Abstract
Different studies have shown that the effect of land use conversion on soil nutrients and soil organic carbon (SOC) is variable, which indicates that more investigations that focus on different specific geographical locations and land use types are required. The objectives of this study were (1) to evaluate the effect of grazing land (GL) conversion into Grevillea robusta plantation and exclosure (EX) on soil nutrients and soil organic carbon (SOC) and (2) to examine the impact of soil organic matter (SOM) on soil nutrients. To achieve these objectives, soil samples were taken from a soil depth of 20 cm (n = 4) in each of the studied land areas. Each soil sample was analysed in a soil laboratory following a standard procedure. Analysis of variance (ANOVA) and Pearson’s correlation coefficient were used for the data analysis. The result indicated that conversion of GL into EX improved the soil electrical conductivity (EC), exchangeable K, cation exchange capacity (CEC), total N and available P (p < 0.05), while the exchangeable Mg, SOC, available K and SOM were decreased (p < 0.05). Conversion of GL into G. robusta improved the soil EC, exchangeable (K, Ca, Mg), CEC, SOC, total N, available K and SOM (p < 0.05). There was a significant relationship between SOM and available P, total N, SOC and EC. There were no significant relationships between SOM and pH, available K and CEC. Finally, the results indicate that both land uses, established in acidic Nitosols, have variable impacts on soil chemical properties and that G. robusta plantation improved most of the soil nutrients and SOC much better than the EX land use.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Land use is characterised by the arrangements, activities and inputs people undertake in a certain land cover type to produce, change or maintain it (FAO 1997a; FAO 1997b). Land use change is a general term for the human modification of the land (Ellis and Pontius 2010). Current rates, extents and intensities of land use/land cover change are far greater than ever before in history and are driving unprecedented changes in ecosystems and environmental processes at local, regional and global scales (Ellis and Pontius 2010). Land use changes, such as forest clearing, cultivation and pasture introduction, are known to result in changes in the soil’s chemical, physical and biological properties (Houghton et al. 1999); the sign and magnitude of these changes vary with land cover and land management (Celik 2005).
Different studies that have focused on the effect of land use change, from natural forest to plantation forest, on soil carbon and soil chemical properties have shown variable results (Ashagrie et al. 2005; Girmay et al. 2008; Liao et al. 2010; Getachew et al. 2012). Some of the findings have argued against using the replacement of natural forests by plantations as a measure of climate change mitigation and soil management (Girmay et al. 2008; Liao et al. 2010), while the results of other studies disagree with this notion (Ashagrie et al. 2005; Getachew et al. 2012). Similarly, the results of studies focusing on the impact of grazing land conversion to plantation forest on soil carbon and soil nutrients are inconsistent. For example, Ussiri et al. (2006) indicated that converting pasture land to Casuarina species and Robinia pseudoacacia forest decreased soil pH and electrical conductivity (EC) after 10 years, while Casuarina species and R. pseudoacacia increased the soil organic carbon by 11 and 42 %, respectively. The soil nitrogen pool was not affected by the land use conversion from pasture to Casuarina species, while it increased by 10 % in the R. pseudoacacia forest over a 10-year time period. Hoogmoed et al. (2012) indicated that there is no evidence for substantial changes in soil carbon, nitrogen or the carbon to nitrogen ratio across three decades of afforestation of pasture lands in the Mediterranean climate. Scott et al. (1999) found a 15 % reduction in the average soil carbon when pasture is converted to pine forest plantations dominated by Pinus radiata. Similarly, Chirino et al. (2010) indicated a decrease in total C, N, P, S and exchangeable Ca and Mg after 5 years of afforestation of grasslands with P. radiata, Eucalyptus nitens and Cupressus macrocarpa. Different studies on the impact of grazing land conversion into exclosure have also shown variable results (Mekuria et al. 2007; Mamo 2008; Abinet 2011).
All of these variable results indicate that more research on the effect of emerging land use changes on soil nutrients and soil organic carbon (SOC) is required because the impact might be related to land use type, history, climate, soil type etc. This research is important for policy recommendations and for making wise decisions regarding the sustainable management of soil resources. Overall, the objectives of this study were (1) to explore the effects of exclosure (EX) and Grevillea robusta plantation that are established in a communal grazing land (GL) on soil nutrients and SOC and (2) to examine the effect of soil organic matter (SOM) on soil chemical properties.
Materials and methods
Site description
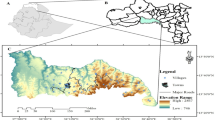
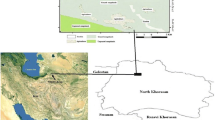
The study was conducted in the Kumbabe locality (specifically in Chora area), found in the Illubabor Zone, southwestern Ethiopia. The site is located at 8° 21′ 16 ″ to 8° 21′ 48″ N latitude and 36° 10′ 47″ to 36° 11′ 22″ E longitude. It has an altitude of 1,800 m above sea level (Fig. 1).
Fifteen years (1997–2011) of climatic data shows that the area has received a unimodal type of rainfall pattern with the highest rainfall occurring between May and September (Fig. 2). The mean annual rainfall was 197.4 ± 13.2 cm. The mean annual temperature was 19 °C, with a mean minimum of 12.7 °C and a mean maximum of 25.6 °C (Fig. 2). Geologically, the area is dominated by the Precambrian basement complex (EMA 1987). The soil mainly consists of Nitosols on the basalt and Lixisols on the basement complex and has a clay loamy texture (Afework et al. 2000; FAO 2006). The pH tends to be slightly acidic to more acidic; it ranges from 4.3 to 6.7 (Kefeni 1991).
The study area was used as a communal grazing land before July 1994 for an unknown period of time. Since August 1994, approximately 13 ha of it has been planted and covered with a G. robusta plantation, while approximately 5 ha of it was exclosed, and grazing was avoided. During the time of our study in the EX area, different woody plants regenerated, including Acacia tortilis (5,625 stems/ha), Grewia ferruginea (625 stems/ha), Maesa lanceolata (1,125 stems/ha), Vernonia thomsoniana (625 stems/ha), Vernonia amygdalina (37 stems/ha), Bersama abyssinica (500 stems/ha), Brucea antidysenterica (225 stems/ha), Clausena anisata (125 stems/ha) and Albizia species (100 stems/ha).
Sampling and analysis
A nested quadrant plot design with an area of 20 m by 20 m was used for soil sample collection. The soil samples were collected at the four corner points and at the centre of the quadrant plot within the top 20 cm of soil depth. This design was used to encompass the small-scale spatial variability of the soils in the study area. The soil samples were collected to a depth of 20 cm because of evidence that showed a greater proportion of SOC, and total nitrogen in the soil of Ethiopia is concentrated between 0 and 20 cm (Singh et al. 2010). Within each land use type (G. robusta, GL and EX), four sampling points were used, with a distance of 100 m between each point and were laid out in a gradient (upslope–downslope). The number of soil samples was restricted to four points (four replications) due to the small size of the EX land use area (5 ha) and to ensure that the number of replications would be equal to those for other land uses for statistical analysis purposes.
The collected soil samples from each plot (at the four corners and the centre of the plot) were thoroughly mixed to form a composite sample. Then, the samples were air-dried in an ambient air-circulating cabinet and sieved through a 2-mm mesh to remove coarse living roots. Soil pH and EC were measured with pH and EC metres, respectively. Exchangeable Ca and Mg were determined by the ethylenediaminetetraacetic acid (EDTA) titration method. Flame photometry was used to determine the exchangeable Na and K content of the soil (Black et al. 1965). The ammonium acetate method was used to determine available K and the cation exchange capacity (CEC) of the soil samples. This method was selected for CEC determination because of its suitability for slightly acidic to neutral soils. The SOC and total N were analysed following Walkley and Black’s titration method and the Kjeldahl method, respectively (Schnitzer 1982). Available P was extracted with 0.1 M sulphuric acid and measured colometrically by the ascorbic acid blue method (Olsen et al. 1954).
Data analysis
The normality of the distribution of the data was evaluated before analysis, and the means were compared for the soil variables that fulfilled the kurtosis. For the soil nutrients that showed significantly non-normal distributions, the Kruskal–Wallis test was used, and their medians were compared. The soil indexes for each plot in the different land use areas were used for comparison, with each plot considered a replication. One-way analysis of variance (ANOVA) was used for the analysis. For those soil variables where there was a significant difference, mean separation was performed using Fisher’s least significant difference (LSD) test with a significance level of p = 0.05. Pearson’s correlation coefficient was employed for the regression analysis, and SigmaPlot programme 10 was used.
Results
Soil nutrients in the different land uses
Statistically, there was a significant difference among the GL, G. robusta and EX soils in their different soil nutrients and SOC (p < 0.05, Fig. 3). The LSD test results indicated a significant difference of the available P (Fig. 3a), available K (Fig. 3b), exchangeable (exch.) Ca (Fig. 3c), exch. K (Fig. 3e), total N (Fig. 3g), SOM (Fig. 3h), CEC (Fig. 3i), EC (Fig. 3j), pH (Fig. 3k) and SOC (Fig. 3l) between the soil of the GL and EX land uses (p < 0.05). However, there were no significant differences in the exch. Mg (Fig. 3d) between the soils of the GL and EX land uses (p > 0.05). The exch. Mg, SOC, available K, OM and C/N in the GL soil were greater than those in the EX soil by 2.37, 3.29, 33.58, 5.69 and 145.44 units, respectively. This result indicates that GL conversion into EX land use did not improve these soil variables.
Mean soil nutrients (available P (a), available K (b), exch. Ca (c), exch. Mg (d), exch. K (E), exch. Na (f), N (g), soil organic matter (h), CEC (i), EC (j), pH (k) and soil organic carbon (l)) in the grazing land (GL), G. robusta plantation (GR) and exclosure area (AC), (p = 0.05). Exch. is exchangeable
There were also significant differences in available K (Fig. 3b), exch. Ca (Fig. 3c), exch. Mg (Fig. 3d), exch. K (Fig. 3e), total N (Fig. 3g), SOM (Fig. 3h), CEC (Fig. 3i), EC (Fig. 3j), pH (Fig. 3k) and OC (Fig. 3l) between the GL and G. robusta land use soils (p < 0.05). However, there were no significant differences in the available P (Fig. 3a) and C/N between the GL and G. robusta land use soils (p > 0.05). Conversion of GL into G. robusta land use has no effect on the available P of the soil. Soil pH decreased from 4.95 to 4.68 after conversion from GL to G. robusta land use. The EC, exch. K, exch. Ca, exch. Mg, CEC, OC, total N, available K and OM in the GL use soil were less than those in the G. robusta plantation soil by 0.013, 0.18, 3.04, 4.18, 5.83, 3.04, 0.005, 33.55 and 5.24 units, respectively.
Finally, there was a significant difference in available P (Fig. 3a), exch. Ca (Fig. 3c), exch. Mg (Fig. 3d), total N (Fig. 3g), SOM (Fig. 3h), EC (Fig. 3j) and SOC (Fig. 3l) between the land use soils of EX and G. robusta. However, there were no significant differences in the available K (Fig. 3b), exch. K (Fig. 3e), CEC (Fig. 3i) and pH (Fig. 3k) between the land use soils of EX and G. robusta. There was no significant difference in soil Na between G. robusta, GL and EX (p > 0.05, Fig. 3f). This result indicated that land use conversion from GL into G. robusta or EX has no effect on soil Na content.
Effect of soil organic matter on different soil variables
The result on the relationship between SOM and different soil variables is presented in Fig. 4. There is a significant relationship between SOM and available P (p = 0.035, Fig. 4a), total N (p = 0.0001, Fig. 4b), SOC (p = 0.0001, Fig. 4c) and EC (p = 0.071, Fig. 4d). These results could indicate that the SOM has an effect on available P, total N, SOC and EC in the studied land uses. There was no significant relationship between SOM and pH (p = 0.701, Fig. 4e), available K (p = 0.558, Fig. 4f) and CEC (p = 0.781). These results could indicate that SOM has no effect on soil pH, available K and CEC.
Discussion
Soil nutrients and soil organic carbon in the different land uses
Soil organic carbon
After conversion of the GL into G. robusta plantation, the SOC increased by 54.8 % over 16 years. This result is consistent with other similar findings (Fisher 1995; Ussiri et al. 2006; Holcomb 2008). Ussiri et al. (2006) found a difference in SOC of 42 and 11 % after conversion of pasture land to R. pseudoacacia and Casuarina species, respectively. Fisher (1995) also found a difference in SOC after conversion of pasture land to Acacia mangium, Vochysia ferruginea and Vochysia guatemalensis. Holcomb (2008) found an increase in SOC upon converting pasture to pine plantation. Contrary to the present results, Powers (2004) found a decrease in SOC after conversion of pasture land to V. guatemalensis plantations. Chirino et al. (2010) also found a decrease in SOC after conversion of grasslands into P. radiata, E. nitens and C. macrocarpa plantations. These differences from the present results could be attributed to differences in plantation species, geographical location, soil type and microenvironment. The increase in SOC in the G. robusta plantation in the present result could also be due to the potential of the species to form a large fraction of soil aggregates and to protect most of the SOC in the soil (Rekha et al. 2010). Overall, the result indicated that conversion of GL into G. robusta plantation improved the SOC in acidic Nitosols.
As a result of the conversion of GL into EX over 16 years, the SOC decreased by 59.45 %. This result is not consistent with other similar study results (Mekuria et al. 2009; Wanga et al. 2011; Girmay and Singh 2012). Mekuria et al. (2009) found an increase in the SOC after conversion of grazing lands into exclosures that were 5, 10 and 15 years old. Wanga et al. (2011) indicated that exclosure of degraded grassland from grazing in China increased the SOC by 34 % over 30 years. Similarly, Girmay and Singh (2012) found that SOC in the exclosure area was higher than that in the grazing land and plantation forest soil. These differences with the present result could be attributed to differences in the age and location of the exclosure areas. Overall, the present result indicated that conversion of GL into EX will reduce SOC in acidic Nitosols.
Soil nitrogen
Holcomb (2008) found an increase in soil N after converting pasture to pine plantation, which is consistent with the present study result. However, the present result for soil N is not consistent with other similar studies (Ussiri et al. 2006; Chirino et al. 2010; Hoogmoed et al. 2012). Ussiri et al. (2006) found no effect on soil N as a result of conversion of pasture land to Casuarina species over 10 years. Hoogmoed et al. (2012) also found that afforestation of pastures in a Mediterranean climate did not induce substantial changes in soil N over three decades. Chirino et al. (2010) indicated that afforestation of grasslands with P. radiata, E. nitens and C. macrocarpa reduced total soil N over 5 years in New Zealand. These differences with the present result could be attributed to differences in the plantation species. Some studies indicated that the impact on soil N in plantations established in pasture land depends on the species (Ussiri et al. 2006). Overall, the present result indicates that G. robusta, a non-nitrogen-fixing species, improved the soil N when established in acidic Nitosols.
Unlike the G. robusta plantation, after conversion of the GL into EX land use, the total soil N decreased by 50 %. This result is not consistent with the findings of Mekuria and Aynekulu (2011). They found higher total soil N in 5-, 10-, 15- and 20-year-old exclosures, which were established in communal grazing lands in northern Ethiopia. Mekuria et al. (2007) also found an increase in soil total N after 10 years of exclosure establishment in a grazing land. Similarly, Abinet (2011) found higher total soil N in a 7-year-old exclosure area established in a free grazing land in southern Ethiopia. Likewise, Girmay and Singh (2012) found higher total soil N in an exclosure area established in a grazing land. The variation from the present study could be attributed to differences in the regenerated woody plants, types of grasses and geographical locations. It could also be a result of differences in urine and dung deposition in the grazing land. Studies indicate that livestock urine and faeces adds N to soils (Beschta et al. 2012).
Soil pH
Ussiri et al. (2006) indicated that converting pasture land to Casuarina species and R. pseudoacacia forest decreased the soil pH in the top 20 cm after 10 years. This result is in agreement with the present study result. However, Boley et al. (2009) indicated no significant differences in the pH of the soil after conversion of grazing land into Tectona grandis plantation, which is not consistent with the present study finding. This variability could be a result of differences in the types of plantation species. Different plantation species affect soil pH differently (Rosoman 1994; Rhoades and Binkley 1996). The lower pH of the soil after conversion of the GL into G. robusta plantation in the present study could also be a result of the acidic nature of the G. robusta leaf. Leaf extracts of G. robusta species were found to be quite acidic (Silas 2011). Conversion of the GL into EX also decreased the pH of the soil. Conversely, Abinet (2011) indicated no significant differences in soil pH after conversion of the grazing land into exclosure in southern Ethiopia over 7 years. The decrease in the soil pH in the EX land use after conversion of GL in the present study could be related to the degree of neutralisation of the soil exchange complex as different species have different effects on the soil pH (Rhoades and Binkley 1996).
EC and CEC
Ussiri et al. (2006) found a decrease in soil EC after converting pasture land to Casuarina species and R. pseudoacacia forest after 10 years. This result is not consistent with the present finding. The present finding on the soil EC (higher EC in the EX than in the GL) is not consistent with the result of Abinet (2011), who found no significant differences on soil EC as a result of the conversion of GL to EX over 7 years. This variability could be attributed to differences in geographical location, the age of the exclosure and/or differences in soil parent material. While the present investigation was conducted in southwestern Ethiopia, where there is higher rainfall, the other analysis was performed in southern Ethiopia, where precipitation is lower. The finding of Abinet (2011) for soil CEC is consistent with the present result.
Available P and K
Chirino et al. (2010) indicated that afforestation of grasslands in New Zealand with P. radiata, E. nitens and C. macrocarpa increased the available P levels under all species after 5 years. This result is not consistent with the present study findings, in which we found no significant differences in the soil’s available P after the conversion of GL into G. robusta plantation (p > 0.05). This difference could be attributed to the high leaf nutrient use efficiency and low nutrient litter production of P in the G. robusta species (Richards and Susanne 2010). In the present finding, conversion of the GL into EX improved the available P in soil by 235 %. This result is consistent with Mekuria and Aynekulu (2011), which showed higher available P in 5-, 10-, 15- and 20-year-old exclosures established in a communal grazing land. Similarly, Mamo (2008) indicated an increase in available P after 9 years of exclosure establishment on a grazing land. In contrast, Tsetargachew (2008) found higher available P in communal grazing land compared with the 20-year-old exclosure established on it. The reason for the difference could be related to differences in the animal manure (cow dung) left in the grazing land. Abinet (2011) reported no significant differences in soil available P after the conversion of GL to EX over 7 years. These differences could also be related to the differences in parent material as it has an effect on the soil P content at the surface as well as in deep horizons (Renneson et al. 2010). Other parameters, such as differences in soil pH or organic matter, which depend on land use, could also be the cause of the differences in the results since they do have an impact on P availability (Renneson et al. 2010). Fisher (1995) looked at the effect of 11 different plantation species on the soil available K, established on grazing land in Costa Rica over 4 years, and found a significant increase in the soil available K in four species (Pithecellobium macradenium, Stryphnodendron microstachyum, Virola koschnyi and V. guatemalensis). This result is consistent with the present finding that the soil available K increased by 195 % after conversion of the GL into G. robusta plantation. Similarly, the soil available K increased by 235 % after the conversion of the GL to EX over 16 years. This result shows that both land uses increased the soil available K.
Soil organic matter
The result of SOM in the present study is not in agreement with that of Dick et al. (2011). They found that soil under the pasture land had higher SOM compared to the Pinus plantation in Brazil. This variability from the present finding could be attributed to differences in the biomass input of the plantation species and the decomposition rate of the below ground-added residue. Contrary to the G. robusta plantation land use, the present finding indicates that conversion of the GL into EX decreased the SOM. This result is not consistent with Mekuria et al. (2007), Mamo (2008) and Abinet (2011) who found an increase in SOM after exclosure establishment in a grazing land. This difference in SOM with the present result could be attributed to differences in nutrient turnover by the regenerated plants and the uptake of soil nutrients. The regenerated woody plant species in the present study area are juveniles, and their leaf turnover is negligible for soil improvement; in fact, they uptake more nutrients for their growth. Berthrong et al. (2009) indicated that trees take up considerable amounts of nutrients from the soils at younger stages.
Effect of SOM on other soil nutrients
The present result indicates that SOM influenced SOC, total N, available P and EC. However, it did not have an impact on the pH, available K and CEC of soil (Fig. 4). The result of the impact of SOM on SOC is consistent with the findings of Singwane and Malinga (2012), who indicated that SOM content and SOC have a direct relationship under the soils of Pinus and Eucalyptus plantations in Swaziland. This result could indicate that in land use conversion, considering a type of land use that has a high SOM turnover in a short period of time could result in a large net gain of C in the soil. Such an activity could have a positive impact on climate change mitigation. Post and Kwon (2000) stated that SOM dynamics might result in enhanced soil C sequestration with changes in land use and soil management.
Du Preez et al. (2011) indicated that loss of SOM has resulted in lower soil N. Albrecht (2007) also indicated that in the later stages of decay of most types of organic matter, N is liberated as ammonia and is subsequently converted into the soluble or nitrate form. The present finding indicates that SOM has a direct, strong and positive relationship with total soil N. Deekor et al. (2012) indicated the content of total soil N was a function of the amount of organic matter. Our result is not in agreement with that of Singwane and Malinga (2012), who found no standard relationship between the proportion of organic matter content and that of soil N. This variability with the present finding could be the result of differences in the land use types and differences in plantation species. The present result indicates that SOM has a significant but inverse relationship with soil available phosphorous. This result is consistent with that of Du Preez et al. (2011), who indicated that SOM does not necessarily result in lower P reserves in the soil. Although Cooperband (2002) showed that SOM has the ability to moderate major changes in the soil pH, the present result indicates that soil pH was not affected by the SOM. Rather, the effect of the land uses on the soil pH might be influenced more by the acidic nature of the soil of the area (Kefeni 1991). The present result also indicates that the SOM has no effect on the available K in the soil.
Conclusion
Our findings, which are specific to land use conversion on acidic Nitosols of GL into EX and G. robusta plantations, showed variable results. The G. robusta plantation that was established in the GL improved the exch. Ca, exch. Mg, SOC, total N and SOM compared to those of the EX land use. In contrast, the EX land use improved the exch. K, CEC and available P of the soil compared to those in the G. robusta plantation. The conversion of the GL into EX decreased the exch. Mg, SOC, total N and SOM in the soil. Overall, the findings indicated that in acidic Nitosols, establishing G. robusta plantation species in a GL than changing it into EX land use could play a big role in enhancing soil carbon sequestration, that is important in climate change mitigation and also in improving SOM which is important in soil management. Finally, we recommend the reasons behind why the soil exch. Mg, SOC, total N and SOM decreased after the conversion of GL into EX has to be investigated.
References
Abinet, T.Y. (2011). The impact of area enclosure on soil quality and farmers perception: the case of Tachignaw Gimbichu enclosure in Shashogo woreda, Southern Ethiopia. M.Sc thesis, Addis Ababa University, Addis Ababa.
Albrecht, W. (2007). Loss of soil organic matter and its restoration. http://wwww.soilandhealth.org/olaglibrary/02120/albrech.usdayrbk/lisom.html. Accessed on 20 December 2012.
Afework, H., Alan, D., & Adrian, W. (2000). Nature, extent and trends in wetland drainage and use in Illubabor Zone, Southwest Ethiopia: a report. Addis Ababa: Ethiopian Wetlands Research.
Ashagrie, Y., Zech, W., & Guggenberger, G. (2005). The transformation of a Podocarpus falcatus dominated natural forest into a monoculture Eucalyptus globulus plantation at Munesa, Ethiopia: soil organic C, N and S dynamics in primary particle and aggregate-size fraction. Agriculture Ecosystem and Environment, 106, 89–98. doi:10.1016/j.agee.2004.07.015.
Berthrong, S. T., Jobbagy, E. G., & Jackson, R. B. (2009). A global meta-analysis of soil exchangeable cations, pH, carbon, and nitrogen with afforestation. Ecological Applications, 19(8), 2228–2241.
Beschta, R. L., Donahue, D. L., Dellasala, D. A., Rhodes, J. J., Karr, J. R., O’Brien, M. H., Fleischner, T. L., & Williams, C. D. (2012). Adapting to climate change on western public lands: addressing the ecological effects of domestic, wild, and feral ungulates. Environmental Management. doi:10.1007/s00267-012-9964-9.
Black, C.A., Evans, D.D., White, J.L., Ensminger, L.E., Cark, F.E. (1965). Methods of soil analysis: part I. Physical and mineralogical properties, including statistics of measurement and sampling. Madison, Wisconsin.
Boley, J. D., Drew, A. P., & Andrus, R. E. (2009). Effects of active pasture, teak (Tectona grandis) and mixed native plantations on soil chemistry in Costa Rica. Forest Ecology and Management, 257, 2254–2261. doi:10.1016/j.foreco.-2009.02.035.
Celik, I. (2005). Land use effects on organic matter and physical properties of soil in a southern Mediterranean highland of Turkey. Soil and Tillage Research, 83, 270–277. doi:10.1016/j.still.2004.08-001.
Chirino, I., Condron, L., McLenaghen, R., & Davis, M. (2010). Effects of plantation forest species on soil properties. In R. J. Gilkes & N. Prakongkep (Eds.), Soil solutions for a changing world (pp. 49–51). Brisbane: Proceedings of the World Congress of Soil Science.
Cooperband, L. (2002). Building soil organic matter with organic amendments: a resource for urban and rural gardeners, small farmers, turf grass managers and large-scale producers. Madison: Center for Integrated Agricultural Systems (CIAS), College of Agricultural and Life Sciences, University of Wisconsin.
Deekor, T. N., Iwara, A. I., Ogundele, F. O., Amiolemen, S. O., & Ita, A. E. (2012). Changes in soil properties under different land use covers in parts of Odukpani, Cross River State, Nigeria. Journal of Environment and Ecology, 3(1), 86–99. doi:10.5296/jee.v3i1.-2265.
Dick, D. P., Leite, S. B., Dalmolin, R. S. D., Almeida, H. C., & Knicker, H. (2011). Pinus afforestation in South Brazilian highlands: soil chemical attributes and organic matter composition. Science in Agriculture (Piracicaba, Braz.), 68(2), 175–181.
Du Preez, C. C., Van Huyssteen, C. W., & Mnkeni, P. N. S. (2011). Land use and soil organic matter in South Africa 2: a review on the influence of arable crop production. South African Journal of Science, 107, 1–8. doi:10.4102/sajs. v107i5/6.358.
Ellis, E., & Pontius, R. (2010). Land use and land cover change, in, Cleveland, C.J. (Eds.),: Encyclopedia of Earth. <http://www.eoearth.org/article/Land-use and land cover change/. Accessed on 12 December 2012.
EMA. (1987). National atlas of Ethiopia. Addis Ababa, Ethiopia.
FAO. (2006). World reference base for soil resources: a framework for international classification, correlation and communication. Rome: FAO.
FAO. (1997a). Forest harvesting in natural forests of the Republic of the Congo: forest harvesting case study no. 7. Rome: FAO.
FAO. (1997b). Environmentally sound forest harvesting: testing the applicability of the FAO model code in the Amazon in Brazil: forest harvesting case study no. 8. Rome: FAO.
Fisher, R. F. (1995). Amelioration of degraded rain forest soils by plantations of native trees. Soil Science Society of American Journal, 59(2), 544–549. doi:10.2136/sssaj1995.036-15995005900020039x.
Getachew, F., Abdulkadir, A., Lemenih, M., & Fetene, A. (2012). Effects of different land use on soil physical and chemical properties in Wondo Genet Area, Ethiopia. New York Science Journal, 5(11), 110–118.
Girmay, G., Singh, B. R., Mitiku, H., Borresen, T., & Lal, R. (2008). Carbon stocks in Ethiopian soils in relation to land use and soil management. Land Degradation and Development, 19, 351–367. doi:10.1002/ldr.844.
Girmay, G., & Singh, B. R. (2012). Changes in soil organic carbon stocks and soil quality: land-use system effects in northern Ethiopia. Acta Agricultura Scandinivica B—Soil and Plant science, 62(6), 519–530. doi:10.1080/09064710.2012.-663786.
Hoogmoed, M., Cunninghama, S. C., Thomson, J. R., Baker, P. J., Beringer, J., & Cavagnaro, T. R. (2012). Does afforestation of pastures increase sequestration of soil carbon in Mediterranean climates? Agriculture, Ecosystem and Environment, 159, 176–183. doi:10.1016/j.agee.2012.07.011.
Houghton, R. A., Hacker, J. L., & Lawrence, K. T. (1999). The U.S. carbon budget: contributions from land use change. Science, 285, 574–578. doi:10.1126/scien-ce.285.5427.574.
Holcomb, J. (2008). Effects of land management on carbon and nitrogen in soils from New Zealand: thesis. Oregon State University; Oregon.
Kefeni, K. (1991). The soils of Iri/Hurumu Area, Illubabor: their genesis, distribution. Addis Ababa: Classification and Agricultural Potential, SCRP.
Liao, C., Luo, Y., Fang, C., & Li, B. (2010). Ecosystem carbon stock influenced by plantation practice: implications for planting forests as a measure of climate change mitigation. PLoS ONE, 5(5), e10867. doi:10.1371/journal. pone. 0010867.
Renneson, M., Dufey, J., Bock, L., & Colinet, G. (2010). Effects of parent material and land use on soil phosphorus forms in Southern Belgium. In R. J. Gilkes & N. Prakongkep (Eds.), Soil solutions for a changing world (pp. 40–43). Brisbane: Proceedings of the World Congress of Soil Science.
Mamo, K. (2008). Enclosure as a viable option for rehabilitation of degraded lands and biodiversity conservation: the case of Kallu Woreda, Southern Wello. Msc thesis, Addis Ababa University, Ethiopia.
Mekuria, W., & Aynekulu, E. (2011). Exclosure land management for restoration of the soils in degraded communal grazing lands in northern Ethiopia. Land Degradation and Development. doi:10.1002/ldr.1146.
Mekuria, W., Veldkamp, E., Haile, M. (2009). Carbon stock changes with relation to land use conversion in the lowlands of Tigray, Ethiopia: paper presented at the Conference on International Research on Food Security, Natural Resource Management and Rural Development. University of Hamburg, Hamburg.
Mekuria, W., Veldkamp, E., Haile, M., Nyssem, J., Muys, B., & Kindeya, G. (2007). Effectiveness of exclosures to restore degraded soils as a result of overgrazing in Tigray, Ethiopia. Journal of Arid Environment, 69, 270–284. doi:10.1016/j.jari-denv.2006.10.009.
Olsen, S.R., Cole, C.V., Watanabe, S., Dean, L.A. (1954). Estimation of available phosphorus in soils by extraction with sodium bicarbonate. USDA circular no. 939, 1–19.
Post, W. M., & Kwon, K. C. (2000). Soil carbon sequestration and land-use change: processes and potential. Global Change Biology, 6, 317–328.
Powers, J. S. (2004). Changes in soil carbon and nitrogen after contrasting land-use transitions in northeastern Costa Rica. Ecosystems, 7, 134–146. doi:10.1007/s10021-003-0123.
Rekha, J., Gupta, S. R., Ravi, K., & Singh, G. (2010). Carbon sequestration in the Grevillea robusta plantation on a reclaimed sodic soil at Karnal in Northern India. Journal of Ecology and Environmental Science, 36, 75–86.
Rhoades, C., & Binkley, D. (1996). Factors influencing the decline in soil pH in Hawaiian Eucalyptus and Albizia plantations. Forest Ecology and Management, 80, 47–56. doi:10.1016/0378-1127(95)03646-6.
Richards, A. E., & Susanne, S. (2010). Complementary resource use by tree species in a rainforest tree plantation. Ecological Applications, 20, 1237–1254. doi:10.1890/0911-80.1.
Rosoman, G. (1994). The plantation effect, an ecoforestry review of the environmental effects of exotic monoculture tree plantations in Aotearoa/New Zealand. Auckland: Greenpeace NZ.
Schnitzer, M. (1982). Total carbon, organic matter. In A. L. Page, R. H. Miller, & D. R. Keeney (Eds.), Methods of soil analysis: part 2 agronomy monograph (2nd edition, Vol. 9, pp. 539–577). Madison: American Society of Agronomy.
Scott, N. A., Tate, K. R., Ford-Robertson, J., & Giltrap, D. J. (1999). Soil carbon storage in plantation forests and pastures: land-use change implications. Tellus, 51B, 326–335. doi:10.1034/j.1600-0889.1999.00015.x.
Silas, E.N. (2011). Buffering capacity and levels of macronutrients found in leaves of selected plants. http://ir-library.ku.ac.ke/ir/handle/123456789/1346. Accessed on 9 December, 2012.
Singh, B.R., Wele, A.D., Lal, R. (2010). Soil carbon sequestration under consequences of agroforestry and agricultural lands in Southern Ethiopia, in: Gilkes R. J., Prakongkep, N. (Eds.),Soil Solutions for a Changing World: Proceedings of the World Congress of Soil Science (pp. 64–66). Brisbane, Australia.
Singwane, S. S., & Malinga, P. (2012). Impact of pine and eucalyptus forest plantations on soil organic matter content in Swaziland: case of Shiselweni Forest. Journal of Sustainable Development in Africa, 14(1), 137–151.
Tsetargachew, A. (2008). Area closure as a strategy for land management: a case study at Kelala Dalacha enclosure in the Central Rift Valley of Ethiopia. Msc thesis, Addis Ababa University, Addis Ababa.
Ussiri, D. A. N., Lal, R., & Jacinthe, P. A. (2006). Soil properties and carbon sequestration of afforested pastures in reclaimed minesoils of Ohio. Soil Science Society of American Journal, 70, 1797–1806. doi:10.2136/sssaj2005.0352.
Wanga, S., Wilkes, A., Zhang, Z., Chang, X., Lang, R., Wange, Y., Niue, H. (2011). Management and land use change effects on soil carbon in northern China’s grasslands: a synthesis. Agriculture, Ecosystem and Environment, 142, 329–340. doi: org/-10.1016/j.agee.2011.06.002.
Acknowledgments
This research was supported by the Interni Grantova Agentura (IGA) of Mendel University in Brno, with a project code of 52/2010. The Bedele Soil Research Center Soil Lab., OARI, is thanked for the chemical analysis of the soil. The Bedele meteorological station is also acknowledged for providing us with the climate data. Thanks also to Mindaye Teshome, Nesiru Hussein, Fantaye Dinka, Simegn Muluneh, Bediru and Sultan (Chora site plantation guards) for all their support in soil sample collection and preparation. Finally, thanks to Tezera Chernet for developing the map of the study area.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alem, S., Pavlis, J. Conversion of grazing land into Grevillea robusta plantation and exclosure: impacts on soil nutrients and soil organic carbon. Environ Monit Assess 186, 4331–4341 (2014). https://doi.org/10.1007/s10661-014-3702-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10661-014-3702-0