Abstract
The present study measured arsenic (As) concentrations in soil, groundwater and rice grain samples in two villages, Sarapur and Chinili, under Chakdaha block, Nadia district, West Bengal, India. This study also included a survey of the two villages to understand the knowledge among villagers about the As problem. Soil and groundwater samples were collected from fields in two villages while rice grain samples were collected from villagers’ houses. The results revealed the presence of As in higher concentrations than the maximum permissible limit of As in drinking water (10 µg L−1 and 50 µg L−1 by WHO and Indian standard, respectively) in groundwater [124.50 ± 1.11 µg L−1 (Sarapur) and 138.20 ± 1.34 µg L−1 (Chinili)]. The level of As in soil was found to range from 47.7 ± 0.14 to 49.3 ± 0.19 mg Kg−1 in Sarapur and from 57.5 ± 0.25 to 62.5 ± 0.44 mg Kg−1 in Chinili which are also higher than European Union maximum acceptable limit in agricultural soil (i.e. 20 mg Kg−1). The analysis of As in rice grains of five varieties, collected from residents of two villages, showed the presence of higher than recommended safe level of As in rice by FAO/WHO (0.2 mg Kg−1). The As concentration order was Gosai (0.95 ± 0.044 mg kg−1), Satabdi (0.79 ± 0.038 mg kg−1), Banskathi (0.60 ± 0.026 mg kg−1), Kunti (0.47 ± 0.018 mg kg−1) and Ranjit (0.29 ± 0.021 mg kg−1). Importantly, Gosai and Satabdi were the most popular varieties being consumed by local people. The data of consumption of rice per day in the survey was used for the measurement of average daily dose and hazard quotient. It was seen that the As hazard was negatively correlated to the age of residents. Therefore, children and toddlers were at higher risk of As exposure than elderly people. In addition, people with skin related As toxicity symptoms were also cited in the two villages. The study emphasized the severity of As problem in remote areas of West Bengal, India where people consume As tainted rice due to lack of awareness about the As problem and associated health issues.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Arsenic (As) is a toxic metalloid distributed throughout the environment. Arsenic contamination in groundwater has surged in the recent past and its exposure is posing a serious threat to health of millions of people worldwide (Gilbert et al. 2015; Podgorski et al. 2017) with the worst effects in the areas of Indo-Gangetic and Ganga–Meghna–Brahmaputra plains (Chakraborti et al. 2013). Some recent studies suggest that contamination has emerged in other parts of the world also where there was no prevalence of As earlier like the Amazon basin, West Africa and some parts of China (Bretzler et al. 2017; Li et al. 2018). The exposure to As through ingestion (drinking water and food) for long time affects the human health and may cause several diseases including cancers, melanosis, skin lesions, hyperkeratosis, lung disease, and peripheral vascular disease (Banerjee et al. 2013; Abdul et al. 2015).
In West Bengal, India, 9 out of total 19 districts are facing the problem of As contamination (Nickson et al. 2000; Chakraborty et al. 2002; Bhattacharya et al. 2010). A number of studies in past few decades analysed As contamination in soil, groundwater and rice grain in the affected regions (Shrivastava et al. 2017; Mondal et al. 2017; Bhowmick et al. 2018; Upadhyay et al. 2018). The studies also included surveys among local people and awareness programs (Hadi 2003; Murray and Sharmin 2015). However, it has been pointed out clearly that more awareness programs and educational camps need to be organized to make people well-informed of the situation and clarify any misconception (George et al. 2013). People, especially those living in rural areas, have no resources, such as piped water supply with effective As removal facilities, even when the problem is known for a few decades now (Basu et al. 2015).
The major route of As contamination for humans is through drinking water and food (Awasthi et al. 2017). Rice grains have greater affinity towards the arsenic accumulation approximately tenfold higher than others major food grains like wheat and barley. Rice is the major crop affected by As contamination. Rice grains have greater affinity towards the As accumulation approximately tenfold higher than others major food grains like wheat and barley (Majumdar and Bose 2017; Awasthi et al. 2017). Recently, Shrivastava et al. (2017) have reported as high as 1 mg kg−1 As levels in rice grains in Chakdaha block of West Bengal while safe As limit for white rice set by WHO is only 0.2 mg kg−1 (Sohn 2014). It is even more concerning to note that the rice consumption rates in As affected regions of Bangladesh and West Bengal, India are much higher, up to 500 g per day (Meharg and Zhao 2012), than that in other parts of the world. The As contamination of groundwater and soil, excessive rice cultivation in two seasons (Boro and Aman) in a year, the potential of rice to accumulate As in high amounts and people’s preference for rice consumption create a unique nexus that makes the exposure of people to As a highly likely process in a region like West Bengal. The humid and warm conditions of the area further exacerbate the problem as people need to drink plenty of water to survive and the demands are mostly met by groundwater that too comes predominantly from shallow aquifers (Biswas et al. 2014; Basu et al. 2015).
The present survey based study was conducted to evaluate ground reality of awareness about As problem among remote people in two villages, Sarapur and Chinili, in Nadia, West Bengal, India. The perception and knowledge of the As problem among villagers was ascertained scientifically through questionnaire based survey. In addition, the quantification of As in groundwater, soil and rice samples was done. The data of As concentrations in rice grains and rice consumption pattern were used to evaluate the HQ in relation to age of villagers.
Materials and methods
Study area
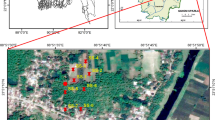
The study was conducted in a part of Chakdaha, a semi-rural municipality area under the Kalyani sub-division in district Nadia, West Bengal, India. Chakdaha is located at 23.08°N 88.52°E whereas the selected study area covers two of its villages at the innermost part with geo-positioning of 23°01′07.8″N 88°39′43.1″E to 23°01′14.3″N 88°38′24.7″E (Sarapur) and 23°01′29.0″N 88°43′41.4″E to 23°01′54.4″N 88°43′14.1″E (Chinili). The total population of Chakdaha block is 405,719 (Census of India 2011).
Survey questionnaire and sample collection
A door-to-door survey was performed by a standardized questionnaire based study in January–May 2017. A total of 105 houses were surveyed and among all individuals, 276 were males and 278 were females including an accountable number of children within an age group of 2–10 years (Supplementary Figure 1). The questionnaire included information like age, sex, educational background, medical history within family, etc. The details of questionnaire are given in Table 1. The individual household’s population was aimed for this study in selected villages after prior discussion with village council members (local self-governance system at village scale). Random sampling method was adopted to select appropriate number of native participants. Therefore on the basis of pilot observation of native residence (Kaccha and Pakka or Hut and Concrete houses), respective families were selected for survey interview. More than 70% interviewed participants were head of their family. Survey interview took around 20–30 min to complete. During the whole survey, one local person helped in obtaining information especially from females and other reluctant participants by having familiarity and by conversation in local language. Apart from personal information, volunteers were asked about their daily rice consumption individually and as a whole for the family to calculate the monthly rice requirement. The source of rice of the volunteers was also questioned; whether locally grown or purchased from market. The study area is a hub of rice production and different varieties are used. In five of the popular rice varieties among local residents, As analysis was performed in grains. These included Gosai, Satabdi, Banskathi, Kunti and Ranjit. The grains of the analysed varieties were collected from household during survey. Groundwater samples were collected from 15 tube wells in triplicates each from the two villages and were analysed for As. We performed the random sampling for groundwater. Further for soil sampling, we collected the agricultural soil from starting points of both villages (5 points of a field, triplicate samples of each point), and similarly midpoint and last point of the village and made the representative samples for these three points as S1, S2, and S3. Soil and groundwater pH and conductivity (EC) was analysed with the help of waterproof Oaklon multi-parameter PCSTestr™ 35 series pH meter. The level of organic matter (%OM) was determined through Walkley–Black method (Walkley and Black 1934). The oxidation reduction potential (ORP) was quantified with the help of handy instrument Aakiro Aquasol digital (AM-ORP-01) after diluting 1 g of soil sample in 10 mL of milli-Q water. The carbonate and bicarbonate test of water samples were done by the titrimetric method of known volume of water to standard H2SO4 using phenolphthalein and methyl orange indicators (Estefan et al. 2013).
Health risk assessment
Based on the survey and respective participation of native residents, Average Daily Dose at (ADD) from As at definite age and Hazard Quotient (HQ) were calculated to evaluate the risk index of the subjected villagers of different age groups as per method given in Shibata et al. (2016).
ADD and HQ were determined by the following equations:
where Crc is concentration of As in specific dietary component [(rice) (mg kg−1)]; IRrc,t (Ingestion Rate) is amount of specific diet at the specific age which varied from 20 to 500 g per day for different age groups and BWt is the average specific body weight of participants at definite age which varied from 3 to 79 kg. The measurement of ADD subsequently resulted in the assessment of the HQ with reference dose (RfD). RfD represents oral reference dose (0.0003 mg kg−1 day−1) for As, given by United States Environmental Protection Agency (USEPA 2005). HQ > 1, denotes that population are at risk of specific hazard (As).
Arsenic analysis and sequential extraction
Arsenic analysis was done in soil, groundwater and rice grain samples following the protocol detailed in Shrivastava et al. (2017). For soil samples, a tri-acid (HNO3, HClO4 and H2SO4) digestion procedure was used. About 0.5 g of dried soil sample was taken in digestion tubes and mixed with 5 mL HNO3 for digestion. The mixtures was permitted to stand overnight in a fume hood in incubation. At next day, the digestion tubes were heated at 60 °C for 2 h in digestion block. Then, the tubes were allowed to cool at room temperature. In continuation, 2 mL HClO4 and 3 mL H2SO4 was added and the tubes were heated again at 160 °C for 2–3 h. After complete digestion, the product was allowed to cool, diluted to 25 mL with milli-Q water, filtered through Whatman 0.45 μm filter paper and stored until further analysis. Plant parts and grains were also subjected to di-acid (HClO4 and H2SO4) digestion under the similar conditions (Majumdar et al. 2018). Earlier to filtration and analysis, the groundwater samples were pre-treated with few drops of 2% HNO3 in the field to slow down the As species transformation. Arsenic analysis was performed by Inductively Coupled Plasma Mass Spectrometry (ICP-MS) (Thermo scientific Q-ICP-MS, X series 2) and the data was processed with Thermo Plasma Lab software. In order to maintain the qualitative accuracy of As, standard reference materials NIST 2711A Montana II were used. Further, sequential extraction process (SEP) was performed as per the protocol given in Sarkar et al. (2017) and modified from Tessier et al. (1979) that allowed extraction of As into five fractions and percentage values were obtained. These fractions were obtained through a series of chemical treatments with different acids and oxidizers resulting in loosened As in the first two fractions; exchangeable (1) and carbonate bound (2), followed by Fe–Mn oxide bound (3), organic matter bound (4) and finally the residual fraction (5). The details of SEP process are given in Table 2.
Statistical analysis
Correlation analysis was performed for determining any possible relationship between HQ and age group distribution of native residents. All experimental analyses were performed in triplicate. One way ANOVA was performed to confirm the data variability and validity of observed results. All the statistical analysis was performed by using Graph Pad Prism 6.
Results
Arsenic in groundwater
In West Bengal, groundwater is the main source of water for daily needs (cooking, drinking, etc.) and for irrigation purpose. Agriculture is mostly groundwater dependent. A huge number of shallow, large-diameter tube wells have been installed for agricultural irrigation. We investigated 30 tube well water samples, 15 from each village, spread across the study area. The analysed tube wells were also being used for water consumption by the people. The average As concentration in water in Sarapur and Chinili was 124.50 µg L−1 and 138.20 µg L−1, respectively (Fig. 1). The As levels were very high and way beyond the permissible limit of As for drinking water set by WHO and also as per Indian standards (10 µg L−1 and 50 µg L−1, respectively). The maximum and minimum As levels in water were 196.44 µg L−1 and 82.15 µg L−1, respectively, in Sarapur and were 189.03 µg L−1 and 92.54 µg L−1, respectively, in Chinili.
Arsenic in soil and physico-chemical parameters of soil
Soil samples were also analysed for As in the selected villages from three selected sites. The total load of As in Chinili soil (57.5–62.5 mg Kg−1) was measured to be higher than that in Sarapur soil (47.7–49.3 mg Kg−1). The top layer of the soil sample from both the villages retained the maximum level of As (60.09 mg Kg−1 and 48.53 mg Kg−1 in Chinili and Sarapur, respectively) (Fig. 2). Soil samples were collected during the winter rice cultivation (Boro) when groundwater is used for irrigation (Shrivastava et al. 2014).
The sequential extraction of As from soil sample was also performed (Fig. 3). Arsenic content was divided into five separate fractions (Tessier et al. 1979) providing information about bound or available fractions of As. The fractions included exchangeable (1), carbonate bound (2), Fe–Mn oxide bound (3), organic matter bound (4) and residual fraction (5). The percentage level of As in soil samples varied in different fractions in both Sarapur and Chinili villages. The exchangeable fraction contained the least As (0.12–0.24% in Sarapur and 0.18–1.64% in Chinili). While, the residual fraction retained the maximum As (12.37–15.27% in Sarapur and 9.88–27.72% in Chinili).
Arsenic mobility in the soil is dependent on the soil physico-chemical parameters (Violante et al. 2010). Soil pH from sarapur and Chinili were measured to be alkaline within a range of 8.2–8.6 and organic matter (%OM) was 1.71 and 1.83, respectively. The ORP of soil was found to range from 230 to 284 and from 227 to 247 in Sarapur and Chinili, respectively, in upper soil layers (0–5 cm). The average EC of the soil solution was 671.89 and 632.55 µS cm−1 (Table 3) whereas the groundwater EC was 281.36 and 305.18 µS cm−1 (Table 4) in Sarapur and Chinili, respectively.
The average pH value was found to be 8.04 and 7.94 in Sarapur and Chinili groundwater samples, respectively (Table 4). The average carbonate (CO−23) and bicarbonate (HCO3−) concentration in groundwater was found to be 76.20 and 173.86 mg L−1, respectively, in Sarapur and 84.46 and 202.53 mg L−1, respectively, in Chinili (Table 4).
Arsenic in rice
The presence of As in food especially rice samples from West Bengal region and its health effects are known (Guha Mazumder et al. 2012; Das et al. 2012; Santra et al. 2013; Rahaman et al. 2013). During survey of the two villages, rice samples (grains of total five rice varieties) were also collected, which were being consumed by the people. Among these varieties, Gosai was the most consumed variety by local residents. This is due to high yield of Gosai, as well as its favourable taste (as per the response of local residents). The average concentration of As in Gosai was 0.95 mg kg−1 (Fig. 4a–e). Satabdi was another popular variety among the local people that showed an average concentration of 0.79 mg kg−1 (Fig. 4a–e). Other three varieties included Banskathi, Kunti and Ranjit; the contribution of these varieties in daily diet of local residents was less as these were not frequently consumed. The average concentration of As in Banskathi (0.60 mg kg−1), Kunti (0.47 mg kg−1) and Ranjit (0.29 mg kg−1) was lower than that of Gosai and Satabdi. The level of As in the analysed grain samples was higher than the safe As limit of 0.2 mg kg−1 set by WHO. No villager consumed a single variety of rice throughout the year and all five tested varieties were being consumed during different times. In some cases, persons belonging to one house were consuming different rice varieties as per their preferences.
Medical conditions of local residents
The medical conditions encountered among local people included skin lesions and hyperkeratosis, lungs and breathing related problems as well as nervous problems. Natives were also observed for any visible superficial effect of As exposure. Many significant skin related symptoms like dermatosis, keratosis and hyperpigmentation were noticed (Fig. 5; Table 5). The visible symptoms of As exposure in natives suggested that they had been exposed to As for long duration.
Discussions
The aquifers of the Bengal Delta are known to be contaminated with As. However, the As level in aquifers and distribution of contaminated aquifers is a highly variable phenomenon (Fendorf et al. 2010). The total As concentration in Chinili soil was higher than that in Sarapur soil. This may be attributable to higher As concentration detected in water samples of Chinili than that of Sarapur (Fig. 1). It is known that high amount of water usage for irrigation purposes leads to build up of As in paddy soil and consequently in rice plants (Shrivastava et al. 2014). It is quite possible that farmers of Chinili and Sarapur might be using variable amount of water for irrigation purposes in their fields that may also lead to different total As build up in soil (Neumann et al. 2011). The top layer of soil (0–5 cm) in both Sarapur and Chinili had higher As concentration than the lower layers (5–10 and 10–15 cm). The application of groundwater for irrigation in Boro season (winter season) is done for 2–3 times. This leads to repeated cycles of flooded and dry conditions in the field that results into a greater built up of As in the top soil layers of 0–5 cm (Barla et al. 2017). The soil of the selected area was characterized in an earlier study (Sarkar et al. 2017) that found the soil to be silty-clay in nature. Such type of soil can retain water in the surface layers for long durations and hence causes As to concentrate mostly in the top soil (Sarkar et al. 2017).
Among the As fractions obtained through sequential extraction of soil, first two fractions, exchangeable and carbonate bound, are considered to be more responsive to redox changes and As available in these fractions becomes easily mobile in the aqueous state and consequently becomes available to the plants (Majumdar et al. 2018). Other three fractions hold As stringently and hence, As contained in these fractions is not available to the plants (Majumdar et al. 2018). A higher percentage of bioavailable As was found in S-2 and S-3 in Chinili while at S-1 in Sarapur. It is well known that soil properties like pH, ORP and OM significantly affect As mobility and bioavailability in the soil via effects on As chemistry, As adsorption/desorption, and redox conditions of soil (Wang and Mulligan 2006; Majumdar and Bose, 2017; Barla et al. 2017). However, the relationship of pH, ORP and OM with As in soil is a complex phenomenon that produces variability in As levels within a field or locality and across large areas (Anwar et al. 2013; Barla et al. 2017). The adsorption of arsenate (AsV) in silty-clays soil is maximum at low pH which decreases quickly at neutral or alkaline pH (Strawn 2018; Gersztyn et al. 2013). Further, pH and ORP have opposite effects on As mobility and bioavailability in soil (Barla et al. 2017). The physico-chemical properties in Sarapur and Chinili showed a mixed pattern in three sites (S1–S3). Hence, a specific reason for variable As bioavailability at three sites could not be ascertained. As suggested above, variable irrigation frequency and amount of irrigation water in two villages might affect not only the total soil As but also soil chemistry and physico-chemical properties. This needs to be studied in further research.
The differences in bioavailable As fraction in Chinili and Sarapur were not reflected in significant variation in As content in rice grain samples. This may be due to the fact that As concentrations in rice grains are dependent on irrigation regime (Spanu et al. 2012), environmental factors (Williams et al. 2006; Rahman et al. 2011; Awasthi et al. 2017) as well as rice genotypes (Bhattacharya et al. 2010; Duan et al. 2017). In our study, rice grain As concentration was found to be higher as compared with some previous studies in Bengal Delta region (Rahman et al. 2009; Rahaman et al. 2013). The As concentrations have also been found to vary in field and market based studies (Meharg et al. 2009; Rahman et al. 2011; Williams et al. 2007) with a maximum level of 1.835 mg kg−1 reported by Meharg and Rahman (2003). However, As usually remains below 1.0 mg kg−1. To have an assessment of As exposure to local residents, the consumption data of water and rice was used to calculate daily average As intake (Davis et al. 2017). The amount of rice consumption ranged from 0.5 to 3.5 kg per day in Sarapur and 0.5 to 7.5 kg per day in Chinli for a family and from 0.142 to 0.700 kg per day per person in Sarapur and 0.142 to 0.750 kg per day per person in Chinili (Supplementary Table 1). Thus, rice constituted the major constituent of diet of local rural people. In fact, rice has been recognized as predominant exposure route to As for humans in earlier studies also (Khan et al. 2009). Hazard quotient (HQ) is an important parameter to calculate risk for a specific hazardous chemical/metal and can also be correlated to age of affected people (Rasheed et al. 2018; Shibata et al. 2016; Singh and Ghosh 2012). The calculation of HQ was found more than 1 in all age groups and indicated a negative correlation (R2 = − 0.647; P ≤ 0.05) with age group distribution as presented in Fig. 6. It suggests that toddlers and children had a greater risk of As poisoning, which is similar to reported in earlier studies (Shibata et al. 2016; Singh and Ghosh 2012). Rice is regularly a baby’s first solid food since long time in India and other parts of world too. The children get exposed regularly to As through rice apart from other sources including mother’s milk (Karagas et al. 2016). Further, children and babies usually consume more food with respect to their total body weight as compared to adult people and hence, As load per kilogram of body weight becomes higher in children than in adult people (Upadhyay et al. 2018; Meharg and Zhao 2012).
It was noticed in the survey that older people used to consume the groundwater or pond surface water more commonly than younger ones. During the survey, female volunteers confirmed that drinking, cooking, bathing, washing clothes, and other household applications are fulfilled by surrounding tube well water; the main source of contamination. Some previous studies showcased that cooking process may effectively reduce significant part of As from the rice grains (Sengupta et al. 2006; Carey et al. 2015). But this is possible when water used for cleaning and cooking has no or very low As. In the study area, the traditional process of rice cooking was common that uses large amount of water for cooking, which may further increase the As content in the cooked rice (Roychowdhury et al. 2002). The subjected study area has been suffering for a long period of time due to high As contamination in ground water (Chowdhury et al. 2000; Nickson et al. 2000) and such condition may deteriorate human health (Das et al. 1995). In the survey, skin related problems were noticed. In addition, other As associated symptoms were also found to be present like respiratory problems involving pulmonary issues and muscle constriction with nerve strain. The primary health impact of As can be observed on skin in the form of roughening and lesions (Milton and Rahman 1999; Maity et al. 2012). Arsenic exposure may cause Diabetes, cardiovascular diseases, nervous system disorders and even cancer. The effect of As are mediated via direct interaction with biomolecules and through impact on DNA repair mechanism, DNA methylation and histone post-translational modifications (Upadhyay et al. 2018; States et al. 2011). A very few of the volunteers were educated enough to know about these effects of As in humans, its presence in water, soil and food and its possible consequences. Most of the natives possessed just a superficial idea about As problem that was basically not sufficient for them to become more cautious. The villagers were unaware of agronomically feasible practices like less water use for irrigation and appropriate rice cultivars/varieties that accumulate less As. Further, farmers were also not aware that more washing steps and less water use for cooking may reduce As concentration in cooked rice; Mostly, participants were aware about the term As without knowing its negative impact on soil health, crop production and quality, and human health.
Conclusions
The study demonstrated the presence of very high As concentrations in water, soil and rice grain samples from the two villages, Sarapur and Chinili. It was clearly evident that people were mostly unaware of As related negative health impacts and were commonly consuming groundwater and locally grown rice grains in their daily routine. Further, a few people were also having symptoms of As toxicity. The study, therefore, demonstrated an alarming lack of awareness and basic knowledge level required to tackle a widespread problem like As. Thus, this study proposes that regular monitoring, and awareness campaigns along with other remedial measures must be exercised to safeguard people against As.
References
Abdul, K. S., Jayasinghe, S. S., Chandana, E. P., Jayasumana, C., & De Silva, P. M. (2015). Arsenic and human health effects: A review. Environmental Toxicology and Pharmacology,40(3), 828–846.
Anwar, H. M., Tareq, S. M., & Ahmed, G. (2013). Is organic matter a source or redox driver or both for arsenic release in groundwater? Physics and Chemistry of the Earth A/B/C,58–60, 49–56.
Awasthi, S., Chauhan, R., Srivastava, S., & Tripathi, R. D. (2017). The journey of arsenic from soil to grain in rice. Frontiers in Plant Sciences,8, 1007.
Banerjee, M., Banerjee, N., Bhattacharjee, P., Mondal, D., Lythgoe Paul, R., Martínez, M., et al. (2013). High arsenic in rice is associated with elevated genotoxic effects in humans. Scientific Reports,3, 2195.
Barla, A., Shrivastava, A., Majumdar, A., Upadhyay, M. K., & Bose, S. (2017). Heavy metal dispersion in water saturated and water unsaturated soil of Bengal delta region, India. Chemosphere,168, 807–816.
Basu, A., Sen, P., & Jha, A. (2015). Environmental arsenic toxicity in West Bengal, India: A brief policy review. Indian Journal of Public Health,59(4), 295.
Bhattacharya, P., Samal, A. C., Majumdar, J., & Santra, S. C. (2010). Accumulation of arsenic and its distribution in rice plant (Oryza sativa L.) in Gangetic West Bengal, India. Paddy and Water Environment,8(1), 63–70.
Bhowmick, S., Pramanik, S., Singh, P., Mondal, P., Chatterjee, D., & Nriagu, J. (2018). Arsenic in groundwater of West Bengal, India: A review of human health risks and assessment of possible intervention options. Science of the Total Environment,612, 148–169.
Biswas, A., Deb, D., Ghose, A., Santra, S. C., & Guha Mazumder, D. N. (2014). Seasonal perspective of dietary arsenic consumption and urine arsenic in an endemic population. Environmental Monitoring and Assessment,186(7), 4543–4551.
Bretzler, A., Lalanne, F., Nikiema, J., Podgorski, J., Pfenninger, N., Berg, M., et al. (2017). Groundwater arsenic contamination in Burkina Faso, West Africa: Predicting and verifying regions at risk. Science of the Total Environment,584, 958–970.
Carey, M., Jiujin, X., Farias, J. G., & Meharg, A. A. (2015). Rethinking rice preparation for highly efficient removal of inorganic arsenic using percolating cooking water. PLoS ONE,10(7), e0131608.
Census of India, West Bengal. (2011). District census handbook, Nadia (Series-20, Part XIIB). http://censusindia.gov.in/2011census/dchb/1910_PART_B_DCHB_NADIA.pdf Accessed March 17, 2019.
Chakraborti, D., Rahman, M. M., Das, B., Nayak, B., Pal, A., Pal, A., et al. (2013). Groundwater arsenic contamination in Ganga–Meghna–Brahmaputra plain, its health effects and an approach for mitigation. Environmental Earth Sciences,70(5), 1993–2008.
Chakraborty, D., Rahaman, M. M., Paul, K., Chowdhury, U. K., Sengupta, M. K., Lodh, D., et al. (2002). Arsenic calamity in the Indian subcontinent what lessons has been learnt? Talanta,58(1), 3–22.
Chowdhury, U. K., Biswas, B. K., Chowdhury, T. R., Samanta, G., Mandal, B. K., Basu, G. C., et al. (2000). Groundwater arsenic contamination in Bangladesh and West Bengal, India. Environmental Health Perspectives,108(5), 393.
Das, D., Chatterjee, A., Mandal, B. K., Samanta, G., Chakraborti, D., & Chanda, B. (1995). Arsenic in ground water in six districts of West Bengal, India: the biggest arsenic calamity in the world. Part 2. Arsenic concentration in drinking water, hair, nails, urine, skin-scale and liver tissue (biopsy) of the affected people. Analyst,120, 917–924.
Das, N., Paul, S., Chatterjee, D., Banerjee, N., Majumder, N. S., Sarma, N., et al. (2012). Arsenic exposure through drinking water increases the risk of liver and cardiovascular diseases in the population of West Bengal, India. BMC Public Health,12(1), 639.
Davis, M. A., Signes-Pastor, A. J., Argos, M., Slaughter, F., Pendergrast, C., Punshon, T., et al. (2017). Assessment of human dietary exposure to arsenic through rice. Science of the Total Environment,586, 1237–1244.
Duan, G., Shao, G., Tang, Z., Chen, H., Wang, B., Tang, Z., et al. (2017). Genotypic and environmental variations in grain cadmium and arsenic concentrations among a panel of high yielding rice cultivars. Rice,10(1), 9.
Estefan, G., Sommer, R., & Ryan, J. (2013). Methods of soil, plant, and water analysis: A manual for the West Asia and North Africa region (pp. 194–196).
Fendorf, S., Michael, H. A., & van Geen, A. (2010). Spatial and temporal variations of groundwater arsenic in South and Southeast Asia. Science,328(5982), 1123–1127.
George, C. M., Factor-Litvak, P., Khan, K., Islam, T., Singha, A., Moon-Howard, J., et al. (2013). Approaches to increase arsenic awareness in Bangladesh: An evaluation of an arsenic education program. Health Education and Behaviour,40(3), 331–338.
Gersztyn, L., Karczewska, A., & Gałka, B. (2013). Influence of pH on the solubility of arsenic in heavily contaminated soils/Wpływ pH na rozpuszczalność arsenu w glebach silnie zanieczyszczonych. Ochrona Srodowiska i Zasobów Naturalnych,24(3), 7–11.
Gilbert, P. J., Polya, D. A., & Cooke, D. A. (2015). Arsenic hazard in Cambodian rice from a market-based survey with a case study of Preak Russey village, Kandal Province. Environmental Geochemistry and Health,37(4), 757–766.
Guha Mazumder, D., Purkayastha, I., Ghose, A., Mistry, G., Saha, C., Nandy, A. K., et al. (2012). Hypertension in chronic arsenic exposure: A case control study in West Bengal. Journal of Environmental Science and Health. Part A,47(11), 1514–1520.
Hadi, A. (2003). Fighting arsenic at the grassroots: experience of BRAC’s community awareness initiative in Bangladesh. Health Policy and Planning,18(1), 93–100.
Karagas, M. R., Punshon, T., Sayarath, V., Jackson, B. P., Folt, C. L., & Cottingham, K. L. (2016). Association of rice and rice-product consumption with As exposure early in life. JAMA Pediatrics,170, 609–616.
Khan, N. I., Bruce, D., Naidu, R., & Owens, G. (2009). Implementation of food frequency questionnaire for the assessment of total dietary arsenic intake in Bangladesh: Part B, preliminary findings. Environmental Geochemistry and Health,31(1), 221–238.
Li, R., Kuo, Y. M., Liu, W. W., Jang, C. S., Zhao, E., & Yao, L. (2018). Potential health risk assessment through ingestion and dermal contact arsenic-contaminated groundwater in Jianghan Plain, China. Environmental Geochemistry and Health,40(4), 1585–1595.
Maity, J. P., Nath, B., Kar, S., Chen, C. Y., Banerjee, S., Jean, J. S., et al. (2012). Arsenic-induced health crisis in peri-urban Moyna and Ardebok villages, West Bengal, India: An exposure assessment study. Environmental Geochemistry and Health,34(5), 563–574.
Majumdar, A., Barla, A., Upadhyay, M. K., Ghosh, D., Chaudhuri, P., Srivastava, S., et al. (2018). Vermiremediation of metal (loid)s via Eichornia crassipes phytomass extraction: A sustainable technique for plant amelioration. Journal of Environmental Management,220, 118–125.
Majumdar, A., & Bose, S. (2017). Toxicogenesis and metabolism of arsenic in rice and wheat plants with probable mitigation strategies. In Knežević, R (Eds.), Arsenic: Risks of exposure, behavior in the environment and toxicology (pp. 149–166). Nova Science Publishers.
Meharg, A. A., & Rahman, M. M. (2003). Arsenic contamination of Bangladesh paddy field soils: Implications for rice contribution to arsenic consumption. Environmental Science and Technology,37(2), 229–234.
Meharg, A. A., Williams, P. N., Adamako, E., Lawgali, Y. Y., Deacon, C., Villada, A., et al. (2009). Geographical variation in total and inorganic arsenic content of polished (white) rice. Environmental Science and Technology,43(5), 1612–1617.
Meharg, A. A., & Zhao, F. J. (2012). Arsenic and rice. New York: Springer.
Milton, A. H., & Rahman, M. (1999). Environmental pollution and skin involvement pattern of chronic arsenicosis in Bangladesh. Journal of Occupational Health,41(4), 207–208.
Mondal, S., Roy, A., Mukherjee, R., Mondal, M., Karmakar, S., Chatterjee, S., et al. (2017). A socio-economic study along with impact assessment for laterite based technology demonstration for arsenic mitigation. Science of the Total Environment,583, 142–152.
Murray, M. P., & Sharmin, R. (2015). Groundwater arsenic and education attainment in Bangladesh. Journal of Health Population and Nutrition,33(1), 20.
Neumann, R. B., St. Vincent, A.P., Roberts, L.C., Badruzzaman, A.B.M., Ali, M.A., et al. (2011). Rice field geochemistry and hydrology: an explanation for why groundwater irrigated fields in Bangladesh are net sinks of arsenic from groundwater. Environmental Science & Technology, 45(6), 2072–2078.
Nickson, R. T., McArthur, J. M., Ravenscroft, P., Burgess, W. G., & Ahmed, K. M. (2000). Mechanism of arsenic release to groundwater, Bangladesh and West Bengal. Applied Geochemistry,15(4), 403–413.
Podgorski, J. E., Eqani, S. A. M. A. S., Khanam, T., Ullah, R., Shen, H., & Berg, M. (2017). Extensive arsenic contamination in high-pH unconfined aquifers in the Indus Valley. Science Advances,3(8), e1700935.
Rahaman, S., Sinha, A. C., Pati, R., & Mukhopadhyay, D. (2013). Arsenic contamination: A potential hazard to the affected areas of West Bengal, India. Environmental Geochemistry and Health,35(1), 119–132.
Rahman, M. M., Asaduzzaman, M., & Naidu, R. (2011). Arsenic exposure from rice and water sources in the Noakhali district of Bangladesh. Water Quality, Exposure and Health,3(1), 1–10.
Rahman, M. M., Owens, G., & Naidu, R. (2009). Arsenic levels in rice grain and assessment of daily dietary intake of arsenic from rice in arsenic-contaminated regions of Bangladesh—Implications to groundwater irrigation. Environmental Geochemistry and Health,31(1), 179–187.
Rasheed, H., Kay, P., Slack, R., & Gong, Y. Y. (2018). Arsenic species in wheat, raw and cooked rice: Exposure and associated health implications. Science of the Total Environment,634, 366–373.
Roychowdhury, T., Uchino, T., Tokunaga, H., & Ando, M. (2002). Survey of arsenic in food composites from an arsenic-affected area of West Bengal, India. Food and Chemical Toxicology,40(11), 1611–1621.
Santra, S. C., Samal, A. C., Bhattacharya, P., Banerjee, S., Biswas, A., & Majumdar, J. (2013). Arsenic in food chain and community health risk: A study in Gangetic West Bengal. Procedia Environmental Sciences,18, 2–13.
Sarkar, S. R., Majumdar, A., Barla, A., Pradhan, N., Singh, S., Ojha, N., et al. (2017). A conjugative study of Typha latifolia for expunge of phyto-available heavy metals in fly ash ameliorated soil. Geoderma,305, 354–362.
Sengupta, M. K., Hossain, M. A., Mukherjee, A., Ahamed, S., Das, B., Nayak, B., et al. (2006). Arsenic burden of cooked rice: traditional and modern methods. Food and Chemical Toxicology,44(11), 1823–1829.
Shibata, T., Meng, C., Umoren, J., & West, H. (2016). Risk assessment of arsenic in rice cereal and other dietary sources for infants and toddlers in the US. International Journal of Environmental Research and Public Health,13(4), 361.
Shrivastava, A., Barla, A., Singh, S., Mandraha, S., & Bose, S. (2017). Arsenic contamination in agricultural soils of Bengal deltaic region of West Bengal and its higher assimilation in monsoon rice. Journal of Hazardous Materials,324, 526–534.
Shrivastava, A., Barla, A., Yadav, H., & Bose, S. (2014). Arsenic contamination in shallow groundwater and agricultural soil of Chakdaha block, West Bengal, India. Frontiers in Environmental Science,2, 50.
Singh, S. K., & Ghosh, A. K. (2012). Health risk assessment due to groundwater arsenic contamination: Children are at high risk. Human and Ecological Risk Assessment: an International Journal,18(4), 751–766.
Sohn, E. (2014). The toxic side of rice. Nature,514, S62–S63.
Spanu, A., Daga, L., Orlandoni, A. M., & Sanna, G. (2012). The role of irrigation techniques in arsenic bioaccumulation in rice (Oryza sativa L.). Environmental Science & Technology.,46(15), 8333–8340.
States, J. C., Barchowsky, A., Cartwright, I. L., Reichard, J. F., Futscher, B. W., & Lantz, R. C. (2011). Arsenic toxicology: Translating between experimental models and human pathology. Environmental Health Perspectives,119(10), 1356–1363.
Strawn, D. G. (2018). Review of interactions between phosphorus and arsenic in soils from four case studies. Geochemical Transactions,19(1), 10.
Tessier, A., Campbell, P. G., & Bisson, M. (1979). Sequential extraction procedure for the speciation of particulate trace metals. Analytical Chemistry,51(7), 844–851.
Upadhyay, M. K., Shukla, A., Yadav, P., & Srivastava, S. (2018). A review of arsenic in crops, vegetables, animals and food products. Food Chemistry,276, 608–618.
USEPA. (2005). United States environmental protection agency. Guidelines for Carcinogen Risk Assessment; EPA/630/P-03/001F; Risk Assessment Forum: Washington, DC, USA.
Violante, A., Cozzolino, V., Perelomov, L., Caporale, A. G., & Pigna, M. (2010). Mobility and bioavailability of heavy metals and metalloids in soil environments. Journal of Soil Science and Plant Nutrition,10(3), 268–292.
Walkley, A., & Black, I. A. (1934). An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Science,37(1), 29–38.
Wang, S., & Mulligan, C. N. (2006). Effect of natural organic matter on arsenic release from soils and sediments into groundwater. Environmental Geochemistry and Health,28(3), 197–214.
Williams, P. N., Islam, M. R., Adomako, E. E., Raab, A., Hossain, S. A., Zhu, Y. G., et al. (2006). Increase in rice grain arsenic for regions of Bangladesh irrigating paddies with elevated arsenic in groundwaters. Environmental Science & Technology,40(16), 4903–4908.
Williams, P. N., Villada, A., Deacon, C., Raab, A., Figuerola, J., Green, A. J., et al. (2007). Greatly enhanced arsenic shoot assimilation in rice leads to elevated grain levels compared to wheat and barley. Environmental Science and Technology,41(19), 6854–6859.
Acknowledgements
The authors are thankful to IISER-K instrumentation core facility. We are highly indebted to Mr. Anshuman Dash a local farmer from Nadia, West Bengal, who helped us to collect the samples and gather personal information about natives in survey. We are also thankful to all other villagers who gave us their precious time directly or indirectly to participate in our survey and in collection of the rice and water samples from their houses. MKU is thankful to Banaras Hindu University for providing Ph.D. fellowship. AB is thankful to CSIR-UGC for SRF fellowship. AM and SB are thankful to MoES [MoES/P.O.(Geosci)/56/2015] for JRF fellowship and project grant for funding, respectively. Funding was provided by Science and Engineering Research Board (Grant No. YSS/2014/000080).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Upadhyay, M.K., Majumdar, A., Barla, A. et al. An assessment of arsenic hazard in groundwater–soil–rice system in two villages of Nadia district, West Bengal, India. Environ Geochem Health 41, 2381–2395 (2019). https://doi.org/10.1007/s10653-019-00289-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10653-019-00289-4