Abstract
Red meat and processed meat consumption has been hypothesized to increase risk of cancer, but the evidence is inconsistent. We performed a systematic review and meta-analysis of prospective studies to summarize the evidence of associations between consumption of red meat (unprocessed), processed meat, and total red and processed meat with the incidence of various cancer types. We searched in MEDLINE and EMBASE databases through December 2020. Using a random-effect meta-analysis, we calculated the pooled relative risk (RR) and 95% confidence intervals (CI) of the highest versus the lowest category of red meat, processed meat, and total red and processed meat consumption in relation to incidence of various cancers. We identified 148 published articles. Red meat consumption was significantly associated with greater risk of breast cancer (RR = 1.09; 95% CI = 1.03–1.15), endometrial cancer (RR = 1.25; 95% CI = 1.01-1.56), colorectal cancer (RR = 1.10; 95% CI = 1.03–1.17), colon cancer (RR = 1.17; 95% CI = 1.09-1.25), rectal cancer (RR = 1.22; 95% CI = 1.01-1.46), lung cancer (RR = 1.26; 95% CI = 1.09–1.44), and hepatocellular carcinoma (RR = 1.22; 95% CI = 1.01-1.46). Processed meat consumption was significantly associated with a 6% greater breast cancer risk, an 18% greater colorectal cancer risk, a 21% greater colon cancer risk, a 22% greater rectal cancer risk, and a 12% greater lung cancer risk. Total red and processed meat consumption was significantly associated with greater risk of colorectal cancer (RR = 1.17; 95% CI = 1.08–1.26), colon cancer (RR = 1.21; 95% CI = 1.09–1.34), rectal cancer (RR = 1.26; 95% CI = 1.09–1.45), lung cancer (RR = 1.20; 95% CI = 1.09-1.33), and renal cell cancer (RR = 1.19; 95% CI = 1.04–1.37). This comprehensive systematic review and meta-analysis study showed that high red meat intake was positively associated with risk of breast cancer, endometrial cancer, colorectal cancer, colon cancer, rectal cancer, lung cancer, and hepatocellular carcinoma, and high processed meat intake was positively associated with risk of breast, colorectal, colon, rectal, and lung cancers. Higher risk of colorectal, colon, rectal, lung, and renal cell cancers were also observed with high total red and processed meat consumption.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Red meat and processed meat as rich sources of saturated fat and heme iron, as well as some carcinogens may be an important dietary risk factors leading to several cancers. Based on the International Agency for Research on Cancer (IARC) Working Group report, red meat (unprocessed) consumption including beef, lamb, veal, pork, mutton, goat, or horse meat may increase risk of colorectal, pancreatic, and prostate cancers, and processed meat including bacon, sausages, salami, hot dogs, or processed turkey may increase risk of colorectal cancer and stomach cancer [1]. However, in pooled analyses of prospective studies, high consumption of red meat or processed meat was not associated with risk of overall prostate cancer [2], renal cell cancer [3], or breast cancer [4]. In contrast, meta-analysis of prospective studies showed that processed meat consumption, but not red meat, was associated with higher breast cancer risk [5]. Higher risk of colorectal cancer, not rectal cancer, was reported with high consumption of both red meat and processed meat in a meta-analysis of prospective studies [6]. Zhao et al. reported in a meta-analysis that risk of pancreatic cancer may increase among men with high intake of red meat and processed meat, but not among women [7]. Furthermore, in meta-analyses of prospective studies, high intake of processed meat, but not red meat, was associated with higher risk of overall prostate cancer [8] and stomach cancer [9], and high intake of red meat, but not processed meat, was associated with higher risk of lung cancer [10] and hepatocellular carcinoma [11]. Processed meat intake was significantly associated with higher risk of renal cell cancer in a dose-response meta-analysis, but not in a meta-analysis comparing the highest versus the lowest category of intake [12]. However, in meta-analyses of prospective studies, red meat or processed meat intake was not associated with risk of ovarian cancer [13], endometrial cancer [14], esophageal cancer [15], bladder cancer [16], leukemia, non-Hodgkin lymphoma [17], or glioma [18]. The existing evidence from meta-analyses on red meat as well as processed meat consumption and cancer incidence is mixed and has several limitations, such as the inclusion of case–control studies, use of some identical studies twice, or inclusion of both cancer mortality and incidence in the meta-analyses. Furthermore, some of the meta-analyses needed to be updated by including newly published articles. This study addresses the gaps identified which will help clarify dietary recommendations for cancer prevention as part of an overall healthy lifestyle. In particular, we integrated the prospective studies into our analysis to identify the associations with various types of cancer incidence hypothesized to be related to consumption of red meat, processed meat, and total red and processed meat, using a systematic review and meta-analysis of prospective studies.
Subjects and methods
Study strategy
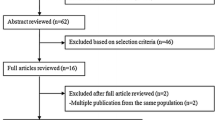
Design, analysis, and interpretation of findings have been done using the Meta-analysis of Observational Studies in Epidemiology (MOOSE) checklist [19]. Two databases, MEDLINE and EMBASE were used to identify publications of prospective studies that reported the associations of red meat, processed meat, or total red and processed meat, with any type of cancer until December 2020. The search string is presented in Supplementary Table S1. In addition, we searched references of related articles to find other relevant publications. We used the definition of IARC Working Group classification for red meat and processed meat [1]. In order to reduce the influence of recall and selection bias, we only selected prospective studies that evaluated the associations of red meat, processed meat, or total red and processed meat consumption as exposures and breast cancer, ovarian cancer, endometrial cancer, prostate cancer, colorectal cancer, colon cancer, rectal cancer, stomach cancer, esophageal squamous-cell carcinoma, esophageal adenocarcinoma, pancreatic cancer, lung cancer, bladder cancer, renal cell cancer, hepatocellular carcinoma, leukemia, non-Hodgkin lymphoma, melanoma, and glioma as endpoints. Case–control, cross-sectional, and ecological studies were excluded. We also excluded reviews and meeting abstracts. Furthermore, if there were several published papers from the same study population, we used the data reported in the most recent paper with the largest number of cancer cases (Fig. 1).
Data extraction
We summarized the characteristics of selected publications including first author, year of publication, study name, country of the study, design of study, follow up years, the total number of participants, number of cancer events, age at baseline, meat variable definition and method of assessment, amount of intake in the highest and the lowest categories, and covariates adjusted in the multivariable models. The relative risks (RRs) and 95% confidence intervals (CI) for the highest versus the lowest categories of intake were extracted from multivariable models with the highest number of covariates.
Data synthesis
We conducted separate analyses on various cancer sites using three exposure variables: “red meat” “processed meat” and “total red and processed meat.” In this meta-analysis, red meat means only unprocessed red meat; processed meat means processed red meat or other types of meat; and total red and processed meat means the sum of red meat and processed meat. The RRs for the highest versus the lowest category of intake of each exposure were pooled using random-effects models (DerSimonian and Laird method [20]). However, the amounts of intake in the highest and the lowest categories were not consistent across the studies. For one study that did not report the highest versus the lowest category of intake [21], assuming a normal distribution, the RRs and 95% CIs for the highest versus the lowest quintiles were calculated using 2.56 as a conversion factor for a one-standard deviation (SD) increase in total red and processed meat intake. For four studies, the RRs and 95% CIs for the highest versus the lowest quartiles or quintiles of intake have been provided via contacting authors [22,23,24,25].
We also calculated the RRs and 95% CI for each 100 g per day intake of red meat, and total red and processed meat, and each 50 g per day intake of processed meat in relation to each cancer, if data were available. We were able to do dose–response analyses for studies that reported the associations with exposures of interest as contentious variables or studies with information as follows: risk estimate, 95% CI or standard error, median intake, number of cases, person-years of follow-up or number of participants, for each exposure category. If ranges of intake were reported for each category of intake, we used the midpoint of the lower and upper bound of intake for each category. For an open-ended highest category, the range of amount of intake was estimated using the range of amount of intake in the previous category. For some of the studies, through correspondence with authors, relevant data (RRs and 95% CIs of exposures of interest as continuous variables, number of participants in each category, number of cases in each category, type of red meat) were provided [23, 24, 26,27,28,29,30,31,32,33,34,35,36,37,38]. For studies that reported servings or times per day of intake, we converted them to g per day as follows: one serving or one time per day of red meat (unprocessed) equal to 120 g, one serving or one time per day of processed meat equal to 50 g, and one serving or one time per day of total red and processed meat equal to 100 g [39].
The RRs and 95% CIs for each study were presented using forest plots in the meta-analysis. To evaluate publication bias, we used visual inspection of a funnel plot [40] and the Begg and Mazumdar test [41]. To evaluate potential heterogeneity among studies, we used the I2 statistic [42]. A two-tailed test at an alpha level of < 0.05 was considered statistically significant. All statistical analyses were conducted using STATA, version 16, software (STATA Corp, College Station, TX).
Results
Study characteristics
We identified 148 publications that examined the associations of red meat, processed meat, and total red and processed meat intake and various sites of cancer in prospective studies: 28 publications for breast cancer [22, 24, 26, 43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67] (Table S2); 8 publications for ovarian cancer [22, 23, 59, 68,69,70,71,72] (Table S3); 8 publications for endometrial cancer [22, 59, 73,74,75,76,77,78] (Table S4); 20 publications for prostate cancer [27, 28, 46, 59, 60, 62, 79,80,81,82,83,84,85,86,87,88,89,90,91,92] (Table S5); 38 publications for colorectal, colon, and rectal cancers [21, 29, 59, 60, 62, 64, 93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112,113,114,115,116,117,118,119,120,121,122,123,124] (Table S6); 10 publications for stomach cancer [59, 64, 70, 96, 125,126,127,128,129,130] (Table S7); 3 publications for esophageal squamous-cell carcinoma and esophageal adenocarcinoma [130,131,132] (Table S8); 16 publications for pancreatic cancer [30,31,32, 59, 133,134,135,136,137,138,139,140,141,142,143,144] (Table S9); 10 publications for lung cancer [33, 46, 59, 60, 64, 70, 145,146,147,148] (Table S10); 8 publications for bladder cancer [34, 59, 149,150,151,152,153,154] (Table S11); 5 publications for renal cell cancer [3, 59, 155,156,157] (Table S12); 7 publications for hepatocellular carcinoma [25, 35, 36, 59, 158,159,160] (Table S13), 4 publications for leukemia [59, 70, 161, 162] (Table S14), 5 publications for non-Hodgkin lymphoma [59, 163,164,165,166] (Table S15), 3 publications for melanoma [37, 59, 70] (Table S16), and 7 publications for glioma [38, 59, 167,168,169,170,171] (Table S17). Nine publications reported the findings for more than one cancer [22, 46, 59, 60, 62, 64, 70, 96, 130]. We did not observe publication bias for the associations of red meat, processed meat, or total red and processed meat intake with various sites of cancer, using visual inspection of a funnel plot or Begg and Mazumdar test (Fig. S1–S3).
Red meat (unprocessed) intake and cancer risk
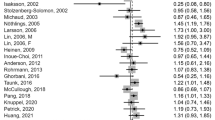
The random-effects summary of RRs comparing the highest versus the lowest category of red meat intake and various cancer types are presented in Fig. 2. Red meat consumption was associated with increased breast cancer, endometrial cancer, colorectal cancer, colon cancer, rectal cancer, lung cancer, and hepatocellular carcinoma risk. The pooled RR comparing the highest versus the lowest category of red meat intake was 1.09 (95% CI = 1.03-1.15; I2 = 51.3%; 15 risk estimates) for breast cancer (Fig. S4), 1.25 (95% CI = 1.01-1.56; I2 = 70.7%; 6 risk estimates) for endometrial cancer (Fig. S6), 1.10 (95% CI = 1.03–1.17; I2 = 28.2%; 22 risk estimates) for colorectal cancer (Fig. S8), 1.17 (95% CI = 1.09-1.25; I2 = 0.0%; 13 risk estimates) for colon cancer (Fig. S9), 1.22 (95% CI = 1.01-1.46; I2 = 49.2%; 11 risk estimates) for rectal cancer (Fig. S10), 1.26 (95% CI = 1.09-1.44; I2 = 25.7%; 7 risk estimates) for lung cancer (Fig. S15), and 1.22 (95% CI = 1.01-1.46; I2 = 0.0%; 6 risk estimates) for hepatocellular carcinoma (Fig. S18). Red meat consumption was not associated with risk of ovarian, prostate, and stomach cancers, as well as esophageal squamous-cell carcinoma, esophageal adenocarcinoma, pancreatic cancer, bladder cancer, renal cell cancer, leukemia, non-Hodgkin lymphoma, melanoma, and glioma (Figs. S5, S7, S11-S14, S16, S17, S19–S22). In dose–response analysis, using available data, each 100 g per day of red meat intake was associated with an 11% higher risk of breast cancer, a 14% higher risk of colorectal cancer, a 17% higher risk of colon cancer, a 26% higher risk of rectal cancer, and a 29% higher risk of lung cancer (Table S18).
Processed meat intake and cancer risk
The pooled RRs comparing the highest versus the lowest category of processed meat intake and various cancer types are presented in Fig. 3. Processed meat consumption was significantly associated with increased risk of breast cancer (pooled RR = 1.06; 95% CI = 1.01–1.12; I2 = 39.0%; 16 risk estimates; Fig. S4), colorectal cancer (pooled RR = 1.18; 95% CI = 1.13–1.24; I2 = 1.9%; 23 risk estimates; Fig. S8), colon cancer (pooled RR = 1.21; 95% CI = 1.13–1.29; I2 = 0.0%; 15 risk estimates; Fig. S9), rectal cancer (pooled RR = 1.22; 95% CI = 1.09-1.36; I2 = 2.4%; 14 risk estimates; Fig. S10), and lung cancer (pooled RR = 1.12; 95% CI = 1.05–1.20; I2 = 1.6%; 10 risk estimates; Fig. S15). Processed meat consumption was not associated with risk of ovarian cancer, endometrial cancer, prostate cancer, stomach cancer, esophageal squamous-cell carcinoma, esophageal adenocarcinoma, pancreatic cancer, bladder cancer, renal cell cancer, hepatocellular carcinoma, leukemia, non-Hodgkin lymphoma, melanoma, and glioma (Figs. S5-S7, S11-S14, S16–S22). In dose–response analysis, using available data, each 50 g per day of processed meat intake was associated with a 16% higher risk of colorectal cancer, a 17% higher risk of colon cancer, a 25% higher risk of rectal cancer, and an 8% higher risk of renal cell cancer (Table S18).
Total red and processed meat intake and cancer risk
The pooled RRs comparing the highest versus the lowest category of total red and processed meat intake and various cancer types are presented in Fig. 4. Total red and processed meat consumption was significantly associated with higher risk of colorectal cancer (pooled RR = 1.17; 95% CI = 1.08–1.26; I2 = 32.7%; 20 risk estimates; Fig. S8), colon cancer (pooled RR = 1.21; 95% CI = 1.09–1.34; I2 = 33.7%; 12 risk estimates; Fig. S9), rectal cancer (pooled RR = 1.26; 95% CI = 1.09–1.45; I2 = 29.4%; 10 risk estimates; Fig. S10), lung cancer (pooled RR = 1.20; 95% CI = 1.09–1.33; I2 = 30.9%; 10 risk estimates; Fig. S15), and renal cell cancer (pooled RR = 1.19; 95% CI = 1.04–1.37; I2 = 0.0%; 4 risk estimates) (Fig. S17). Findings were similar for the associations between total red and processed meat intake and colorectal cancer (pooled RR = 1.19; 95% CI = 1.11–1.29; I2 = 23.3%; 19 risk estimates), colon cancer (pooled RR = 1.29; 95% CI = 1.19–1.40; I2 = 0.0%; 11 risk estimates), and rectal cancer (pooled RR = 1.30; 95% CI = 1.11–1.53; I2 = 25.5%; 9 risk estimates), when the study by Nguyen et al. [21] was excluded; as mentioned above, we calculated the RRs and 95% CIs of the highest versus the lowest quintile of intake for that study using 2.56 as a conversion factor for a one-SD increase in total red and processed meat intake, assuming a normal distribution. Based on results from one study, high total red and processed meat intake was associated with higher risk of esophageal squamous-cell carcinoma (RR= 1.79; 95% CI = 1.07-3.01; Fig. S12). High intake of total red and processed meat was suggestively associated with higher risk of pancreatic cancer (pooled RR = 1.13; 95% CI = 1.00-1.27; I2 = 41.2%; 12 risk estimates; Fig. S14). Total red and processed meat consumption was not associated with risk of breast cancer, ovarian cancer, endometrial cancer, prostate cancer, stomach cancer, esophageal adenocarcinoma, bladder cancer, hepatocellular carcinoma, leukemia, non-Hodgkin lymphoma, melanoma, and glioma (Figs. S4–S7, S11, S13, S16, S18–S22). Each 100 g per day of total red and processed meat intake was associated with a 12% higher risk of endometrial cancer, an 18% higher risk of colorectal cancer, a 25% higher risk of colon cancer, a 25% higher risk of rectal cancer, and a 35% higher risk of lung cancer (Table S18).
Discussion
This comprehensive systematic review and meta-analysis study shows that high intake of red meat as well as high intake of processed meat are associated with increased risk of breast cancer, colorectal cancer, colon cancer, rectal cancer, and lung cancer. In Addition, high red meat intake was associated with higher risk of endometrial cancer and hepatocellular carcinoma. Furthermore, significant positive associations were observed between high total red and processed meat consumption and risk of colorectal cancer, colon cancer, rectal cancer, lung cancer, and renal cell cancer. Based on results from one study, high total red and processed meat intake was associated with higher risk of esophageal squamous-cell carcinoma. High intake of total red and processed meat was suggestively associated with higher risk of pancreatic cancer.
Evidence on biological mechanisms explaining the association between high red meat and processed meat intake and cancer is mounting. Processing meat, which includes curing and smoking, produces carcinogens such as N-nitroso compounds (NOC) and polycyclic aromatic hydrocarbons (PAHs) [1, 172]. Cooking meat, especially at high temperature like grilling, barbecuing, or frying, also results in the production of known or suspected carcinogenic substances, such as heterocyclic aromatic amines (HAAs) and PAHs [173]. 2-Amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP), one of the most frequent HAAs formed during cooking meat, has been shown to be potently estrogenic, with the potential to increase cell proliferation, migration, and invasion [174, 175]. Additionally, PAHs may cause cancer through the DNA adduct formation and interference with apoptosis [176]. A positive dose–response association between meat doneness levels and risk of breast cancer was observed in the Iowa Women’s Health Study; consistently consuming very well-done hamburger, beef steak, and bacon was associated with a 4.62 times higher risk of breast cancer, compared with consuming rare or medium-done meat [177].
Heme iron from red and processed meat has been found to induce cytotoxicity and promote apoptosis and hyperplasia of epithelial cell, to induce lipid peroxidation, and free radical and DNA adduct formation in epithelial cells, as well as to catalyze N-nitroso compound formation, which can promote carcinogenesis [178]. High animal protein intake has been shown to be associated with a significantly higher risk of inflammatory bowel disease [179]; inflammatory bowel diseases are associated with elevated colorectal cancer risk [180]. In addition, animal-derived products contain antibiotics which may adversely affect the compositions and functions of the gut microbiota [181] and may affect local immunity as well as systemic immune responses [181]. The combined effects of these factors promote carcinogenesis [182]. Protein from animal sources is rich in dietary branched-chain and aromatic amino acids, which have been found to increase risk of insulin resistance [183, 184]. Insulin resistance has been linked to an increased risk of various cancers, such as breast cancer, prostate cancer, colorectal cancer, and endometrial cancer [185,186,187,188,189,190]. The high intake of red meat elevates insulin-like growth factor I (IGF-I) levels [191, 192]. IGF-I plays significant roles in cell growth, cancer development, and tumor progression [193]. Several studies have suggested that high circulating IGF-I has been associated with greater risk of colorectal, prostate, and breast malignancies [194,195,196,197]. The circulating level of IGF-I was lower among women with a plant-based (vegan) diet, compared with those with a meat-eating or lacto-ovo-vegetarian diet [198]. The increased release of bile acids, cholecystokinin, and prostaglandins from the high total fat content of meat also increases the risk of colorectal and pancreatic cancers [199,200,201]. Further, Persistent Organic Pollutants (POPs), which accumulate in animal fat cells [202, 203], may adversely affect endocrine pathways and increase risk of various chronic diseases, including cancer [203]. They are also present in lipoproteins and have been shown to be higher in participants with cancer compared to healthy individuals [204].
Higher red meat and processed meat consumption has been associated with increased oxidative stress and plasma concentrations of inflammatory biomarkers, including C-reactive protein [205, 206]. Nonhuman sialic acid N-glycolylneuraminic acid (Neu5Gc) and methionine, which are abundant in red meat, have been shown to promote chronic inflammation contributing to cancer development and tumor progression [207, 208]. Decreased proliferation of numerous cancer cell lines has been reported with reduced methionine consumption [209].
The most widely used growth promoting sex hormones in cattle raised in the US are estrogens, including estradiol-17β [210]. Estradiol-17β was reported as a carcinogen by the IARC [211], primarily due to its effects in increasing risk of breast cancer and endometrial cancer [212, 213]. Estrogens have also been shown in the development and progression of prostate cancer as well [214]. Some metabolites of estrogens may lead to the free radical formation, epigenetic, immunotoxic, and inflammatory changes, as well as, genotoxicity and hyperprolactinemia [214]. Therefore, consumption of meat from animals treated with hormones is likely to positively influence carcinogenesis [210].
Increased red meat or processed meat consumption has been shown to be associated with shorter telomere length [215, 216], which in turn result in chromosomal instability and promoting cancer risk [217, 218]. Indeed, studies have shown inverse associations between telomere length and risk of cancer incidence and mortality [219,220,221].
The multiple, but not exhaustive, cancer-promoting mechanisms cited above, appear to support our findings of increased risk of the various cancer types with increased red meat and processed meat consumption. However, current evidence does not support a significant positive association between high red meat and/or high processed meat intakes and risk of several cancers including ovarian cancer, prostate cancer, stomach cancer, esophageal adenocarcinoma, bladder cancer, leukemia, non-Hodgkin lymphoma, melanoma, and glioma. The lack of significant associations for some sites of cancer may be due to the small number of available studies or no biological relationship between red meat, processed meat, and those cancers. Further investigations are warranted to confirm the safety of red and processed meat intake for prevention of those cancers.
The strength of this meta-analysis should be noted. We analyzed comprehensively all the prospective studies which evaluated the risk of cancer in relation to red meat, processed meat, and total red and processed meat consumption. We only selected prospective studies for this meta-analysis, so recall and selection biases were minimized. Despite wide variations in the amount of red meat and processed meat intake as well as population characteristics across studies, for the majority of cancer sites, there was low to moderate heterogeneity across studies using the I2 statistic. This supports the external validity of pooling RRs of studies from different populations.
The limitations of this meta-analysis should be noted as well. In the interpretation of findings, publication bias should be considered even though we did not observe publication bias using visual inspection of a funnel plot or Begg and Mazumdar test for either red meat, processed meat, or total red and processed meat. Residual confounding is possible even though major cancer risk factors were controlled in most of the studies. The under- or over-reporting of the amount of red meat and processed meat items, as well as measurement errors, are possible when dietary intake was assessed using the food frequency questionnaire (FFQ). However, this bias may be observed in both cases and non-cases; therefore, it is more likely to attenuate findings. Moreover, we pooled RRs in the highest versus the lowest levels of intake, but levels of intake may be different across studies. We observed almost similar associations using dose–response analysis, though data from all of the studies were not included for dose–response analysis. Even though both intakes of red meat and processed meat were associated with higher risk of breast cancer, we did not observe a significant association with total red and processed meat intake; thus, further studies are needed. The significant positive association of total red and processed meat with esophageal squamous-cell carcinoma was based on only one study, and hence, more studies are needed to confirm this finding. In addition, most of the findings related to associations of red meat and processed meat were from studies in North America and Europe, generalizability to other regions with differences across social and physical environments, as well as sociodemographic factors such as race and ethnicity may be an important issue.
It is important to note that our findings provide robust evidence that high intake of red meat or processed meat increase risk of breast cancer, endometrial cancer, colon cancer, rectal cancer, esophageal squamous-cell carcinoma, lung cancer, renal cell cancer, and hepatocellular carcinoma in addition to colorectal cancer and pancreatic cancer previously reported by IARC [1]. Furthermore, this study supports dietary guidelines promoting limiting or avoiding red meat and processed meat intake to decrease cancer risk as proposed by the World Cancer Research Fund and American Institute for Cancer Research (WCRF/AICR) [222, 223]. Findings also support the American Cancer Society (ACS) dietary guidelines to limit or eliminate red meat and processed meat intake [224]. Evidence on the protective effects of high intake of wholefoods, plant-based diets, including legumes which are healthy substitutes for meat [24, 225,226,227,228], and the adverse effects of high red meat and processed meat intake, as well as supporting mechanisms for their actions on the risk of various types of cancer, continue to increase. Reduction of red meat and processed meat consumption has been shown to not only reduce cancer risk, but also other diseases such as diabetes and cardiovascular diseases [229,230,231,232]. Given the current available evidence, it seems prudent for practitioners and public health officials to encourage the adoption of a lifestyle that incorporates limiting or avoiding red meat and processed meat intake to decrease cancer risk burden in the US and worldwide.
Conclusion
Findings from this comprehensive systematic review and meta-analysis of prospective studies provide evidence that lower consumption of red meat and processed meat could be a key modifiable lifestyle factor in reducing the types of cancer identified in this paper.
References
IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Red meat and processed meat. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, No. 114. Lyon (FR): International Agency for Research on Cancer; 2018.
Wu K, Spiegelman D, Hou T, Albanes D, Allen NE, Berndt SI, et al. Associations between unprocessed red and processed meat, poultry, seafood and egg intake and the risk of prostate cancer: a pooled analysis of 15 prospective cohort studies. Int J Cancer. 2016;138(10):2368–82.
Lee JE, Spiegelman D, Hunter DJ, Albanes D, Bernstein L, van den Brandt PA, et al. Fat, protein, and meat consumption and renal cell cancer risk: a pooled analysis of 13 prospective studies. J Natl Cancer Inst. 2008;100(23):1695–706.
Missmer SA, Smith-Warner SA, Spiegelman D, Yaun SS, Adami HO, Beeson WL, et al. Meat and dairy food consumption and breast cancer: a pooled analysis of cohort studies. Int J Epidemiol. 2002;31(1):78–85.
Farvid MS, Stern MC, Norat T, Sasazuki S, Vineis P, Weijenberg MP, et al. Consumption of red and processed meat and breast cancer incidence: a systematic review and meta-analysis of prospective studies. Int J Cancer. 2018;143(11):2787–99.
Zhao Z, Feng Q, Yin Z, Shuang J, Bai B, Yu P, et al. Red and processed meat consumption and colorectal cancer risk: a systematic review and meta-analysis. Oncotarget. 2017;8(47):83306–14.
Zhao Z, Yin Z, Pu Z, Zhao Q. Association between consumption of red and processed meat and pancreatic cancer risk: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2017;15(4):486–93.
Bylsma LC, Alexander D. A review and meta-analysis of prospective studies of red and processed meat, meat cooking methods, heme iron, heterocyclic amines and prostate cancer. Nutr J. 2015;14:125.
Kim SR, Kim K, Lee SA, Kwon SO, Lee JK, Keum N, Park SM. Effect of red, processed, and white meat consumption on the risk of gastric cancer: an overall and dose-response meta-analysis. Nutrients. 2019;11(4):826.
Xue XJ, Gao Q, Qiao JH, Zhang J, Xu CP, Liu J. Red and processed meat consumption and the risk of lung cancer: a dose-response meta-analysis of 33 published studies. Int J Clin Exp Med. 2014;7(6):1542–53.
Luo J, Yang Y, Liu J, Lu K, Tang Z, Liu P, Liu L, Zhu Y. Systematic review with meta-analysis: meat consumption and the risk of hepatocellular carcinoma. Aliment Pharmacol Ther. 2014;39(9):913–22.
Zhang SJ, Wang QW, He JJ. Intake of red and processed meat and risk of renal cell carcinoma: a meta-analysis of observational studies. Oncotarget. 2017;8(44):77942–56.
Khodavandi A, Alizadeh F, Razis AFA. Association between dietary intake and risk of ovarian cancer: a systematic review and meta-analysis. Eur J Nutr. 2021;60(4):1707-36.
Ju W, Keum N, Lee DH, Kim YH, Kim SC, Ding EL, et al. Red meat intake and the risk of endometrial cancer: meta-analysis of observational studies. World J Meta-Anal. 2015;3(2):125–32.
Zhao Z, Wang F, Chen D, Zhang C. Red and processed meat consumption and esophageal cancer risk: a systematic review and meta-analysis. Clin Transl Oncol. 2020;22(4):532–45.
Crippa A, Larsson SC, Discacciati A, Wolk A, Orsini N. Red and processed meat consumption and risk of bladder cancer: a dose-response meta-analysis of epidemiological studies. Eur J Nutr. 2018;57(2):689–701.
Sergentanis TN, Ntanasis-Stathopoulos I, Tzanninis IG, Gavriatopoulou M, Sergentanis IN, Dimopoulos MA, Psaltopoulou T. Meat, fish, dairy products and risk of hematological malignancies in adults - a systematic review and meta-analysis of prospective studies. Leuk Lymphoma. 2019;60(8):1978–90.
Wei Y, Zou D, Cao D, Xie P. Association between processed meat and red meat consumption and risk for glioma: a meta-analysis from 14 articles. Nutrition. 2015;31(1):45–50.
Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000;283(15):2008–12.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–88.
Nguyen S, Li H, Yu D, Gao J, Gao Y, Tran H, et al. Adherence to dietary recommendations and colorectal cancer risk: results from two prospective cohort studies. Int J Epidemiol. 2020;49(1):270–80.
Dunneram Y, Greenwood DC, Cade JE. Diet and risk of breast, endometrial and ovarian cancer: UK Women’s Cohort Study. Br J Nutr. 2019;122(5):564–74.
Rice MS, Poole EM, Willett WC, Tworoger SS. Adult dietary fat intake and ovarian cancer risk. Int J Cancer. 2020;146(10):2756–72.
Farvid MS, Cho E, Chen WY, Eliassen AH, Willett WC. Dietary protein sources in early adulthood and breast cancer incidence: prospective cohort study. BMJ. 2014;348:g3437.
Luu HN, Neelakantan N, Geng TT, Wang R, Goh GB, Clemente JC, et al. Quality diet indexes and risk of hepatocellular carcinoma: Findings from the Singapore Chinese Health Study. Int J Cancer. 2021;148(9):2102–14.
Bravi F, Decarli A, Russo AG. Risk factors for breast cancer in a cohort of mammographic screening program: a nested case–control study within the FRiCaM study. Cancer Med. 2018;7(5):2145–52.
Wright ME, Bowen P, Virtamo J, Albanes D, Gann PH. Estimated phytanic acid intake and prostate cancer risk: a prospective cohort study. Int J Cancer. 2012;131(6):1396–406.
Layne TM, Graubard BI, Ma X, Mayne ST, Albanes D. Prostate cancer risk factors in black and white men in the NIH-AARP Diet and Health Study. Prostate Cancer Prostatic Dis. 2019;22(1):91–100.
Jones P, Cade JE, Evans CEL, Hancock N, Greenwood DC. The Mediterranean diet and risk of colorectal cancer in the UK Women's Cohort Study. Int J Epidemiol. 2017;46(6):1786–96.
Taunk P, Hecht E, Stolzenberg-Solomon R. Are meat and heme iron intake associated with pancreatic cancer? Results from the NIH-AARP diet and health cohort. Int J Cancer. 2016;138(9):2172–89.
Pang Y, Holmes MV, Guo Y, Yang L, Bian Z, Chen Y, et al. Smoking, alcohol, and diet in relation to risk of pancreatic cancer in China: a prospective study of 0.5 million people. Cancer Med. 2018;7(1):229–39.
Quist AJL, Inoue-Choi M, Weyer PJ, Anderson KE, Cantor KP, Krasner S, et al. Ingested nitrate and nitrite, disinfection by-products, and pancreatic cancer risk in postmenopausal women. Int J Cancer. 2018;142(2):251–61.
Pirie K, Peto R, Green J, Reeves GK, Beral V, Million Women Study C. Lung cancer in never smokers in the UK Million Women Study. Int J Cancer. 2016;139(2):347–54.
Xu X. Processed meat intake and bladder cancer risk in the prostate, lung, colorectal and ovarian (PLCO) cohort. Cancer Epidemiol Biomarkers Prev. 2019;28(12):1993–1997.
Luo X, Sui J, Yang W, Sun Q, Ma Y, Simon TG, et al. Type 2 Diabetes Prevention Diet and Hepatocellular Carcinoma Risk in US Men and Women. Am J Gastroenterol. 2019;114(12):1870–7.
Ma Y, Yang W, Li T, Liu Y, Simon TG, Sui J, et al. Meat intake and risk of hepatocellular carcinoma in two large US prospective cohorts of women and men. Int J Epidemiol. 2019;48(6):1863–71.
Yen H, Li WQ, Dhana A, Li T, Qureshi A, Cho E. Red meat and processed meat intake and risk for cutaneous melanoma in white women and men: Two prospective cohort studies. J Am Acad Dermatol. 2018;79(2):252–7.
Kuan AS, Green J, Kitahara CM, De Gonzalez AB, Key T, G KR, et al. Diet and risk of glioma: combined analysis of 3 large prospective studies in the UK and USA. Neuro Oncol. 2019;21(7):944–52.
Han MA, Zeraatkar D, Guyatt GH, Vernooij RWM, El Dib R, Zhang Y, et al. Reduction of red and processed meat intake and cancer mortality and incidence: A systematic review and meta-analysis of cohort studies. Ann Intern Med. 2019;171(10):711–20.
Sterne JA, Egger M. Funnel plots for detecting bias in meta-analysis: guidelines on choice of axis. J Clin Epidemiol. 2001;54(10):1046–55.
Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;50(4):1088–101.
Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–60.
Mills PK, Beeson WL, Phillips RL, Fraser GE. Dietary habits and breast cancer incidence among Seventh-day Adventists. Cancer. 1989;64(3):582–90.
Toniolo P, Riboli E, Shore RE, Pasternack BS. Consumption of meat, animal products, protein, and fat and risk of breast cancer: a prospective cohort study in New York. Epidemiology. 1994;5(4):391–7.
Key TJ, Sharp GB, Appleby PN, Beral V, Goodman MT, Soda M, Mabuchi K. Soya foods and breast cancer risk: a prospective study in Hiroshima and Nagasaki, Japan. Br J Cancer. 1999;81(7):1248–56.
Gilsing AM, Weijenberg MP, Goldbohm RA, Dagnelie PC, van den Brandt PA, Schouten LJ. Vegetarianism, low meat consumption and the risk of lung, postmenopausal breast and prostate cancer in a population-based cohort study. Eur J Clin Nutr. 2016;70(6):723–9.
Holmes MD, Colditz GA, Hunter DJ, Hankinson SE, Rosner B, Speizer FE, et al. Meat, fish and egg intake and risk of breast cancer. Int J Cancer. 2003;104(2):221–7.
van der Hel OL, Peeters PHM, Hein DW, Doll MA, Grobbee DE, Ocke M, et al. GSTM1 null genotype, red meat consumption and breast cancer risk (The Netherlands). Cancer Causes Control. 2004;15(3):295–303.
Kabat GC, Miller AB, Jain M, Rohan TE. Dietary iron and heme iron intake and risk of breast cancer: a prospective cohort study. Cancer Epidemiol Biomarkers Prev. 2007;16(6):1306–8.
Ferrucci LM, Cross AJ, Graubard BI, Brinton LA, McCarty CA, Ziegler RG, et al. Intake of meat, meat mutagens, and iron and the risk of breast cancer in the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial. Br J Cancer. 2009;101(1):178–84.
Larsson SC, Bergkvist L, Wolk A. Long-term meat intake and risk of breast cancer by oestrogen and progesterone receptor status in a cohort of Swedish women. Eur J Cancer. 2009;45(17):3042–6.
Pala V, Krogh V, Berrino F, Sieri S, Grioni S, Tjonneland A, et al. Meat, eggs, dairy products, and risk of breast cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort. Am J Clin Nutr. 2009;90(3):602–12.
Genkinger JM, Makambi KH, Palmer JR, Rosenberg L, Adams-Campbell LL. Consumption of dairy and meat in relation to breast cancer risk in the Black Women’s Health Study. Cancer Causes Control. 2013;24(4):675–84.
Hastert TA, Beresford SA, Patterson RE, Kristal AR, White E. Adherence to WCRF/AICR cancer prevention recommendations and risk of postmenopausal breast cancer. Cancer Epidemiol Biomarkers Prev. 2013;22(9):1498–508.
Pouchieu C, Deschasaux M, Hercberg S, Druesne-Pecollo N, Latino-Martel P, Touvier M. Prospective association between red and processed meat intakes and breast cancer risk: modulation by an antioxidant supplementation in the SU.VI.MAX randomized controlled trial. Int J Epidemiol. 2014;43(5):1583–92.
Nomura SJ, Dash C, Rosenberg L, Yu J, Palmer JR, Adams-Campbell LL. Adherence to diet, physical activity and body weight recommendations and breast cancer incidence in the Black Women's Health Study. Int J Cancer. 2016;139(12):2738–52.
Inoue-Choi M, Sinha R, Gierach GL, Ward MH. Red and processed meat, nitrite, and heme iron intakes and postmenopausal breast cancer risk in the NIH-AARP Diet and Health Study. Int J Cancer. 2016;138(7):1609–18.
van den Brandt PA, Schulpen M. Mediterranean diet adherence and risk of postmenopausal breast cancer: results of a cohort study and meta-analysis. Int J Cancer. 2017;140(10):2220–31.
Knuppel A, Papier K, Fensom GK, Appleby PN, Schmidt JA, Tong TYN, et al. Meat intake and cancer risk: Prospective analyses in UK Biobank. Int J Epidemiol. 2020;49(5):1540–52.
Diallo A, Deschasaux M, Latino-Martel P, Hercberg S, Galan P, Fassier P, et al. Red and processed meat intake and cancer risk: Results from the prospective NutriNet-Santé cohort study. Int J Cancer. 2018;142(2):230–7.
Lo JJ, Park YM, Sinha R, Sandler DP. Association between meat consumption and risk of breast cancer: Findings from the Sister Study. Int J Cancer. 2020;146(8):2156–65.
Beslay M, Srour B, Deschasaux M, Fouche E, Naud N, Bacquie V, et al. Anxiety is a potential effect modifier of the association between red and processed meat consumption and cancer risk: findings from the NutriNet-Santé cohort. Eur J Nutr. 2021;60(4):1887–96.
Barrios-Rodríguez R, Toledo E, Martinez-Gonzalez MA, Aguilera-Buenosvinos I, Romanos-Nanclares A, Jiménez-Moleón JJ. Adherence to the 2018 World Cancer Research Fund/American Institute for Cancer Research recommendations and breast cancer in the project. Nutrients. 2020;12(7):2076.
Wie GA, Cho YA, Kang HH, Ryu KA, Yoo MK, Kim YA, et al. Red meat consumption is associated with an increased overall cancer risk: a prospective cohort study in Korea. Br J Nutr. 2014;112(2):238–47.
Kang JH, Peng C, Rhee JJ, Farvid MS, Willett WC, Hu FB, et al. Prospective study of a diabetes risk reduction diet and the risk of breast cancer. Am J Clin Nutr. 2020;112(6):1492–503.
Marcondes LH, Franco OH, Ruiter R, Ikram MA, Mulder M, Stricker BH, et al. Animal foods and postmenopausal breast cancer risk: a prospective cohort study. Br J Nutr. 2019;122(5):583–91.
Heath AK, Muller DC, van den Brandt PA, Papadimitriou N, Critselis E, Gunter M, et al. Nutrient-wide association study of 92 foods and nutrients and breast cancer risk. Breast Cancer Res. 2020;22(1):5.
Larsson SC, Wolk A. No association of meat, fish, and egg consumption with ovarian cancer risk. Cancer Epidemiol Biomarkers Prev. 2005;14(4):1024–5.
Kiani F, Knutsen S, Singh P, Ursin G, Fraser G. Dietary risk factors for ovarian cancer: the Adventist Health Study (United States). Cancer Causes Control. 2006;17(2):137–46.
Cross AJ, Leitzmann MF, Gail MH, Hollenbeck AR, Schatzkin A, Sinha R. A prospective study of red and processed meat intake in relation to cancer risk. PLoS Med. 2007;4(12):e325.
Merritt MA, Tzoulaki I, van den Brandt PA, Schouten LJ, Tsilidis KK, Weiderpass E, et al. Nutrient-wide association study of 57 foods/nutrients and epithelial ovarian cancer in the European Prospective Investigation into Cancer and Nutrition study and the Netherlands Cohort Study. Am J Clin Nutr. 2016;103(1):161–7.
Gilsing AM, Weijenberg MP, Goldbohm RA, van den Brandt PA, Schouten LJ. Consumption of dietary fat and meat and risk of ovarian cancer in the Netherlands Cohort Study. Am J Clin Nutr. 2011;93(1):118–26.
Kabat GC, Miller AB, Jain M, Rohan TE. Dietary iron and haem iron intake and risk of endometrial cancer: a prospective cohort study. Br J Cancer. 2008;98(1):194–8.
van Lonkhuijzen L, Kirsh VA, Kreiger N, Rohan TE. Endometrial cancer and meat consumption: a case-cohort study. Eur J Cancer Prev. 2011;20(4):334–9.
Genkinger JM, Friberg E, Goldbohm RA, Wolk A. Long-term dietary heme iron and red meat intake in relation to endometrial cancer risk. Am J Clin Nutr. 2012;96(4):848–54.
Arem H, Gunter MJ, Cross AJ, Hollenbeck AR, Sinha R. A prospective investigation of fish, meat and cooking-related carcinogens with endometrial cancer incidence. Br J Cancer. 2013;109(3):756–60.
Merritt MA, Tzoulaki I, Tworoger SS, De Vivo I, Hankinson SE, Fernandes J, et al. Investigation of dietary factors and endometrial cancer risk using a nutrient-wide association study approach in the EPIC and Nurses’ Health Study (NHS) and NHSII. Cancer Epidemiol Biomarkers Prev. 2015;24(2):466–71.
Yamamoto A, Harris HR, Vitonis AF, Chavarro JE, Missmer SA. A prospective cohort study of meat and fish consumption and endometriosis risk. Am J Obstet Gynecol. 2018;219(2):178 e1–e10.
Severson RK, Nomura A, Grove JS, Stemmermann GN. A prospective study of demographics, diet, and prostate cancer among men of Japanese. Cancer Res. 1989;49(7):1857–60.
Mills PK, Beeson WL, Phillips RL, Fraser GE. Cohort study of diet, lifestyle, and prostate cancer in Adventist men. Cancer. 1989;64(3):598–604.
Le Marchand L, Kolonel LN, Wilkens LR, Myers BC, Hirohata T. Animal fat consumption and prostate cancer: a prospective study in Hawaii. Epidemiology. 1994;5(3):276–82.
Veierød MB, Laake P, Thelle DS. Dietary fat intake and risk of prostate cancer: a prospective study of 25,708 Norwegian men. Int J Cancer. 1997;73(5):634–8.
Michaud DS, Augustsson K, Rimm EB, Stampfer MJ, Willett WC, Giovannucci E. A prospective study on intake of animal products and risk of prostate cancer. Cancer Causes Control. 2001;12(6):557–67.
Cross AJ, Peters U, Kirsh VA, Andriole GL, Reding D, Hayes RB, et al. A prospective study of meat and meat mutagens and prostate cancer risk. Cancer Res. 2005;65(24):11779–84.
Rodriguez C, McCullough ML, Mondul AM, Jacobs EJ, Chao A, Patel AV, et al. Meat consumption among Black and White men and risk of prostate cancer in the Cancer Prevention Study II Nutrition Cohort. Cancer Epidemiol Biomarkers Prev. 2006;15(2):211–6.
Neuhouser ML, Barnett MJ, Kristal AR, Ambrosone CB, King I, Thornquist M, Goodman G. (n-6) PUFA increase and dairy foods decrease prostate cancer risk in heavy smokers. J Nutr. 2007;137(7):1821–7.
Park SY, Murphy SP, Wilkens LR, Henderson BE, Kolonel LN. Fat and meat intake and prostate cancer risk: the multiethnic cohort study. Int J Cancer. 2007;121(6):1339–45.
Rohrmann S, Platz EA, Kavanaugh CJ, Thuita L, Hoffman SC, Helzlsouer KJ. Meat and dairy consumption and subsequent risk of prostate cancer in a US cohort study. Cancer Causes Control. 2007;18(1):41–50.
Allen NE, Key TJ, Appleby PN, Travis RC, Roddam AW, Tjønneland A, et al. Animal foods, protein, calcium and prostate cancer risk: the European Prospective Investigation into Cancer and Nutrition. Br J Cancer. 2008;98(9):1574–81.
Koutros S, Cross AJ, Sandler DP, Hoppin JA, Ma X, Zheng T, et al. Meat and meat mutagens and risk of prostate cancer in the Agricultural Health Study. Cancer Epidemiol Biomarkers Prev. 2008;17(1):80–7.
Sinha R, Park Y, Graubard BI, Leitzmann MF, Hollenbeck A, Schatzkin A, et al. Meat and meat-related compounds and risk of prostate cancer in a large prospective cohort study in the United States. Am J Epidemiol. 2009;170(9):1165–77.
Agalliu I, Kirsh VA, Kreiger N, Soskolne CL, Rohan TE. Oxidative balance score and risk of prostate cancer: results from a case-cohort study. Cancer Epidemiol. 2011;35(4):353–61.
Bostick RM, Potter JD, Kushi LH, Sellers TA, Steinmetz KA, McKenzie DR, et al. Sugar, meat, and fat intake, and non-dietary risk factors for colon cancer incidence in Iowa women (United States). Cancer Causes Control. 1994;5(1):38–52.
Kato I, Akhmedkhanov A, Koenig K, Toniolo PG, Shore RE, Riboli E. Prospective study of diet and female colorectal cancer: the New York University Women’s Health Study. Nutr Cancer. 1997;28(3):276–81.
Singh PN, Fraser GE. Dietary risk factors for colon cancer in a low-risk population. Am J Epidemiol. 1998;148(8):761–74.
Knekt P, Jarvinen R, Dich J, Hakulinen T. Risk of colorectal and other gastro-intestinal cancers after exposure to nitrate, nitrite and N-nitroso compounds: a follow-up study. Int J Cancer. 1999;80(6):852–6.
Pietinen P, Malila N, Virtanen M, Hartman TJ, Tangrea JA, Albanes D, et al. Diet and risk of colorectal cancer in a cohort of Finnish men. Cancer Causes Control. 1999;10(5):387–96.
Järvinen R, Knekt P, Hakulinen T, Rissanen H, Heliovaara M. Dietary fat, cholesterol and colorectal cancer in a prospective study. Br J Cancer. 2001;85(3):357–61.
Flood A, Velie EM, Sinha R, Chaterjee N, Lacey JV Jr, Schairer C, et al. Meat, fat, and their subtypes as risk factors for colorectal cancer in a prospective cohort of women. Am J Epidemiol. 2003;158(1):59–68.
English DR, MacInnis RJ, Hodge AM, Hopper JL, Haydon AM, Giles GG. Red meat, chicken, and fish consumption and risk of colorectal cancer. Cancer Epidemiol Biomarkers Prev. 2004;13(9):1509–14.
Lin J, Zhang SM, Cook NR, Lee IM, Buring JE. Dietary fat and fatty acids and risk of colorectal cancer in women. Am J Epidemiol. 2004;160(10):1011–22.
Wei EK, Giovannucci E, Wu K, Rosner B, Fuchs CS, Willett WC, et al. Comparison of risk factors for colon and rectal cancer. Int J Cancer. 2004;108(3):433–42.
Chao A, Thun MJ, Connell CJ, McCullough ML, Jacobs EJ, Flanders WD, et al. Meat consumption and risk of colorectal cancer. JAMA. 2005;293(2):172–82.
Larsson SC, Rafter J, Holmberg L, Bergkvist L, Wolk A. Red meat consumption and risk of cancers of the proximal colon, distal colon and rectum: the Swedish Mammography Cohort. Int J Cancer. 2005;113(5):829–34.
Norat T, Bingham S, Ferrari P, Slimani N, Jenab M, Mazuir M, et al. Meat, fish, and colorectal cancer risk: the European Prospective Investigation into cancer and nutrition. J Natl Cancer Inst. 2005;97(12):906–16.
Tiemersma EW, Kampman E, Bueno de Mesquita HB, Bunschoten A, van Schothorst EM, Kok FJ, et al. Meat consumption, cigarette smoking, and genetic susceptibility in the etiology of colorectal cancer: results from a Dutch prospective study. Cancer Causes Control. 2002;13(4):383–93.
Berndt SI, Platz EA, Fallin MD, Thuita LW, Hoffman SC, Helzlsouer KJ. Genetic variation in the nucleotide excision repair pathway and colorectal cancer risk. Cancer Epidemiol Biomarkers Prev. 2006;15(11):2263–9.
Kabat GC, Miller AB, Jain M, Rohan TE. A cohort study of dietary iron and heme iron intake and risk of colorectal cancer in women. Br J Cancer. 2007;97(1):118–22.
Butler LM, Wang R, Koh WP, Yu MC. Prospective study of dietary patterns and colorectal cancer among Singapore Chinese. Br J Cancer. 2008;99(9):1511–16.
Lee SA, Shu XO, Yang G, Li H, Gao YT, Zheng W. Animal origin foods and colorectal cancer risk: a report from the Shanghai Women’s Health Study. Nutr Cancer. 2009;61(2):194–205.
Etemadi A, Abnet CC, Graubard BI, Beane-Freeman L, Freedman ND, Liao L, et al. Anatomical subsite can modify the association between meat and meat compounds and risk of colorectal adenocarcinoma: Findings from three large US cohorts. Int J Cancer. 2018;143(9):2261–70.
Ollberding NJ, Wilkens LR, Henderson BE, Kolonel LN, Le Marchand L. Meat consumption, heterocyclic amines and colorectal cancer risk: the Multiethnic Cohort Study. Int J Cancer. 2012;131(7):E1125–33.
Gilsing AM, Schouten LJ, Goldbohm RA, Dagnelie PC, van den Brandt PA, Weijenberg MP. Vegetarianism, low meat consumption and the risk of colorectal cancer in a population based cohort study. Sci Rep. 2015;5:13484.
Mehta SS, Arroyave WD, Lunn RM, Park YM, Boyd WA, Sandler DP. A prospective analysis of red and processed meat consumption and risk of colorectal cancer in women. Cancer Epidemiol Biomarkers Prev. 2020;29(1):141–150.
Barrubés L, Babio N, Hernandez-Alonso P, Toledo E, Ramirez Sabio JB, Estruch R, et al. Association between the 2018 WCRF/AICR and the low-risk lifestyle scores with colorectal cancer risk in the PREDIMED study. J Clin Med. 2020;9(4):1215.
O'Sullivan DE, Metcalfe A, Hillier TWR, King WD, Lee S, Pader J, et al. Combinations of modifiable lifestyle behaviours in relation to colorectal cancer risk in Alberta's Tomorrow Project. Scientific reports. 2020;10(1):20561.
Mejborn H, Moller SP, Thygesen LC, Biltoft-Jensen A. Dietary intake of red meat, processed meat, and poultry and risk of colorectal cancer and all-cause mortality in the context of dietary guideline compliance. Nutrients. 2020;13(1):32.
Sellers TA, Bazyk AE, Bostick RM, Kushi LH, Olson JE, Anderson KE, et al. Diet and risk of colon cancer in a large prospective study of older women: an analysis stratified on family history (Iowa, United States). Cancer Causes Control. 1998;9(4):357–67.
Islam Z, Akter S, Kashino I, Mizoue T, Sawada N, Mori N, et al. Meat subtypes and colorectal cancer risk: A pooled analysis of 6 cohort studies in Japan. Cancer Sci. 2019;110(11):3603–14.
Takachi R, Tsubono Y, Baba K, Inoue M, Sasazuki S, Iwasaki M, et al. Red meat intake may increase the risk of colon cancer in Japanese, a population with relatively low red meat consumption. Asia Pac J Clin Nutr. 2011;20(4):603–12
Bernstein AM, Song M, Zhang X, Pan A, Wang M, Fuchs CS, et al. Processed and unprocessed red meat and risk of colorectal cancer: analysis by tumor location and modification by time. PLoS One. 2015;10(8):e0135959.
Nomura SJ, Dash C, Rosenberg L, Yu J, Palmer JR, Adams-Campbell LL. Is adherence to diet, physical activity, and body weight cancer prevention recommendations associated with colorectal cancer incidence in African American women? Cancer Causes Control. 2016;27(7):869–79.
Hastert TA, White E. Association between meeting the WCRF/AICR cancer prevention recommendations and colorectal cancer incidence: results from the VITAL cohort. Cancer Causes Control. 2016;27(11):1347–59.
Wada K, Oba S, Tsuji M, Tamura T, Konishi K, Goto Y, et al. Meat consumption and colorectal cancer risk in Japan: The Takayama study. Cancer Sci. 2017;108(5):1065–70.
Nomura A, Grove JS, Stemmermann GN, Severson RK. A prospective study of stomach cancer and its relation to diet, cigarettes, and alcohol consumption. Cancer Res. 1990;50(3):627–31.
Galanis DJ, Kolonel LN, Lee J, Nomura A. Intakes of selected foods and beverages and the incidence of gastric cancer among the Japanese residents of Hawaii: a prospective study. Int J Epidemiol. 1998;27(2):173–80.
Sauvaget C, Lagarde F, Nagano J, Soda M, Koyama K, Kodama K. Lifestyle factors, radiation and gastric cancer in atomic-bomb survivors (Japan). Cancer Causes Control. 2005;16(7):773–80.
Gonzalez CA, Jakszyn P, Pera G, Agudo A, Bingham S, Palli D, et al. Meat intake and risk of stomach and esophageal adenocarcinoma within the European Prospective Investigation Into Cancer and Nutrition (EPIC). J Natl Cancer Inst. 2006;98(5):345–54.
Larsson SC, Bergkvist L, Wolk A. Processed meat consumption, dietary nitrosamines and stomach cancer risk in a cohort of Swedish women. Int J Cancer. 2006;119(4):915–9.
Keszei AP, Schouten LJ, Goldbohm RA, van den Brandt PA. Red and processed meat consumption and the risk of esophageal and gastric cancer subtypes in The Netherlands Cohort Study. Ann Oncol. 2012;23(9):2319–26.
Cross AJ, Freedman ND, Ren J, Ward MH, Hollenbeck AR, Schatzkin A, et al. Meat consumption and risk of esophageal and gastric cancer in a large prospective study. Am J Gastroenterol. 2011;106(3):432–42.
Jakszyn P, Lujan-Barroso L, Agudo A, Bueno-de-Mesquita HB, Molina E, Sanchez MJ, et al. Meat and heme iron intake and esophageal adenocarcinoma in the European Prospective Investigation into Cancer and Nutrition study. Int J Cancer. 2013;133(11):2744–50.
Stolzenberg-Solomon RZ, Pietinen P, Taylor PR, Virtamo J, Albanes D. Prospective study of diet and pancreatic cancer in male smokers. Am J Epidemiol. 2002;155(9):783–92.
Isaksson B, Jonsson F, Pedersen NL, Larsson J, Feychting M, Permert J. Lifestyle factors and pancreatic cancer risk: a cohort study from the Swedish Twin Registry. Int J Cancer. 2002;98(2):480–2.
Michaud DS, Giovannucci E, Willett WC, Colditz GA, Fuchs CS. Dietary meat, dairy products, fat, and cholesterol and pancreatic cancer risk in a prospective study. Am J Epidemiol. 2003;157(12):1115–25.
Nothlings U, Wilkens LR, Murphy SP, Hankin JH, Henderson BE, Kolonel LN. Meat and fat intake as risk factors for pancreatic cancer: the multiethnic cohort study. J Natl Cancer Inst. 2005;97(19):1458–65.
Larsson SC, Hakanson N, Permert J, Wolk A. Meat, fish, poultry and egg consumption in relation to risk of pancreatic cancer: a prospective study. Int J Cancer. 2006;118(11):2866–70.
Heinen MM, Verhage BAJ, Goldbohm RA, van den Brandt PA. Meat and fat intake and pancreatic cancer risk in the Netherlands Cohort Study. Int J Cancer. 2009;125(5):1118–26.
Rohrmann S, Linseisen J, Nothlings U, Overvad K, Egeberg R, Tjonneland A, et al. Meat and fish consumption and risk of pancreatic cancer: results from the European Prospective Investigation into Cancer and Nutrition. Int J Cancer. 2013;132(3):617–24.
Ghorbani Z, Pourshams A, Malekshah AF, Sharatkhah M, Poustchi H, Hekmatdoost A. Major dietary protein sources in relation to pancreatic cancer: a large prospective study. Arch Iran Med. 2016;19(4):248–56.
McCullough ML, Jacobs EJ, Shah R, Campbell PT, Wang Y, Hartman TJ, et al. Meat consumption and pancreatic cancer risk among men and women in the Cancer Prevention Study-II Nutrition Cohort. Cancer Causes Control. 2018;29(1):125–33.
Petrick JL, Castro-Webb N, Gerlovin H, Bethea TN, Li S, Ruiz-Narvaez EA, et al. A prospective analysis of intake of red and processed meat in relation to pancreatic cancer among african american women. Cancer Epidemiol Biomarkers Prev. 2020;29(9):1775–83.
Zhang ZQ, Li QJ, Hao FB, Wu YQ, Liu S, Zhong GC. Adherence to the 2018 World Cancer Research Fund/American Institute for Cancer Research cancer prevention recommendations and pancreatic cancer incidence and mortality: a prospective cohort study. Cancer Med. 2020;9(18):6843–53.
Huang BZ, Wang S, Bogumil D, Wilkens LR, Wu L, Blot WJ, et al. Red meat consumption, cooking mutagens, NAT1/2 genotypes and pancreatic cancer risk in two ethnically diverse prospective cohorts. Int J Cancer. 2021. https://doi.org/10.1002/ijc.33598.
Tasevska N, Cross AJ, Dodd KW, Ziegler RG, Caporaso NE, Sinha R. No effect of meat, meat cooking preferences, meat mutagens or heme iron on lung cancer risk in the prostate, lung, colorectal and ovarian cancer screening trial. Int J Cancer. 2011;128(2):402–11.
Linseisen J, Rohrmann S, Bueno-de-Mesquita B, Buchner FL, Boshuizen HC, Agudo A, et al. Consumption of meat and fish and risk of lung cancer: results from the European Prospective Investigation into Cancer and Nutrition. Cancer Causes Control. 2011;22(6):909–18.
Cai H, Sobue T, Kitamura T, Ishihara J, Sawada N, Iwasaki M, et al. Association between meat and saturated fatty acid intake and lung cancer risk: The Japan Public Health Center-based prospective study. Int J Cancer. 2020;147(11):3019–28.
Gnagnarella P, Maisonneuve P, Bellomi M, Rampinelli C, Bertolotti R, Spaggiari L, et al. Red meat, Mediterranean diet and lung cancer risk among heavy smokers in the COSMOS screening study. Ann Oncol. 2013;24(10):2606–11.
Chyou PH, Nomura AM, Stemmermann GN. A prospective study of diet, smoking, and lower urinary tract cancer. Ann Epidemiol. 1993;3(3):211–6.
Nagano J, Kono S, Preston DL, Moriwaki H, Sharp GB, Koyama K, et al. Bladder-cancer incidence in relation to vegetable and fruit consumption: a prospective study of atomic-bomb survivors. Int J Cancer. 2000;86(1):132–8.
Michaud DS, Holick CN, Giovannucci E, Stampfer MJ. Meat intake and bladder cancer risk in 2 prospective cohort studies. Am J Clin Nutr. 2006;84(5):1177–83.
Larsson SC, Johansson JE, Andersson SO, Wolk A. Meat intake and bladder cancer risk in a Swedish prospective cohort. Cancer Causes Control. 2009;20(1):35–40.
Ferrucci LM, Sinha R, Ward MH, Graubard BI, Hollenbeck AR, Kilfoy BA, et al. Meat and components of meat and the risk of bladder cancer in the NIH-AARP Diet and Health Study. Cancer. 2010;116(18):4345–53.
Jakszyn P, Gonzalez CA, Lujan-Barroso L, Ros MM, Bueno-de-Mesquita HB, Roswall N, et al. Red meat, dietary nitrosamines, and heme iron and risk of bladder cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC). Cancer Epidemiol Biomarkers Prev. 2011;20(3):555–9.
Fraser GE, Phillips R, Beeson WL. Hypertension, antihypertensive medication and risk of renal carcinoma in California Seventh-Day Adventists. Int J Epidemiol. 1990;19(4):832–8.
Daniel CR, Cross AJ, Graubard BI, Park Y, Ward MH, Rothman N, et al. Large prospective investigation of meat intake, related mutagens, and risk of renal cell carcinoma. Am J Clin Nutr. 2012;95(1):155–62.
Rohrmann S, Linseisen J, Overvad K, Wurtz AML, Roswall N, Tjonneland A, et al. Meat and fish consumption and the risk of renal cell carcinoma in the European prospective investigation into cancer and nutrition. Int J Cancer. 2015;136(5):E423–31.
Freedman ND, Cross AJ, McGlynn KA, Abnet CC, Park Y, Hollenbeck AR, et al. Association of meat and fat intake with liver disease and hepatocellular carcinoma in the NIH-AARP cohort. J Natl Cancer Inst. 2010;102(17):1354–65.
Li WQ, Park Y, McGlynn KA, Hollenbeck AR, Taylor PR, Goldstein AM, et al. Index-based dietary patterns and risk of incident hepatocellular carcinoma and mortality from chronic liver disease in a prospective study. Hepatology. 2014;60(2):588–97.
Fedirko V, Trichopolou A, Bamia C, Duarte-Salles T, Trepo E, Aleksandrova K, et al. Consumption of fish and meats and risk of hepatocellular carcinoma: the European Prospective Investigation into Cancer and Nutrition (EPIC). Ann Oncol. 2013;24(8):2166–73.
Ross JA, Kasum CM, Davies SM, Jacobs DR, Folsom AR, Potter JD. Diet and risk of leukemia in the Iowa Women’s Health Study. Cancer Epidemiol Biomarkers Prev. 2002;11(8):777–81.
Saberi Hosnijeh F, Peeters P, Romieu I, Kelly R, Riboli E, Olsen A, et al. Dietary intakes and risk of lymphoid and myeloid leukemia in the European Prospective Investigation into Cancer and Nutrition (EPIC). Nutr Cancer. 2014;66(1):14–28.
Chiu BC, Cerhan JR, Folsom AR, Sellers TA, Kushi LH, Wallace RB, et al. Diet and risk of non-Hodgkin lymphoma in older women. JAMA. 1996;275(17):1315–21.
Zhang S, Hunter DJ, Rosner BA, Colditz GA, Fuchs CS, Speizer FE, et al. Dietary fat and protein in relation to risk of non-Hodgkin’s lymphoma among women. J Natl Cancer Inst. 1999;91(20):1751–8.
Rohrmann S, Linseisen J, Jakobsen MU, Overvad K, Raaschou-Nielsen O, Tjonneland A, et al. Consumption of meat and dairy and lymphoma risk in the European Prospective Investigation into Cancer and Nutrition. Int J Cancer. 2011;128(3):623–34.
Daniel CR, Sinha R, Park Y, Graubard BI, Hollenbeck AR, Morton LM, et al. Meat intake is not associated with risk of non-Hodgkin lymphoma in a large prospective cohort of U.S. men and women. J Nutr. 2012;142(6):1074–80.
Mills PK, Preston-Martin S, Annegers JF, Beeson WL, Phillips RL, Fraser GE. Risk factors for tumors of the brain and cranial meninges in Seventh-Day Adventists. Neuroepidemiology. 1989;8(5):266–75.
Rollison DE, Helzlsouer KJ. Processed meat consumption and adult gliomas in a Maryland cohort. Cancer Causes Control. 2004;15(1):99–100.
Michaud DS, Holick CN, Batchelor TT, Giovannucci E, Hunter DJ. Prospective study of meat intake and dietary nitrates, nitrites, and nitrosamines and risk of adult glioma. Am J Clin Nutr. 2009;90(3):570–7.
Ward HA, Gayle A, Jakszyn P, Merritt M, Melin B, Freisling H, et al. Meat and haem iron intake in relation to glioma in the European Prospective Investigation into Cancer and Nutrition study. Eur J Cancer Prev. 2018;27(4):379–83.
Dubrow R, Darefsky AS, Park Y, Mayne ST, Moore SC, Kilfoy B, et al. Dietary components related to N-nitroso compound formation: a prospective study of adult glioma. Cancer Epidemiol Biomarkers Prev. 2010;19(7):1709–22.
Turesky RJ. Mechanistic evidence for red meat and processed meat intake and cancer risk: a follow-up on the International Agency for Research on Cancer evaluation of 2015. Chimia (Aarau). 2018;72(10):718-24.
Cross AJ, Sinha R. Meat-related mutagens/carcinogens in the etiology of colorectal cancer. Environ Mol Mutagen. 2004;44(1):44–55.
Lauber SN, Ali S, Gooderham NJ. The cooked food derived carcinogen 2-amino-1-methyl-6-phenylimidazo[4,5-b] pyridine is a potent oestrogen: a mechanistic basis for its tissue-specific carcinogenicity. Carcinogenesis. 2004;25(12):2509–17.
Lauber SN, Gooderham NJ. The cooked meat-derived mammary carcinogen 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine promotes invasive behaviour of breast cancer cells. Toxicology. 2011;279(1–3):139–45.
Pratt MM, John K, MacLean AB, Afework S, Phillips DH, Poirier MC. Polycyclic aromatic hydrocarbon (PAH) exposure and DNA adduct semi-quantitation in archived human tissues. Int J Environ Res Public Health. 2011;8(7):2675–91.
Zheng W, Gustafson DR, Sinha R, Cerhan JR, Moore D, Hong CP, et al. Well-done meat intake and the risk of breast cancer. J Natl Cancer Inst. 1998;90(22):1724–9.
Gamage SMK, Dissabandara L, Lam AK, Gopalan V. The role of heme iron molecules derived from red and processed meat in the pathogenesis of colorectal carcinoma. Crit Rev Oncol Hematol. 2018;126:121–8.
Jantchou P, Morois S, Clavel-Chapelon F, Boutron-Ruault MC, Carbonnel F. Animal protein intake and risk of inflammatory bowel disease: The E3N prospective study. Am J Gastroenterol. 2010;105(10):2195–201.
Stidham RW, Higgins PDR. Colorectal Cancer in Inflammatory Bowel Disease. Clin Colon Rectal Surg. 2018;31(3):168–78.
Becattini S, Taur Y, Pamer EG. Antibiotic-induced changes in the intestinal microbiota and disease. Trends Mol Med. 2016;22(6):458–78.
Ong HS, Yim HCH. Microbial factors in inflammatory diseases and cancers. Adv Exp Med Biol. 2017;1024:153-174.
Chen Z, Franco OH, Lamballais S, Ikram MA, Schoufour JD, Muka T, et al. Associations of specific dietary protein with longitudinal insulin resistance, prediabetes and type 2 diabetes: The Rotterdam Study. Clin Nutr (Edinburgh, Scotland). 2020;39(1):242–9.
Lynch CJ, Adams SH. Branched-chain amino acids in metabolic signalling and insulin resistance. Nat Rev Endocrinol. 2014;10(12):723–36.
Bruning PF, Bonfrer JM, van Noord PA, Hart AA, de Jong-Bakker M, Nooijen WJ. Insulin resistance and breast-cancer risk. Int J Cancer 1992;52(4):511–16.
Lifshitz K, Ber Y, Margel D. Role of metabolic syndrome in prostate cancer development. Eur Urol Focus. 2021;S2405-4569(21)00128-0.
Cirillo F, Catellani C, Sartori C, Lazzeroni P, Amarri S, Street ME. Obesity, insulin resistance, and colorectal cancer: Could miRNA dysregulation play a role? Int J Mol Sci. 2019;20(12):2922.
Arcidiacono B, Iiritano S, Nocera A, Possidente K, Nevolo MT, Ventura V, et al. Insulin resistance and cancer risk: an overview of the pathogenetic mechanisms. Exp Diabetes Res. 2012;2012:789174.
Fernandez CJ, George AS, Subrahmanyan NA, Pappachan JM. Epidemiological link between obesity, type 2 diabetes mellitus and cancer. World J Methodol. 2021;11(3):23–45.
Mu N, Zhu Y, Wang Y, Zhang H, Xue F. Insulin resistance: a significant risk factor of endometrial cancer. Gynecol Oncol. 2012;125(3):751–7.
Kaklamani VG, Linos A, Kaklamani E, Markaki I, Koumantaki Y, Mantzoros CS. Dietary fat and carbohydrates are independently associated with circulating insulin-like growth factor 1 and insulin-like growth factor-binding protein 3 concentrations in healthy adults. J Clin Oncol. 1999;17(10):3291–8.
Larsson SC, Wolk K, Brismar K, Wolk A. Association of diet with serum insulin-like growth factor I in middle-aged and elderly men. Am J Clin Nutr. 2005;81(5):1163-7.
Weroha SJ, Haluska P. The insulin-like growth factor system in cancer. Endocrinol Metab Clin North Am. 2012;41(2):335–50.
Davies M, Gupta S, Goldspink G, Winslet M. The insulin-like growth factor system and colorectal cancer: clinical and experimental evidence. Int J Colorectal Dis. 2006;21(3):201–8.
Renehan AG, Zwahlen M, Minder C, O’Dwyer ST, Shalet SM, Egger M. Insulin-like growth factor (IGF)-I, IGF binding protein-3, and cancer risk: systematic review and meta-regression analysis. Lancet (London, England). 2004;363(9418):1346–53.
Shi R, Yu H, McLarty J, Glass J. IGF-I and breast cancer: a meta-analysis. Int J Cancer. 2004;111(3):418–23.
Rowlands MA, Gunnell D, Harris R, Vatten LJ, Holly JM, Martin RM. Circulating insulin-like growth factor peptides and prostate cancer risk: a systematic review and meta-analysis. Int J Cancer. 2009;124(10):2416–29.
Allen NE, Appleby PN, Davey GK, Kaaks R, Rinaldi S, Key TJ. The associations of diet with serum insulin-like growth factor I and its main binding proteins in 292 women meat-eaters, vegetarians, and vegans. Cancer Epidemiol Biomarkers Prev. 2002;11(11):1441–8.
Ocvirk S, Wilson AS, Appolonia CN, Thomas TK, O'Keefe SJD. Fiber, fat, and colorectal cancer: New insight into modifiable dietary risk factors. Curr Gastroenterol Rep. 2019;21(11):62.
Reddy BS. Nutritional factors and colon cancer. Crit Rev Food Sci Nutr. 1995;35(3):175–90.
Appel MJ, Meijers M, Van Garderen-Hoetmer A, Lamers CB, Rovati LC, Sprij-Mooij D, et al. Role of cholecystokinin in dietary fat-promoted azaserine-induced pancreatic carcinogenesis in rats. Br J Cancer. 1992;66(1):46–50.
Hernández AR, Boada LD, Mendoza Z, Ruiz-Suárez N, Valerón PF, Camacho M, et al. Consumption of organic meat does not diminish the carcinogenic potential associated with the intake of persistent organic pollutants (POPs). Environ Sci Pollut Res Int. 2017;24(5):4261–73.
Guo W, Pan B, Sakkiah S, Yavas G, Ge W, Zou W, et al. Persistent organic pollutants in food: contamination sources, health effects and detection methods. Int J Environ Res Public Health. 2019;16(22):4361.
Ljunggren SA, Helmfrid I, Salihovic S, van Bavel B, Wingren G, Lindahl M, et al. Persistent organic pollutants distribution in lipoprotein fractions in relation to cardiovascular disease and cancer. Environ Int. 2014;65:93–9.
Azadbakht L, Esmaillzadeh A. Red meat intake is associated with metabolic syndrome and the plasma C-reactive protein concentration in women. J Nutr. 2009;139(2):335–9.
Ley SH, Sun Q, Willett WC, Eliassen AH, Wu K, Pan A, et al. Associations between red meat intake and biomarkers of inflammation and glucose metabolism in women. Am J Clin Nutr. 2014;99(2):352–60.
Samraj AN, Pearce OM, Laubli H, Crittenden AN, Bergfeld AK, Banda K, et al. A red meat-derived glycan promotes inflammation and cancer progression. Proc Natl Acad Sci USA. 2015;112(2):542–7.
Chaturvedi P, Kamat PK, Kalani A, Familtseva A, Tyagi SC. High methionine diet poses cardiac threat: a molecular insight. J Cell Physiol. 2016;231(7):1554–61.
Cellarier E, Durando X, Vasson MP, Farges MC, Demiden A, Maurizis JC, et al. Methionine dependency and cancer treatment. Cancer Treat Rev. 2003;29(6):489–99.
Andersson AM, Skakkebaek NE. Exposure to exogenous estrogens in food: possible impact on human development and health. Eur J Endocrinol. 1999;140(6):477–85.
Schneider HPG, Mueck AO, Kuhl H. IARC monographs program on carcinogenicity of combined hormonal contraceptives and menopausal therapy. Climacteric. 2005;8(4):311–6.
Feigelson HS, Henderson BE. Estrogens and breast cancer. Carcinogenesis. 1996;17(11):2279–84.
Greenwald P, Caputo TA, Wolfgang PE. Endometrial cancer after menopausal use of estrogens. Obstet Gynecol. 1977;50(2):239–43.
Nelles JL, Hu WY, Prins GS. Estrogen action and prostate cancer. Expert Rev Endocrinol Metab. 2011;6(3):437–51.
O’Callaghan NJ, Toden S, Bird AR, Topping DL, Fenech M, Conlon MA. Colonocyte telomere shortening is greater with dietary red meat than white meat and is attenuated by resistant starch. Clin Nutr. 2012;31(1):60–4.
Nettleton JA, Diez-Roux A, Jenny NS, Fitzpatrick AL, Jacobs DR Jr. Dietary patterns, food groups, and telomere length in the Multi-Ethnic Study of Atherosclerosis (MESA). Am J Clin Nutr. 2008;88(5):1405–12.
Rudolph KL, Millard M, Bosenberg MW, DePinho RA. Telomere dysfunction and evolution of intestinal carcinoma in mice and humans. Nat Genet. 2001;28(2):155–9.
Calado RT, Young NS. Telomere diseases. N Engl J Med. 2009;361(24):2353–65.
Willeit P, Willeit J, Mayr A, Weger S, Oberhollenzer F, Brandstatter A, et al. Telomere length and risk of incident cancer and cancer mortality. JAMA. 2010;304(1):69–75.
Shen G, Huang JY, Huang YQ, Feng YQ. The Relationship between Telomere Length and Cancer Mortality: Data from the 1999–2002 National Healthy and Nutrition Examination Survey (NHANES). J Nutr Health Aging. 2020;24(1):9–15.
Bao Y, Prescott J, Yuan C, Zhang M, Kraft P, Babic A, et al. Leucocyte telomere length, genetic variants at the TERT gene region and risk of pancreatic cancer. 2017;66(6):1116-22.
https://www.aicr.org/resources/media-library/10-cancer-prevention-recommendations/.
https://www.wcrf.org/wp-content/uploads/2021/01/Recommendations.pdf.
Rock CL, Thomson C, Gansler T, Gapstur SM, McCullough ML, Patel AV, Andrews KS. American Cancer Society Guidelines on Nutrition and Physical Activity for Cancer Prevention. CA Cancer J Clin. 2020;70, 245–71.
Budhathoki S, Sawada N, Iwasaki M, Yamaji T, Goto A, Kotemori A, et al. Association of animal and plant protein intake with all-cause and cause-specific mortality in a Japanese Cohort. JAMA Intern Med. 2019;179(11):1509–18.
Zhu B, Sun Y, Qi L, Zhong R, Miao X. Dietary legume consumption reduces risk of colorectal cancer: evidence from a meta-analysis of cohort studies. Sci Rep. 2015;5:8797.
Li J, Mao QQ. Legume intake and risk of prostate cancer: a meta-analysis of prospective cohort studies. Oncotarget. 2017;8(27):44776–44784.
Papandreou C, Becerra-Tomas N, Bullo M, Martinez-Gonzalez MA, Corella D, Estruch R, et al. Legume consumption and risk of all-cause, cardiovascular, and cancer mortality in the PREDIMED study. Clin Nutr. 2019;38(1):348–56.
Tian S, Xu Q, Jiang R, Han T, Sun C, Na L. Dietary protein consumption and the risk of type 2 diabetes: a systematic review and meta-analysis of cohort studies. Nutrients. 2017;9(9):982.
Kim K, Hyeon J, Lee SA, Kwon SO, Lee H, Keum N, et al. Role of total, red, processed, and white meat consumption in stroke incidence and mortality: a systematic review and meta-analysis of prospective cohort studies. J Am Heart Assoc. 2017;6(9):e005983.
Micha R, Michas G, Mozaffarian D. Unprocessed red and processed meats and risk of coronary artery disease and type 2 diabetes--an updated review of the evidence. Curr Atheroscler Rep. 2012;14(6):515–24.
Kwok CS, Gulati M, Michos ED, Potts J, Wu P, Watson L, et al. Dietary components and risk of cardiovascular disease and all-cause mortality: a review of evidence from meta-analyses. Eur J Prev Cardiol. 2019;26(13):1415–29.
Acknowledgment
We thank Drs. Demetrius Albanes, Hannah Arem, Eunyoung Cho, Yashvee Dunneram, Brian Z. Huang, Petra Jones, Rena R. Jones, Christiana Kartsonaki, Ai Seon Kuan, Tricia Li, Hung N. Luu, Kirstin Pirie, Antonio Giampiero Russo, Veronica Wendy Setiawan, Rachael Stolzenberg-Solomon, Siân Sweetland, Mary K. Townsend, Shelley S. Tworoger, Margaret E. Wright, Xin Xu, and Xuehong Zhang, for kindly providing additional data for this meta-analysis.
Funding
There is no funding source for this study.
Author information
Authors and Affiliations
Contributions
Maryam S. Farvid: Study concept and design, data extraction and statistical analysis, interpretation of data, drafting of manuscript, critical revision of the manuscript for important intellectual content, and approval of the final manuscript for submission. Elkhansa Sidahmed: Helped with data extraction, interpretation of data, critical revision of the manuscript for important intellectual content, and approval of the final manuscript for submission. Nicholas D. Spence: Helped with interpretation of data, critical revision of the manuscript for important intellectual content, and approval of the final manuscript for submission. Kingsly Mante Angua: Helped with data extraction, and approval of the final manuscript for submission. Bernard A. Rosner: Helped with the interpretation of data and approval of the final manuscript for submission. Junaidah B. Barnett: Helped with the interpretation of data and writing the discussion section, critical revision of the manuscript for important intellectual content, and approval of the final manuscript for submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors of this study have no conflict of interest or any financial disclosures to make.
Data sharing
Data are available to share.
Ethical approval
NA.
Patient and public involvement
NA.
Transparency
All authors affirm that the manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned (and, if relevant, registered) have been explained.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Farvid, M.S., Sidahmed, E., Spence, N.D. et al. Consumption of red meat and processed meat and cancer incidence: a systematic review and meta-analysis of prospective studies. Eur J Epidemiol 36, 937–951 (2021). https://doi.org/10.1007/s10654-021-00741-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10654-021-00741-9