Abstract
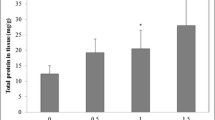
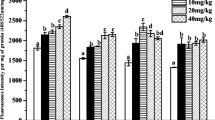
Contamination by 2,4,6-trinitrotoluene (TNT) is a global environmental problem at sites of former explosive production, handling, or storage, and could have deleterious consequences for human and ecological health. We investigated its sublethal effects to Eisenia fetida, using two nonspecific biomarkers. In coelomocytes of earthworms exposed 24, 48, or 72 h, we evaluated DNA damage (comet assay) and neutral red retention time (NRRT), using the filter paper contact test. Both percentage of damage (D%) and calculated damage index showed significant DNA damage at almost all concentrations, at all time points assayed. Along exposure time, two different patterns were observed. At the lower TNT concentrations (0.25–0.5 μg/cm2) an increased DNA migration at 48 h, with a decrease close to initial levels after 72 h exposure, was observed. This decrease could be attributed to activation of the DNA repair system. At higher concentrations (1.0–2.0 μg/cm2), the high DNA damage observed remained constant during the 72 h exposure, suggesting that the rate of DNA repair was not enough to compensate such damage. Analysis of NRRT results showed a significant interaction between time and treatment. After 48 h, a significant decrease was observed at 4.0 μg/cm2. After 72 h, NRRT presented a concentration-dependent decrease, significantly different with respect to control at 0.5, 1.0, 2.0, and 4.0 μg/cm2. The two assayed methods, performed on the same sample, showed clear responses to sublethal TNT exposure in E. fetida, providing sensitive unspecific biomarkers of cell injury and DNA damage.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Achtnich, C., & Lenke, H. (2001). Stability of immobilized 2,4,6-trinitrotoluene metabolites in soil under long-term leaching conditions. Environmental Toxicology and Chemistry, 20, 280–283.
Ateeq, B., Abul Farah, M., & Ahmad, W. (2005). Detection of DNA damage by alkaline single cell gel electrophoresis in 2,4-dichlorophenoxyacetic-acid- and butachlor-exposed erythrocytes of Clarias batrachus. Ecotoxicology and Environmental Safety, 62, 348–354.
Batzias, F., & Siontorou, C. (2006). A knowledge-based approach to environmental biomonitoring. Environmental Monitoring and Assessment, 123, 167–197.
Belden, J. B., Ownby, D. R., Lotufo, G. R., & Lydy, M. J. (2005). Accumulation of trinitrotoluene (TNT) in aquatic organisms: Part 2—Bioconcentration in aquatic invertebrates and potential for trophic transfer to channel catfish (Ictalurus punctatus). Chemosphere, 58, 1161–1168.
Bolt, H. M., Degen, G. H., Dorn, S. B., Plöttner, S., & Harth, V. (2006). Genotoxicity and potential carcinogenicity of 2,4,6-TNT trinitrotoluene: Structural and toxicological considerations. Reviews on Environmental Health, 21, 217–228.
Burch, S. W., Fitzpatrick, L. C., Goven, A. J., Venables, B. J., & Giggleman, M. A. (1999). In vitro earthworm Lumbricus terrestris coelomocyte assay for use in terrestrial toxicity identification evaluation. Bulletin of Environmental Contamination and Toxicology, 62, 547–554.
Burlinson, B., Tice, R. R., Speit, G., Agurell, E., Brendler-Schwaab, S. Y., Collins, A. R., et al. (2007). Fourth international workgroup on genotoxicity testing: Results of the in vivo Comet assay workgroup. Mutation Research, 627, 31–35.
Bursch, W. (2001). The autophagosomal - lysosomal compartment in programmed cell death. Cell Death and Differentiation, 8, 560–581.
Casabé, N. B., Oneto, M. L., Fuchs, J. S., Sánchez-Rivas, C., & Kesten, E. M. (2003). Nitrocompuestos aromáticos explosivos: Biotransformación, toxicidad y remediación. Acta Bioquímica Clínica Latinoamericana, 37, 47–58.
Casabé, N. B., Piola, L., Fuchs, J. S., Oneto, M. L., Pamparato, L., Basack, S., et al. (2007). Ecotoxicological assessment of the effects of glyphosate and chlorpyrifos in an Argentine soya field. Journal of Soils and Sediments, 7, 232–239.
Fuchs, J. S., Oneto, M. L., Casabé, N. B., Gómez Segura, O., Tarulla, R., Vaccarezza, M., et al. (2001). Ecotoxicological characterization of a disposal lagoon from a munition plant. Bulletin of Environmental Contamination and Toxicology, 67, 696–703.
Fugère, N., Brousseau, P., Krzysztof, K., Coderre, D., & Fournier, M. (1996). Heavy metal-specific inhibition of phagocytosis and different in vitro sensitivity of heterogeneous coelomocytes from Lumbricus terrestris. Toxicology, 109, 157–166.
Gastaldi, L., Ranzato, E., Caprì, F., Hankard, P., Pérès, G., Canesi, L., et al. (2007). Application of a biomarker battery for the evaluation of the sublethal effects of pollutants in the earthworm Eisenia andrei. Comparative Biochemistry and Physiology C-Pharmacology Toxicology & Endocrinology, 146, 398–405.
Gong, P., Guan, X., Inouye, L. S., Pirooznia, M., Indest, K. J., Athow, R. S., et al. (2007). Toxicogenomic analysis provides new insights into molecular mechanisms of the sublethal toxicity of 2,4,6-trinitrotoluene in Eisenia fetida. Environmental Science & Technology, 41, 8195–8202.
Guecheva, T., Henriques, J. A., & Erdtmann, B. (2001). Genotoxic effects of copper sulphate in freshwater planarian in vivo, studied with the single-cell gel test (comet assay). Mutation Research, 497, 19–27.
Hwang, H. M., Wade, T. L., & Sericano, J. L. (2004). Destabilized lysosomes and elimination of polycyclic aromatic hydrocarbons and polychlorinated biphenyls in eastern oysters (Crassostrea virginica). Environmental Toxicology and Chemistry, 23, 1991–1995.
ISO (2008). Soil quality—avoidance test for testing the quality of soils and effects of chemicals on behaviour—Part 1: Test with earthworms (Eisenia fetida and Eisenia andrei). ISO 17512–1. Geneva: International Organization for Standardization.
Kumagai, Y., Kikushima, M., Nakai, Y. Y., Shimojo, Z. M., & Kunimoto, M. (2004). Neuronal Nitric Oxide Synthase (NNOS) catalyzes one-electron reduction of 2,4,6-trinitrotoluene, resulting in decreased nitric oxide production and increased nNOS gene expression: Implication for oxidative stress. Free Radical Biology and Medicine, 37, 350–357.
Kumaravel, T., Vilhar, B., Faux, S., & Jha, A. (2009). Comet assay measurements: A perspective. Cell Biology and Toxicology, 25, 53–64.
Lachance, B., Renoux, A. Y., Sarrazin, M., Hawari, J., & Sunahara, G. I. (2004). Toxicity and bioaccumulation of reduced TNT metabolites in the earthworm Eisenia andrei exposed to amended forest soil. Chemosphere, 55, 1339–1348.
Lee, R. F., & Steinert, S. (2003). Use of the single cell gel electrophoresis/comet assay for detecting DNA damage in aquatic (marine and freshwater) animals. Mutation Research, 544, 43–64.
Lewis, T. A., Newcombe, D. A., & Crawford, R. L. (2004). Bioremediation of soils contaminated with explosives. Journal Environmental Management, 70, 291–307.
Maboeta, M. S., Reinecke, S. A., & Reinecke, A. J. (2003). Linking lysosomal biomarker and population responses in a field population of Aporrectodea caliginosa (Oligochaeta) exposed to the fungicide copper oxychloride. Ecotoxicology and Environmental Safety, 56, 411–418.
Maleri, R. A., Fourie, F., Reinecke, A. J., & Reinecke, S. A. (2008). Photometric application of the MTT- and NRR-assays as biomarkers for the evaluation of cytotoxicity ex vivo in Eisenia andrei. Soil Biology & Biochemistry, 40, 1040–1048.
Manerikar, R. S., Apte, A. A., & Ghole, V. S. (2008). In vitro and in vivo genotoxicity assessment of Cr(VI) using comet assay in earthworm coelomocytes. Environmental Toxicology and Pharmacology, 25, 63–68.
Martín-Díaz, M. L., Tuberty, S. R., McKenney, C. L., Blasco, J., Sarasquete, C., & Dellvalls, T. A. (2006). The use of bioaccumulation, biomarkers and histopathology diseases in Procambarus clarkii to establish bioavailability of Cd and Zn after a mining spill. Environmental Monitoring and Assessment, 116, 169–184.
Moore, M. N., Allen, J. I., & Mc Veigh, A. (2006). Environmental prognostics: An integrated model supporting lysosomal stress responses as predictive biomarkers of animal health status. Marine Environmental Research, 61, 278–304.
Neuwoehner, J., Schofer, A., Erlenkaemper, B., Steinbach, K., Hund-Rinke, K., & Eisentraeger, A. (2007). Toxicological characterization of 2,4,6-trinitrotoluene, its transformation products, and two nitramine explosives. Environmental Toxicology and Chemistry, 26, 1090–1099.
OECD (1984). Guideline for testing of chemicals N°. 207. Earthworm, acute toxicity tests. Paris: Organization for Economic Cooperation and Development.
Ownby, D., Belden, J., Lotufo, G., & Lydy, M. (2005). Accumulation of trinitrotoluene (TNT) in aquatic organisms: Part 1—Bioconcentration and distribution in channel catfish (Ictalurus punctatus). Chemosphere, 58, 1153–1159.
Qiao, M., Chen, Y., Wang, C. X., Wang, Z., & Zhu, Y. G. (2007). DNA damage and repair process in earthworm after in-vivo and in-vitro exposure to soils irrigated by wastewaters. Environmental Pollution, 148, 141–147.
Reinecke, S. A., & Reinecke, A. J. (2004). The comet assay as biomarker of heavy metal genotoxicity in earthworms. Archives of Environmental Contamination and Toxicology, 46, 208–215.
Renoux, A. Y., Sarrazin, M., Hawari, J., & Sunahara, G. I. (2000). Transformation of 2,4,6-trinitrotoluene in soil in the presence of the earthworm Eisenia Andrei. Environmental Toxicology and Chemistry, 19, 1473–1480.
Roberts, B. L., & Dorough, H. W. (1984). Relative toxicities of chemicals to the earthworm Eisenia foetida. Environmental Toxicology and Chemistry, 3, 67–78.
Robidoux, P. Y., Svendsen, C., Sarrazin, M., Hawari, J., Thiboutot, S., Ampleman, G., et al. (2002). Evaluation of tissue and cellular biomarkers to assess 2,4,6-trinitrotoluene (TNT) exposure in earthworms: Effects-based assessment in laboratory studies using Eisenia andrei. Biomarkers, 7, 306–321.
Scott-Fordsmand, J. J., & Weeks, J. M. (2000). Biomarkers in earthworms. Reviews of Environmental Contamination and Toxicology, 165, 117–159.
Sims, J., & Steevens, J. (2008). The role of metabolism in the toxicity of 2,4,6-trinitrotoluene and its degradation products to the aquatic amphipod Hyalella azteca. Ecotoxicology and Environmental Safety, 70, 38–46.
Singh, N. P., McCoy, M. T., Tice, R. R., & Schneider, E. L. (1988). A simple technique for quantitation of low levels of DNA damage in individual cells. Experimental Cell Research, 175, 123–130.
Sun, L. W., Qu, M. M., Li, Y. Q., Wu, Y. L., Chen, Y. G., Kong, Z. M., et al. (2004). Toxic effects of aminophenols on aquatic life using the zebrafish embryo test and the comet assay. Bulletin of Environmental Contamination and Toxicology, 73, 628–634.
Svendsen, C., & Weeks, J. M. (1997). Relevance and applicability of a simple earthworm Biomarker of copper exposure. I. Links to ecological effects in a laboratory study with Eisenia andrei. Ecotoxicology and Environmental Safety, 36, 72–79.
Svendsen, C., Spurgeon, D., Hankard, P., & Weeks, J. (2004). A review of lysosomal membrane stability measured by neutral red retention: is it a workable earthworm biomarker? Ecotoxicology and Environmental Safety, 57, 20–29.
Weeks, J. M., & Svendsen, C. (1996). Neutral red retention by lysosomes from earthworm (Lumbricus rubellus) coelomocytes: A simple biomarker of exposure to soil copper. Environmental Toxicology and Chemistry, 15, 1801–1805.
Xiao, N. W., Jing, B., Ge, F., & Liu, X. H. (2006). The fate of herbicide acetochlor and its toxicity to Eisenia fetida under laboratory conditions. Chemosphere, 62, 1366–1373.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fuchs, J., Piola, L., González, E.P. et al. Coelomocyte biomarkers in the earthworm Eisenia fetida exposed to 2,4,6-trinitrotoluene (TNT). Environ Monit Assess 175, 127–137 (2011). https://doi.org/10.1007/s10661-010-1499-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10661-010-1499-z