Abstract
Arsenic (As) may occur in surface freshwater ecosystems as a consequence of both natural contamination and anthropogenic activities. In this paper, As concentrations in muscle samples of 10 fish species, sediments and surface water from three naturally contaminated rivers in a central region of Argentina are reported. The study area is one of the largest regions in the world with high As concentrations in groundwater. However, information of As in freshwater ecosystems and associated biota is scarce. An extensive spatial variability of As concentrations in water and sediments of sampled ecosystems was observed. Geochemical indices indicated that sediments ranged from mostly unpolluted to strongly polluted. The concentration of As in sediments averaged 6.58 μg/g ranging from 0.23 to 59.53 μg/g. Arsenic in sediments barely followed (r = 0.361; p = 0.118) the level of contamination of water. All rivers showed high concentrations of As in surface waters, ranging from 55 to 195 μg/L. The average concentration of As in fish was 1.76 μg/g. The level of contamination with As differed significantly between species. Moreover, the level of bioaccumulation of As in fish species related to the concentration of As in water and sediments also differed between species. Whilst some fish species seemed to be able to regulate the uptake of this metalloid, the concentration of As in the large catfish Rhamdia quelen mostly followed the concentration of As in abiotic compartments. The erratic pattern of As concentrations in fish and sediments regardless of the invariable high levels in surface waters suggests the existence of complex biogeochemical processes behind the distribution patterns of As in these naturally contaminated ecosystems.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Arsenic (As) is a highly ubiquitous and potentially toxic element. It may occur in surface freshwater ecosystems as a consequence of both natural contamination and anthropogenic activities. Human exposure to As can occur through a variety of routes, although the consumption of groundwater is the main route of exposure for most people. Health problems because of the presence of As in groundwater have been recognised in many parts of the world, including Bangladesh, West Bengal, Taiwan, Inner Mongolia, Mexico, Hungary, Argentina and Chile, as well as more localised occurrences related to mining activity and geothermal sources (Smedley and Kinniburgh 2002). Dietary intake may also be important. By the year 1993, the US Food and Drug Administration indicated that fish and other seafood accounted for 90 % of the total food As exposure with all other foods accounting for the remaining 10 %. Detrimental effects of arsenic on human health are varied and well known. Arsenic has been classified as a human carcinogen by the U.S. Environmental and Protection Agency (USEPA 1997) and the International Association for Research on Cancer (IARC 1987). More recently, As was ranked first on the Superfund List of Hazardous Substances (ATSDR 2007). The long-term exposure to ingested inorganic As has been found to induce blackfoot disease, a unique peripheral vascular disease that ends with dry gangrene and spontaneous amputation of affected extremities (Chen et al. 2001). In Argentina, people exposed to high level of As in drinking water develop an endemic disease known as Regional Endemic Chronic Hydroarsenism (HACRE) (Biagini et al. 1978).
The Chaco-Pampa Plain is one of the largest regions in the world (ca. one million km2) with the presence of As in groundwater (Smedley and Kinniburg 2002). The geographic distribution of arsenic and trace elements in lotic ecosystems of this region was already evaluated (Rosso et al. 2011b). These authors revealed that natural pollution by As is a regional problem in surface drainages. Nevertheless, results reporting the level of arsenic pollution in sediments (Schenone et al. 2007) and biota of freshwater ecosystems are scarce. Moreover, little is known about whether this regional distribution of the As concentration in water is paralleled with As concentration in sediments and freshwater biota. Several fish species of this region support important recreational fisheries and some of them are part of the dietary intake of many people. Therefore, concentrations of As in edible tissues of such species should be of public concern. On the other hand, exposure of fish to high levels of As may be relevant for fisheries and conservation management. Indeed, in vivo exposure of fish to As disturbed testicular and ovarian development (Shukla and Pandey 1984a, b).
The main objective of this study was to determine the concentration of As in water, sediments and fish species in three rivers with different degree of natural contamination by As. The level of As accumulation in fish tissues in relation to contamination level of sediments and water was also explored. The suitability of fish species for biomonitoring programmes and the risk of consuming fish according to the latest evaluation of joint FAO/WHO committee are discussed.
Materials and methods
Study area
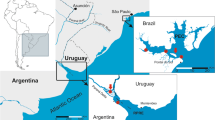
Three rivers of the Pampa Plain (Fig. 1) where the level of As was reported to be high (Rosso et al. 2011b) were selected for this study. In these rivers, the concentration of As in sediments, water and fish species was evaluated. Sampled ecosystems are characterised by alkaline waters (pH always above 8) and high conductivities (Rosso et al. 2011b). One of these rivers, the Sauce Grande, belongs to a zone that Rosso et al. (2011a) described as a high-risk zone in relation to the concentration of As (range 0.12–1.13 mg/L) and fluoride (range 0.57–1.64 mg/L) in surface waters. This river heads at the Ventania Hills, enters the plain, forms the Sauce Grande Lake and finally reaches the Atlantic Ocean. Along this continuum, three sampling stations were selected (Fig. 1): upstream (Las Oscuras, LO) and downstream (Valle Hermoso, VH) to the lake and in the Sauce Grande lake itself (SG). The Salado River (RS), located in the low-risk zone (As range 0.07–0.11 mg/L; F range 0.15–0.75 mg/L), and the Quequén Salado River (QS), which flows throughout the boundary between the high- and low-risk zones, were also surveyed. Each river was visited four times during the years 2008 and 2009: October 2008, March 2009, May 2009 and November 2009.
Map of the study area showing locations of sampling sites. Square: Salado River (RS); circle: Quequén Salado River (QS); triangle: Sauce Grande River. Detail of sampling sites along the continuum in the Sauce Grande River is also displayed at the bottom of the figure. White arrow denotes river main flow direction
Samples collection and treatment
Surface water samples for total dissolved As determination were collected manually at 0.5 m depth with 0.5-L polyethylene-tereftalate bottles. Samples were immediately transported to the laboratory and filtered through Acetate Plus Gamafil® filters (0.45 μm) with vacuum. A subsample of 100 mL filtered water was acidified to pH < 2. These samples were stored in darkness at 4 °C up to the analysis. Collection, preservation, preparation, pretreatment and analyses of water samples were conducted according to APHA (1995). Physicochemical attributes of water, including pH, temperature (°C), conductivity (μS/cm) and turbidity (NTU), were measured in situ at each site and date using a multi-parameter probe Horiba U-53G. Sediment samples were collected manually using an adaptation of the pedal corer described by Parada (2008). Samples were properly labelled in the field and transported to the laboratory at 4 °C. Water and sediment samples were collected at three different points within each site. These samples were separately pooled to obtain one pooled sample of sediment and one pooled sample of water (Keith 1991; Mudrock and Macknight 1994). This procedure was repeated three times in order to obtain water and sediment samples by triplicate.
Fish were caught using different fishing gears with complementary selectivity and at different times of the day. This maximised the capture of different species. For each species, individuals within similar size (a reliable surrogate of age in fish of the same population) range were selected for further analyses. Only fish in good body and health conditions were kept for analyses. Fish samples were first washed with deionised water. Wet samples of white muscle of fish and sediments were dried at 105 °C to constant weight. Subsamples of 2 g were then digested with concentrated (65 %) nitric acid (EPA, Method 3050B). Finally, samples were ashed at 500 °C. Ashes were recovered using a known volume of ultra pure Millipore water and acidified with concentrated nitric acid to 10 %v/v. Arsenic was determined by ICP-OES using a Perkin–Elmer Optima 2000 DV and wavelength set at 193 nm. Perkin–Elmer Pure atomic spectroscopy standard solution was used for calibration. Arsenic content in water samples was expressed in μg/L (or ppb). Arsenic contents in sediment and fish samples were expressed in μg/g (or ppm). The ICP-OES detection limit for As was 10 μg/L.
Geochemical indices
The interpretation of data on accumulation of elements in tissues of freshwater biota should be based on natural concentrations, which compose the biogeochemical background (Kuznetsova et al. 2002). For that reason, the level of As in sediments was evaluated with two different indices, the Håkanson’s (1980) contamination factor (CF) and the Müller’s (1979) geoaccumulation index (Igeo). The CF is defined as the relationship between the concentration of the element in surface sediments and its average natural background level (NBL). The NBL of an element in the sediment is defined as the average global value of this element in the Earth crust, as proposed by Turekian and Wedephol (1961), considering different types of sediments. According to Håkanson’s classification, contamination degrees can be delimited as: low (CF < 1), moderate (1 < CF < 3), considerable (3 < CF < 6) and high (CF > 6).
The Igeo index was calculated using the following equation: Igeo = log2 Cn/1.5 Cb, where Cn is the concentration of the element n determined in sediments, Cb is the average concentration at the NBL of this element, and 1.5 is the factor used to minimise lithological variations of the NBL. According to Igeo classification, pollution degrees can be delimitated as: very strongly polluted (Igeo > 5), strongly to very strongly polluted (Igeo = 4–5), strongly polluted (Igeo = 3–4), moderately to strong polluted (Igeo = 2–3), moderately polluted (Igeo = 1–2), unpolluted to moderately polluted (Igeo = 0–1) and unpolluted (Igeo < 0).
Data analysis
Spearman rank correlation coefficients were applied to explore the relationships between As concentrations in fish species, water and sediment. The same coefficients were applied to explore relationships between As and water chemistry. The Kruskal–Wallis one-way ANOVA on ranks was used to test for differences in As concentration in abiotic (water and sediments) compartments and fishes between sampled rivers. Each single fish species was analysed separately. Then, the same test was performed on the entire set of As determinations in fish tissues of each site. The rationale behind this analysis was to use and interpret the fish data as a third (biotic) compartment. This approach aimed to characterise not only the level of As contamination in single fish species but also improved the characterisation of the pollution condition in sampled ecosystems. If significant differences were found with the ANOVA, multi-comparisons were performed with the Kruskal–Wallis multiple-comparison Z-value test (Daniel 1978). When a fish species was caught only at two sites, comparisons were made using the Mann–Whitney test.
Results
Physicochemical parameters of water
Sampled rivers were characterised by alkaline waters (Table 1). The pH values fluctuated between 7.9 and 9.9. Water conductivity in surface water was found to be in the range of 1,550–10,200 μS/cm. Water turbidity also varied greatly from almost nil (1 NTU) to more than 500 NTU. Mean water temperature did not surpassed 20 °C ranging from 11 °C in autumn to 25.7 °C in spring months. The pH (r = 0.62; p < 0.05) and turbidity (r = 0.59; p < 0.05) of water were found to be significantly correlated with the concentration of As.
Arsenic in abiotic and biotic compartments
All rivers showed high (albeit variable) concentrations of As in surface waters. Arsenic concentration in surface waters of sampled ecosystems ranged from 55 to 195 μg/L, averaging 125.95 μg/L (Table 1). The one-way ANOVA showed that the level of contamination varied significantly between sampling sites (df: 4; χ2: 15.271 and p value: 0.00417). Particularly, the concentration of As in the Sauce Grande Lake and downstream to the lake (Valle Hermoso) was higher than the value recorded in the Salado River and upstream (Las Oscuras) to the lake (Table 2 and Fig. 2).
Concentrations of As in environmental (water and sediments) and biological (fishes) compartments from river ecosystems of the Pampa Plain. For each site, the mean (filled circle) and one standard deviation (upper and lower lines) are reported. Site codes: 1RS Salado River, 2QS Quequén Salado River, 3LO Las Oscuras (upstream to the Sauce Grande Lake), 4SG Sauce Grande Lake, 5VH Valle Hermoso (downstream to the Sauce Grande Lake)
Arsenic in sediments barely followed (r = 0.361 and p = 0.118) the level of contamination of water. For instance, the Quequén Salado, a river otherwise highly enriched with As in surface waters, showed an extremely low level of As contamination of sediments (Table 1 and Fig. 2). It was remarkable the difference between the concentration of As in sediments of the Sauce Grande Lake when compared with other sites, including those located in the same river, upstream (Las Oscuras) and downstream (Valle Hermoso) to the lake. Sediments in Salado River, Quequén Salado and Las Oscuras were almost unpolluted. Conversely, geochemical indices indicated that sediments in Sauce Grande and Valle Hermoso were moderately to highly polluted with As (Table 1). The average concentration of As in edible tissues of fish from the Pampa Plain was 1.76 μg/g (n = 74). Fish samples collected in sites located in the more contaminated Sauce Grande River displayed higher concentrations of As. Particularly, As concentrations in fishes from Las Oscuras and Valle Hermoso were higher than samples from Quequén Salado (Table 2 and Fig. 2).
Arsenic in fish species
Large differences in the concentration of As between some species were observed. Moreover, two groups may be clearly and significantly (χ 2 = 38.48 and p = 0.000014) defined in relation to the level of As concentration in fish species. A high-impacted group conformed by Pimelodella laticeps, Cheirodon interruptus and Jenynsia multidentata and a low-impacted group represented by Bryconamericus iheringii, Oligosarcus jenynsii, Rhamdia quelen and Odontesthes bonariensis (Fig. 3). Comparatively, even the low-impacted group species of the Pampa Plain showed As concentrations among the highest reported worldwide (Table 3).
Concentrations of As in muscle of fish species of the Pampa Plain. For each species, the mean (filled circle) and one standard deviation (upper and lower lines) are reported. Species codes: Ae: Astyanax eigenmanniorum; Af: Australoheros facetus; Bi: Bryconamericus iheringii; Ci: Cheirodon interruptus; Cv: Cyphocharax voga; Jm: Jenynsia multidentata; Ob: Odontesthes bonariensis; Oj: Oligosarcus jenynsii; Pl: Pimelodella laticeps; Rq: Rhamdia quelen
Statistical analysis significantly discriminated between Pimelodella laticeps and Cheirodon interruptus with higher concentration of As than Bryconamericus iheringii, Odontesthes bonariensis and Oligosarcus jenynsii. In addition, the small Cyprinodontiformes Jenynsia multidentata showed higher concentrations of As than the inland silverside Odontesthes bonariensis. The scaleless species collected in this study also differed significantly regarding their level of contamination with As. The level of natural contamination with As in the small catfish Pimelodella laticeps was higher than the concentration observed in the large Rhamdia quelen. Interestingly, the pattern in As accumulation in these species in relation with the concentration of As in water and sediments also differed. The level of As in Pimelodella laticeps was consistently high regardless of the level of natural pollution of water and sediments of the site in which it was collected. Indeed, As concentration in Pimelodella laticeps was not related at all with water and sediment concentrations (Table 4) and did not show significant differences among sampling sites (Table 2). Conversely, Rhamdia quelen showed significant differences in As concentration among sites with different degree of natural pollution by As (Table 2). Accordingly, the As concentration in this species was marginally related (p < 0.10) with As concentrations in water and sediments (Table 4).
None of the species showed a significant relationship between the concentration of As in muscle and in water. On the other hand, two species showed significant correlations (p < 0.05) between the concentration of As in their tissues and the corresponding level in sediments. One of these species, Cyphocharax voga, is a detritivore which largely rely on bottom sediments for feeding. This explanation did not seem to be as straightforward for the small characid Astyanax eigenmanniorum which actively prey upon small invertebrates and plant remains. Nevertheless, in line with these relationships, the level of As in Cyphocharax voga and Astyanax eigenmanniorum was significantly higher (Table 2) in those rivers where sediments showed higher concentrations of this element (Table 1).
Discussion
Water chemistry and As
Streams of the Pampa Plain have a large range of physical and chemical conditions. Regional differences may be due to variations in water availability, parent material (Feijoó and Lombardo 2007) and land use practices (Rosso and Fernández Cirelli 2012). A common feature of Pampa Plain streams is the invariable alkaline pH (almost constantly above 8) that characterises their surface waters (Feijoó and Lombardo 2007; Rosso et al. 2011a, b). Similarly, groundwaters range from neutral (6.99) to alkaline (8.66) resulting from silicate and carbonate reactions (Smedley et al. 2002). The high pHs, together with the presence of young age As-rich loess sediments and slow groundwater flow, have enabled the accumulation of the high concentrations of As in solution without significant opportunity for flushing of the aquifer to enable their removal (Smedley et al. 2002; Blanco et al. 2006). This explains why groundwater As commonly correlates positively with pH. Interestingly, the same positive correlation was observed in this survey between As in surface waters and pH. An explanation behind the observed relationship between As and water turbidity does not seem to be as straightforward. The probable role of surface run-off contributing to both sediment (increasing turbidity) and As loads should be evaluated. Soil As and As-charged agro-chemicals could be participating in this process.
Pollution condition of sampled ecosystems
Arsenic concentrations in water samples were above the World Health Organisation (WHO 2008) and United State Environmental Protection Agency (USEPA) guidelines for drinking water (10 μg/L) in all sites during all collection campaigns. Guide levels for the protection of the aquatic biota (15 μg/L) and drinking water (10 μg/L) proposed by local authorities (SRH 2006) were also surpassed in all samples. An extensive spatial variability of As concentrations in water and sediments of sampled ecosystems was observed. This closely resembles the complex pattern in the geographic distribution of As earlier recorded in surface drainages (Rosso et al. 2011b) and groundwater (Paoloni et al. 2005; Paoloni et al. 2009) of this region.
The Sauce Grande River was by far the most naturally polluted ecosystem. Particularly, Sauce Grande and Valle Hermoso sites showed consistently high levels of As in water, sediment and fish samples. A similar condition, but of lower magnitude, was identified in the Salado River. Upstream to the Sauce Grande Lake (Las Oscuras), sediments and water were relatively unpolluted with As. These results suggested that the altered river channel morphology driven by the lake may influence the accumulation of As in depositional zones of the ecosystem. Higher concentrations of As in sediments at depositional zones within a river ecosystem were already reported (Casper et al. 2004). The enriched sediments, in turn, may behave as an important source of As for downstream environments and their associated biota. This was clearly reflected in our results (see the continuum Las Oscuras, Sauce Grande and Valle Hermoso in Table 1).
Among the freshwater biota, fish species usually actively alternate between available habitats within a river network. In the Sauce Grande River, this implies that fish species would alternate between high- and low-impacted sites. Accordingly, in spite of the low concentrations of As upstream to the lake (Las Oscuras), fish inhabiting this river reach showed high concentrations of this metalloid in muscle, probably previously accumulated in higher polluted reaches. On the other hand, “local” uptake of As in Las Oscuras is also possible since bioaccumulation can develop at any metal concentration without minimum level requirements (Amiard et al. 1987; Marcovecchio 2004). Nevertheless, many animals are able to detect and avoid toxicants (Atchison et al. 1996). Therefore, evaluating an organism’s behaviour is integral in determining its degree of exposure (Weber 1997).
A particular situation was highlighted in the Quequén Salado River. Water was highly enriched with As, but the level of this element in sediments and fish samples was low. This is not an isolated and fortuitous result. The content of a metal in water may only reflect short-term events and does not necessarily mean that biota and sediments are polluted with this metal (Sager and Pucsko 1991). Moreover, metals in sediments and in organisms with a long life cycle, as fish, usually reflect trends of their content in whole aquatic ecosystems and water catchment area of many years and appear to be weakly related with their content in water (Gladyshev et al. 2001). This has strong implications for monitoring programmes (Ion et al. 1997). In this respect, when planning monitoring programmes for As, it is important to remind that biomagnification does not occur; rather, As concentrations decrease as trophic level increases (Culioli et al. 2009b).
Geochemical indices indicated that sediments of sampled rivers ranged from mostly unpolluted to strongly polluted. Particularly, the most naturally polluted sediments reported in this study (27.8 μg/g in SG) largely surpassed the level (10.4 μg/g) of As contamination in sediments of a river draining a copper processing industry in UK (Casper et al. 2004) and almost paralleled (29.9 μg/g) the level of contamination of the highly impacted Yangtze River in China (Yang et al. 2008). However, this pollution condition must be considered only as preliminary. In sediments, metals can be present in a number of chemical forms, exhibiting different physical and chemical behaviours with respect to chemical interactions, mobility, biological availability and potential toxicity (Singh et al. 2005). This explains why the suitability of using total metal contents as a criterion for the classification of sediments as polluted from a legal point of view has been debated (Keller and Hammer 2004). An adequate criterion for environmental risk assessment must include both total metal content and bioavailable metal fraction (Liu et al. 2009). In spite of all these caveats, bioavailability of As from sediments of sampled rivers did not seem to be impaired. There were low polluted sediments with highly contaminated fish (Las Oscuras), but the highly polluted sediments always showed highly contaminated fish (Sauce Grande, Valle Hermoso and Salado River).
As expected, concentrations of As in fish species of the Pampa Plain were much higher than As concentrations found in fish (0.005–0.201 μg/g) inhabiting pristine aquatic ecosystems (Gutleb et al. 2002). On average, a fish from the Pampa Plain showed higher (1.76 μg/g; n = 74) As in muscle than the upper limit suggested for several freshwater fish species (Donohue and Abernathy 1999). These authors reported that the total As in freshwater fish tissues ranged from 0.007 to 1.46 μg/g. Interestingly, the average concentration of As in fish from the Pampa Plain was still higher than the lower limit of the range (0.94–15.1 μg/g) reported for cultured fish from blackfoot disease areas in Taiwan (Liao and Ling 2003).
Pollution condition of sampled species
Some trace metals are naturally required in small amounts by fish as constituents of enzymes and are essential for healthy development. On the other hand, many trace metals are acutely or chronically toxic to fish and other organisms (Bryan 1971). Particularly, McIntyre and Linton (2011) suggested that effects appear to occur when fish tissue concentrations of As reach 2–5 μg/g. Following our results, three species Jenynsia multidentata, Cheirodon interruptus and Pimelodella laticeps should be at risk and conservation efforts as well as ecotoxicological studies should be addressed.
Fish can be precise indicators in estimating the status of pollution of water environments, and several species have been widely used in monitoring programmes (Håkanson 1984; Chevreuil et al. 1995; Andres et al. 2000; Szarek-Gwiazda and Amirowicz 2006). Nevertheless, not all fish species are useful bioindicators for all the trace metals: species able to regulate their body levels of any metal (at least partly) should be rejected as bioindicators for this metal (Bryan 1984). In the arsenic-rich ecosystems of the Pampa Plain, Odontesthes bonariensis and Bryconamericus iheringii showed low concentrations of As and negative and low correlations coefficients with abiotic compartments. These results suggested that these species may be able to regulate the level of As in muscle. Conversely, the concentration of As in the scaleless silurid Rhamdia quelen closely followed the level of As in water and sediments. Perhaps, the intimate interaction between this large catfish and sediments in addition to its intimate contact with water (due to the lack of scales) may help to explain this pattern. Irrespective of this, regulation of As acquisition seemed to be almost nil. This makes Rhamdia quelen, which is a common species in freshwater ecosystems of the Pampa Plain, a good candidate for biomonitoring programmes. A biomonitor species should make it possible to determine the “quantity” of the environment or its change (Markert et al. 1997). The detritivore Cyphocharax voga and the small characid Astyanax eigenmanniorum are promissory candidates for biomonitoring programmes as well. Both species showed significant correlation coefficients with As in sediments and accordingly showed significant differences in their level of contamination between sites with different degree of sediment pollution by As. As Cyphocharax voga, other bottom-dwelling fishes also intimately reflect the metal concentrations in surface sediments (Marcovecchio 1988; Oyewale and Musa 2006). Pimelodella laticeps showed consistently high levels of As in spite of the variable level of natural contamination of sites in which it was collected. This means that this species is able to integrate concentrations of As through time, and as a consequence, it should be considered a good candidate for bioindicator analysis. A bioindicator species should provide information on the “quality” of the environment or its change (Markert et al. 1997).
When considering fish results, it is mandatory to remark that only muscle samples were processed in order to quantify total As concentrations. As known, content of metals in other tissues and organs of fishes, for instance, livers, kidneys and gills, can be substantially higher and correlate better with the contents in water and sediments (Moiseenko 1999). Similarly, intrinsic factors such as growth size, mass, age, gender, sexual maturity, physiology and stress have shown to influence the accumulation of trace metals in marine organisms (Phillips and Rainbow, 1993). The role of these factors in trace metal accumulation has not been deeply examined in fish outside marine ecosystems. Particularly, Maher et al. (1999) reported that As concentrations were found not to be related to the gender of the fish. Maher et al. (1999) further reported that As concentrations were found not to be related to the age of fish, and where significant correlations with mass were found, these were negative and did not explain much of the variability.
The inland silverside Odontesthes bonariensis is by far the most consumed species among those sampled in this study. Fortunately, the level of As in muscle of this species was below the maximum suggested for human consumption by several international authorities such as Hong Kong (2.3 μg/g; Hong Kong Government Hong Kong Government. Food Adulteration (Metallic Contamination) Regulations. Laws of Hong Kong 2 1987), New Zealand and Australia (2.0 μg/g; Australia New Zealand Food Authority 1999) and Croatia (2.0 μg/g; Has-Schön et al. 2008). However, the level of As in Odontesthes bonariensis of Quequén Salado was slightly over the limit suggested by United States (1.2 μg/g; USEPA 2000). Overall, As concentrations in most fish species were higher than the maximum allowed concentration in United States. Only three species showed mean As concentrations over 2 μg/g, a maximum adopted by most countries.
In a review of the latest scientific evidence from epidemiological data, the lower limit on the benchmark dose of inorganic arsenic for a 0.5 % increased incidence of lung cancer was determined to be 3.0 μg/kg body weight (210 μg in a 70-kg adult) per day (FAO/WHO, 2010). Following the latest (period 2005–2010) estimation of fish consumption (13.9 g/day of fish for a 70-kg adult) by local authorities, when consuming the most polluted Odontesthes bonariensis (1.23 μg/g), a person would receive around 17 μg of As per day. This value is one order of magnitude lower than the lower limit mentioned above. Nevertheless, this value only reflects total As acquisition. Interesting and diverse aspects relevant to arsenic toxicity, arsenic biotransformations and the determination of arsenic species in freshwater fish were already highlighted (Jankong et al. 2007). Approximately 90 % of As in fish is organic As (Mandal and Suzuki 2002) that is of low toxicity (IPCS, 2001). Arsenobetaine is the dominant species in marine fish, but speciation in freshwater fish is much more variable (McIntyre and Linton 2011). Indeed, Jankong et al. (2007) demonstrated that although arsenate was the major extractable arsenical in freshwater fish from uncontaminated waters, dimethylarsinate was by far the dominant arsenic species in fish from contaminated sites. Moreover, some results indicated that there is a correlation between fish family and the arsenic speciation pattern (Šlejkovec et al. 2004).
The inland silverside, Odontesthes bonariensis, is one of the few species of the family Atherinopsidae inhabiting freshwater ecosystems. Most of its relatives are marine species. Therefore, whether this species behaves as a marine or freshwater species in relation to biotransformation and accumulation of different arsenic species is an interesting task that deserves further research.
References
Amiard, J. C., Amiard-Triquet, C., Berthet, B., & Metayer, C. (1987). Comparative study of the patterns of bioaccumulation of essential (Cu, Zn) and non-essential (Cd, Pb) trace metals in various estuarine and coastal organisms. Journal of Experimental Marine Biology and Ecology, 106, 73–89.
Andres, S., Ribeyre, F., Tourencq, J. N., & Boudou, A. (2000). Interspecific comparison of cadmium and zinc contamination in the organs of four fish species along a polymetallic pollution gradient (Lot River, France). Science of the Total Environment, 248, 11–25.
APHA (American Public Health Association). (1995). Standard Methods for Examination of Water and Wastewater (19th ed.). Washington: American Public Health Association.
Atchison, G. J., Sandheinrich, M. B., & Bryan, M. D. (1996). Effects of environmental stressors on interspecific interactions of aquatic animals. In M. C. Newman & C. H. Jagoe (Eds.), Quantitative ecotoxicology: A hierarchical approach (pp. 319–345). Boca Raton: Lewis.
ATSDR (Agency for Toxic Substances and Disease Registry). (2007). CERCLA Priority List of Hazardous Substances. http://www.atsdr.cdc.gov/cercla/05list.htmlN.
Australia New Zealand Food Authority. (1999). Food Standards Code in the Gazette, Australia New Zealand Food Authority.
Biagini, R., Rivero, M., Salvador, M., & Córdoba, S. (1978). Hidroarsenicismo crónico y cáncer de pulmón. Archivos Argentinos de Dermatología, 28, 151–158.
Blanco, M. C., Paoloni, J. D., Morrás, H. J. M., Fiorentino, C. E., & Sequeira, M. (2006). Content and Distribution of Arsenic in Soils, Sediments and Groundwater Environments of the Southern Pampa Region, Argentina. Environmental Toxicology, 21, 561–574.
Bryan, G. W. (1971). The effects of heavy metals (other than mercury) on marine and estuarine organisms. Proceedings of the Royal Society of London, 177, 389–410.
Bryan, G. W. (1984). Pollution due to heavy metals and their compounds. Marine Ecology, 5, 1289–1431.
Casper, S. T., Mehra, A., Farago, M. E., & Gill, R. A. (2004). Contamination of surface soils, river water and sediments by trace metals from copper processing industry in the Churnet River Valley, Staffordshire, UK. Environmental Geochemistry and Health, 26, 59–67.
Chen, C. J., Hsueh, Y. M., Tseng, M. P., Lin, Y. C., Hsu, L. I., & Chou, W. L. (2001). Individual susceptibility to arseniasis. In W. R. Chappell, C. O. Abernathy, & C. L. Calderon (Eds.), Arsenic exposure and health effects. Oxford: Elsevier.
Cheung, K. C., Leung, H. M., & Wong, M. H. (2008). Metal concentrations of common freshwater and marine fish from the Pearl River Delta, South China. Archives of Environmental Contamination and Toxicology, 54, 705–715.
Chevreuil, M., Carru, A.-M., Chesterikoff, A., Boët, P., Tales, E., & Allardi, J. (1995). Contamination of fish from different areas of the river Seine (France) by organic (PCB and pesticides) and metallic (Cd, Cr, Cu, Fe, Mn, Pb and Zn) micropollutants. Science of the Total Environment, 162, 31–42.
Ciardullo, S., Aureli, F., Raggi, A., & Cubadda, F. (2010). Arsenic speciation in freshwater fish: Focus on extraction and mass balance. Talanta, 81, 213–221.
Culioli, J.-L., Calendini, S., Mori, C., & Orsini, A. (2009a). Arsenic accumulation in a freshwater fish living in a contaminated river of Corsica, France. Ecotoxicology and Environmental Safety, 72, 1440–1445.
Culioli, J.-L., Fouquoirea, A., Calendini, S., Mori, C., & Orsini, A. (2009b). Trophic transfer of arsenic and antimony in a freshwater ecosystem: A field study. Aquatic Toxicology, 94, 286–293.
Daniel, W. W. (1978). Applied nonparametric statistics. Boston: Houghton Mifflin Company.
Das, H. K., Mitra, A. K., Sengupta, P. K., Hossain, A., Islam, F., & Rabbani, G. H. (2004). Arsenic concentrations in rice, vegetables, and fish in Bangladesh: a preliminary study. Environment International, 30, 383–387.
Donohue, J. M., & Abernathy, C. O. (1999). Exposure to inorganic arsenic from fish and shellfish. In W. R. Chappell, C. O. Abernathy, & R. L. Calderon (Eds.), Arsenic exposure and health effect. Oxford: Elsevier.
FAO/WHO. (2010). Summary and conclusions of the 72 meeting of the Joint FAO/WHO Expert Committee on Food Additives. Rome, Food and Agriculture Organization of the United Nations; Geneva, World Health Organization (JECFA/72/SC; http://www.who.int/foodsafety/chem/summary72_rev.pdf).
Feijoó, C. S., & Lombardo, R. J. (2007). Baseline water quality and macrophyte assemblages in Pampean streams: a regional approach. Water Research, 41, 1399–1410.
Gladyshev, M. I., Gribovskaya, I. V., Moskvicheva, A. V., Muchkina, E. Y., Chuprov, S. M., & Ivanova, E. A. (2001). Content of metals in compartments of ecosystem of a siberian pond. Archives of Environmental Contamination and Toxicology, 41, 157–162.
Gutleb, A. C., Helsberg, A., & Mitchell, C. (2002). Heavy metal concentrations in fish from a pristine rainforest valley in Peru: A baseline study before the start of oil-drilling activities. Bulletin of Environment Contamination and Toxicology, 69, 523–529.
Håkanson, L. (1980). An ecological Risk index for aquatic pollution control: a sedimentological approach. Water Research, 14, 975–1001.
Håkanson, L. (1984). Metals in fish and sediments from the River Kolbäcksån water system, Sweden. Archiv für Hydrobiologie, 101, 373–400.
Has-Schön, E., Bogut, I., Rajkovic, V., Bogut, S., Cacic, M., & Horvatic, J. (2008). Heavy metal distribution in tissues of six fish species included in human diet, inhabiting freshwaters of the nature park ‘‘Hutovo Blato’’ (Bosnia and Herzegovina). Archives of Environmental Contamination and Toxicology, 54, 75–83.
Has-Schön, E., Bogut, I., & Strelec, I. (2006). Heavy metal profile in five fish species included in human diet, domiciled in the end flow of river Neretva (Croatia). Archives of Environmental Contamination and Toxicology, 50, 545–551.
Hong Kong Government. (1987). Food Adulteration (Metallic Contamination) Regulations. Laws of Hong Kong 2; Chapter 132.
IARC (International Association for Research on Cancer). (1987). IARC monographs on the evaluation of carcinogenic risks of chemicals to humans. Suppl. 7: Overall evaluations of Carcinogenicity: Updating of IARC Monographs. IARC, Lyon. 1:42.
Ion, J., de Lafontaine, Y., Dumont, P., & Lapierre, L. (1997). Contaminant levels in St Lawrence River yellow perch (Perca flavescens): spatial variation and implications for monitoring. Canadian Journal of Fisheries and Aquatic Sciences, 54, 2930–2946.
IPCS. (2001). Arsenic and arsenic compounds, 2nd ed. Geneva, World Health Organization, International Programme on Chemical Safety (Environmental Health Criteria 224; http://whqlibdoc.who.int/ehc/WHO_EHC_224.pdf).
Jankong, P., Chalhoub, C., Kienzl, N., Goessler, W., Francesconi, K. A., & Visoottiviseth, P. (2007). Arsenic accumulation and speciation in freshwater fish living in arsenic-contaminated waters. Environmental Chemistry, 4, 11–17.
Keith, L. H. (1991). Environmental Sampling and Analysis: A Practical Guide. Boca Raton: Lewis Publ/CRC Press.
Keller, C., & Hammer, D. (2004). Metal availability and soil toxicity after repeated croppings of Thlaspi caerulescens in metal contaminated soils. Environmental Pollution, 131, 243–254.
Kuznetsova, A. I., Zarubina, O. V., & Leonova, G. A. (2002). Comparison of Zn, Cu, Pb, Ni, Cr, Sn, Mo concentrations in tissues of fish (roach and perch) from lake Baikal and Bratsk reservoir, Russia. Environmental Geochemistry and Health, 24, 205–213.
Liao, C. M., & Ling, M. P. (2003). Assessment of Human Health Risks for Arsenic Bioaccumulation in Tilapia (Oreochromis mossambicus) and Large-Scale Mullet (Liza macrolepis) from Blackfoot Disease Area in Taiwan. Archives of Environmental Contamination and Toxicology, 45, 264–272.
Ling, M. P., Hsu, H. T., Shie, R. H., Wu, C. C., & Hong, Y. S. (2009). health risk of consuming heavy metals in farmed tilapia in central Taiwan. Bulletin of Environment Contamination and Toxicology, 83, 558–564.
Liu, J., Li, Y., Zhang, B., Cao, J., Cao, Z., & Domagalski, J. (2009). Ecological risk of heavy metals in sediments of the Luan River source water. Ecotoxicology, 18, 748–758.
Maher, W., Goessler, W., Kirby, J., & Raber, G. (1999). Arsenic concentrations and speciation in the tissues and blood of sea mullet Mugil cephalus from Lake Macquarie NSW, Australia. Marine Chemistry, 68, 169–182.
Mandal, B. K., & Suzuki, K. T. (2002). Arsenic round the world: A review. Talanta, 58, 201–235.
Marcovecchio, J. E. (1988). Estudio comparativo de la distribución de los metales traza mercurio, cadmio y zinc en organismos de dos zonas estuariales de Argentina: Bahía Blanca y desembocadura del Río de la Plata. Unpublished Ph.D. Thesis, UNMDP (Argentina). pp. 222.
Marcovecchio, J. E. (2004). The use of Micropogonias furnieri and Mugil liza as bioindicators of heavy metals pollution in La Plata river estuary, Argentina. Science of the Total Environment, 323, 219–226.
Markert, B., Oehlmann, J., & Roth, M. (1997). General aspects of heavy metal monitoring by plants and animals. In K. S. Subramanian, & G. V. Iyengar (Eds.), Environmental biomonitoring: Exposure assessment and specimen banking (pp. 18–29). Washington: Am Chem Soc Symp Ser No. 654.
McIntyre, D. O., & Linton, T. K. (2011). Fish Physiology, 31, (PART B.). Elsevier. doi:10.1016/S1546-5098(11)31028-X.
Moiseenko, T. I. (1999). Estimation of an ecological danger under pollution of waters by metals. Vodnyie Resursy, 26, 186–197.
Mora, M. A., Papoulias, D., Nava, I., & Buckler, D. R. (2001). A comparative assessment of contaminants in fish from four resacas of the Texas, USA–Tamaulipas, Mexico border region. Environment International, 27, 15–20.
Mudrock, A., & Macknight, S. D. (1994). Techniques for aquatic sediments sampling. Boca Raton: Lewis Publ.
Müller, G. (1979). Schwermetallen in den Redimen des rheins. Veranderrugen Seit. Umschau, 79, 778–783.
Oyewale, A. O., & Musa, I. (2006). Pollution assessment of the lower basin of Lakes Kainji/Jebba, Nigeria: heavy metal status of the waters, sediments and fishes. Environmental Geochemistry and Health, 28, 273–281.
Paoloni, J. D., Sequeira, M. E., Espósito, M. E., Fiorentino, C. E., & Blanco, M. C. (2009). Arsenic in water resources of the Southern Pampa Plains, Argentina. Journal of Environmental and Public Health. doi:10.1155/2009/216470.
Paoloni, J. D., Sequeira, M. E., & Fiorentino, C. E. (2005). Mapping of arsenic content and distribution in groundwater in the Southeast Pampa Argentina. Journal of Environmental Health, 67(8), 50–53.
Parada, J. M. (2008). A pedal corer for the quantitative sampling of sediments and benthic organisms in submerged areas accessible on foot. Hydrobiologia, 610, 351–354.
Phillips, D. J. H., & Rainbow, P. S. (1993). Biomonitoring of Trace Aquatic Contaminants (pp. 1–371). London: Elsevier.
Rashed, M. N. (2001). Monitoring of environmental heavy metals in fish from Nasser Lake. Environment International, 27, 27–33.
Rosso, J. J., & Fernández Cirelli, A. (2012). Effects of land use on environmental conditions and macrophytes in prairie lotic ecosystems. Limnologica. http://dx.doi.org/10.1016/j.limno.2012.06.001.
Rosso, J. J., Puntoriero, M. L., Troncoso, J. J., Volpedo, A. V., & Fernández Cirelli, A. (2011a). Occurrence of Fluoride in Arsenic-rich Surface Waters: a Case Study in the Pampa Plain, Argentina. Bulletin of Environmental Contamination and Toxicology, 87, 409–413.
Rosso, J. J., Troncoso, J. J., & Fernández Cirelli, A. (2011b). Geographic Distribution of Arsenic and Trace Metals in Lotic Ecosystems of the Pampa Plain, Argentina. Bulletin of Environment Contamination and Toxicology, 86, 129–132.
Sager, M., & Pucsko, R. (1991). Trace element concentrations of oligochaetes and relations to sediment characteristics in the reservoirs at Altenworth/Austria. Hydrobiologia, 226, 39–49.
Schenone, N., Volpedo, A. V., & Fernández Cirelli, A. (2007). Trace metal contents in water and sediments in Samborombón Bay wetland, Argentina. Wetlands Ecology and Management, 15, 303–310.
Shukla, J. P., & Pandey, K. (1984a). Impaired spermatogenesis in arsenic treated freshwater fish Colisa fasciatus (Bl. and Sch.). Toxicology Letters, 21, 191–195.
Shukla, J. P., & Pandey, K. (1984b). Impaired ovarian functions in arsenic treated freshwater fish Colisa fasciatus (Bl. and Sch.). Toxicology Letters, 20, 1–3.
Singh, K. P., Mohan, D., Singh, V. K., & Malik, A. (2005). Studies on distribution and fractionation of heavy metals in Gomti river sediments—A tributary of the Ganges, India. Journal of Hydrology (Amst), 312, 14–27.
Šlejkovec, Z., Bajc, Z., & Doganoc, D. Z. (2004). Arsenic speciation patterns in freshwater fish. Talanta, 62, 931–936.
Smedley, P., & Kinniburg, D. (2002). A review of the source, behaviour and distribution of arsenic in natural waters. Applied Geochemistry, 17, 517–568.
Smedley, P. L., Nicolli, H. B., Macdonald, D. M. J., Barros, A. J., & Tullio, J. O. (2002). Hydrogeochemistry of arsenic and other inorganic constituents in groundwaters from La Pampa, Argentina. Applied Geochemestry, 17, 259–284.
SRH (Subsecretaria de Recursos Hídricos de Argentina). (2006). Niveles Guía de Calidad de Aguas. http://hidricos.obraspublicas.gov.ar/calidad_del_agua_actividades.htm. Accessed 11 July 2011.
Szarek-Gwiazda, E., & Amirowicz, A. (2006). Bioaccumulation of trace elements in roach, silver bream, rudd, and perch living in an inundated opencast sulphur mine. Aquatic Ecology, 40, 221–236.
Turekian, K. K., & Wedephol, K. H. (1961). Distribution of the elements in some major units of the earth’s crust. Geological Society of American Chemistry, 51(7), 844–851.
US Food and Drug Administration (USFDA). (1993). Guidance document for arsenic in shellfish. Washington: US Food and Drug Administration.
USEPA (U.S. Environmental Protection Agency). (1997). IRIS (Integrated Risk Information System). On-line database maintained in Toxicology Data Network (TOXNET). Bethesda MD: National Library of Medicine.
USEPA (U.S. Environmental Protection Agency). (2000). Guidance for assessing chemical contaminant, data for use in fish advisories. Volume 2. Risk assessment and fish consumption limits (3rd ed.). DC: Washington.
Weber, D. N. (1997). Mechanisms of behavioral toxicology: An integrated approach. American Zoologist, 37, 343–345.
WHO. (2008). Guidelines for drinking-water quality, 3rd edition incorporating 1st and 2nd addenda. Vol. 1. Recommendations (pp. 306–308b). Geneva: World Health Organization. (http://www.who.int/water_sanitation_health/dwq/GDW12rev1and2.pdf).
Yang, Z., Wang, Y., Shen, Z., Niu, J., & Tang, Z. (2008). Distribution and speciation of heavy metals in sediments from the mainstream, tributaries, and lakes of the Yangze River catchment of Wuhan, China. Journal of Hazardous Materials, 166(2–3), 1186–1194.
Acknowledgments
Authors are indebted to the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) and the Universidad de Buenos Aires (UBA) for financial support. We thank Juan José Troncoso for his technical assistance in sample processing. We also thank Alejandro Travi, Diego Laura, Hugo Carrizo and Luis Rey Ocampo for their collaboration in the field. Pablo Blanco and his troop at the Sauce Grande Lake were crucial for logistic aspects of this investigation.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Rosso, J.J., Schenone, N.F., Pérez Carrera, A. et al. Concentration of arsenic in water, sediments and fish species from naturally contaminated rivers. Environ Geochem Health 35, 201–214 (2013). https://doi.org/10.1007/s10653-012-9476-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10653-012-9476-9