Abstract
Experiments were conducted for 3 seasons, 2007–2008, 2008–2009 and 2009–2010 in a wheat field planted with a cultivar susceptible to powdery mildew in Langfang City, Hebei Province, China. Plants were inoculated with Blumeria graminis f. sp. tritici (Bgt) and conidia of Bgt in the air were trapped using volumetric spore samplers. Disease severity was recorded weekly. The relationships between airborne conidial concentrations and meteorological factors, as well as disease index were analyzed. Conidia were first detected about 20 days after inoculation in all three seasons, and then increased gradually with time. The highest conidial concentrations in the air were observed in mid-May 2008 and 2009 and late May 2010 at growth stage (GS) 10.5.4. The concentrations of Bgt conidia after inoculation (GS 5) to milky ripe (GS 11.1) in the air were positively correlated with temperature, solar radiation, and negatively with relative humidity and vapor pressure deficit (VPD). Prediction models of Bgt conidial concentrations in the air based on meteorological factors were constructed using multiple regression analysis. Time series analysis, using autoregressive integrated moving average (ARIMA) (p, d, q) models, showed that each of the three season’s data can be fitted with simple ARIMA (1, 0, 0) models. Conidial concentrations within the canopy were significantly higher than those above the canopy (P < 0.01). The weekly-accumulated mean hourly conidia per cubic metre of air significantly (P < 0.01) correlated with disease index in all three seasons.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Powdery mildew of wheat, caused by the obligate fungal parasite B. graminis f. sp. tritici (Bgt), is widespread throughout the world, especially in the regions where rain occurs early in the season and temperatures are relatively low, such as the regions with maritime climate and the cooler regions with a humid continental climate (Bennett 1984). Yield losses of 10–15% can occur from natural infections in wheat (Bowen et al. 1991; Lipps and Madden 1989). In China, wheat powdery mildew has tended to be severe since the 1970s. The area of wheat production affected by powdery mildew was over 12.0 million ha in 1990 and 1991, causing an estimated grain yield losses of 1.44 and 0.77 million tons, respectively (Liu and Shao 1994).
Symptoms of powdery mildew can appear at any time on leaves, sheaths, stems and heads after seedlings emerge. The disease begins as small white circular patches of fungal mycelium often surrounded by chlorosis, most visible on the underside of the leaves. As the diseased area expands, tissue around older pustules dies and turns brown. Powdery mildew is prevalent on lower leaves and can develop on upper leaves when it is severe. Applications of host resistance and fungicides are the two main disease management strategies (Wolfe 1984), but due to the emergence of virulent races of the pathogen, varieties often lose their resistance in a relatively short period of time. So far, application of fungicide is still efficient and essential for disease management wherever yield potential is high (Schepers et al. 1996).
The optimal temperatures for development of powdery mildew ranged from 15 to 22°C and infection and disease development decline when temperature is >25°C (Mathre 1982). Infections of barley powdery mildew can occur when temperature is as low as 5°C (Ward and Manners 1974). It is assumed (Aust and von Hoyningen-Huene 1986) that powdery mildew favours relatively dry atmospheric conditions and mild temperatures. Although rains can promote spore germination and infection, they can also apparently reduce conidia density in the air. Therefore, the effects of rain on powdery mildew vary in different regions. For example, in China, rain is favourable to powdery mildew in the dry northern areas such as in Wendeng, Shandong province; if rainfall in the last twenty-day period of April is more than 50 mm, severe disease would occur. But in southern areas such as in Wanxian, Sichuan province, disease severity is reduced when rainfall exceeds 77 mm in March.
Development of the disease is polycyclic within a wheat growing season. Production of conidia is optimal at 20°C and declines rapidly above and below this temperature (Ward and Manners 1974). Conidia germinate most rapidly at 97 to 100% RH. However, germ tube growth and appressorium production are greatly reduced below 92% RH (Friedrich and Boyle 1993).
Wind is considered to be the major factor influencing dispersion of conidia of Bgt. Airborne conidia of Bgt disperse infections, facilitating a progressive increase in disease development locally, and ultimately the spread of disease epidemics regionally. Given the key role played by airborne conidia in disease spread, it is important to understand the temporal dynamics of Bgt inoculum in the air during the wheat growing seasons for disease prediction and evaluation. The conidial concentration in the air can be used as an indicator to estimate the possible trends of disease epidemics. Aerobiological studies could lead to a better understanding of the biology and epidemiology of an airborne fungal pathogen, and to the development of a disease management program to reduce unnecessary fungicide applications and the risk of the disease (Govi 1992). Measurement of pseudothecial maturation combined with monitoring of airborne ascospores, on a regional basis, has been suggested to improve regional forecasts for stem canker (West et al. 1999) and molecular diagnostics combined with air sampling can now allow quantification of many airborne plant pathogens (West et al. 2008). Smith and Davies (1973) studied relationships between weather conditions and spore trap catches of barley mildew. The effect of meteorological factors on dispersal of other powdery mildew conidia such as apple powdery mildew (Sutton and Jones 1979; Xu et al. 1995), grape powdery mildew (Willocquet and Clerjeau 1998), cherry powdery mildew (Grove 1998) and strawberry powdery mildew (Blanco et al. 2004) have been studied. However, there are few studies on the relations between conidial dispersal and powdery mildew in wheat.
The objectives of this study were: (i) to study the dynamics of Bgt conidial concentrations in the air; (ii) to determine the relationship between environment conditions and the concentrations of Bgt conidia in the air; and (iii) to determine the relationship between concentration of Bgt conidia and disease index in wheat.
Materials and methods
Experimental field and inoculation
The experiments were carried out in 3 wheat growing seasons, 2007–2008, 2008–2009 and 2009–2010 in the same field at the Langfang Experimental Station of the Chinese Academy of Agricultural Sciences (39°30′42″N, 116°36′07″E) in Hebei Province, China. The field has been cropped with a winter wheat/soybean rotation since 2002. Flooding irrigations were performed six times during each season and fertilizers were applied twice. Weeds were controlled by hand weeding and no fungicides were used. The experimental field was maintained according to conventionally agronomic practices for wheat production in China. Each year in the first week of October (on 6 October 2007, 5 October 2008 and 6 October 2010, respectively), experimental fields were planted with cultivar Jingshuang 16, which was highly susceptible to wheat powdery mildew. Plants were grown in rows with 0.25 m of row space, and the plot was 6 × 20 m.
In order to produce conidial spores for field experiments, seedlings of the cultivar Jingshuang 16, growing in a greenhouse were inoculated. About 50 seeds of Jingshuang 16 were sown in a 10-cm pot. Ten days after seed sowing, plants were inoculated by spreading spores on the leaves. The inoculated plants were incubated under 18°C for 7 days. In spring of each year (24 March 2008, 27 March 2009 and 2 April 2010, respectively), the prepared pots with infected seedlings were placed in the experimental plot to initiate disease development. One pot of the infected seedlings was placed in every five rows.
Sampling and quantification of Bgt conidia in the air
In the experimental field, two 7-day Volumetric Spore Samplers (Burkard Manufacturing. Co. Ltd., Ricksmanworth, Hertfordshire, UK) were established to collect conidia of Bgt in the air. The traps were placed in the center of the field 2 weeks after inoculation, and operated at a constant flow rate of 10 l of air per minute. In order to determine the difference in conidial concentrations between inside and above the canopy, two sampling orifices were 1.6 m and 0.6 m above the ground in all three seasons, respectively. The distance between the spore traps was 2 m. Melinex tape coated with petroleum jelly was placed on the trap drum. The tape was replaced every 7 days and cut into 7 sections, each 48 mm long, representing a single day. Under a compound microscope (×400), four longitudinal traverses of the 48 mm tape surface were examined to determine the concentration of conidia in air in terms of number of conidia per cubic metre of air sampler per day.
Weather data collection
Meteorological data were collected using a Dynamet weather station (Dynamax, Inc., Houston, Texas, USA) placed about 50 m away from the experimental field. A data logger (CR1000, Campbell Scientific, Logan, UT) was used to record weather data at 1-h average. Air temperature (T) and relative humidity (RH) were recorded with a CS500 temperature and relative humidity probe (Campbell Scientific, Logan, UT) at a height of 1.6 m. Precipitation (P) was measured with a tipping bucket rain gage (TE525, Campbell Scientific). A silicon radiation sensor (LI-200SZ, Li-Cor, Lincoln, NE) was used to measure solar radiation (SR). Wind speed was recorded by 3 Cup Anemometer AC (RM Young Model 03001, RM Young Co., Traverse City, MI), positioned at 2 m above the ground. Daily average temperature, vapor pressure deficit (VPD), RH and wind speed, rainfall and solar radiation were then calculated.
Disease assessment
At various growth stages (GS, Feekes scale: Large 1954), disease was assessed at 7-day intervals from about 1 month after inoculation (GS 5) to milky ripe stage (GS 11.1). Powdery mildew severity was assessed by using a 0-to-9 scale, in which 0 = no disease and 9 = more than 90% of plant area was diseased (Sheng and Duan 1991). Each time, 18 positions each with 20 stems selected randomly within the plot were investigated, respectively. The average disease index (DI) was calculated by using the following equation:
Where: n0, n1......n9 are the number of plants with severity as 0, 1…9, respectively.
Data analysis
The concentration of conidia in the air determined for each spore trap sample was logarithm (log10) transformed for data analysis.
Time series analysis
Autoregressive integrated moving average (ARIMA) models, describing a wide range of time series, were built to predict Bgt spore concentrations. ARIMA modeling generally proceeds in three stages: preliminary analyses to identify which of the above models is suitable, estimation of parameters and final validation (Box and Jenkins 1976). Before time series analysis, the Box–Cox transformation, originally proposed by Box and Cox (1964), was used to make the data more normal and with less skewness.
Correlation and regression analysis
The correlations between daily and hourly meteorological variables and respective daily and hourly conidial concentrations in the air were analyzed using the CORR procedure of SAS (SAS Institute Inc, Cary, NC, USA, 1996). The differences in spore concentration between inside and above the canopy were compared by using t-test of SAS. The exponential regression between disease severity and accumulated weekly mean conidial concentration was performed by using the nonlinear CORR procedure of SAS. Parallel curve analysis was conducted to examine the effects of years.
Results
Disease epidemics in the field
The disease indices of wheat powdery mildew in the experimental field exceeded 80 in all 3 seasons on the last disease assessment date (GS 11.1), although disease epidemic patterns differed among seasons (Fig. 1). Developments of wheat powdery mildew were similar in 2007–2008 and 2008–2009 seasons. Disease developed rapidly from 23 April (GS 5) to 21 May (GS 10.5.4) in 2008 and from 21 April (GS 5) to 19 May (GS 10.5.4) in 2009. Disease index increased from 9.7, 5.0 to 74.5, 76.5 while the mean temperature and RH were 16.7°C, 58.4% and 18.6°C, 49.3% during this period of time, respectively. The disease developed slower when temperature was higher than 23°C in both seasons. Disease in the 2009–2010 season occurred later and developed more slowly from 27 April (GS 5) to 5 May (GS 10) with mean temperature at 11.4°C. Disease index increased only by 3.55 from GS 5 to GS 10, compared to 18.78 and 14.55 in 2007–2008 and 2008–2009 seasons respectively. Then disease developed quickly before 27 May (GS 10.5.4) with mean temperature at 19.1°C and RH at 45.1% in the 2009–2010 season. After that, disease development became slower.
Seasonal dynamics of conidial concentration in relation to weather variables
In all three seasons, conidial concentrations in the air were associated with increased resulting disease development. A clear peak of conidial concentration was observed in each season, and after then the concentration declined gradually (Fig. 2).
Daily mean temperature (T), relative humidity (RH), rainfall and number of Bgt spores per cubic metre of air after inoculation in the growing seasons of wheat in Langfang for 2007–2008, 2008–2009 and 2009–2010 season. Concentrations of conidia were determined with Burkard volumetric spore samplers operated continuously
During the 2007–2008 season, spores of Bgt in the air were first detected on 15 April (22 days after inoculation) and continuously detected until 1 June except on 21 April. Rain (>5 mm) occurred on 20 April. Conidial concentrations increased gradually as season progressed until the highest conidial concentration in air was observed on 18 May (55 days after inoculation, GS 10.5.4) when temperature was 19.6°C, RH was 39.7% and no rain occurred, but RH was 80.6% on 17 May. Then conidial concentrations decreased as temperature was higher than 22°C from 20 to 28 May.
In the 2008–2009 season, spores were first observed on 16 April (20 days after inoculation). Conidial concentrations in the air increased with time until the highest concentration reached on 16 May (50 days after inoculation, GS 10.5.4) when the temperature was 20.2°C, RH was 26.6% and no rainfall occurred. But RH increased to 65.8% on 15 May. After then, the conidial concentration decreased through 2 June when temperature was higher than 22°C from 17 to 27 May except on 21 May.
In the 2009–2010 season, spores were first observed on 22 April (20 days after inoculation), and then increased gradually with time. The highest concentration was observed on 31 May (59 days after inoculation, GS 10.5.4) when temperature was 21.0°C, RH was 62.7%, and no rain occurred. Then spore concentrations decreased through 12 June. Rainfall exceeded 15 mm in 4 h on 1 June, and temperature was higher than 23°C from 5 to 9 June. The concentrations of conidia in the air were much higher during the third season (2009–2010) than those in the first (2007–2008) and the second (2008–2009) season.
There was a significant and positive correlation between temperature and log10 conidial concentration in the 3 seasons, while the relationship between log10 (conidial concentration +1) and rainfall was insignificant (Table 1). The log10 (conidial concentration +1) was significantly and negatively correlated with WS in the 2007–2008 and 2009–2010 seasons, as well as with VPD in 2007–2008 and 2008–2009 seasons. RH and SR were negatively and positively correlated with log10 conidial concentration +1) in the 2008–2009 season, respectively. On the basis of these correlation results, the prediction models for spore concentrations based on meteorological variables were built using multiple stepwise regression analysis (Table 2).
After transformation by Box–Cox transformation, the data for all three seasons can be fitted with a simple autoregressive model ARIMA (1, 0, 0) (Table 2). Compared with models based on meteorological variables, the R2-values for ARIMA models were increased in all 3 seasons.
Diurnal periodicity of conidial concentration and relations with weather variables
Correlation analyses of daily spore concentration and environmental factors showed two general patterns or types of spore dispersal. In type 1 (Fig. 3a), representing the situation when rainfall occurred or air relative humidity was high, conidial concentrations were abundant throughout the day with no discernable periodicity, and no significant correlations between log10 conidial concentration and hourly environmental factors were found (Table 3). In type 2 (Fig. 3b), spore concentrations in daytime were higher than that at night, with a peak in mid-day. Hourly temperature, wind speed and solar radiation were usually higher and relative humidity was lower during mid-day. The log10 (conidial concentration +1) was significantly correlated with hourly environmental factors, including temperature, RH, SR and WS (Table 3).
Two types of hourly concentrations of airborne conidia of Bgt in the air determined by Burkard volumetric spore sampler (a: type 1; b: type 2). In type 1, hourly conidial concentrations in the air were with little or no correlation with hourly environmental variables when rainfall occurred or air relative humidity was high. In type 2, hourly conidial concentrations in the air were highly correlated with hourly environmental variables
Conidial concentrations within or above the canopy
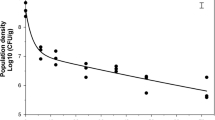
The concentrations of conidia within the canopy were significantly higher than those above the canopy in all the seasons assessed by t- test (t =7.362, P =0.0001; t =6.372, P =0.0001 and t =5.653, P =0.0001 for 2007–2008, 2008–2009, 2009–2010 season, respectively) (Fig. 4). The mean concentrations of conidia were 2.1 spores m−3 and 1.0 spores m−3 at the heights of 0.6 m and 1.6 m above ground during the 2007–2008 season, respectively, and those were 2.6 spores m−3 at the height of 0.6 m and 0.7 spores m−3 at the height of 1.6 m during the 2008–2009 season. In the 2009–2010 season, the mean concentrations of conidia were 9.6 spores m−3 at 0.6 m above ground and 3.1 spores m−3 at the height of 1.6 m above ground. In all the three seasons, significant positive correlations were found between spore concentrations at 0.6 m and those at 1.6 m above ground.
Relationship between disease index and conidial concentration in the air
Disease index was significantly correlated with the weekly-accumulated concentration of conidia per cubic metre of air at all three seasons (P = 0.0057, 0.003, 0.0004 in 2007–2008, 2008–2009 and 2009–2010, respectively). Parallel curve analysis showed that there was no significant difference (P = 0.1895 for slope and P = 0.6056 for intercept, respectively) in the fitted equations for disease index and accumulated weekly mean hourly conidia of Bgt per cubic metre of air between 2007 and 2008 and 2008–2009 seasons. Therefore a significant (P = 0.0001) exponential relation was found between disease index and weekly-accumulated mean hourly concentration of airborne conidia per cubic metre for 2007–2008 and 2008–2009 season with the R2-value as 0.87 (Fig. 5a). The exponential relationship between disease index and weekly-accumulated mean hourly concentration of conidia per cubic metre air for the 2009–2010 season was also significant (P = 0.0004, R2 = 0.96) (Fig. 5b).
Relationship between disease index and weekly-accumulated number of Bgt spores per cubic metre of air at Langfang in 2007–2008 and 2008–2009 season (a) and 2009–2010 season (b). Parallel curve analysis showed no significant difference (P = 0.1895 for slop factor and P = 0.6056 for intercept, respectively) between 2007–2008 and 2008–2009 season in the fitted equations between disease index and accumulated weekly mean hourly conidia of Bgt per cubic metre of air. Thus, the data were incorporated
Discussion
Concentration of Bgt conidia in the air increased with disease development, and then decreased after reaching its maximum level in all three seasons. This was consistent to the findings of the previous studies (Jenkyn 1974; Friedrich 1995), in which they found that the dispersal of Bgt conidia parallels with powdery mildew development, although conidial concentrations fluctuated daily. In early growing stages, the disease developed quickly under favorable weather conditions from lower leaves to higher leaves, which was related to the increasing conidial concentration in the air. Later in the growing season, as temperatures became higher, usually above 23°C, the disease declined, that led a reduction of spore production. Mycelium on older leaves died and sporulation stopped, so the conidial concentration was greatly reduced.
Although spores can be transported long-distance by winds, the turbulent nature of wind dilutes most of the spores as they move down-wind. The corollary of gust penetration is the gradual ejection of air from the canopy after gusts, which can be responsible for the upward transport of spores. In another phenomenon, called outward interactions, air moves upwards at speeds greater than the local average wind speed (Aylor 1990). These complicated flow patterns have consequences for spore removal and transport within and out of the canopy. Wheat powdery mildew is prevalent on lower leaves and can develop on upper leaves when it is severe which implies that the possibility of up-draught spread of the disease through the canopy. Therefore, we consider that almost the spores captured by spore traps in our studies came from local fields, and those that came from long-distance transport accounted for a low proportion.
Smith and Davies (1973) reported that spore catches of barley mildew increased rapidly with increasing in maximal daily temperatures above 19°C. We obtained that the peak concentration of wheat powdery mildew spores in air was in mid to late May with average daily temperature at about 20°C which was the optimal temperature for powdery mildew sporulation (Ward and Manners 1974).
It has been proved that raindrops can apparently reduce conidial concentration in the air (Paulus 1990). Sivapalan (1993) demonstrated that rain can cause physical damage to mildews, and the correlation between rainfall and strawberry powdery mildew conidia was negative and significant (Blanco et al. 2004). However, in our study, only 4 rainy days occurred in the 2007–2008 season when spores were trapped. The relationships between conidial concentration and rainfall were negative but not significant in all seasons.
The correlation between RH and spore concentration was significantly negative in the 2007–2008 season but not in the 2008–2009 and 2009–2010 seasons. Although high RH is needed for spore germination and production, it might not be as well for spore liberation. Also, the changes in relative humidity can trigger spore liberation (Adams et al. 1986). It was found in our study that the peaks of conidial concentrations in the air in all three seasons were followed by RH changes from 80.6%, 65.8%, 52.8% to 39.7%, 26.6%, 62.7% in 2007–2008, 2008–2009 and 2009–2010 respectively.
Many studies reported the effects of wind on powdery mildew spore release and wind was the main factor that related with spore release. The wind speed for conidia release ranged from 0.6 to 2.0 m s−1, but wind speed within plant canopies seldom reached 0.5 m s−1. However, brief gusts of high speeds apparently occur in canopies (Aylor and Lukens 1974). But the daily mean wind speed was little relevant to spore dispersal occurring during very short period of time, and daily mean wind speed indicated little about gustiness, which was very important in spore liberation. In our study, the correlation between daily conidial number and average daily wind speed was negative but not significant, while there was significant positive correlation between hourly conidial number and hourly wind speed, and similar results were found on apple powdery mildew (Xu et al. 1995).It was found that most of the released spores were deposited on leaves nearby, and the half distance of the dispersal gradient for Bgt was found to be 5.8 m generated from the exponential model (Cooke et al. 2006). However, the half-distance is likely to change according to wind speed and turbulence and the density of the crop canopy. Fried et al. (1979) found a power law model to determine the deposition gradients for wheat powdery mildew, and the slope of the gradients was correlated with the wind speed.
Linear models are usually used to predict spore concentrations, but these models, only using meteorological variables, may produce results with a low prediction level. And our results also confirmed this (Table 2). Other variables better reflecting the series of factors affecting spore production and dispersing should be taken into account. Based on this, we used ARIMA to predict spore concentrations. The daily data showed that the dynamics of Bgt spore concentrations in the air in all 3 seasons were similar and the proposed ARIMA models were more accurate compared with linear models.
Three general patterns of spore dispersal were observed on apple powdery mildew (Sutton and Jones 1979). In our study, two types of diurnal periodicity of conidial concentration in air were found. One was highly correlated with environmental variables, while the other with little or no correlation with environmental variables when rainfall occurred or air relative humidity was high. This indicates that relative humidity has confounding effects on conidial concentration. It is favourable for spore germination, infection and production but not for spore liberation. Wind also has confounding effects on conidial concentration. It is necessary for spore liberation and dispersal; however, it also has an influence on deposition of spores on the crop and the ground. When spores are released in wind gusts their inertial impaction efficiency is enhanced because they are travelling at relatively high wind speeds (McCartney and Bainbridge 1987).
The concentrations of Bgt conidia were much greater within the canopy than those above the canopy. Aylor and Francis (1989) indicated that concentrations of spores were generally greater inside than above the canopy and decreased monotonically with height above the canopy. Additionally, spore concentrations within and above the crop were affected by the conditions at the site where spores were formed and liberated (Last 1955; Eversmeyer et al. 1973). Because wheat powdery mildew developed from lower leaves to upper leaves, plant density and canopy structure influenced the dynamics of spores escape from the canopy, especially when disease was most prevalent on lower leaves. The exponential equations describing the relationship between disease indexes and weekly-accumulated mean hourly concentration of airborne conidia per cubic metre were significantly different among the three seasons. The variation in environmental conditions which can affect both the development of disease and airborne conidial concentrations may be the main reason for this difference. For example, average daily temperature in March and April in 2010 was lower than those in 2008 and 2009. The effects were not only on powdery mildew development, but also on wheat growth delaying inoculation in 2010.
Although many prediction models for wheat powdery mildew have been constructed for disease management (Rossi and Giosuè 2003; Audsley et al. 2005), few of them considered inoculum amount, while inoculum was an inevitable factor in the development of this polycyclic disease. Xu et al. (2000) derived models for infection of strawberry flowers by Botrytis cinerea by inoculum only, by weather variables only, and by both inoculum and weather variables, and models using both weather variables and inoculum gave the best predictions. Mathematically, the cumulative spore counts are synonymous with disease severities (Zadoks 1974), and Jeger (1984) developed models to describe the relationship between apple powdery mildew progress and the cumulative numbers of trapped spores. In this study, prediction models based on inoculum amount were constructed and they indicated that predictive models for wheat powdery mildew based on airborne conidia are feasible for disease management and fungicide application.
References
Adams, G. C., Gottwald, T. G., & Leach, C. M. (1986). Environmental factors initiating liberation of conidia of powdery mildews. Phytopathology, 7, 1239–1245.
Audsley, E., Milne, A., & Paveley, N. (2005). A foliar disease model for use in wheat disease management decision support systems. Annals of Applied Biology, 147, 161–172.
Aust, H. J., & von Hoyningen-Huene, J. (1986). Microclimate in relation to epidemics of powdery mildew. Annual Review of Phytopathology, 24, 491–510.
Aylor, D. E. (1990). The role of intermittent wind in the dispersal of fungal pathogens. Annual Review of Phytopathology, 28, 73–92.
Aylor, D. E., & Francis, J. F. (1989). Dispersion of spores released from an elevated line source within a wheat canopy. Boundary-Layer Meteorology, 46, 251–273.
Aylor, D. E., & Lukens, R. J. (1974). Liberation of Helminthosporium maydis spores by wind in field. Phytopathology, 64, 1136–1138.
Bennett, F. G. A. (1984). Resistance to powdery mildew in wheat: a review of its use in agriculture and breeding programmes. Plant Pathology, 33, 279–300.
Blanco, C., Santos, B. D. L., Barrau, C., Arroyo, F. T., Porras, M., & Romero, F. (2004). Relationship among concentrations of Sphaerotheca macularis conidia in the air, environmental conditions, and the incidence of powdery mildew in strawberry. Plant Disease, 88, 878–881.
Bowen, K. L., Everts, K. L., & Leath, S. (1991). Reduction in yield of winter wheat in North Carolina due to powdery mildew and leaf rust. Phytopathology, 81, 503–511.
Box, G. E. P., & Cox, D. R. (1964). An analysis of transformations. Journal of the Royal Statistical Society, Series B, 26, 211–252.
Box, G. E. P., & Jenkins, G. M. (1976). Time series analysis, forecasting, and control. San Francisco: Holden-Day.
Cooke, B. M., Jones, D. G., & Kaye, B. (Eds.). (2006). The epidemiology of plant diseases. Netherlands: Springer.
Eversmeyer, M. G., Kramer, C. L., & Burleigh, J. R. (1973). Vertical spore concentrations of three wheat pathogens above a wheat field. Phytopathology, 63, 211–218.
Fried, P. M., Mackenzie, D. R., & Nelson, R. R. (1979). Dispersal gradients from a point source of Erysiphe graminis f. sp. tritici on Chancellor winter wheat and four multilines. Phytopathologische Zeitschrift, 95, 140–150.
Friedrich, S. (1995). Calculation of conidial dispersal of Erysiphe graminis within naturally infected plant canopies using hourly meteorological input parameters. Zeitschrift für Pflanzenkrankheiten und Pflanzenschutz, 102, 337–347 (in German).
Friedrich, S., & Boyle, C. (1993). Wirkung unterschiedlicher Luftfeuchten auf die Production und Keimung der Konidien von Erysiphe graminis f. sp. tritici in vitro. Journal of Plant Diseases and Protection, 100, 180–188. in German.
Govi, G. (1992). Aerial diffusion of phytopathogenic fungi. Aerobiologia, 8, 84–93.
Grove, G. G. (1998). Meteorological factors affecting airborne conidia concentrations and the latent period of Podosphaera clandestina on sweet cherry. Plant Disease, 82, 741–746.
Jeger, M. J. (1984). Relating disease progress to cumulative numbers of trapped spores: apple powdery mildew and scab epidemics in sprayed and unsprayed orchard plots. Plant Pathology, 33, 517–523.
Jenkyn, J. F. (1974). A comparison of seasonal changes in deposition of spores of Erysiphe graminis on different trapping surfaces. Annals of Applied Biology, 76, 257–267.
Large, E. C. (1954). Growth stages in cereals. Illustration of the Feekes scale. Plant Pathology, 3, 128–129.
Last, F. T. (1955). The spore content of air within and above mildew infested cereal crops. Transactions of the British Mycological Society, 38, 453–464.
Lipps, P. E., & Madden, L. V. (1989). Effect of fungicide application timing on control of powdery mildew and grain yield of winter wheat. Plant Disease, 73, 991–994.
Liu, W. C., & Shao, Z. R. (1994). Epidemiology, occurrence and analysis of wheat powdery mildew in recent years. Plant Protection Technology and Extension, 6, 17–20 (in Chinese).
Mathre, D. E. (1982). Compendium of barley diseases. St. Paul: APS.
McCartney, H. A., & Bainbridge, A. (1987). Deposition of Erysiphe graminis conidia on a barley crop. I: sedimentation and impaction. Journal of Phytopathology, 118, 243–257.
Paulus, A. O. (1990). Fungal diseases of strawberry. HortScience, 25, 885–889.
Rossi, V., & Giosuè, S. (2003). A dynamic simulation model for powdery mildew epidemics on winter wheat. Bulletin OEPP/EPPO Bulletin, 33, 389–396.
Schepers, H., Bouma, E., Frahm, J., Wolk, T., & Secher, B. J. M. (1996). Control of fungal diseases in winter wheat with appropriate dose rates and weather-based decision support systems. Bulletin EPPO/OEPP Bulletin, 26, 623–630.
Sheng, B. Q., & Duan, X. Y. (1991). Improvement of scale 0–9 method for scoring adult plant resistance to powdery mildew of wheat. Beijing Agricultural Sciences, 9, 38–39 (in Chinese).
Sivapalan, A. (1993). Effects of impacting rain drops on the growth and development of powdery mildew fungi. Plant Pathology, 42, 256–263.
Smith, L. P., & Davies, R. R. (1973). Weather conditions and spore trap catches of barley mildew. Plant Pathology, 22, 1–10.
Sutton, T. B., & Jones, A. L. (1979). Analysis of factors affecting dispersal of Podosphaera leucotricha conidia. Phytopathology, 69, 380–383.
Ward, S. V., & Manners, J. G. (1974). Environmental effects on the quantity and viability of conidia produced by Erysiphe graminis. Transactions of the British Mycological Society, 62, 119–128.
West, J. S., Biddulph, J. E., Fitt, B. D. L., & Gladders, P. (1999). Epidemiology of Leptosphaeria maculans in relation to forecasting stem canker severity on winter oilseed rape in the UK. Annals of Applied Biology, 135, 535–546.
West, J. S., Atkins, S. D., Emberlin, J., & Fitt, B. D. L. (2008). PCR to predict risk of airborne disease. Trends in Microbiology, 16, 380–387.
Willocquet, L., & Clerjeau, M. (1998). An analysis of the effects of environmental factors on conidial dispersal of Uncinula necator (grape powdery mildew) in vineyards. Plant Pathology, 47, 227–233.
Wolfe, M. S. (1984). Trying to understand and control powdery mildew. Plant Pathology, 33, 451–466.
Xu, X. M., Butt, D. J., & Ridout, U. S. (1995). Temporal patterns of airborne conidia of Podosphaera leucotricha, causal agent of apple powdery mildew. Plant Pathology, 44, 944–955.
Xu, X. M., Harris, D. C., & Berrie, A. M. (2000). Modeling infection of strawberry flowers by Botrytis cinerea using field data. Phytopathology, 90, 1367–1374.
Zadoks, J. C. (1974). The role of epidemiology in modern phytopathology. Phytopathology, 64, 918–923.
Acknowledgments
This work was financially supported by National Natural Science Foundation of China (31171793), National Basic Research Program of China (2010CB951503) and Special Fund for Agro-scientific Research in the Public Interest (No.3-15). The authors thank Dr Xiangming Xu, East Malling Research, Kent, UK and Dr Jonathan S. West, Rothamsted Research, Harpenden, UK for their constructive comments to improve the quality of this manuscript prior to submission.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cao, X., Duan, X., Zhou, Y. et al. Dynamics in concentrations of Blumeria graminis f. sp tritici conidia and its relationship to local weather conditions and disease index in wheat. Eur J Plant Pathol 132, 525–535 (2012). https://doi.org/10.1007/s10658-011-9898-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-011-9898-8