Abstract
Thirty-two brownfield sites from the city of Wolverhampton were selected from those with a former industrial use, wasteland or areas adjacent to industrial processes. Samples (<2 mm powdered soil fraction) were analysed, using inductively coupled plasma–atomic emission spectrometry (ICP–AES) for 20 elements. Loss on ignition and pH were also determined. A five-step chemical sequential extraction technique was carried out. Single leach extraction with 0.12 M hydrochloric acid of Pb, Cu and Zn in soil was determined as a first approximation of the bioavailability in the human stomach. Some of the sites were found to have high concentrations of the potentially toxic elements Pb, Zn, Cu and Ni. The partitioning of metals showed a high variability, however a number of trends were determined. The majority of Zn was partitioned into the least chemically stable phases (steps 1, 2 and 3). The majority of Cu was associated with the organic phase (step 4) and the majority of Ni was fractionated into the residue phase (step 5). The majority of Pb was associated with the residue fraction (step 5) followed by Fe–Mn oxide fraction (step 3). The variability reflects the heterogeneous and complex nature of metal speciation in urban soils with varied historic histories. There was a strong inverse linear relationship between the metals Ni, Zn and Pb in the readily exchangeable phase (step 1) and soil pH, significant at P < 0.01 level. There was a significant increase (P < 0.05) in the partitioning of Cu, Ni and Zn into step 4 (the organic phase) in soils with a higher organic carbon content (estimated by loss on ignition). Copper was highly partitioned into step 4 as it has a strong association with organics in soil but this phase was not important for the partitioning of Ni or Zn. The fractionation of Ni, Cu and Zn increased significantly in step 3 when the total metal concentration increases (P < 0.01). The Fe–Mn oxide fraction becomes more important in soils elevated in these metals, possibly due to the scavenging of metals by oxides. Cu and Pb extracted by HCl was statistically similar to the sum of the metals in steps 1 to 4 (P < 0.01) and HCl available Zn was statistically similar to the sum of Zn in steps 1 to 3 (P < 0.01). Step 4 (the organic phase) was not an important phase for Zn, so it was concluded that any Cu, Zn and Pb present in soil in a nonresidue phase would be potentially available for uptake into the human system once soil has been ingested.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Urban geochemical mapping in the UK has been described and discussed by Fordyce et al. (2005) for 21 cities, including the city of Wolverhampton, which is known to exhibit high concentrations of potentially harmful elements (PHEs) (Kelly et al. 1996; Kelly 1997; Fordyce et al. 2005).
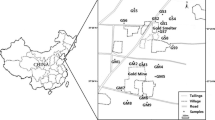
Wolverhampton, with a population of around 236,600, lies to the north east of Birmingham in the West Midlands, UK (Fig. 1) and covers an area of 6,945 hectares. The geology of Wolverhampton is divided by the Western Boundary Fault that separates Triassic sandstones in the north-northwest and Carboniferous coal measures in the southeast (Hains 1969). The Carboniferous coal bands have been extensively worked, leaving a modified landscape and a disrupted soil profile (Bridge et al. 1997). The Triassic sandstones and Permian–Carboniferous sandstone and mudstone sequences to the north of the Western Boundary Fault are covered by a layer of locally derived glacial till, up to 12 m thick in places (Hains et al. 1969; Bridge et al. 1997).
The region has had mining and metal processing activities since the 16th century, which were geographically constrained to the local coal and iron stone resources. There are no major river systems in the city; and the building of a canal network in the late 18th century and a rail network in the mid 19th century provided important transport links for industry. Linear industry grew adjacent to the infrastructure served by canal wharfs and rail lines. Urban development during the industrial revolution of the 18th and 19th centuries was responsible for the conversion of the agricultural market town into a regionally important manufacturing and commercial centre. The landscape was still predominantly rural, until urban development schemes during the 1920s and 1930s attracted manufacturing companies such as bicycle, vehicle and aircraft producers along with the associated component industries, and residential development to house the workforce (Upton 1998). Residential housing in this region has since developed over, or adjacent to, sites of former mining and metal processing industries. The industrial activity was dominated by the manufacturing of steel, metal component parts, metal plating, galvanising and the paint and pigment industry (Upton 1998). Industrial development has declined in the UK over the last 40 years, a trend reflected in Wolverhampton, but the region has modernised towards electronics and component-based industries. Metal-based industries still play an important role in the local economy with the UK’s only Cu refinery located a few kilometres to the east of the Wolverhampton administrative boundary (Upton 1998; EA 2001). A regional road-building scheme in the southeast has improved transport links with the West Midlands and the rest of the UK; redevelopment of former industrial areas and the canal network, used now for recreational activities, are part of the ongoing regional development scheme.
Former industries, many now closed, and the land and buildings used for other, including residential, purposes, have left a legacy of contamination by potentially hazardous elements (PHEs). Atmospheric and terrestrial contamination enhances the concentrations of many PHEs in the urban environment. Thus the geological input of trace metals into urban soils has been exceeded by anthropogenic input through industrialisation and urbanisation, resulting in elevated metal concentrations (Thornton 1991; Facchinelli et al. 2001; Fordyce et al. 2005). The geochemical signature of urban ground, which is often disturbed and reused as land use changes, is usually very different from that of the surrounding rural areas, and soils frequently have an elevated pH due to the large amounts of cement and concrete used in construction. Urban gardens and parks, which might appear undisturbed, show enhanced concentrations of PHEs from atmospheric emissions from industrial and utility point sources and traffic, urban runoff, littering and changes in land use (Ander et al. 2001; Fordyce et al. 2005).
The impact of specific human activities has been reported in a number of surveys. The proximity to industrial activity such as factories, scrap metal yards, battery plants, waste ground and repair garages was found to significantly increase the level of Pb in adjacent garden soils and house dusts (for example: Davies et al. 1987; Rieuwerts et al. 2000; Ander et al. 2001; Fordyce et al. 2005). Kelly et al. (1996) reported a spatial variation in metal concentration from surface and subsurface soils in Wolverhampton, higher values being reported in areas on or adjacent to former or contemporary industrial activity. The influence and nature of the adjacent road, the property age and the degree of recent redecoration, particularly the contribution of Pb-rich paint chips from stripping and sanding of internal and external surfaces, were found to be significant factors influencing Pb levels in and around UK houses (Thornton et al. 1985; Davies et al. 1987; Culbard et al. 1988).
The classical infectious diseases of childhood have been overtaken by a group of conditions to which pollutants in the environment may be contributing (ATSDR 2002), examples include asthma (CDC 1998), childhood cancer (Zahm and Devesa 1995) and autism or autistic spectrum disorder (ASD) (Palmer et al. 2006). Elevated ASD prevalence in urban areas versus rural areas (Palmer et al. 2006; Williams et al. 2006) suggests an environmental contribution, and an association between heavy metal exposure and ASD has been suggested (Lidsky and Schneider 2005). Nataf et al. (2006) have demonstrated that coproporphyrin levels were elevated in children with autistic disorder relative to control groups. Precoproporphyrin, a specific indicator of heavy-metal toxicity, was also elevated in autistic disorder. That chronic exposure to lead and mercury can contribute to developmental and behavioural disabilities in children is well documented (for example, Myers et al. 1997; Landrigan et al. 2002; Szpir 2006). For lead, impairment can occur even at relatively low levels (Canfield et al. 2003). Although currently children’s blood lead should be below 10 μg/dL, Canfield et al. (2003) suggest that “there may be no threshold for the adverse consequences of lead exposure” and they estimate that considerably more children are adversely affected by exposure to environmental Pb than had been estimated. Manganese in excess affects the adult brain but has also been implicated in diminished intellectual function in children (Wasserman et al. 2006).
Thus the enhanced concentrations of many PHEs in the urban environment and their bioavailability are of interest, particularly in the area of the health of children. Fordyce et al. (2005) point out that the potential hazards posed by PHEs depend on a number of factors including concentration, chemical form and bioavailability. This study aims to assess the concentrations of Pb, Cu, Zn and Ni and the factors influencing their variability and bioavailability in soil sampled from Wolverhampton, UK. This paper reports the results for samples from 32 brownfield sites, sampled under a National Environmental Research Council (NERC) URGENT project as detailed in the paper by Thornton et al. in this volume, which describes investigations of mineralogy and chemical speciation, plant uptake and Pb isotopic source apportionment studies by a research consortium from Imperial College, London, NERC Isotope Geosciences Laboratory, British Geological Survey and the University of Nottingham.
Metals form chemically stable species and associations with major soil constituents, which leads to accumulation in soil (Davies 1990). The least chemically stable fraction is considered the most available to environmentally sensitive receptors. Assessing metal availability is an important part of an exposure and risk assessment; identifying the factors affecting the available portion are therefore important parameters in the assessment of risk. The physicochemical form of a metal in soil is an important control to mobility and bioavailability (West et al. 1999). Metals are regarded as stable if they are incorporated into the silicate lattice or become associated to secondary authigenic precipitates such as carbonates, amorphous Fe–Mn oxides and insoluble phosphates, which fix the metals in soil. Metals become strongly bonded to the surface layer of organic matter, Fe–Mn oxides and clay minerals. Surface adsorption can lead to the metals becoming incorporated into the host by chemosorption. Weaker electrostatic bonding with soil particles is less chemically stable and metals are regarded as more available. Assessments of metal availability are made by measuring the portion of total metal in a soluble or readily exchangeable phase (Ure et al. 1993). Soluble metal species occur as free metal ions [Mn+] and neutral molecules of organic and inorganic compounds (Thornton et al. 1985). It is the free metal ion that is regarded as the most available for plant uptake; the activity has been predicted by total metal concentration and pH for [Ni2+], [Cu2+], [Zn2+] and [Pb2+] (Japony and Young 1994; Sauvé et al. 1996; Ge et al. 2000). Soil pH, redox potential, the number of ion exchange sites and competing cations for ion exchange sites, are all factors that have been found to influence the availability of metals in soils.
Chemical sequential extraction methods are used to determine the distribution of metals in operationally defined phases (Tessier et al. 1979; Rauret et al. 1999). This study uses a method of Tessier et al. (1979) for the partitioning of metals in river sediments, and adapted for the determination of multielement analysis in soil samples analysed by inductively coupled plasma-atomic emission spectrometry (Li et al. 1995a, b). The limitations of chemical extractions to assess metal speciation occur due to the simplification of an open system by a reagent-defined experiment. The partitioning of elements are therefore dependent on the procedures used and are referred to as “operationally defined” (Tessier et al. 1979; Rauret et al. 1999).
Single-chemical extraction techniques assess the proportion of total metal that is available for a specific environmental receptor. The first approximation of Pb that is bioaccessible in the stomach of the human gastrointestinal tract is assessed by a weak HCl solution (Gibson and Farmer 1984; Gulson et al. 1994).
Methods
Thirty-two brownfield sites, including two allotments, were sampled in 1999–2000 for the NERC URGENT project “Studies into metal speciation and bioavailability to assist risk assessment and remediation of brownfield sites in urban areas” (grant reference number GST/03/2251). A composite surface soil (0–15 cm) sample was taken from 20–25 subsamples from a 20 m × 20 m square using a Dutch auger. To determine total metal concentration the soil samples were air dried at 30°C for 2–3 days, disaggregated, sieved to <2 mm and milled to a fine powder for acid digestion with HNO3–HClO4 (Thompson and Walsh 1983). Soil pH was determined from the <2 mm fraction in a 10:1 ratio of deionised water to soil. The standard method of loss on ignition (LOI) was used to estimate organic carbon content. A known weight of the <2 mm soil is placed in 105°C to expel moisture, weighed again and placed at 450°C to burn off organic matter. The weight loss is expressed as a percentage of the moisture free soil. The samples underwent wet digestion in HNO3–HClO4 using the method of Thompson and Walsh (1983) before the determination of the “total” concentrations of Li, Na, K, Be, Mg, Ca, Sr, Ba, La, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Cd, Pb, P, S and As by inductively coupled plasma–atomic emission spectrometry (ICP–AES). Total concentrations and results of extractions are reported in this paper for the elements Ni, Cu, Zn and Pb.
Sequential chemical extractions follow the established method of Tessier et al. (1979) modified for analysis of soil samples by ICP–AES by Li et al. (1995a, b). Five chemical reagents determine the fraction of an element brought into solution under different chemical conditions. Step 1, 0.5 M MgCl2 at pH 7.0; step 2, 1.0 M CH3CO2Na at pH 5.0; step 3, 0.04 M NH2·OH·HCl-in 25% CH3CO2H; step 4, 30% H2O2 at pH 4.0; and step 5, 70% HNO3–60% HClO4–40% HF. The first step extracts the metals in readily exchangeable sites by a weak electrolyte solution of magnesium chloride under neutral pH conditions. The step 2 reagent, sodium acetate at pH 5.0, dissolves carbonate species and releases ions specifically adsorbed onto pH-dependent exchange sites. The third step uses hydroxylammonium chloride to reduce amorphous oxide and hydroxides of Fe, Mn and Al, with an addition of acetic acid to keep the dissolved component in solution. The fourth step oxidises organic matter and sulphides using a hot hydrogen peroxide attack at pH 4.0. The last step dissolves the residue phases by an attack of nitric, perchloric and hydrofluoric acids; this step dissolves the clay fraction, phosphates, residue organic material, sulphides, slag material, alloys and other chemically resistant compounds from anthropogenic sources.
Single-chemical extraction approximates the fraction extracted during soil ingestion in the human large intestine. A solution of 0.12 M HCl is agitated with soil in a 10:1 acid to soil ratio at room temperature for 2 h (Gulson et al. 1994; Rieuwerts et al. 2000). Elemental determination of the total, sequential and single extraction solutions was carried out using inductively coupled plasma–atomic emission spectrometry (ICP–AES).
A standard quality control procedure for total and sequential extractions was followed after Thompson and Howarth (1976) and Gill and Ramsey (1997). Certified reference materials, blanks and random duplicate samples were incorporated into the analytical run. Two soil reference materials from the National Institute of Standards and Technology were used: SRM 2709 and SRM 2710. The total element analysis gave an acceptable level of bias and precision (<5%) for all elements analysed. The majority of elements show a recovery of less than 100%, in the sequential extraction procedure, which is partially accounted for by a loss of material during the sequential analytical procedure, which involves several stages of washing and the transferal of material (Rubio and Ure 1993). Reagent blank measurements were used to determine the detection limit for elements in each step. A value of three times the standard deviation of the instrumental detection limit for duplicate reagent blank measurements was taken as the analytical detection limit. Samples with a low total concentration of Pb have had the values replaced by the analytical detection limit in steps 1 and 2, limiting the determination of metal partitioning between the first two steps for samples with a low total Pb concentration.
Results
Total concentrations of Ni, Cu, Zn and Pb
Descriptions of the sites are given in Table 1, descriptive statistics for the concentrations of Ni, Cu, Zn and Pb in the 32 urban brownfield soils are given in Table 2 and results for pH and LOI are given in Table 3. Table 2 shows that the concentrations of the metals Ni, Cu, Zn and Pb are positively skewed and some values are anomalously high. Geochemical data are typically strongly skewed, the existence of unusually high concentrations is a very common characteristic, and outliers may often originate from another population from the main body of data (Reimann and Filzmoser 2000; Reimann et al. 2002). Samples with a high metal concentration are of most interest to environmental investigations, and therefore it is important to retain these values in the data set.
Box plot diagrams indicating the ranges of Ni, Cu, Zn and Pb concentrations in the Wolverhampton brownfield soils are shown in Fig. 2. The site W6 was classified as an extreme value for the elements Ni, Cu and Zn (an extreme value has a concentration >3 times the variance of the box) and an outlier for the element Pb (an outlying value has a concentration >1.5 times the variance of the box). Site W3 was classified an outlier for Cu and Zn. Site W7 was an outlier for Cu, Zn and Pb, whereas site W10 was classified as an outlier in the distribution of Pb and W29 on outlier for Cu only. In Fig. 3 the nine sites with the highest concentrations of Ni, Cu, Zn and Pb are represented.
Soil properties
Values for pH and loss on ignition are given in Table 3. Soil pH ranges from 4.2 to 8.2 with a median value of 6.4. Loss on ignition is an approximation of the percentage of organic carbon in soil. The organic content of urban brownfield soils is between 3% and 51% with a median of 9%. Both parameters are important variables in modelling the behaviour of trace metal solubility and bioavailability in soils.
Sequential extractions
The partitioning of Ni, Cu, Zn and Pb across the five-step extraction scheme is shown in Table 4 and illustrated in Fig. 4. The geometric means and ranges are nonparametric (Kolmogorov–Smirnov test). Percentages of the total extracted in each step were used to construct the box plots shown in Fig. 4. Box plots are useful in nonparametric statistics because they use the 25th median and 75th percentiles to illustrate the box and thus the distribution around the central tendency. As the data are skewed, the median values are different from the geometric means, thus orders shown in the figure and the table may be different. This is especially true for zinc because it has a very great variability, which was found to be high over all steps of the sequential extraction. The majority of Zn was partitioned into steps 3 and 5 and decreased in the order 3, 5 > 2 > 4 > 1. The majority of Pb was partitioned into step 5 and 3 decreasing in the order 5 > 3 > 4 > 2 > 1. The majority of Ni is partitioned into step 5 (>70%). The majority of Cu was partitioned into step 4, followed by step 5.
Extraction by 0.12 M HCl
Samples from 30 brownfield sites were extracted with 0.12 M HCl. The two allotment sites were not included since it was possible that soil treatment and importation by the allotment holders would influence the bioavailability. Results for the extraction of Cu, Zn and Pb by 0.12 M HCl for the 30 sites are given in Table 5. More than 25% of the samples for Ni were on or below the analytical detection limit, therefore Ni is not included in the table.
Discussion
Table 2 and Figs. 2 and 3 show that the concentrations of Cu, Zn, Ni and Pb vary greatly in the sites investigated. The four sites with the highest total concentrations of these four metals were W6, W7, W10 and W3. W6, now designated as a nature reserve, was previously a landfill site and used for dumping dredged canal sediment. W7 was the site of a metal works. Site W10 was used formerly by an enamelling works and a tyre factory. W3, now designated as a recreation area, was a former colliery site. Thus the concentrations of these elements in soils are highly influenced by historical land use, and all the sites are within areas of former or contemporary industrial activity.
The UK government’s contaminated land exposure assessment (CLEA) model (DEFRA-EA 2002a) considers the exposure to contaminants from the direct and indirect ingestion and inhalation of soils (DEFRA-EA 2002b) and has defined generic soil guideline values (SGV) for As, Cd, Cr, Pb, Hg, Ni and Se (DEFRA-EA 2002c). Values for Ni and Pb are shown Table 6. Eleven of the 32 sites exceed the SGV of 50 μg/g for Ni and three sites exceed the 450 μg/g for Pb (DEFRA-EA 2002d).
The available or labile metal pool is a generic term that is used to describe the fraction of metal that is potentially available for uptake into a biological receptor. It has been defined (Young et al. 2005) as that metal in the solid phase of the soil that is immediately capable of buffering the metal ion activity in the soil solution. These authors point out that the functions of the labile pool may include slow interactions with nonlabile phases or metal associations, and the control over bioavailability following direct ingestion. Chemical extraction schemes are often used to quantify labile metal pools, and multistep sequential extractions enable the identification of the main chemical associations of metals in soil. Despite limitations in the selectivity of exact soil phases, the partitioning of metals is of value in the assessment of metal mobility and persistence in contaminated soil (Gibson and Farmer 1986; Young et al. 2005).
The partitioning of metals across the five-step extraction scheme has high variability in urban soils, and the trends found in this work were comparable to those in published surveys. Gibson and Farmer (1986) reported the majority of Zn partitioned into the carbonate, moderately reducible and residual phases, Cu into the organic phase and Pb into the moderately reducible and residual phases. These authors concluded that the distinct partitioning pattern of heavy metals in urban soils was of value in establishing the main chemical associations of the heavy metals in the solid phase. In the urban brownfield soils of Wolverhampton a higher proportion of Zn was found to occur in the relatively soluble phases, steps 1 and 2, as compared with Cu, Pb and Ni, which were associated with the more resistant phases (Table 4, Fig. 4).
Correlation coefficients were calculated between the results of the sequential extractions and the soil pH, organic carbon content (LOI), and total metal concentrations using Spearman’s correlation coefficient (r′), as this is suitable for nonparametric data, and the results are shown in Table 7. A strong positive relationship between two variables is indicated by two asterisks at the 99% significance level or P < 0.01. A weaker but still significant relationship is indicated by a single, at the 95% significance level or P < 0.05. The sign of the coefficient indicates the direction of the relationship. The partitioning of Ni, Cu, Zn and Pb in step 1 increases as soil pH decreases. This relationship is significant, implying that in more-acidic soils there is a higher proportion of exchangeable metals. The partitioning of Zn into step 2 increases in soils that are more alkaline. This may be the competitive adsorption of Zn in soils with a high pH at the expense of other cations.
The loss on ignition of the soil (LOI) approximates the amount of organic matter, which is an important property in the binding of trace metals in soil (Plant and Raiswell 1983; Rieuwerts et al. 1998). The step 4 reagent, hydrogen peroxide, is designed to break down organic-metal compounds (Tessier et al. 1979; Li et al. 1995a). The partitioning of Ni, Cu and Zn increases in step 4 as the organic carbon content of soil increases. It is inferred that the organic phase becomes more important for Ni, Cu and Zn in organic-rich soil. This is particularly the case for Cu, where a high percentage of total Cu is partitioned into step 4, the organic phase (median 48%, range 10–84%). Soils high in organic carbon have a positive relationship with total Pb; however, as there was no significant correlation between Pb in step 4 and the organic carbon content, it is suggested that it is possible that Pb-organic complexes were not extracted by step 4 reagents, and so the occurrence of the species may be underestimated by this method.
To test if there was a greater partitioning into one phase in more contaminated soil a correlation between the total metal concentration and the partitioning of metals was performed (Table 8). The partitioning of Ni, Cu and Zn increases in step 3, the Fe–Mn oxide phase, in soils elevated in these metals. The partitioning of Pb in step 1 appears to decrease in soils more contaminated with these metals.
The partitioning coefficient of metal into the readily exchangeable fraction (step 1) has been calculated from
There is a strong inverse relationship between the partitioning of Zn into step 1 and soil pH (P = 0.00). The partitioning of Ni into step 1 also has a strong inverse relationship with soil pH whilst Pb and Cu have a weaker relationship, although they are still significant at 95% at the significance level (P = 0.03 and P = 0.05, respectively). The partitioning of metal into the readily exchangeable fraction may be a useful measure of potential bioavailability and mobility in soil.
The brownfield sites represent a range of land-use types and reflect the variability of metal partitioning in urban soils. The patterns of fractionation for Pb are shown in Fig. 5. It is apparent that very little of the Pb is solubilised in step 1. Lead in soil from site W21 (rough ground, adjacent to metal works and light industrial park) is all solubilised in steps 1–4, whereas that from site W22 from the same area (Table 1) has a different pattern of fractionation, demonstrating inhomogeneity in both intersite and intrasite soils (see also Thornton et al. 2008 in this volume).
Partitioning of Pb into five steps for 30 brownfield sites in Table 1 (two allotments not included)
The patterns of fractionation for two sites (W6 and W14) are shown in detail in Fig. 6. These two sites are designated as nature reserves and are used for recreation, including play sites for young children. Soil from the site W6 has high concentrations of Cu, Ni, Zn and Pb (Fig. 3 and Table 2). The majority of total Pb (768 μg/g) was found in the least available residual fraction. The total Cu concentration (1663 μg/g) was found to be fractionated into the most available steps 1, 2 and 3; however, the Fe–Mn oxide phase, step 3 was found to be an important phase for Ni, Cu and Zn. W14 is adjacent to a railway, managed as a nature reserve with vegetation cover of trees and grasses. The soil has a low pH (5.8) and an estimated 50% organic carbon content. Only a small proportion of the Pb is in the residual fraction. Nickel and Zn were found to be comparatively high in step 1. The partitioning of metals into the organic fraction (step 4) was found to be more important than the Fe–Mn oxide fraction, particularly for Ni, Cu and Pb.
Extraction with 0.12 M hydrochloric acid has been used to approximate bioaccessible Pb, the fraction of total Pb available for absorption in the human stomach from ingested soil. Single leach extraction schemes have been designed to model human stomach acidity; however they are not necessarily a true representation of the bioavailable fraction (Davis et al. 1994). Lead binds strongly to humic materials in soil, and is less soluble in the acidic conditions in the stomach than is the Pb bound to Fe and Mn oxides (Chaney et al. 1988). In the alkaline conditions in the duodenum both OM and Fe oxides strongly bind Pb (Chaney et al. 1988). A better assessment of Pb bioavailability would be obtained using the physiologically based extraction test (PBET) (Ruby et al. 1996), where Pb is extracted in both acid and alkaline conditions simulating those in both stomach and duodenum. However in this work only the acidic conditions were simulated.
There are significant positive correlations between the concentration of metal extracted by 0.12 M HCl (Table 5) and the total concentration of the elements. Spearman’s correlation coefficients were high and positive; Cu r′ = 0.93 (P < 0.01), Zn r′ = 0.96 (P < 0.01) and Pb r′ = 0.94 (P < 0.01). The proportion of Cu and of Pb extracted by 0.12 M HCl was found to be statistically similar to the sum of Cu and Pb in steps 1, 2, 3 and 4, respectively; the proportion of 0.12 M HCl extractable Zn was statistically similar to the sum of Zn in steps 1, 2 and 3. The results are presented in Table 9, with significance indicated at the P < 0.01 level. Step 4 was not an important phase for Zn (median 9%) therefore it can be concluded that the availability of Cu, Zn and Pb once soil had been ingested into the GI tract was equivalent to the nonresidual phases (step 5); all other species are potentially available for uptake.
The integrated exposure uptake biokinetic (IEUBK) model for Pb blood levels in children was developed for the US Environmental Protection Agency to estimate the blood lead (PbB) levels in children exposed to Pb contamination from several sources (IEUBK model 2002). The IEUBK model was run [using model default values as recommended (IEUBK 2002)] and using site-specific “total” Pb concentration in soil from 30 urban brownfield sites. In the first run the default value for bioavailability (30%) was used. In the second run the site-specific HCl availability value was used. The results show blood Pb (PbB) level (in μg/dL) for a hypothetical child, or population of children, between the ages of 1 and 2 years. Blood Pb levels in children should not exceed 10 μg/dL, (US EPA 1994).
Using the default estimate of bioavailability (30%), it was found that children exposed to the urban brownfield soils in this study would not exceed blood lead levels of 10 μg/dL. However, when the experimental values of the availability from the HCl extractions for each site was used, there were three sites where blood lead levels were predicted to exceed the 10 μg/dL threshold value. The sites were W6, W10 and W24. Sites W6 and W10 have high total soil Pb concentration (PbTotal 768 and 732 μg/g respectively), and exceed the SGV for residential development (DEFRA-EA 2002d). The sites have measured HCl availability of 35% and 39%, respectively. W24 has a moderately high PbTotal 401 μg/g and a high HCl extractability of 97%. Although the mean Pb concentration at site W24 does not exceed the SGV for residential development, the high bioavailability means that the Pb is a potential hazard should the site be redeveloped for residential purposes in the future. Since lead exposure even at relatively low levels below 10 μg/dL can pose a hazard to children, leading to intellectual impairment (Canfield et al. 2003), it appears necessary to carry out bioavailability tests for Pb, in addition to total concentrations, before brownfield sites in urban areas are developed for residential housing.
Conclusions
The high variability of partitioning across the five-step schemes can be attributed to the variability of anthropogenic activity controlling metal source and the soil properties. Soils with higher total Zn have a greater partitioning in the more environmentally available phases. Soils with a higher total Ni, Cu and Pb have higher concentrations of these metals in the residue phases, suggesting that the anthropogenic input is in a chemically resistant form or that the anthropogenic metals form insoluble solid phases. Strong organic-metal complexes are released during oxidation of organic matter in step 4, thus the partitioning of Ni, Cu and Zn increases in step 4 as the organic carbon (measured by LOI) content of soil increases, particularly for Cu. where a high percentage of total Cu was partitioned into step 4, the organic phase (median 48%, range 10–84%).
The partitioning of Ni, Cu, Zn and Pb in step 1 increases as soil pH decreases, whereas the partitioning of Zn into step 2 increases with soil pH. Using HCl availability, there were three sites where children exposed to the soil by hand-to-mouth activity would be predicted to exceed the 10 μg/dL threshold blood lead concentration. The sites were W6, W10 and W24 (Table 1).
It can be concluded from the results that estimates of PHE content, concentration and bioavailability is site specific, and depends on the historical land use. The potential bioavailability and bioaccessibility of the elements studied here is dependent on soil properties, such as carbon content and pH. It is recommended that bioavailability tests, particularly for Pb, be carried out before brownfield sites in urban areas are developed for residential housing.
References
Ander, E. L., Smith, B., Fordyce, F. M., & Rawlins, B. G. (2001). Trace elements. In B. J. D’Arcy, J. B. Ellis, R. C. Ferrier, A. Jenkins, & R. Dils (Eds.), Diffuse pollution impacts (pp. 85–94). Lavenham, Surrey: CIWEM, Terence Dalton Publishers.
ATSDR. (2002). Children’s environmental health. Public Health and the Environment, 1(1/2) and articles therein.
Bridge, D. Mc. C., Brown, M. J., & Hooker, P. J. (1997). Wolverhampton urban environmental survey: An integrated geoscientific case study. BGS Technical Report. WE/95/49.
Canfield, R. L., Henderson, J., Cory-Slechta, D. A., Cox, C., Jusko, T. A., & Lanphear, B. P. (2003). Intellectual impairment in children with blood lead concentrations below 10 μg per deciliter. The New England Journal of Medicine, 348, 1517–1526.
CDC. (1998). Centers for disease control. Surveillance for asthma—United States, 1960–1995. Morbidity and Mortality Weekly Report, 47(SS–1), 1–27.
Chaney, R. L., Mielke, H. W., & Sterret, S. B. (1988). Speciation, mobility and bioavailability of soil lead. In B. E. Davies & B. G. Wixon (Eds.), Lead in soil: Issues and guidelines. Northwood: Science Reviews Limited.
Culbard, E. B., Thornton, I., Watt, J., Wheatley, M., Moorcroft, S., & Thompson, M. (1988). Metal contamination in British urban dust and soils. Journal of Environmental Quality, 17, 226–234.
Davies, B. E. (1990). Lead. In B. J. Alloway (Ed.), Heavy metals in soil. Glasgow: Blackie.
Davies, D. J. A., Watt, J. M., & Thornton, I. (1987). Lead levels in Birmingham dust and soils. The Science of the Total Environment, 67, 177–185.
Davis, A., Ruby, M. V., & Bergstrom, P. D. (1994). Factors controlling lead bioavailability in the Butte mining district, Montana, USA. Environmental Geochemistry and Health, 16(3/4), 147–157.
DEFRA-EA. (2002a). Contaminated land exposure assessment model: Technical basis and algorithms. R&D Report, CLR10. Department of the Environment, Food and Rural Affairs and the Environment Agency, Bristol.
DEFRA-EA. (2002b). Contaminants in soils: Collation of toxicological data and intake values for humans. R&D Report, CLR9. Department of the Environment, Food and Rural Affairs and the Environment Agency, Bristol.
DEFRA-EA. (2002c). Assessment of risks to human health from land contamination: An overview of the development of the soil guideline values and related research. R&D Report, CLR7. Department of the Environment, Food and Rural Affairs and the Environment Agency, Bristol.
DEFRA-EA. (2002d). Soil guideline values for arsenic contamination. R&D Report, SGV1. Department of the Environment, Food and Rural Affairs and the Environment Agency, Bristol.
EA. (2001). Environment agency: Your environment, pollution inventory. http://environmentagency.gov.uk
Facchinelli, A., Sacchi, E., & Mallen, L. (2001). Multivariate statistical and GIS-based approach to identify heavy metal source in soils. Environmental Pollution, 114, 313–324.
Fordyce, F. M., Brown, S. E., Ander, E. L., Rawlins, B. G., O’Donnell, K. E., Lister, T. R., et al. (2005). GSUE: Urban geochemical mapping in Great Britain. Geochemistry: Exploration, Environment Analysis, 5, 325–336.
Ge, Y., Murray, P., & Hendershot, W. H. (2000). Trace metal speciation and bioavailability in urban soils. Environmental Pollution, 107, 137–144.
Gibson, M. J., & Farmer, J. G. (1984). Chemical partitioning of trace metal contaminants in urban street dirt. Science of the Total Environment, 33, 49–57.
Gibson, M. J., & Farmer, J. G. (1986). Multi-step sequential chemical extraction of heavy metals from urban soils. Environmental Pollution, 11, 117–135.
Gill, R., & Ramsey, M. H. (1997). What a geochemical analysis means. In R. Gill (Ed.), Modern analytical geochemistry. Longman.
Gulson, B. L., Davis, J. J., Mizon, K. J., Krosch, M. J., Law, A. J., & Howarth, D. (1994). Lead bioavailability in the environment of children: Blood lead levels can be elevated in a mining community. Archives of Environmental Health, 49, 326–331.
Hains, Ba. A. (1969). British regional geology: Central England (3rd ed.). London: HMSO.
IEUBK Model. (2002). Available from: http:www.epa.gov/superfund/programs/lead/ieubk.htm
Japony, M., & Young, S. D. (1994). The solid ↔ solution equilibria of lead and cadmium in polluted soils. European Journal of Soil Science, 45, 59–70.
Kelly, J. J. P. (1997). Influence of geology and anthropogenic activity on the geochemistry of urban soils. PhD Thesis. Imperial College, London.
Kelly, J. J. P., Thornton, I., & Simpson, P. R. (1996). Urban geochemistry: A study of the influences of anthropogenic activity on the heavy metal content of soils in traditionally industrial and non-industrial areas of Britain. Applied Geochemistry, 11, 363–370.
Landrigan, P. J., Schechter, C. B., Lipton, J. M., Fahs, M. C., & Schwartz, J. (2002). Environmental pollutants and disease in American children: Estimates of morbidity, mortality, and costs for lead poisoning, asthma, cancer, and developmental disabilities. Environmental Health Perspectives, 110, 721–728.
Li, X., Coles, B. J., Ramsey, M. H., & Thornton, I. (1995a). Sequential extraction of soils for multielement analysis by ICP–AES. Chemical Geology, 124, 109–123.
Li, X., Coles, B. J., Ramsey, M. H., & Thornton, I. (1995b). Chemical partitioning of the new National Institute of Standard and Technology Standard Reference Materials (SRM 2709–2711) by sequential extraction using inductively coupled plasma atomic emission spectrometry. Analyst, 120, 1415–1419.
Lidsky, T. I., & Schneider, J. S. (2005). Autism and autistic symptoms associated with childhood lead poisoning. Journal of Applied Research, 5, 80–87.
Myers, G. J., Davidson, P. W., Weitzman, M., & Lanphear, B. P. (1997). Contribution of heavy metals to developmental disabilities in children. Mental Retardation and Developmental Disabilities Research Reviews, 3, 239–245.
Nataf, R., Skorupk, C., Amet, L., Lam, A., Springbett, A., & Lathe, R. (2006). Porphyrinuria in childhood autistic disorder: Implications for environmental toxicity. Toxicology and Applied Pharmacology, 214, 99–108.
Palmer, R. F., Blanchard, S., Stein, Z., Mandell, D., & Miller, C. (2006). Environmental mercury release, special education rates, and autism disorder: An ecological study of Texas. Health & Place, 12, 203–209.
Plant, J. A., & Raiswell, R. (1983). Principles of environmental geochemistry. In I. Thornton (Ed.), Applied environmental geochemistry (pp. 1–40). London: Academic.
Rauret, G., López-Sánchez, J. F., Sahuquillo, A., Rubio, R., Davidson, C., Ure, A., et al. (1999). Improvement of the BCR three step sequential extraction procedure prior to the certification of new sediment and soil reference materials. Journal of Environmental Monitoring, 1, 57–61.
Reimann, C., & Filzmoser, P. (2000). Normal and lognormal data distribution in geochemistry: Death of a myth. Consequences for the statistical treatment of geochemical and environmental data. Environmental Geology, 39, 1001–1014.
Reimann, C., Filzmoser, P., & Garrett, R. G. (2002). Factor analysis applied to regional geochemical data problems and possibilities. Applied Geochemistry, 17, 185–206.
Rieuwerts, J. S., Farago, M. E., Cikrt, M., & Bencko, V. (2000). Differences in lead bioavailability between a smelting and mining area. Water Air and Soil Pollution, 122, 203–229.
Rieuwerts, J. S., Thornton, I., Farago, M. E., & Ashmore, M. R. (1998). Factors influencing metal bioavailability in soils: preliminary investigations for the development of a critical loads approach for metals. Chemical Speciation and Bioavailability, 10(2), 61–75.
Rubio, R., & Ure, A. M. (1993). Approaches to sampling and sample pretreatments for metal speciation in soils and sediments. International Journal of Analytical Chemistry, 51, 205–217.
Ruby, M. V., Davis, A., Schoof, R., Eberle, S., & Sellstone, C. M. (1996). Estimation of lead and arsenic bioavailability using a physiologically based extraction test. Environmental Science and Technology, 30, 422–430.
Sauvé, S., Cook, N., Hendershot, W. H., & McBride, M. B. (1996). Linking plant tissue concentrations and soil copper pools in urban contaminated soils. Environmental Pollution, 94(2), 153–157.
Szpir, M. (2006). Focus: New thinking on neurodevelopment. Environmental Health Perspectives, 14(2), A101–A107.
Tessier, A., Campbell, P. G. C., & Bisson, M. (1979). Sequential extraction procedure for the speciation of particulate trace metals. Analytical Chemistry, 5(7), 844–851.
Thompson, M., & Howarth, R. J. (1976). Duplicate analysis in geochemical practice. Part 1: Theoretical approach and estimation of analytical reproducibility. Analyst, 101, 690–698
Thompson, M., & Walsh, J. N. (1983). A handbook of inductively coupled plasma spectrometry. New York: Blackie.
Thornton, I. (1991). Metal contamination of soils in urban areas. In P. Bullock & P. J. Gregory (Eds.), Soils in the urban environment (pp. 47–75). Oxford: Blackwell Scientific.
Thornton, I., Culbard, E., Moorcroft, S., Watt, J., Wheatley, M., & Thompson, M. (1985). Metals in urban dust and soils. Environmental Technology Letters, 6, 137–144.
Thornton, I., Farago M. E., Thums C. R., Parrish R. R., McGill R. A. R., Breward N., et al. (2008). Urban geochemistry: research strategies to assist risk assessment and remediation of brownfield sites in urban areas. Environmental Geochemistry and Health. doi:10.1007/s10653-008-9182-9
Upton, C. (1998). A history of Wolverhampton. Chichester: Phillimore.
Ure, A. M., Quevaubiller, P., Muntau, H., & Griepink, B. (1993). Speciation of heavy metals in soils and sediments. An account of the improvement and harmonisation of extraction techniques undertaken under the auspices of the BCR of the Commission of the European Communities. International Journal of Environmental Analytical Chemistry, 51, 135–151.
US EPA. (1994). United States Environmental Protection Agency. Revised soil lead guidelines for CERCLA sites and RCRA corrective action facilities (pp. 4–27). Office of Solid Waste and Emergency Response Directive #9200.
Wasserman, G. A., Liu, X., Parvez, F., Ahsan, H., Levy, D., Factor-Litvak, P., et al. (2006). Water manganese exposure and children’s intellectual function in Araihazar, Bangladesh. Environmental Health Perspectives, 114, 124–129.
West, L. J., Stewart, D. I., Duxbury, J. R., & Johnson, R. S. (1999). Toxic metal mobility and retention at industrially contaminated sites. In R. Metcalfe & C. A. Rochelle (Eds.), Chemical contaminant of waste in the geosphere (Vol. 157, pp. 241–264). Geological Society Special Publication.
Williams, J. G., Higgins, J. P., & Brayne, C. E. (2006). Systematic review of prevalence studies of autism spectrum disorders. Archives of Disease in Childhood, 91, 8–15.
Young, S. D., Zhang, H., Tye, A. M., Maxted, A., Thums, C., & Thornton, I. (2005). Characterizing the availability of metal in contaminated soils. 1. The solid phase: Sequential extraction and isotopic dilution. Soil Use and Management, 21, 450–458.
Zahm, S. H., & Devesa, S. S. (1995). Childhood cancer: Overview of incidence trends and environmental carcinogens. Environmental Health Perspectives, 103(suppl 6), 177–184.
Acknowledgements
The work was carried out for the NERC URGENT project “Studies into metal speciation and bioavailability to assist risk assessment and remediation in urban brownfield sites” Grant No. GST/03/2251. The authors thank members of the research consortium from the NERC Isotope Geosciences Laboratory, British Geological Survey and the University of Nottingham, especially Scott Young, Andy Tye (University of Nottingham) and Alex Fergusson (British Geological Survey) for the collection and preparation of soils. Catherine R. Thums thanks the Research Communication Foundation for a grant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thums, C.R., Farago, M.E. & Thornton, I. Bioavailability of trace metals in brownfield soils in an urban area in the UK. Environ Geochem Health 30, 549–563 (2008). https://doi.org/10.1007/s10653-008-9185-6
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10653-008-9185-6