Abstract
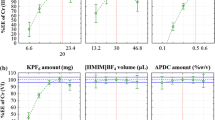
Dispersive liquid–liquid microextraction followed by inductively coupled plasma-optical emission spectrometry has been investigated for determination of Cd(II) ions in water samples. Ammonium pyrrolidine dithiocarbamate was used as chelating agent. Several factors influencing the microextraction efficiency of Cd (II) ions such as extracting and dispersing solvent type and their volumes, pH, sample volume, and salting effect were optimized. The optimization was performed both via one variable at a time, and central composite design methods and the optimum conditions were selected. Both optimization methods showed nearly the same results: sample size 5 mL; dispersive solvent ethanol; dispersive solvent volume 2 mL; extracting solvent chloroform; extracting solvent volume 200 \(\upmu \)L; pH and salt amount do not affect significantly the microextraction efficiency. The limits of detection and quantification were 0.8 and 2.5 ng L − 1, respectively. The relative standard deviation for five replicate measurements of 0.50 mg L − 1 of Cd (II) was 4.4%. The recoveries for the spiked real samples from tap, mineral, river, dam, and sea waters samples ranged from 92.2% to 104.5%.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Agency of Toxic Substances and Disease Registry (1999). Toxicological profile for cadmium. Available at http://www.atsdr.cdc.gov/toxprofiles/tp5.html. Accessed 1 May 2008.
Anthemidis, A. N., Zachariadis, G. A., & Stratis, J. A. (2003). Development of an on-line solvent extraction system for electrothermal atomic absorption spectrometry utilizing a new gravitational phase separator: Determination of cadmium in natural waters and urine samples. Journal of Analytical Atomic Spectrometry, 18, 1400–1403.
Araujo, P. W., & Brereton, R. G. (1996). Experimental design II, optimization. Trends in Analytical Chemistry, 15, 63–70.
Atsumi, K., Minami, T., & Uada, J. (2005). Determination of cadmium in spring water by graphite-furnace atomic absorption spectrometry after coprecipitation with ytterbium hydroxide. Analytical Science, 21, 647–650.
Batterham, G. J., Munksgaard, N. C., & Parry, D. L. (1997). Determination of trace metals in sea-water by inductively coupled plasma mass spectrometry after off-line dithiocarbamate solvent extraction. Journal of Analytical Atomic Spectrometry, 12, 1277–1280.
Berijani, S., Assadi, Y., Anbia, M., Milani Hosseini, M. R., & Aghaee, E. (2006). Dispersive liquid–liquid microextraction combined with gas chromatography-flame photometric detection: Very simple, rapid and sensitive method for the determination of organophosphorus pesticides in water. Journal of Chromatography A, 1123, 1–9.
Chen, J. R., Xiao, S. M., Wu, X. H., Fang, K. M., & Liu, W. H. (2005). Determination of lead in water samples by graphite furnace atomic absorption spectrometry after cloud point extraction. Talanta, 67, 992–996.
Fatahi, N., Assadi, Y., Milani Hosseini, M. R., & Zeini Jahromi, E. (2007). Determination of chlorophenols in water samples using simultaneous dispersive liquid–liquid microextraction and derivatization followed by gas chromatography-electron-capture detection. Journal of Chromatography A, 1157, 23–29.
Gemperline, P. (2006) Practical guide to chemometrics, 2nd ed. Boca Raton: CRC.
Hinkelmann, K., & Kempthorne, O. (2007). Design and analysis of experiments. Introduction to experimental design (2nd edn, Vol. 1). Hoboken: Wiley.
International Agency for Research on Cancer (IARC) (1997). Monographs on the evaluation of carcinogenic risks to humans. Beryllium, cadmium, mercury, and exposures in the glass manufacturing industry. Summary of data reported and evaluation (Vol. 58). Available at http://www.monographs.iarc.fr/ENG/Monographs/vol58/volume58.pdf. Accessed 1 May 2008.
Jarup, L. (2003). Hazards of heavy metal contamination. British Medical Bulletin, 68, 167–182.
Jarup, L., & Alfven, T. (2004). Low level cadmium exposure, renal and bone effects-the OSCAR study. Biometals, 17, 505–509.
Jarup, L., Berglund, M., Elinder, C. G., Nordberg, G., & Vahter, M. (1998). Health effects of cadmium exposure-a review of the literature and a risk estimate. Scandinavian Journal of Work, Environment & Health, 24, 1–51.
Jiang, H., Qin, Y., & Hu, B. (2008). Dispersive liquid phase microextraction (DLPME) combined with graphite furnace atomic absorption spectrometry (GFAAS) for determination of trace Co and Ni in environmental water and rice samples. Talanta, 74, 1160–1165.
Li, X., Schramel, P., Wang, Z., Grill, P., Yedile, A., & Kettrup, A. (1996). Determination of trace amounts of Sb, Pb, Tl in water samples by inductively coupled plasma atomic emission spectrometry after a diantipyrylmethane-iodide third phase preconcentration. Microchimica Acta, 124, 171–178.
Liang, P., & Sang, H. (2008). Determination of trace lead in biological and water samples with dispersive liquid–liquid microextraction preconcentration. Analytical Biochemistry, 380, 21–25.
Lundstedt, T., Seifert, E., Abramo, L., Thelin, B., Nystrom, A., Pettersen, J., et al. (1998). Experimental design and optimization. Chemometrics and Intelligent Laboratory Systems, 42, 3–40. doi:10.1016/S0169-7439(98)00065-3.
Melek, E., Tuzen, M., & Soylak, M. (2006). Flame atomic absorption spectrometric determination of cadmium (II) and lead (II) after their solid phase extraction as dibenzyldithiocarbamate chelates on Dowex Optipore V-493. Analytica Chimca Acta, 578, 213–219.
Myers, R. H., & Montgomery, D. C. (2002). Response surface methodology. New York: Wiley.
Ojeda, C. B., & Rojas, F. S. (2009). Separation and preconcentration by dispersive liquid–liquid microextraction procedure: A review. Chromatographia, 69, 11–12. doi:10.1365/s10337-009-1104-1.
Rezaee, M., Assadi, Y., & Hosseini, M. M. (2006). Determination of organic compounds in water using dispersive liquid–liquid microextraction. Journal of Chromatography A, 1116, 1–9.
Stephan, D. D., Werner, J., & Yeater, R. P. (1998) Essential regression and experimental design for chemists and engineers. MS Excel Add-in Software Package.
Waalkes, M. P. (2003). Cadmium carcinogenesis. Mutation Research, 10, 107–120.
Wuilloud, R. G., Martinis, E. M., Olsina, R. A., & Altamirano, J. C. (2008). Sensitive determination of cadmium in water samples by room temperature ionic liquid-based preconcentration and electrothermal atomic absorption spectrometry. Analytica Chimica Acta, 628, 41–48.
Yamini, Y., Hejazi, L., & Mohammadi, D. E. (2003). Column system using diaion HP-2MG for determination of some metal ions by flame atomic absorption spectrometry. Microchimica Acta, 142, 21–25.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Salahinejad, M., Aflaki, F. Optimization and determination of Cd (II) in different environmental water samples with dispersive liquid–liquid microextraction preconcentration combined with inductively coupled plasma optical emission spectrometry. Environ Monit Assess 177, 115–125 (2011). https://doi.org/10.1007/s10661-010-1622-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10661-010-1622-1