Abstract
In many countries obesity has increased dramatically during the last decades, while there has been a parallel decrease in smoking. The objective of the present study was to estimate the net effect on premature mortality of these trends. A simulation model was developed to estimate the expected number of deaths between ages 19–56 years for cohorts of young men (n = 50,000), depending on inputs of obesity and smoking prevalence. The model was populated with nationwide data of Swedish men performing mandatory military conscription tests between 1969 and 2005. Risk equations for all cause mortality with smoking and obesity status as predictors were developed based on the 1969–1970 conscription cohort (n = 45,920; 2,897 deaths, median follow-up 38 years). It was found that between 1969 and 2005, the prevalence of smoking decreased from 58.6 to 23.2%, while overweight increased from 5.7 to 15.6% and obesity from 0.8 to 5.5%. As a result of these trends, a 14% (CI95% 6, 21%) reduction of premature deaths between ages 19 and 56 years was forecasted for men aged 19 year in 2004–2005 compared to men aged 19 years in 1969–1970 (2,679 vs. 3,116 deaths). However, one-third of the survival benefit from reduced smoking during the period was offset due to the parallel increase in obesity. This study shows that despite large increases in overweight and obesity, a continued decline in premature deaths among Swedish males is expected due to reduced smoking during the last four decades.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obesity has increased dramatically in recent decades and is today highly prevalent, with an estimated worldwide prevalence in 2005 of 20.3% in economically developed countries [1]. In Swedish adolescents, overweight has tripled, obesity quintupled and morbid obesity increased tenfold from the period 1969–1974 compared to 2000–2005 [2]. With the strong association between obesity status and subsequent cardiovascular disease [3], disability pension [4, 5] and premature death [6–8], it will likely be several decades until the full burden of recent obesity trends on healthcare and society is manifested.
In contrast to the trends of increasing obesity, the prevalence of smoking, a factor also strongly associated with cardiovascular disease, disability pension [9] and premature death [10], has decreased substantially during the last decades in several industrialized countries [11–13]. Smoking and obesity in adolescents, arguably the two most important modifiable risk factors in this age group, independently increase premature mortality risk, but the combined effects of these simultaneous opposite trends on life expectancy is unknown.
The aim of this study was to estimate the net effect on future premature deaths due to trends in obesity and smoking in adolescent Swedish men.
Materials and methods
A Markov model [14] with Monte Carlo simulation was developed to simulate the effects on expected number of premature deaths between age 19 and 56 years based on inputs of obesity and smoking status from a nationwide cohort study of Swedish men. The time horizon of the model was chosen in order to avoid extrapolation outside the observed data. The model was constituted by two health states (“Alive” and “Dead”) with possible annual transitions from “Alive” to “Dead”. Cycles of 1 year until cohort participants had reached age 56 years or died were utilized. In each cycle, individuals of the cohort were subjected to time-dependent risks of death, based on obesity and smoking status at age 19 years, and age at cycle (i.e. 19–56 years, with increasing risk of death with older age). Simulations were run for 10,000 iterations, and empirical 95% confidence intervals were retrieved from the sorted simulation results as the 250th and 9,750th values.
Input data
The model input data consisted of prevalence of smoking and obesity, as well as obesity-, smoking-, and age-specific annual risk of death. The data were retrieved from the nationwide Swedish Military Service Conscription Register, which has been presented in greater detail elsewhere [15]. Briefly, military conscription was mandatory for all Swedish men during the study period, and is normally completed during the last year of high-school (age 18–19 years). At conscription the young men go through a 2-day test including medical examinations.
Based on the unique personal identification number assigned to each Swedish citizen, a dataset was created by register linkage between the Military Service Conscription Register, Register of the Total Population, and Population and Housing Censuses. In order to be included in the current study, subjects were required to have performed their military conscription tests between 16 and 20 years of age, and to have data recorded on measured weight and height as well as self-reported smoking habits at conscription (i.e. complete case analysis was used). The age restriction was used to keep the sample homogenous by age.
Risk equation input data
The risk equations employed in the model were derived from the cohort performing military conscription tests in 1969–1970. A proportional hazards regression model using the parametric Weibull distribution [16] was used to model absolute annual risks of premature death for each one-year cycle in the simulation model. Participants were censored at emigration, death or end of follow-up, whichever came first. Except for the predictors smoking and obesity status, the models were adjusted for household socio-economic index (white collar, blue collar, self-employed, other). The information on socio-economic index was extracted from the Population and Housing Census 1970 and thus represents household socio-economic index during childhood. Data on alcohol consumption and highest attained education were also available, but due to high degree of collinearity with socio-economic position these variables were not used simultaneously. The estimates for BMI- and smoking categories were unaffected when these covariates were also used in the regression model.
Body mass index (BMI) was used as proxy for fatness. It was defined as weight in light clothing (kg) divided by height (m) squared, without shoes, and categorized into the World Health Organization-defined BMI categories underweight (<18.5), normal weight (18.5–24.9), overweight (25–29.9) and obesity (≥30) [17]. To minimize errors of misclassifications, exclusion limits were applied for height (≤150 or ≥210 cm), weight (≤40 or ≥150 kg) and BMI (≤15 or ≥60 kg/m2). Smoking status was categorized into three levels (non-smoker/1–10/>10 daily cigarettes) using a questionnaire.
Risk factor prevalence input data
Prevalence of obesity and smoking were retrieved from the 1969–1970 (n = 45,920) and 2004–2005 (n = 56,411) conscription cohorts. To predict distribution over obesity status categories for future cohorts, data from each individual year from 1969 to 2005 (n = 1.6 million) were used. The predictions were performed with logistic regression using year of military conscription testing as predictor. The future prevalence was then calculated according to the following formula:
where α is the intercept on the log odds scale and β is the log odds ratio for the obesity status category, as has been done previously [2].
Regarding smoking, individual participant data were obtained from the Military Service Conscription Register for the 1969–1970 cohort. Thereafter information on smoking at the time of military conscription was registered and reported by the Swedish Federation of Alcohol and Drug Awareness [18], but not made available for register linkage for research purposes. Therefore smoking prevalence data for 2004–2005 were taken from the Swedish Federation of Alcohol and Drug Awareness. “Daily smoking” was set as “>10 cigarettes/day”, and “Practically daily smoking” and “More seldom smoking” as “1–10 cigarettes/day”.
Simulations
The model was run for hypothetical cohorts of 50,000 men in order to enable direct comparisons between cohorts using several input scenarios. Firstly, the expected number of premature deaths from 19 to 56 years was estimated for the cohort from 2004 to 2005 and compared with the 1969–1970 cohort. Secondly, the isolated effects of changing trends in smoking (smoking = 2004–2005, BMI distribution = 1969–1970) or BMI distribution (smoking = 1969–1970, BMI distribution = 2004–2005) were estimated. Thirdly, a cohort consisting exclusively of normal weight non-smokers was simulated. Finally, several future cohorts were simulated under the assumption of unaltered smoking habits since 2004–2005 but a hypothetical continuing increase of overweight and obesity according to the prevalence projections.
Statistical analyses were conducted in Stata (version 10.0, College Station, Texas, USA), while the simulation model was built in Microsoft Excel 2003 with Visual Basic for Applications.
Results
Of 50,398 men in the cohort performing military conscription in 1969–1970, 45,920 met the inclusion criteria [7]. The participant characteristics are shown in Table 1. During 1.7 million person-years of observation (median 37.7 years) 2,897 individuals died. Overweight and obesity compared to normal weight, and smoking compared to non-smoking, were significantly associated with increased risk of premature death, while underweight was not (Table 2). The relative risks estimated by use of the parametric survival models employed in the simulation were almost identical to the estimates from the semi-parametric Cox models used in a previous study on the same cohort (data not shown) [7]. The data followed a Weibull distribution and no violations of the proportional hazards assumption were detected when investigated graphically.
During the period from 1969–1970 to 2004–2005, overweight almost tripled and obesity increased sevenfold among young Swedish men, while smoking more than halved from 59 to 23% (Table 2).
Model fit
Using the smoking and obesity prevalence in 1969–1970 as inputs for the risk equations, the simulation model was found to fit well to the observed data, producing only small discrepancies in number of deaths during a time horizon of 37 years and a cohort size of 45,920 individuals, as in the dataset (Appendix, Table A1). The model underestimated the total number of deaths by 1.2% (2,861 vs. 2,897) compared to reality.
Expected outcome for 2004–2005 and 1969–1970 cohorts
When using the model to simulate cohorts of 50,000 men from 19 to 56 years, a total of 3,116 (CI95% 2,941, 3,301) deaths were expected to occur for the 1969–1970 cohort (Fig. 1). Despite higher prevalence of overweight and obesity in the 2004–2005 cohort, 14% (6, 21%) fewer deaths were expected over the same time horizon due to decreases in smoking.
Total number of expected deaths in simulated cohorts of 50,000 men during the period from age 19 to 56 years stratified by obesity status for different scenarios. Effect of altered obesity = smoking prevalence from 1969 to 1970, obesity prevalence from 2004 to 2005. Effect of altered smoking = smoking prevalence from 2004 to 2005, obesity prevalence from 1969 to 1970. Normal weight non-smokers = full cohort normal weight non-smokers. Error bars represent 95% empirical confidence intervals
Isolated effects of smoking and BMI trends
Keeping all other parameters constant and only altering the BMI distribution in the 1969–1970 cohort to that of 2004–2005 resulted in a 7% (−1, 16%) increase in the number of expected deaths (Fig. 1, ‘Effect of altered obesity’). The corresponding effect of only altering smoking status patterns predicted 20% (15, 25%) fewer deaths (Fig. 1, ‘Effect of altered smoking’).
Potential for preventive efforts
If all 19 year old Swedish men had been normal weight non-smokers, and all other risk factors being similar, 33% (28, 38%) fewer deaths would be expected until age 56 years compared with the cohort of 1969–1970 (Fig. 1, ‘Normal weight non-smokers’). Compared to the observed cohort characteristics in 2004–2005, 22% (17, 28%) fewer deaths would be expected.
Future mortality
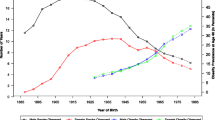
For the hypothetical case of an unabated obesity trend, the resulting number of deaths from age 19 to 56 years for future cohorts is shown in Fig. 2. The calculations were based on the assumption of unaltered smoking prevalence at conscription from 2004 to 2005, but increasing overweight and obesity according to extrapolation of the trends observed in the Swedish military conscription data from 1969 to 2005. It was estimated that the survival benefits associated with the successful decrease in smoking during the last four decades would be offset by the effect of increasing overweight and obesity within three decades. In 2030, a young Swedish male cohort was predicted to consist of 2.6% underweight, 48.6% normal weight, 29.7% overweight and 19.1% obese individuals, and to experience 3,140 (2,860, 3,440) deaths from age 19 to 56 years, compared to 3,116 (2,940, 3,300) for the lean but heavily smoking cohort of 1969–1970 (Fig. 2).
Observed and predicted distribution over BMI categories in Swedish military conscription cohorts (left panel). Predicted number of deaths during the period from age 19 to 56 years (right panel; dashed line = deaths in the 1969–70 cohort; bar charts = projected deaths in future cohorts with unaltered smoking prevalence from 2004 to 2005 but increasing prevalence of overweight and obesity according to predicted prevalences in left panel)
Discussion
By employing a simulation model, we estimated the effects of changes in obesity and smoking prevalence on premature death during the main part of productive age in Swedish men. Male cohorts today were predicted to enjoy reductions in premature mortality from age 19 to 56 years compared to male cohorts born 35 years earlier, independent of health care improvements. Despite substantial increases in adolescent overweight and obesity the past 35 years, their effect on future premature death was predicted to be offset by health benefits from concurrent decreases in smoking. However, approximately one-third of the survival gains from reduced smoking was estimated to be counteracted by the increases in overweight and obesity.
Based on observed declining mortality, some public health officials in Sweden have argued that either the “alarms of an obesity bomb” are grossly exaggerated, or that the detrimental health effects of obesity have been counteracted by a simultaneous improvement of another risk factor [19]. The present study supports the latter. Although obesity was modeled with an increased risk for premature death similar to that of smoking >10 cigarettes/day, male Swedish cohorts of today were predicted to experience fewer premature deaths compared to a cohort from the early 1970’s, despite a trebled and sevenfold increased prevalence of overweight and obesity, respectively. This is explained by the more than halved smoking prevalence during the last decades, and by the fact that the prevalence of obesity was very low to begin with. If the obesity trend does not level off but continues along the same trajectory as between 1969 and 2005, and smoking prevalence remains constant, the survival benefits of the last four decades’ successful anti-smoking campaigns were estimated to be offset for young adults in 2030. By then about 30% of the young men were predicted to be overweight, and 20% obese. However, this projection is purely hypothetical and should be interpreted with caution, especially since three recent studies indicate that increases in childhood obesity are levelling off in Sweden [20–22]. If the prevalence rise of overweight and obesity will be less steep in reality, it will take longer time to attain the projected prevalences and corresponding number of deaths predicted by the model.
A recent study by Stewart et al. [23] investigated the effects of extrapolated increases in obesity and decreases in smoking, as hitherto observed in the US, on life expectancy. They concluded that in the future in the US, the negative effects on health of increasing obesity will outweigh the positive effects of decreasing smoking. Although similar to the secondary analysis of the present study, a comparison is not straightforward since different questions were addressed.
Forecasts generated by simulation models are often afflicted by a high degree of uncertainty, partly due to unknown future factors and partly due to the mix of sources providing input data. Having all input data from one single data source covering practically the whole Swedish male population was an advantage of the present study.
This study also used data with a long follow-up period and BMI measured early in life, when chronic diseases associated with weight loss are rare, thus minimizing the effect of reverse causation impacting the risk equations [24]. Unfortunately, the data provided information on obesity and smoking status only at the beginning of the follow-up period, with no repeated measurements. Obesity in childhood has been found to be associated with obesity in adulthood [25, 26], but smoking continuation, cessation or initiation could be harder to predict from smoking habits in late adolescence. It is, however, likely that smoking cessation during follow-up was more common in this cohort than smoking initiation, resulting in some decline in smoking prevalence over time [27]. If this is the case, the association between smoking and mortality would be somewhat underestimated. Also, this study analyzed premature mortality, defined as deaths before age 56 years. A large proportion of deaths occur after this age, and if the relation between the risk factors smoking and obesity in terms of mortality risk changes with age, this could have an impact on the results of this study. Smoking is likely to incur increased mortality risk also in higher ages, but there are indications that this is not always the case for overweight [28]. It is therefore possible that the truncation of the analysis at age 56 years could imply an overestimation of the obesity related deaths in relation to smoking related deaths.
Another limitation of the present study was the assumption that each predictor’s associated risk for death has remained unaltered over time. A heavily smoking, obese individual at conscription today was assumed to be subjected to the same risk of death as in the early 1970’s. This assumption is not totally valid as risks have changed over time, e.g. due to more effective treatments, which would lead to an overestimated number of deaths for present cohorts. It is also possible that risks have changed as the excess weight of obese individuals today may be attributable to obesogenic environmental influences to a greater extent. However, it is not clear how it would affect the obesity related mortality risk whether an individual’s obesity has its origin primarily in genetic or environmental factors. On the other hand, the mortality in the later cohorts could be underestimated if the observed decrease in prevalence of smoking is in fact due to a shift towards usage of moist snuff (snus) rather than becoming tobacco-free [29], although it has been suggested that health gains can still be realized by current smokers switching to snus [30]. It is also probable that current cohorts quit smoking at a lower rate than older cohorts, while gaining weight at a higher rate. Lacking longitudinal data on these parameters, we assumed these rates to be unchanged over time.
In conclusion, despite large increases in overweight and obesity during the last four decades, a decline in premature deaths among Swedish males is expected due to health gains from reduced smoking. However, approximately a third of the survival gains from reduced smoking were estimated to be counteracted by increases in overweight and obesity.
Abbreviations
- BMI:
-
Body mass index
- HR:
-
Hazard ratio
- CI:
-
Confidence interval
- SD:
-
Standard deviation
References
Kelly T, Yang W, Chen CS, Reynolds K, He J. Global burden of obesity in 2005 and projections to 2030. Int J Obes (Lond). 2008;32(9):1431–7.
Neovius M, Teixeira-Pinto A, Rasmussen F. Shift in the composition of obesity in young adult men in Sweden over a third of a century. Int J Obes (Lond). 2008;32(5):832–6.
Falkstedt D, Hemmingsson T, Rasmussen F, Lundberg I. Body mass index in late adolescence and its association with coronary heart disease and stroke in middle age among Swedish men. Int J Obes (Lond). 2007;31(5):777–83.
Rissanen A, Heliovaara M, Knekt P, Reunanen A, Aromaa A, Maatela J. Risk of disability and mortality due to overweight in a Finnish population. BMJ. 1990;301(6756):835–7.
Neovius M, Kark M, Rasmussen F. Association between obesity status in young adulthood and disability pension. Int J Obes (Lond). 2008;32(8):1319–26.
Sonne-Holm S, Sorensen TI, Christensen U. Risk of early death in extremely overweight young men. Br Med J (Clin Res Ed). 1983;287(6395):795–7.
Neovius M, Sundstrom J, Rasmussen F. Combined effects of overweight and smoking in late adolescence on subsequent mortality: nationwide cohort study. BMJ. 2009;338:b496.
Whitlock G, Lewington S, Sherliker P, Clarke R, Emberson J, Halsey J, et al. Body-mass index and cause-specific mortality in 900,000 adults: collaborative analyses of 57 prospective studies. Lancet. 2009;373(9669):1083–96.
Neovius K, Neovius M, Rasmussen F. The combined effects of overweight and smoking in late adolescence on subsequent disability pension: a nationwide cohort study. Int J Obes (Lond). 2009;34(1):75–82.
Doll R, Peto R, Boreham J, Sutherland I. Mortality in relation to smoking: 50 years’ observations on male British doctors. BMJ. 2004;328(7455):1519.
Center for Disease Control and Prevention (CDC). National Health Interview Survey 1965–2007. www.cdc.gov. Accessed 20090923.
UK National Statistics. www.statistics.gov.uk. Accessed 20090923.
McGee R, Williams S, Stanton W. Smoking among New Zealand adolescents 1960–1993. N Z Med J. 1995;108(995):85–7.
Sonnenberg FA, Beck JR. Markov models in medical decision making: a practical guide. Med Decis Mak. 1993;13(4):322–38.
Andreasson S, Allebeck P, Romelsjo A. Alcohol and mortality among young men: longitudinal study of Swedish conscripts. Br Med J (Clin Res Ed). 1988;296(6628):1021–5.
Carroll KJ. On the use and utility of the Weibull model in the analysis of survival data. Control Clin Trials. 2003;24(6):682–701.
WHO. Obesity: preventing and managing the global epidemic. Report of a WHO consultation. World Health Organ Tech Rep Ser. 2000;894(I–xii):1–253.
CAN. Drug habits among conscripts 2005 (Mönstrandes drogvanor 2005). Report 95. Stockholm, 2006.
Asplund K. Alarms of obesity bomb grossly exaggerated (Larmen om fetmabomb är kraftigt överdrivna). Dagens Nyheter 2008-08-12. Accessible at http://www.dn.se/opinion/debatt/larmen-om-fetmabomb-ar-kraftigt-overdrivna-1.716654. 2008.
Bergstrom E, Blomquist HK. Is the prevalence of overweight and obesity declining among 4-year-old Swedish children? Acta Paediatr. 2009;98(12):1956–8.
Sjoberg A, Lissner L, Albertsson-Wikland K, Marild S. Recent anthropometric trends among Swedish school children: evidence for decreasing prevalence of overweight in girls. Acta Paediatr. 2008;97(1):118–23.
Sundblom E, Petzold M, Rasmussen F, Callmer E, Lissner L. Childhood overweight and obesity prevalences levelling off in Stockholm but socioeconomic differences persist. Int J Obes (Lond). 2008;32(10):1525–30.
Stewart ST, Cutler DM, Rosen AB. Forecasting the effects of obesity and smoking on US life expectancy. N Engl J Med. 2009;361(23):2252–60.
Lawlor DA, Hart CL, Hole DJ, Davey Smith G. Reverse causality and confounding and the associations of overweight and obesity with mortality. Obesity (Silver Spring). 2006;14(12):2294–304.
Guo SS, Chumlea WC. Tracking of body mass index in children in relation to overweight in adulthood. Am J Clin Nutr. 1999;70(1):145S–8S.
van Dam RM, Willett WC, Manson JE, Hu FB. The relationship between overweight in adolescence and premature death in women. Ann Intern Med. 2006;145(2):91–7.
Rasmussen F, Tynelius P, Kark M. Importance of smoking habits for longitudinal and age-matched changes in body mass index: a cohort study of Swedish men and women. Prev Med. 2003;37(1):1–9.
Walter S, Kunst A, Mackenbach J, Hofman A, Tiemeier H. Mortality and disability: the effect of overweight and obesity. Int J Obes (Lond). 2009;33(12):1410–8.
Boffetta P, Straif K. Use of smokeless tobacco and risk of myocardial infarction and stroke: systematic review with meta-analysis. BMJ. 2009;339:b3060.
Gartner CE, Hall WD, Vos T, Bertram MY, Wallace AL, Lim SS. Assessment of Swedish snus for tobacco harm reduction: an epidemiological modelling study. Lancet. 2007;369(9578):2010–4.
Conflict of interest statement
None.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
See Table 3.
Rights and permissions
About this article
Cite this article
Neovius, K., Rasmussen, F., Sundström, J. et al. Forecast of future premature mortality as a result of trends in obesity and smoking: nationwide cohort simulation study. Eur J Epidemiol 25, 703–709 (2010). https://doi.org/10.1007/s10654-010-9485-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10654-010-9485-x