Abstract
Voles are often considered as harmful pests in agriculture and silviculture. Then, the knowledge of their abundance may be of considerable economical importance. Commonly used methods in the monitoring of vole abundances are relatively laborious, expensive, and spatially quite restricted. We demonstrate how the mean clutch size of the tawny owl Strix aluco may be cost-effectively used to predict relative densities of voles over large areas. Besides installing a number of suitable nest boxes, this vole monitoring system primarily includes only the inspection of the nest boxes and counting the number of tawny owl eggs found two times during a few weeks period in spring. Our results showed a considerable agreement between the fluctuations in the mean clutch size of tawny owls and the late spring abundance indices of small voles (Myodes, Microtus) in our study areas in southern Finland. The mean clutch size of the tawny owl reflected spring vole abundance over the spatial range examined, suggesting its suitability for general forecasting purposes. From the pest management point of view, an additional merit of the present method is that it may increase numbers of vole-eaters that provide biological control of vole populations.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Voles and other rodents are commonly considered as pests in agriculture (Myllymäki 1979; Stenseth et al. 2003; Brown et al. 2007) and silviculture (Gill 1992; Baxter and Hansson 2001; Huitu et al. 2009). Therefore, the knowledge of their abundance may be of considerable economical importance (Stenseth et al. 2003; Huitu et al. 2009; Jacob and Tkadlec 2010; Jacob et al. 2014; Jareno et al. 2014). Consequently, in terms of pest control, monitoring of small rodents has been conducted at large scales in various parts of the world (e.g., Huitu et al. 2009, Witmer et al. 2009, Jareno et al. 2014). Such projects, in general, provide regional data on the abundance of voles. Knowledge about higher vole abundance can allow agriculturalists and silviculturalists to anticipate greater damage in the coming season and make appropriate mitigation measures. However, due to the well-known spatiotemporal heterogeneity in the occurrence of voles (e.g., Goszczyński et al. 1993; Petty and Fawkes 1997; Romanowski and Zmihorski 2009), the network of sampling areas in a large-scale monitoring project does not necessarily cover representatively all local situations. Therefore, more detailed local data, being more instructive and more usable in practical actions, would be preferable.
Snap trapping is a commonly used, relatively easy, and quite suitable method for monitoring vole abundances (e.g., Hansson and Hoffmeyer 1973; Redpath et al. 1995; Christensen and Hörnfeldt 2003). Live trapping is a more laborious but also more informative alternative (Flowerdew et al. 2003; Sibbald et al. 2006; Huitu et al. 2009). Besides direct monitoring by trapping, also various indirect methods based on different signs of voles in the field have been used (e.g., Hansson 1979, 1986; Lambin et al. 2000; Gervais 2010; Krebs et al. 2012; Jareno et al. 2014). In many countries, very simple vole indices are collected which are based on counts of burrow entrances (e.g., Lisická et al. 2007). Such indices are probably relatively easy to collect but their reliability should be evaluated. There may be many practical problems such as how the burrows are identified, how to separate the new and old burrows, how do the burrows reflect the numbers of pest species, etc.
So, it seems that all these methods are somewhat problematic and their spatial coverage is in practice relatively limited. As a tool for monitoring of small mammals, the analysis of food remains of vole-eating birds of prey (e.g., Glue 1971; Solonen and Karhunen 2002; Balčiauskienė 2005; Balčiauskienė and Naruševičius 2006; Sibbald et al. 2006) may be spatially more comprehensive than other indirect methods or direct trapping, but the collecting of samples and picking and identifying prey remains is still quite laborious.
Variation in breeding parameters of vole-eating birds of prey has been commonly explained by the fluctuating abundance of small voles (smaller than Arvicola, weighing less than 100 g, such as Myodes and Microtus spp.) (e.g., Linkola and Myllymäki 1969; Korpimäki 1984, 1992; Lõhmus 1999; Solonen 2005, 2010). It may be practical then to use breeding parameters of vole-eating birds of prey as a method to monitor vole abundance over large spatial scales in a cost-effective way (cf. Petty and Fawkes 1997). Population dynamics of vole-eating species of birds of prey commonly monitored by both amateur and professional ornithologists (Kovács et al. 2008; Saurola 2008) provide useful cues of cyclic abundance fluctuations of small voles over large spatial scales (e.g., Lõhmus 1999; Sundell et al. 2004). Particularly, Tengmalm’s owl (boreal owl) Aegolius funereus (Linnaeus 1758) has been shown to be suitable as such an indicator (e.g., Korpimäki 1984, 1992; Solonen 2004; Hörnfeldt et al. 2005). The occurrence of nomadic Tengmalm’s owls is, however, highly variable both spatially and temporally, and, in addition, recently, this earlier common species has become rare or occasional breeder in several parts of its range probably due to some unfavorable changes in environmental conditions (e.g., Korpimäki and Hakkarainen 2012). In such areas, at least, a more stationary, resident vole-eater might be a more suitable indicator of vole abundance.
In this paper, we examine if the mean clutch size of the tawny owl Strix aluco Linnaeus 1758 can be used as a proxy for the occurrence of voles (cf. Petty and Fawkes 1997). Clutch size has shown to be such a relatively easily available breeding parameter that commonly vary in accordance with food supply in vole-eating birds of prey (e.g., Korpimäki 1984; Korpimäki and Hakkarainen 1991; Solonen 2005). The tawny owl is a widespread bird of prey of rural and urban habitats in Europe (Mikkola 1983; Cramp 1985). It commonly occupies habitats in the vicinity of human settlements. A suitable nesting cavity is an essential prerequisite for the tawny owl’s breeding, which is otherwise strictly governed by an adequate availability of food. In our study areas in southern Finland, the availability of food for breeding tawny owls is particularly affected by the synchronous abundance fluctuations of small voles (Solonen 2004, 2010; Sundell et al. 2004), the bank vole Myodes glareolus (Schreber 1780) and field vole Microtus agrestis (Linnaeus 1761). The bank vole is a common pest in silviculture (e.g., Huitu et al. 2009) and the field vole in agriculture (e.g., Myllymäki 1979) over a wide range in Europe and Asia (Shenbrot and Krasnov 2005). Though their main habitats differ (bank voles mainly occupy forests and field voles fields), there is also considerable spatial overlap between the species, and their cyclic abundance fluctuations show considerable synchrony (e.g., Hansson 2002; Huitu et al. 2004; Solonen and Ahola 2010; Korpela et al. 2013). Recently, however, the regularity of the vole cycles has diminished, making the forecasting of vole abundance more difficult than before (e.g., Hörnfeldt et al. 2005; Korpela et al. 2013).
Our reasoning goes as follows: When owls lay large clutches (due to favorable food conditions based on abundant vole supply), there may exist plenty of voles still in late spring, whereas when small clutches prevail (due to bad food conditions based on poor vole supply), voles are probably scanty also in late spring. First, we show the general suitability of our data for demonstrating a vole abundance indicator by examining if there are significant positive relationships between our autumn vole indices (derived from snap trappings) and the mean clutch size of tawny owls in the next spring breeding season. Then, we study if the mean clutch size of tawny owls reflects vole abundance in late spring (during the last phases and after the breeding of owls). Such a relationship over a large spatial scale should imply general suitability of the indicator for forecasting purposes.
The autumn vole densities and the winter survival of voles are the basic elements determining the clutch size of vole-eating owls. So, if the clutch size should be a good indicator of vole abundance, there should be a significant relationship between the clutch size and both the autumn and spring densities of voles. Although the data on autumn densities of voles were lacking, the clutch size of vole-eating birds of prey should indicate the order of magnitude of vole densities both in the preceding autumn and current spring. After evaluating the indicator of vole abundance, we conduct a simple cost-benefit analysis between the methods used (snap trapping vs. mean clutch size of the tawny owl). We also formulate some recommendations for the effective use of the indicator.
Material and methods
Study areas and the field work
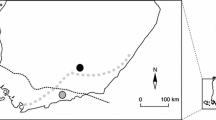
Our two local study areas on owls and voles were situated in Uusimaa, between the southern coast of Finland and the northern border of the hemiboreal zone (Solonen 2010). They mainly consisted of low-lying rural habitats of mixed fields and forests. The hemiboreal zone halfway between the temperate and subarctic boreal zones is characterized by mixed coniferous forests, relatively cold winters, and mild summers. Both data sets were collected following similar guidelines.
We monitored population dynamics of the tawny owl in a western study area of ca. 250 km2 (1981–2012; Karell et al. 2009) and in an eastern study area of ca. 500 km2 (1986–2000; Solonen and Karhunen 2002). The areas were situated about 50 km apart from each other. Nest boxes for the tawny owl were installed in trees ca. 3–4 m above the ground in suitable habitats (parks, gardens, forest edges, etc.) relatively uniformly over the study areas. The number of nest boxes was ca. 100 in the western study area and ca. 300 in the eastern study area. Thus, the densities of nest boxes in the study areas were about 0.4/km2 and 0.6/km2, respectively. Minimum dimensions of the wooden nest boxes were approximately as follows: height 60 cm, floor diameter 30 cm, and entrance diameter 12 cm. Because owls do not build a nest, the floor of the nest boxes was covered by a ca. 10 cm layer of sawdust or rotten wood. All the nest boxes were checked usually during the most probable incubation period of tawny owls in April, and the active nests found were re-examined after 1–3 weeks in order to determine the clutch size. The annual number of tawny owl nests found averaged 18 ± 9 (SD, n = 32) and 25 ± 8 (SD, n = 15) in our western study area and our eastern study area, respectively.
We characterized the general abundance of voles during the breeding season of owls by the results of snap trappings. We caught voles in both autumn and spring at three sites in two periods of time between 1981 and 2012. The longer-term trapping in two western study sites (situated 13 km apart in Lohja, 60° 16′ N, 24° 12′ E and Kirkkonummi, 60° 13′ N, 24° 24′ E), situating in our western owl monitoring area covered the whole study period. The smaller-scale trapping effort in our easternmost study site (Sipoo, 60° 18′ N, 25° 10′ E) in the eastern owl monitoring area was conducted between 1986 and 2000. In the western study sites, we conducted snap trappings along four transects (two in Lohja and two in Kirkkonummi) (Solonen and Ahola 2010). At each line, we used 16 points of three traps, the points located about 25 m from each other, during two 24-h trapping periods (totalling 384 trap nights in each trapping). On the assumption of a minimum radius of about 25 m of effective catching, the spatial coverage of trapping in these two sites totalled about 0.1 km2. In the eastern study site (Sipoo), we trapped small mammals at 30 standard points of three snap traps along a 1.5-km transect (total of 90 traps) throughout a 24-h period (Solonen and Karhunen 2002). The spatial coverage of the effort can then be estimated as about 0.1 km2. The results (abundance of voles) were expressed as catch indices, indicating the number of individuals caught per 100 trap nights. We conducted the autumn trappings before the first snowfalls in October–November and the spring trappings after the snow melt in May.
Statistical methods
To evaluate the indictor of vole abundance, we fitted the data to linear mixed-effects models (Pinheiro and Bates 2000), using the statistical package “nlme” in R (version 2.14.1; R Development Core Team 2011; Venables et al. 2014). When examining the general suitability of our data for the present purpose, the response variable was the mean clutch size (reflecting productivity) of the tawny owl. The explanatory variables included the vole abundance index of the preceding autumn (fixed effect) and year (random effect). When evaluating the predictor of vole abundance, the response variable was the spring vole abundance index. The explanatory variables included the mean clutch size of the tawny owl (fixed effect) and year (random effect). The fixed effects were centered to zero mean, describing average conditions.
Results
The relationship between the preceding autumn vole abundance and the mean clutch size of the tawny owl was significantly positive in our western study area and nearly significantly positive in the eastern one (Table 1). Particularly, the effect of the field vole abundance seemed to be considerable.
The mean clutch size of the tawny owl reflected significantly positively the late spring abundance of voles in the western study area (Table 2, Fig. 1a) where the trapping effort was 384 trap nights/0.1 km2. In the eastern study area, where the effort was only 90 trap nights/0.1 km2, the respective relationship was not significant (Table 2, Fig. 1b). According to a simple linear model of the most significant relationship studied (Fig. 1), the mean clutch sizes of three, four, and five eggs approximately corresponded spring vole abundance indices 0.5, 2.0, and 3.5, respectively.
An approximate cost-benefit analysis showed that the basic costs of vole monitoring by the mean clutch size of tawny owls may be about twofold compared to those of snap trapping (Table 3). However, their annual costs in h/km2 might be orders of magnitude less than in snap trapping.
Discussion
Evaluation of the vole indicator
On the basis of the present study, there was a considerable agreement between the abundance fluctuations of small voles in autumn and variations in the mean clutch size of neighboring tawny owls in the next spring. The positive relationship between the autumn vole abundance and mean clutch size of tawny owls was observable over the spatial range examined. This suggests a close association between the vole and owl populations studied, confirming the general suitability of our data for demonstrating a vole abundance indicator. The mean clutch size of the tawny owl in turn indicated well the relative abundance of voles later in spring. The mean clutch size of the tawny owl reflected spring vole abundance over the spatial range examined, suggesting its suitability for general forecasting purposes. Only the most local and minor catches of voles were not significantly predictable on the basis of clutch size of owls.
Owls probably scan voles quite evenly throughout their hunting range, in any case much more evenly and wider ranging than is possible in conventional trapping projects (e.g., Balčiauskienė and Naruševičius 2006). Our results and the cost-benefit analysis conducted (Table 3) suggest that effective monitoring of local vole abundance by snap trapping required an annual effort of more than 100 h/km2. The respective value for the owl clutch size method was approximately only a percentage of this. Even the clutch size of one pair of owls may give a rough estimate of the local vole abundance. A larger number of clutches was, of course, preferable. The number of clutches available depends both on the densities of tawny owls in the district and on the (minimum) size of the area to be monitored. General densities of tawny owls in southern Finland are about 5–10 pairs/100 km2 while local densities may be as high as 40–50 pairs/100 km2 (Solonen 1993). Depending on breeding densities, the area effectively monitored by owls may be more or less discontinuous. However, it gives a representative general picture of the relative abundance of voles in the surroundings.
Spatial and temporal heterogeneity of the environmental conditions no doubt affects the efficiency of the mean clutch size of the tawny owl to indicate vole abundance. When voles are scanty, only low numbers of owls breed and only the best territories, providing enough alternative prey, are occupied by breeding birds (e.g., Solonen and Karhunen 2002). When there are plenty of voles, the conditions allow more pairs to breed and the distribution of occupied territories covers the potential nesting territories—even the less preferred ones—more evenly. The inclusion of the clutch sizes of the less preferred, poor nesting territories somewhat decreases the mean clutch size. The higher clutch sizes of the preferred territories in good vole years as compared to those of poor vole years, however, keep the annual difference between the mean clutch sizes still considerable. Thus, the wide range of variation and largely parallel fluctuations in the mean clutch size of owls and in the spring abundance index of voles suggest that the mean clutch size works properly as an indicator of relative abundance of voles.
The importance of small voles in the diet of tawny owls vary temporally and spatially (e.g., Goszczyński et al. 1993; Balčiauskienė and Naruševičius 2006; Romanowski and Zmihorski 2009), affecting the relationship between the mean clutch size of owls and the abundance of voles. In our eastern study area, 26.7 % of the prey items of breeding tawny owls were small voles (Solonen and Karhunen 2002). Variation between nests was, however, considerable (range 2.7–67.7 %, mean 22.5 ± 14.3 SD, n = 51). Before breeding, when there are less alternative prey available than later in the season, the proportion of small voles in prey is probably higher. Therefore, it can be expected that the clutch size of tawny owls largely indicated the abundance of small voles.
Among the vole-eating birds of prey owls, in general, begin breeding early in spring. Therefore, their clutch size provides forecasts on vole abundance earlier than that of many other vole-eaters. In a large part of Europe and in some neighboring areas as well, the tawny owl fulfills also various other criteria of a good indicator species (e.g., Ellenberg 1981; Weiss 1981; Solonen and Lodenius 1990; Chausson et al. 2014). Among others, tawny owl populations can be effectively manipulated and controlled by providing nest boxes which enables an easy sampling of various kinds of data (such as breeding parameters, individual characteristics, diet). Another advantage of the increased population of owls is the enhancing of biological control of pests (cf., e.g., Paz et al. 2013). In any case, owls eat considerable numbers of voles (e.g., Linkola and Myllymäki 1969; Solonen and Karhunen 2002; Balčiauskienė and Naruševičius 2006).
Outside the breeding range of the tawny owl, some ecologically similar species may serve as a corresponding indicator species. The species monitored should be the key prey species of the indicator species in the district considered. So, in practice, the monitoring system outlined here could and should be modified according to the local or regional conditions.
For an earlier forecasting of spring vole abundances, the numbers of over-wintering vole-eaters (such as buzzards, kestrels, and owls) provide a promising alternative indicator (e.g., Cheveau et al. 2004). The number of occupied territories or nests of tawny owls could also provide an earlier indicator of spring vole abundance than the mean clutch size. The censusing of hooting owls over a wide range is, however, a relatively hard task compared to the checking of nests in a nest box population. Therefore, it was considered to be out of the scope of the present study focused to find a simple, easy, and cost-effective indicator of vole abundance.
Recommendations
The present study exemplifies the potential of breeding parameters of vole-eating birds of prey as indicators of general abundance of small voles in spring. It calls for collaboration between land owners (agriculturalists, silviculturalists) and (professional and amateur) ornithologists who are interested in birds of prey, for instance, in ringing them. An additional merit of the present method is that it may increase numbers of vole-eaters that act as biological control of vole populations.
Based on our results, a simple but comprehensive vole monitoring system, using the mean clutch size of the tawny owl as an indicator of vole abundance, could be introduced as follows. (1) Install a number of suitable nest boxes for tawny owls throughout the monitoring area (as described in the present “Material and methods” section). In many areas, the proper density of nest boxes might be about 1/km2, and the suitable number of boxes at least 20–30. Possible earlier installed nest boxes in the surroundings are worth to take into consideration. (2) Check the nest boxes and count the number of eggs (at least) in two evenings (to lessen the risk of disturbance) separated by about 1–3 weeks during the early phases of (local and annual) breeding season of owls so that at least one visit occurs during the incubation period. (3) After the fledging of young, it is possible to analyze the composition of owls’ diet, including the occurrence of voles, from the prey remains in the nest bottom litter. This provides a usable additional indicator of local vole abundance if more detailed information is needed.
References
Balčiauskienė, L. (2005). Analysis of Tawny Owl (Strix aluco) food remains as a tool for long-term monitoring of small mammals. Acta Zoologica Lituanica, 15(2), 85–89.
Balčiauskienė, L., & Naruševičius, V. (2006). Coincidence of small mammal trapping data with their share in the Tawny Owl diet. Acta Zoologica Lituanica, 16(2), 93–101.
Baxter, R., & Hansson, L. (2001). Bark consumption by small rodents in the northern and southern hemispheres. Mammal Review, 31, 47–59.
Brown, P. R., Huth, N. I., Banks, P. B., & Singleton, G. R. (2007). Relationship between abundance of rodents and damage to agricultural crops. Agriculture, Ecosystems & Environment, 120(2–4), 405–415.
Chausson, A., Henry, I., Ducret, B., Almasi, B., & Roulin, A. (2014). Tawny Owl Strix aluco as an indicator of Barn Owl Tyto alba breeding biology and the effect of winter severity on Barn Owl reproduction. Ibis, 156, 433–441.
Cheveau, M., Drapeau, P., Imbeau, L., & Bergeron, Y. (2004). Owl winter irruptions as an indicator of small mammal population cycles in the boreal forest of eastern North America. Oikos, 107(1), 190–198.
Christensen, P., & Hörnfeldt, B. (2003). Long-term decline of vole populations in northern Sweden: a test of the destructive sampling hypothesis. Journal of Mammalogy, 84(4), 1292–1299.
Cramp, S. (Ed.) (1985). The birds of the Western Palearctic, Vol. 4. Oxford University Press: Oxford.
Ellenberg, H. (1981). Was ist ein Bioindikator? Sind Greifvögel Bioindikatoren? Ökologie der Vögel, 3 (Sonderheft), 83–99.
Flowerdew, J. R., Shore, R. F., Poulton, S. M. C., & Sparks, T. H. (2003). Live trapping to monitor mammals in Britain. Mammal Review, 34(1–2), 31–50.
Gervais, J. A. (2010). Testing sign indices to monitor voles in grasslands and agriculture. Northwest Science, 84, 281–288.
Gill, R. M. A. (1992). A review of damage by mammals in north temperate forests. 2. Small mammals. Forestry, 65, 281–308.
Glue, D. E. (1971). Avian predator pellet analysis and the mammalogist. Mammal Review, 1, 53–62.
Goszczyński, J., Jabloński, P., Lesiński, G., & Romanowski, J. (1993). Variation in diet of tawny owl Strix aluco L. along an urbanization gradient. Acta Ornithologica, 27, 113–123.
Hansson, L. (1979). Field signs as indicators of vole abundance. Journal of Applied Ecology, 16, 339–347.
Hansson, L. (1986). Vole snow trails and tunnels as density indicators. Acta Theriologica, 31(29), 401–408.
Hansson, L. (2002). Cycles and traveling waves in rodent dynamics: a comparison. Acta Theriologica, 47, 9–22.
Hansson, L., & Hoffmeyer, I. (1973). Snap and live trap efficiency for small mammals. Oikos, 24, 477–478.
Huitu, O., Kiljunen, N., Korpimäki, E., Koskela, E., Mappes, T., Pietiäinen, H., Pöysä, H., & Henttonen, H. (2009). Density-dependent vole damage in silviculture and associated economic losses at a nationwide scale. Forest Ecology and Management, 258, 1219–1224.
Huitu, O., Norrdahl, K., & Korpimäki, E. (2004). Competition, predation and interspecific synchrony in cyclic small mammal communities. Ecography, 27, 197–206.
Hörnfeldt, B., Hipkiss, T., & Eklund, U. (2005). Fading out of vole and predator cycles? Proceedings of the Royal Society London, B, 271, 2045–2049.
Jacob, J., Manson, P., Barfkneft, R., & Fredricks, T. (2014). Common vole (Microtus arvalis) ecology and management: implications for risk assessment of plant protection products. Pest Management Science, 70(6), 869–878.
Jacob, J. & Tkadlec, E. (2010). Rodent outbreaks in Europe: dynamics and damage. In G. Singleton, S. Belmain, P. R. Brown & B. Hardy (Eds.), Rodent outbreaks: Ecology and impacts (pp. 207–224. IRRI (International Rice Research Institute): Los Banos (Philippines).
Jareno, D., Vinuela, J., Luque-Larena, J. J., Arroyo, L., Arroyo, B., & Mougeot, F. (2014). A comparison of methods for estimating common vole (Microtus arvalis) abundance in agricultural habitats. Ecological Indicators, 36, 111–119.
Karell, P., Ahola, K., Karstinen, T., Zolei, A., & Brommer, J. E. (2009). Population dynamics in a cyclic environment: consequences of cyclic food abundance on tawny owl reproduction and survival. Journal of Animal Ecology, 78(5), 1050–1062.
Korpela, K., Delgado, M., Henttonen, H., Korpimäki, E., Koskela, E., Ovaskainen, O., Pietiäinen, H., Sundell, J., Yoggoz, N., & Huitu, O. (2013). Nonlinear effects of climate on boreal rodent dynamics: mild winters do not negate high-amplitude cycles. Global Change Biology, 19, 697–710.
Korpimäki, E. (1984). Population dynamics of birds of prey in relation to fluctuations in small mammal populations in western Finland. Annales Zoologici Fennici, 21, 287–293.
Korpimäki, E. (1992). Population dynamics of Fennoscandian owls in relation to wintering conditions and between-year fluctuations of food. In C. A. Galbraith, I. R. Taylor & S. Percival (Eds.), The ecology and conservation of European owls. UK Nature Conservation No. 5: 1–10. Joint Nature Conservation Committee: Peterborough.
Korpimäki, E., & Hakkarainen, H. (1991). Fluctuating food supply affects the clutch size of Tengmalm’s owl independent of laying date. Oecologia, 85, 543–552.
Korpimäki, E. & Hakkarainen, H. (2012). The Boreal Owl. Ecology, behaviour and conservation of a forest-dwelling predator. Cambridge University Press: Cambridge.
Kovács, A., Mammen, U. C. C., & Wernham, C. V. (2008). European monitoring for raptors and owls: state of the art and future needs. Ambio, 37(6), 408–412.
Krebs, C. J., Bilodeau, F., Reid, D., Gauthier, G., Kenney, A. J., Gilbert, S., Duchesne, D., & Wilson, D. J. (2012). Are lemming winter nest counts a good index of population density? Journal of Mammalogy, 93(1), 87–92.
Lambin, X., Petty, S. J., & MacKinnon, J. L. (2000). Cyclic dynamics in field vole populations and generalist predation. Journal of Animal Ecology, 69, 106–118.
Linkola, P., & Myllymäki, A. (1969). Der Einfluss der Kleinsäugerfluktuationen auf das Brüten einiger kleinsäugerfressender Vögel im südlichen Häme, Mittelfinnland 1952–1966. Ornis Fennica, 46, 45–78.
Lisická, L., Losík, J., Zejda, J., Heroldová, M., Nesvadbová, J., & Tkadlec, E. (2007). Measurement error in a burrow index to monitor relative population size in the common vole. Folia Zoologica, 56, 169–176.
Lõhmus, A. (1999). Vole-induced regular fluctuations in the Estonian owl populations. Annales Zoologici Fennici, 36, 167–178.
Mikkola, H. (1983). Owls of Europe. Poyser: Calton.
Myllymäki, A. (1979). Importance of small mammals as pests in agriculture and stored products. In D. M. Stoddart (Ed.), Ecology of small mammals (pp. 239–279). Chapman & Hall: London.
Paz, A., Jareño, D., Arroyo, L., Viñuela, J., Arroyo, B. E., Mougeot, F., Luque-Larena, J. J., & Fargallo, J. A. (2013). Avian predators as a biological control system of common vole (Microtus arvalis) populations in NW Spain: experimental set-up and preliminary results. Pest Management Science, 69(3), 444–450.
Petty, S. J. & Fawkes, B. L. (1997). Clutch size variation in Tawny Owls (Strix aluco) from adjacent valley systems: can this be used as a surrogate to investigate temporal and spatial variations in vole density? In J. R. Duncan, D. H. Johnson, T. H. Nicholls (Eds.), Biology and conservation of owls of the Northern Hemisphere: 2nd international symposium. Gen. Tech. Rep. NC-190: 315–324. St. Paul, MN, U.S. Dept. of Agriculture, Forest Service, North Central Forest Experiment Station.
Pinheiro, J. C., & Bates, D. M. (2000). Mixed-effects models in S and S-Plus. Springer: New York.
R Development Core Team (2011). R: a language and environment for statistical computing. Vienna, Austria, R Foundation for Statistical Computing. ISBN 3–900051–07–0. Available at: http://www.R-project.org
Redpath, C. J., Thirgood, S. J., & Redpath, S. M. (1995). Evaluation of methods to estimate field vole abundance in the uplands. Journal of Zoology, 237, 49–57.
Romanowski, J., & Zmihorski, M. (2009). Seasonal and habitat variation in the diet of the tawny owl (Strix aluco) in Central Poland during unusually warm years. Biologia, 64, 365–369.
Saurola, P. (2008). Monitoring birds of prey in Finland: a summary of methods, trends, and statistical power. Ambio, 37(6), 413–419.
Shenbrot, G. I. & Krasnov, B. R. (2005). An atlas of the geographic distribution of the arvicoline rodents of the world (Rodentia, Muridae: Arvicolinae). Pensoft: Sofia.
Sibbald, S., Carter, P. & Poulton, S. (2006). Proposal for a national monitoring scheme for small mammals in the United Kingdom and the Republic of Eire. The Mammal Society Research report No. 6. ISBN: 0 906282 60 8.
Solonen, T. (1993). Spacing of birds of prey in southern Finland. Ornis Fennica, 70, 129–143.
Solonen, T. (2004). Are vole-eating owls affected by mild winters in southern Finland? Ornis Fennica, 81, 65–74.
Solonen, T. (2005). Breeding of Finnish birds of prey in relation to variable winter food and weather conditions. Memoranda Societatis pro Fauna et Flora Fennica, 81, 19–31.
Solonen, T. (2010). Reflections of winter season large-scale climatic phenomena and local weather conditions in abundance and breeding frequency of vole-eating birds of prey. In P. K. Ulrich, & J. H. Willett (Eds.), Trends in ornithology research (pp. 95–119). Nova Science Publishers: New York.
Solonen, T., & Ahola, P. (2010). Intrinsic and extrinsic factors in the dynamics of local small-mammal populations. Canadian Journal of Zoology, 88, 178–185.
Solonen, T., & Karhunen, J. (2002). Effects of variable feeding conditions on the Tawny Owl Strix aluco near the northern limit of its range. Ornis Fennica, 79, 121–131.
Solonen, T., & Lodenius, M. (1990). Feathers of birds of prey as indicators of mercury contamination in southern Finland. Holarctic Ecology, 13, 229–237.
Stenseth, N. C., Leirs, H., Skonhoft, A., Davis, S. A., Pech, R. P., Andreassen, H. P., Singleton, G. R., Lima, M., Machang’u, R. S., Makundi, R. H., Zhang, Z., Brown, P. R., Shi, D., & Wan, X. (2003). Mice, rats, and people: the bio-economics of agricultural rodent pests. Frontiers in Ecology and the Environment, 1, 367–375.
Sundell, J., Huitu, O., Henttonen, H., Kaikusalo, A., Korpimäki, E., Pietiäinen, H., Saurola, P., & Hanski, I. (2004). Large-scale spatial dynamics of vole populations in Finland revealed by the breeding success of vole-eating avian predators. Journal of Animal Ecology, 73, 167–178.
Venables, W. N., Smith, D. M. & the R Development Core Team (2014). An introduction to R. Version 3.1.1. The R Project for Statistical Computing. Available at: http://www.r-project.org
Weiss, J. (1981). Die Eignung des Waldkauzes (Strix aluco L.) als mögliger Umweltgűtezeiger. Ökologie der Vögel, 3(Sonderheft), 101–110.
Witmer, G., Snow, N., Humberg, L., & Salmon, T. (2009). Vole problems, management options, and research needs in the United States. In J. R. Boulanger (Ed.), Proceedings of the 13th wildlife damage management conference (pp. 235–249). NY: Available at: http://digitalcommons.unl.edu/icwdm_wdmconfproc/140Saratoga.
Acknowledgments
Various people participated in the fieldwork with the authors, including Juhani Ahola, Pentti Ahola, Arto Laesvuori, Jari Pynnönen, Kimmo af Ursin, and Martti Virolainen. Lisa Solonen checked the English.
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Solonen, T., Ahola, K. & Karstinen, T. Clutch size of a vole-eating bird of prey as an indicator of vole abundance. Environ Monit Assess 187, 588 (2015). https://doi.org/10.1007/s10661-015-4783-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-015-4783-0