage agricultural systems on sediment yield in two large catchments in Southern Brazil. Journal of Soils and Sediments, 14, 1287–1297.
ESRI (2011). ArcGIS desktop: release 10. Redlands, CA: Environmental Systems Research Institute.
Ferreira, D. F. (2011). SISVAR: a computer statistical analysis system. Ciencia e Agrotecnologia (Science and Agrotechnology), 35, 1039–1042.
Fink, J. R., Inda, A. V., Bavaresco, J., Barrón, V., Torrent, J., & Bayer, C. (2016). Adsorption and desorption of phosphorus in subtropical soils as affected by management system and mineralogy. Soil and Tillage Research, 155, 62–68.
Fink, J. R., Inda, A. V., Bayer, C., Torrent, J., & Barrón, V. (2014). Mineralogy and phosphorus adsorption in soils of south and central-west Brazil under conventional and no-tillage systems. Acta Scientiarum Agronomy, 36, 379–387.
Gatiboni, L. C., Kaminski, J., & Santos, D. R. (2005). Modificações nas formas de fósforo do solo após extrações sucessivas com Mehlich-1, Mehlich-3 e resina trocadora de ânions (Changes in soil phosphorus forms after successive extractions with Mehlich-1, Mehlich-3 and anion exchange resin). Revista Brasileira de Ciência do Solo (Brazilian Journal of Soil Science), 29, 363–371.
Gatiboni, L. C., Thomas, J. S., Schmitt, D. E., Cassol, P. C., & Oliveira, C. M. B. (2015). Soil phosphorus thresholds in evaluating risk of environmental transfer to surface waters in Santa Catarina, Brazil. Revista Brasileira de Ciência do Solo (Brazilian Journal of Soil Science), 39, 1225–1234.
Guardini, R., Comin, J. J., Rheinheimer, D., Gatiboni, C., Tiecher, T., Schmitt, D., et al. (2012). Phosphorus accumulation and pollution potential in a Hapludult fertilized with pig manure. Revista Brasileira de Ciência do Solo (Brazilian Journal of Soil Science), 36, 1333–1342.
House, W. A., & Denison, F. H. (2000). Factors influencing the measurement of equilibrium phosphate concentrations in river sediments. Water Research, 34, 1187–1200.
IUSS Working Group WRB. (2015). World reference base for soil Resources 2014, update 2015 International soil classification system for naming soils and creating legends for soil maps. World Soil Resources Reports No. 106. Rome: FAO.
Jan, J., Borovec, J., Kopacek, J., & Hejzlar, J. (2015). Assessment of phosphorus associated with Fe and Al (hydr) oxides in sediments and soils. Journal of Soils and Sediments, 15, 1620–1629.
Jarvie, H. P., Sharpley, A. N., Scott, T., Haggard, B., Bowes, M., & Massey, L. (2012). Within-river phosphorus retention: accounting for a missing piece in the watershed phosphorus puzzle. Environmental Science and Technology, 46, 13284–13292.
Jarvie, H. P., Neal, C., & Withers, P. J. A. (2006). Sewage-effluent phosphorus: a greater risk to river eutrophication than agricultural phosphorus? Science of the Total Environment, 360, 246–253.
Jing, Z., Min, L. I., Shuang, L. I. U., Yujin, L. I. U., Liqiang, Z., Qi, C. A. O., & Dezhi, S. U. N. (2012). Seasonal variations and bioavailability of inorganic phosphorus in soils of Yeyahu wetland in Beijing, China. International Journal of Sediment Research, 26, 181–192.
Erikkison, K. A., Petter, J., & Hesterberg, D. (2015). Phosphorus speciation of clay fractions from long-term fertility experiments in Sweden. Geoderma, 241-242, 68–74.
Lopez, P., Lluch, X., Vidal, M., & Morgui, J. A. (1996). Adsorption of phosphorus on sediments of the Balearic Islands (Spain) related to their composition, estuarine. Coast and Shelf Science, 42, 185–196.
Kaiser, D. E., Mallarino, A. P., Haq, M. U., & Allen, B. L. (2009). Runoff phosphorus loss immediately after poultry manure application as influenced by the application rate and tillage. Journal of Environmental Quality, 38, 299–308.
Kim, J., Li, W., Philips, B. L. & Grey, C. P. (2011). Phosphate adsorption on the iron oxyhydroxides goethite (α-FeOOH), akaganeite (β-FeOOH), and lepidocrocite (γ-FeOOH): a 31P NMR study. Energy and Environmental Science, 4, 4298–4305.
Koski-Vähälä, J., & Hartikainen, H. (2001). Assessment of the risk of phosphorus loading due to resuspended sediment. Journal of Environmental Quality, 30, 960–966.
Kröger, R., Lizotte, R. E., Shields, F. D., & Usborne, E. (2012). Inundation infl uences on bioavailability of phosphorus in managed wetland sediments in agricultural landscapes. Journal of Environmental Quality, 41, 604–612.
Upreti, K., Joshi, S. R., McGrath, J., & Jaisi, D. P. (2015). Factors controlling phosphorus mobilization in a coastal plain tributary to the Chesapeake Bay. Soil Science Society of Ameriian Journal, 79, 826–837.
Meng, J., Yu, Z., Yao, Q., Bianchi, T. S., Paytan, A., Zhao, B., et al. (2015). Distribution, mixing behavior, and transformation of dissolved inorganic phosphorus and suspended particulate phosphorus along a salinity gradient in the Changjiang Estuary. Marine Chemistry, 168, 124–134.
Mckean, S. J., & Warren, G. P. (1996). Determination of phosphate desorption characteristics in soils using successive resin extractions. Communication in Soil Science and Plant Analysis, 27, 2397–2417.
Muggler, C. C., Pape, T., & Buurman, P. (1997). Laser grain—size determination in soil genetic studies 2. Clay content, clay formation, and aggregation in some Brazilian Oxisols. Soil Science, 162, 219–228.
Némery, J., & Garnier, J. (2007). Typical features of particulate phosphorus in the Seine estuary (France). Hydrobiologia, 588, 271–290.
Maier, C. (2013). Variabilidade intra-evento da origem das fontes de sedimentos em uma bacia hidrográfica rural. (Doctoral Thesis), Water Resources and Environmental Sanitation. Brazil: Universidade Federal do Rio Grande do Sul (in Portuguese).
Mehra, O. P., & Jackson, M. L. (1960). Iron oxide removal from soils and clays by a dithionitecitrate system buffered with sodium bicarbonate. Clay Minerals, 7, 317–327.
Minella, J. P. G., Merten, G. H., Alessandra, C. & Barros, P. D. (2015). Long-term sediment yield from small catchment in Southern Brazil affected by land use and soil management changes. EGU General Assembly 2015. Geophysical Research Abstracts, 17, 10803.
Murphy, J., & Riley, J. P. (1962). A modified single solution method for the determination of phosphate in natural waters. Analytica Chimica Acta, 27, 31–36.
Olsen, S. R., & Sommers, L. E. (1982). Phosphorus. In A. L. Page, R. H. Miller, & Y. Q. R. Keene (Eds.), Methods of soil analysis, part 2, second ed. Chemical and microbiological properties (pp. 403–430). Madison: Soil Science Society of America.
Parfitt, R. L. (1978). Anion adsorption by soils and soil materials. Advances in Agronomy, 30, 01–46.
Pellegrini, J. B. R., Santos, D. R., Gonçalves, C. S., Copetti, A. C. C., Bortoluzzi, E. C., & Tessier, D. (2010). Impacts of anthropic pressures on soil phosphorus availability, concentration, and phosphorus forms in sediments in a Southern Brazilian watershed. Journal of Soils and Sediments, 10, 451–460.
Peterse, N. M., Bleuten, W., & Jorgensen, S. E. (2003). Contribution of point sources and diffuse sources to nitrogen and phosphorus loads in lowland river tributaries. Journal of Hydrology, 271, 213–225.
Rheinheimer, D. S., Anghinoni, I., & Kaminski, J. (2000). Depleção do fósforo inorgânico de diferentes frações provocada pela extração sucessiva com resina em diferentes solos e manejos (Depletion of inorganic phosphorus different fractions caused by successive extractions with resin in different soils and managements). Revista Brasileira de Ciência do Solo (Brazilian Journal of Soil Science), 24, 345–354.
Rheinheimer, D. S., & Anghinoni, I. (2001). Distribuição do fósforo inorgânico em sistemas de manejo de solo (Distribution of inorganic phosphorus fraction in soil management systems). Brazilian Journal of Agricultural Research, 36, 151–160.
Rheinheimer, D. S., Gatiboni, L. C., & Kaminski, J. (2008). Fatores que afetam a disponibilidade do fósforo e omanejo da adubação fosfatada emsolos sob sistema plantio direto (Factors affecting the phosphorus availability and the fertilization management in no tillage system). Ciência Rural, 38, 576–586.
Schwertmann, U. (1964). Differenzierung der eisenoxide des bodens durch extraction mit ammoniumoxalat-losung. Z. Pflanzenernähr. Bodenkd. Journal of Plant Nutrition and Soil Science, Germany, 105, 194–202.
Scalenghe, R., Edwards, A. C., & Barberis, E. (2007). Phosphorus loss in overfertilized soils: the selective P partitioning and redistribution between particle size separates. European Journal of Agronomy, 27, 72–80.
Sharpley, A., Jarvie, H. P., Buda, A., May, L., Spears, B., & Kleinman, P. (2013). Phosphorus legacy: overcoming the effects of past management practices to mitigate future water quality impairment. Journal of Environmental Quality, 42, 1308–1326.
Sharpley, A. N. (2003). Plowing to decrease surface stratification of phosphorus in manured soils. Journal of Environmental Quality, 32, 1375–1384.
Sposito, G. (2008). The chemistry of the soils, (2nd ed). New York: Oxford University Press.
Sprague, L. A., & Gronberg, J. A. M. (2012). Relating management practices and nutrient export in agricultural watersheds of the United States. Journal of Environmental Quality, 41, 1930–1950.
Su, J., Bochove, E. V., Auclair, J., Thériault, G., Denault, J., Bossé, C., et al. (2014). Phosphorus algal availability and release potential in suspended and streambed sediments in relation to sediment and catchment characteristics. Agriculture, Ecosystem and Environment, 188, 169–179.
Tiecher, T., Caner, L., Minella, J. P. G., Bender, M. A., & dos Santos, D. R. (2015). Tracing sediment sources in a subtropical rural catchment of Southern Brazil by using geochemical tracers and near-infrared spectroscopy. Soil and Tillage Research, 155, 478–491.
Tiessen, H., Stewart, W. B., & Cole, C. V. (1984). Pathways of phosphorus transformations in soils of differing pedogenesis. Soil Science Society of American Journal, 48, 853–858.
Torrent, J., & Delgado, A. (2001). Using phosphorus concentration in the soil solution to predict phosphorus desorption to water. Journal of Environmental Quality, 30, 1829–1835.
Tu, C., Zheng, C. R., & Chen, K. M. (2002). Effect of heavy metals on phosphorus retention by typic udic ferrisols: equilibrium and kinetics. Pedosphere, 12, 15–24.
Walkley, A., & Black, I. A. (1934). An examination of the Degtjareff method for determining soil organic matter and a proposed modification of the chromic acid titration method. Soil Science, 37, 29–38.
Withers, P. J. A., & Jarvie, H. (2008). Delivery and cycling of phosphorus in rivers: a review. Science of the Total Environment, 400, 379–395.
Withers, P. J. A., Dijk, K. C. V., Neset, T. S., Nesme, T., Oenema, O., Rubæk, G. H., et al. (2015). Stewardship to tackle global phosphorus inefficiency: the case of Europe. Ambio, 44(s), 193–206.
Zhang, W., Tang, W., Zhang, H., Bi, J., Jin, X., Li, J., et al. (2014). Characterization of biogenic phosphorus in sediments from the multi-polluted Haihe River, China, using phosphorus fractionation. Ecological Engineering, 71, 520–526.
Zhang, J. Z., & Huang, X. (2007). Relative importance of solid-phase phosphorus and iron on the sorption behavior of sediments. Environmental Science & Technology, 41, 2789–2795.
Zhu, B., Wang, Z., & Zhang, X. (2011). Phosphorus fractions and release potential of ditch sediments from different land uses in a small catchment of the upper Yangtze River. Journal of Soils & Sediments, 12, 278–290.
Acknowledgments
The study was supported in part by the Academy of Sciences for the Developing World (TWAS) and the Brazilian National Council for Scientific and Technological Development (CNPq) under process number 190334/2011-8 to first author and project entitle “Production systems aimed at increasing the availability and improving the water quality–Mais Água” number (550009/20132). We thank all the farmers of the Guaporé region, RS for their cooperation and access to their land and Alexandre Torian for transport facilities and help during all field campaigns.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Supplementary Table 1
(DOC 66 kb)
Rights and permissions
About this article
Cite this article
Zafar, M., Tiecher, T., de Castro Lima, J.A.M. et al. Phosphorus seasonal sorption-desorption kinetics in suspended sediment in response to land use and management in the Guaporé catchment, Southern Brazil. Environ Monit Assess 188, 643 (2016). https://doi.org/10.1007/s10661-016-5650-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-016-5650-3