Abstract
The seven-spot ladybird beetle, Coccinella septempunctata, is a major natural enemy of aphids in the field and in greenhouses in China and is part of integrated pest management (IPM). Imidacloprid, a highly efficient insecticide that not only kills aphids at lethal concentrations, but also can cause various sublethal effects in nontarget organisms. To strengthen IPM and its sustainability, it is important assessing possible side effects on natural enemies. When the effects of sublethal concentrations (LC5 and 10%LC5) of imidacloprid on C. septempunctata were evaluated, the adult longevity was shortened by 23.97 and 28.68 %, and the fecundity reduced by 52.81 and 56.09 % compared to control population. In the F1 generation (i.e., the progeny of the exposed individuals), the juvenile development was slower by 1.44 days and 0.66 days, and the oviposition period was shortened by 10 and 13 days, respectively. The fecundity of the F1 generation decreased by 17.88, 44.03 and 51.69 % when exposed to 1%LC5, 10%LC5, and LC5, respectively. The results of demographical growth estimates showed that the intrinsic rate of increase (r m ) and net reproductive rate (R 0 ) were lower in C. septempunctata populations that had been exposed to sublethal concentrations of imidacloprid. The results emphasize the importance of assessing side effects of low imidacloprid concentrations on such predator species, even at the transgenerational level.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Generalist arthropod predators are able to feed on many phytophagous insects and mites in cultivated crops (Bompard et al. 2013; Mirande et al. 2015; Pérez-Hedo and Urbaneja 2015; Saeed et al. 2015; Biondi et al. 2016). The seven-spot ladybird beetle, Coccinella septempunctata L. (Coleoptera: Coccinellidae), is one of the beneficial arthropod predators that are potential biological control agents of aphids worldwide (Zhang et al. 2011; Lu et al. 2012). Both larvae and adults of C. septempunctata are known to be effective predators on aphids that infest a variety of plants, which is why the species was selected as an effective natural enemy in an integrated pest management (IPM) programme of aphid pests (Yu et al. 2014a; 2014b). However, in most cropping systems aphid populations are suppressed primarily through broad-spectrum chemical insecticides, which can seriously harm beneficial arthropods (Lu et al. 2012). The success of IPM programmes rely partially on optimised applications of selective insecticides, i.e. that are less harmful to natural enemies (Desneux et al. 2007; Stark et al. 2007; Guedes et al. 2016). However, this approach requires extensive knowledge of the side effects of pesticides on physiological and behavioural traits of these arthropod biological control agents (Desneux et al. 2004; Bengochea et al. 2014; Biondi et al. 2012b; 2015; Zhang et al. 2015).
Aphid natural enemies inhabiting the crop environment are usually exposed to pesticides either topically in the form of spray droplets (Desneux et al. 2006; He et al. 2012), through contact with residues on foliage when foraging for hosts/prey (Torres and Ruberson 2004; Biondi et al. 2012a), or through feeding on pesticide-contaminated extra-floral nectars (Gontijo et al. 2014; Moscardini et al. 2015) and/or pesticide-contaminated hosts/prey (Ahmad et al. 2003; Cabral et al. 2011). Insecticide exposure can result in acute toxicity and/or sublethal effects (physiological and/or behavioural effects on individuals that survive exposure to the toxicant at sublethal concentrations or doses, Desneux et al. 2007). Such effects may have important consequences in exposed individuals, and at the population level (Stark and Banks 2003; Ali et al. 2012; Abbes et al. 2015). Therefore, a demographic analysis of effects of insecticides, estimating their total effect on insect populations, is often crucial for a complete insecticide risk assessment.
Neonicotinoid insecticides act selectively on the insects’ nicotinic acetylcholine receptors, and the use of these insecticides rose sharply during the 1980s (Tomizawa and Casida 2001; Pan et al. 2014). These agrochemicals have been widely used to control various sap-sucking pests (Tomizawa and Casida 2001) and recently they have been tested on chewing insects with contrasting results (Chen et al. 2015; van Herk et al. 2015). Nevertheless, their ecotoxicological profile has been recently under scrutiny(Blacquiere et al. 2012; Decourtye et al. 2013; Sanchez-Bayo 2014; Sanchez-Bayo et al. 2016). Imidacloprid, a neonicotinoid insecticide, is registered in about 120 countries for use in over 140 crops. As a broad-spectrum systemic insecticide, it has proved extremely effective in controlling pests when applied as a foliar spray, as a seed treatment, and through irrigation (Palumbo et al. 2001; Liang et al. 2012). In China, imidacloprid is currently applied for controlling sucking insects in field crops, orchards and vegetables grown in greenhouses (He et al. 2013; Qu et al. 2015).
Several studies have reported the impact of neonicotinoid insecticides on predators belonging to the Coccinellidae family. These studies focused on acute toxicity and on the impairment of physiological and behavioural biological traits (Smith and Krischik 1999; Vincent et al. 2000; Youn et al. 2003; Lucas et al. 2004; Papachristos and Milonas 2008; Moser and Obrycki 2009; Eisenback et al. 2010; Cabral et al. 2011; He et al. 2012; Fogel et al. 2013; Yu et al. 2014). The acute toxicity of various insecticides at field rates and mechanisms of resistance to them have been investigated in C. septempunctata (Bozsik 2006; Ji et al. 2011), and the toxic effects of dimethoate and hexaflumuron on the predatory capacity and biological traits of C. septempunctata have also been reported (Singh et al. 2004; Yu et al. 2014a; 2014b). However, the potential sublethal effects of imidacloprid on C. septempunctata and the potential changes at population level have not been documented yet. In this context, we aimed at assessing the sublethal effects of imidacloprid at three low concentrations on the development, fecundity and demographic parameters of C. septempunctata.
Materials and methods
Insect rearing
Adults of C. septempunctata were collected from experimental cotton fields (39°95′N, 116°28′E) of the Beijing Academy of Agriculture and Forestry Sciences (BAAFS), Beijing, China, during May 2013. The ladybird beetles were transported to the laboratory and reared on the pea aphid, Aphis craccivora Koch, at the Laboratory of Natural Enemies Research, Institute of Plant and Environment Protection, BAAFS. The environmental conditions were 25 ± 2 °C; 65 % RH; 12D:12L (L-100, Suntech, Beijing, China). Predators were maintained in custom-built culturing cages (50 cm × 50 cm × 60 cm; 45 mesh plastic fabric on aluminium frames) at a density of 40 pairs to a cage and fed daily on pea aphids grown on fresh seedlings of broad bean (Vicia faba L. ‘LinCan-5’) (Tan et al. 2014).
Insecticide and assessment of acute toxicity
Technical grade imidacloprid (95 %) was provided by Jiangsu Changlong Chemical Co. Ltd, China. The chemical’s toxicity was assessed using the method described by He et al. (2012) which involves exposure to dry insecticide residues in glass tubes. Preliminary experiments were carried out to establish the range of concentrations to be tested using six concentrations, beginning with 6.25 mg L–1 and progressively doubling each concentration to reach 200 mg L–1for the bioassay. An aliquot of 820 μL of the solution of the insecticide in acetone was placed in each glass tube (14.5 cm long and 1.5 cm in diameter). Then, these tubes were immediately rotated using a micro-rotator (American Wheaton Company) until the acetone had fully evaporated. Twenty 5-day-old adults of C. septempunctata were exposed to each concentration, each treatment being replicated three times. Controls were treated only with acetone. The ladybird beetles were then reared in the laboratory under the same conditions described above. Mortality was recorded after 24 h—individuals that failed to respond after being pushed with a fine paint brush were considered dead (He et al. 2012). This insecticide exposure methodology was used in all the experiments.
Three sublethal concentrations were chosen, namely 4.837 mg L–1(LC5), 0.484 mg L–1(10 % of LC5) and 0.048 mg L–1 (1 % of LC5) (see the result section) for assessing sublethal effects. The toxicity of these three concentrations was checked on 5-day-old adults of C. septempunctata, using the method described above, but with nine replicates (with 20 ladybird beetles each). All the experiments were carried out in an incubator (Sanyo, M251H, Osaka, Japan) under the following conditions— temperature, 25 ± 2 °C; relative humidity, 65 %;12 h of light alternating with 12 h of darkness. Mortality was assessed following 24 h of exposure.
Sublethal effects of imidacloprid sublethal concentrations on the parental generation (F0)
The three aforementioned sublethal concentrations were used for assessing the effects of imidacloprid on the development and fecundity of C. septempunctata. Exposure to the insecticide was performed using the method described previously. Pairs of 10-day-old males and virgin females were placed inside new and clean plastic cups (9 cm deep and 7 cm in diameter) and fed daily on fresh leaves infested with 50/100 mixed instars of A. craccivora. The number of offspring was recorded daily until the female died. Dead males were promptly replaced. Three replicates were carried out, each with 20 pairs of ladybird beetles.
Trangenerational effects of imidacloprid sublethal concentrations on C. septempunctata F1 generation
To estimate the potential transgenerational impact of imidacloprid sublethal concentrations on C. septempunctata population dynamic, the survival and longevity of the progeny (F1) of ladybird adults previously exposed to the three concentrations of imidacloprid was monitored. The eggs laid on leaves from F0 individuals within 24 h following the exposure were collected and transferred to clean Petri dishes (9 cm in diameter) for use in the life table studies. Each newly hatched first instar was moved individually to a plastic cup (7 cm in diameter and 9 cm deep) and reared in a growth chamber under the same conditions mentioned above and fed daily with fresh leaves infested with 100 aphids. Survival and development were recorded daily until pupation. Newly emerged adults were paired and moved to a new plastic cup. The number of offspring (F2) was also recorded daily until the female died. Dead males were promptly replaced.
Data analysis
The LC50 value was determined through a log-probit model (Finney 1971) and the using software PoLo Plus ver. 2.0 (Leora Software, Petaluma, CA, USA). The dose–mortality relationship was considered valid, if the observed values and the predicted values did not deviate from each other significantly (P < 0.05). Datasets of the various biological traits recorded were first tested for normality and homogeneity of variance using the Kolmogorov–Smirnov D test and the Cochran test, respectively, and were transformed if necessary. Untrasformed data are presented. Within each dataset one-way analysis of variance (ANOVA) followed by Tukey’s honestly significant difference (HSD) for multiple comparisons were carried out using ProStat (Poly Software International, Pearl River, NY, USA).
Raw data on the development, survival and daily fecundity were analysed using the software TWOSEX-MS Chart (Chi 2012), according to the age-stage, two-sex life table theory (Chi and Liu 1985; Chi 1988). The age-stage-specific survival rate (S xj ) (x=age, j=stage) is the probability that a newly laid egg will survive to age x and stage j; the reproductive value (V xj ) is the contribution of an individual of age x and stage j to the future population; and life expectancy (e xj ) is the time that an individual of age x and stage j is expected to live. The age-specific survival rate (lx) is the proportion of individuals in the initial cohort alive at a given age; the female age-specific fecundity (f x ), and the age-specific fecundity (m x ) is the mean number of female progeny produced per female.The estimate demographic parameters are as follows: (1) net reproductive rate (R 0 =∑l x m x ), (2) mean generation time (T=∑xl x m x /R 0 ), (3) intrinsic rate of natural increase (r m =ln(R0)/T), and (4) finite rate of increase (λ=exp(rm). The above-mentioned demographic parameters were obtained from TWOSEX-MS Chart, which includes a routine for estimating the standard error of demographic parameters using the bootstrap technique.
Results
Imidacloprid concentration–mortality relationship
Based on the log-probit regression analysis, the LC50 of imidacloprid for C. septempunctata was estimated at 36.753 mg L–1 (95 % confidence interval: 30.31–44.60 mg L–1). The data fitted the linear model with the regression equation Y=1.873X + 2.073 (χ 2 = 1.78, P = 0.193). The LC5 of imidacloprid for C. septempunctata was 4.838 mg L–1, as calculated based on the regression equation. The predator mortality at 0.048 mg L–1 (1 % of LC5), 0.484 mg L–1(10 % of LC5) and 4.837 mg L–1(LC5), were 3.33 ± 0.19 %, 6.67 ± 0.24 %, and 7.78 ± 0.24 %, respectively (corrected mortality; Abbott 1925). These concentrations of imidacloprid were considered sublethal because they induced no statistically significant (F 3, 34 = 1.735; P = 0.179) mortality in the experimental population when compared to the control population (2.22 ± 0.12 %) (Desneux et al. 2007).
Sublethal effects on longevity and fecundity in the parental generation (F0)
The effects of the three sublethal concentrations of imidacloprid on longevity and fecundity of C. septempunctata in the parental generation are shown in Fig. 1. The pre-oviposition period of C. septempunctata exposed to 0.484 mg L–1(12.10 ± 1.16 days) and 4.837 mg L–1(11.80 ± 1.609 days) of imidacloprid increased significantly compared to that in the control population (9.30 ± 1.38 days) (Fig. 1A; F 3, 78 = 22.93, P < 0.001); whereas the oviposition period decreased significantly after exposed to these corresponding concentrations (Fig. 1B; F 3, 78 = 456.28, P < 0.001). A similar trend was observed in the longevity of adults (Fig. 1C; F 3, 78 = 362.63, P < 0.001). In addition, the fecundity (eggs per female) was lowered significantly, (Fig. 1D; F 2, 78 = 371.46, P < 0.001).
Effect of sublethal imidacloprid concentrations (1 % of LC5, 10 % of LC5 and LC5) on the pre-oviposition (A) and oviposition periods (B), longevity (C), and fecundity (D)of Coccinella septempunctata (F0) adults exposed to dry insecticide residues. The results are means (± SE). Histograms bearing different letters are significantly different at P < 0.05 (one-way ANOVA followed by Tukey’s HSD test)
Sublethal effects on development, longevity and fecundity of the F1 generation
The effects of the three sublethal concentrations of imidacloprid on the length of the development period of each instars and fecundity of C. septempunctata in the F1 generation are reported in Table 1. There were no significant differences between the control and the various treatments except for the first instars, in which exposure to 0.484 mg L–1 of imidacloprid resulted in significantly slower development than that in other groups. This translated to an overall slower juvenile development when exposed to this concentration (F 3, 168 = 15.813, P < 0.001). The longevity of adults was significantly shorter at all the three concentrations than that of adults in the control population (F 3, 168 = 39.207, P < 0.001). Overall, the total longevity of C. septempunctata was significantly lower in 4.837 mg L−1 imidacloprid treatment than that in the control (F 3, 168 = 33.467, P < 0.001).
The adult pre-oviposition period (APOP) refers to the beginning of reproduction and remained unaffected by exposure to imidacloprid, as did the total pre-oviposition period (TPOP), which refers to the period from the beginning of the life table study to laying the first egg (Table 1). However, the oviposition period was significantly shortened (F 3, 98 = 44.38, P < 0.001) in case of exposure to imidacloprid at all the three sublethal concentrations (Table 1). Furthermore, fecundity was also affected adversely, being significantly decreased by 17.88, 44.04 and 51.69 % at the three concentrations compared to that in the control (F 3, 98 = 81.99, P < 0.001).
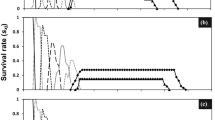
Effects on survival rate, life expectancy, reproduction value and fecundity of the F1 generation
The negative effect of imidacloprid was obvious when the age-stage survival rate in adults (Fig. 2) and life expectancy (e xj ) (Fig. 3) were plotted against the concentrations. Imidacloprid decreased the life expectancy, the decrease being gradual, for example, 25-day-old females and males that had not been exposed to imidacloprid were expected to live 51.70 and 48.88 days on average and those exposed to 4.837 mg L–1 imidacloprid, 39.78 and 37.75 days. The reproductive value (V xj ) is the contribution of individual of age x and stage j to the future population (Chi and Su 2006). Females near the peak of their reproductive abilities contributed more to the population than those at other ages and stages did. Exposure to sublethal concentrations of imidacloprid decreased the reproductive values gradually: 34-day-old females not exposed to imidacloprid had a markedly higher reproductive value, 232.30, when compared to 155.44 in those exposed to 4.837 mg L–1 imidacloprid concentration (LC5) (Fig. 4). The age-specific survival rate (l x ), female age-stage-specific fecundity (f x ), and age-specific fecundity of total population (m x ) are reported in Fig. 5. The curve of l x is a simplified version of S xj . Exposure to sublethal concentrations of imidacloprid lowered the survival rate and fecundity: in the control population, the maximum female age-stage-specific fecundity (f x ) was 35.95 eggs (at 40 days), compared to 25.52 eggs (at 37 days) and 24.51 eggs (at 37 dayss) in insects exposed to imidacloprid at 0.484 and 4.837 mg L–1, respectively.
Effects on the F1 generation demographic parameters
The demographic parameters of C. septempunctata estimated using the bootstrap technique are presented in Table 2. The intrinsic rate of increase (r m) was significantly lower in predators exposed to imidacloprid at 0.484 and 4.837 mg L–1 than in those in the control population. The finite rate of increase (λ) showed the same trend. The net reproduction rate (R 0) at 0.484 and 4.837 mg L–1 was significantly lower than in the control population, and so was the mean generation time (T) at 4.837 mg L–1. Moreover, the T values at the two lower imidacloprid concentrations were significantly higher than the T value in the control population.
Discussion
When insecticides are applied as foliar sprays in the field to suppress various arthropod pests, natural enemies of those pests are also likely to be exposed to the insecticides when foraging on the treated plants, with various direct and indirect effects. In the present study, sublethal concentrations of imidacloprid (LC5, 10 and 1 % of LC5) adversely affected the biological performances of the progeny of the exposed predators, i.e. through transgenerational effects. The adverse effects included a longer juvenile development duration, shorter lifespans and lower fecundity.
Imidacloprid is a neonicotinoid insecticide and is an agonist of insect nicotinic acetylcholine receptors. The toxicity symptoms in insects include loss of coordination, tremors and paralysis. In our study, the juvenile development duration was significantly longer in the beetles exposed to sublethal concentrations of imidacloprid, similarly to other studies in which imidacloprid extended development time of immature Hippodamia undecimnotata (Papachristos and Milonas 2008). An hypothesis that could explain this phenomenon is that imidacloprid may affect the fitness of F1 insects by reducing the feeding activity of their parents because ladybird beetles were unable to move or feed properly after being exposed to sublethal concentration(s) of imidacloprid (Nauen et al. 1998; Ramirez-Romero et al. 2008; Han et al. 2010; He et al. 2013). The anti-feeding properties of imidacloprid induce inhibition in absorption of nutrients, or a disruption of the hormonal balance in insects (Gerami et al. 2005; Han et al. 2012). Moreover, the decrease in resource acquisition, and in particular energy, in toxicant-contaminated organisms may match with recent theoretical models based on Dynamic Energy Budget theories, in which all toxicological data (from all stages and times) can be integrated in a time-independent parameters on the energy budget framework (Baas et al. 2010; Jager and Zimmer 2012).
Reproduction-related traits represent typical levels of insect populations in life-table studies (Stark et al. 2004; Desneux et al. 2007). In the present study, fecundity in parental and in F1 generations decreased through exposure to sublethal concentrations of imidacloprid (Fig. 1D, Table 1). This observation is consistent with earlier laboratory studies reporting lower fecundity in adult Encarsia inaron and Eretmocerus mundus following exposure to sublethal concentrations of imidacloprid (Sohrabi et al. 2012, 2013). Moreover, the number of eggs laid by the predatory coccinellid H. undecimnotata exposed to imidacloprid was reduced by one-third compared to that in the control population (Papachristos and Milonas 2008). A reduction in the fecundity of Rodolia cardinalis females exposed to imidacloprid was also reported by Grafton-Cardwell and Gu (2003). Similar results have been reported with another neonicotinoid insecticide, namely acetamiprid, which lowered the fecundity of female Eriopis connexa by 22-44 % and their fertility by 37-45 % (Fogel et al. 2013).
Reproduction in arthropods is largely regulated by neurohormones; neurohormonal imbalance resulting from the toxic effects of an insecticide may affect normal reproductive functions (Desneux et al. 2007). Parental reproductive physiology may be disrupted when the neurosecretory system is negatively affected by toxicants. Imidacloprid specifically targets the nicotinic acetylcholine receptors in insects (Palumbo et al. 2001). As a consequence, behavioural effects in predators are likely because sophisticated nervous activity is required in the process of prey detection by predators (Desneux et al. 2007). More recently, He et al. (2012) reported that sublethal concentrations of imidacloprid may decrease the reproductive capacity in S. japonicum by affecting their feeding capacity.
Demographical estimates are one of the most useful tools for studying the dynamics of insect populations in an ecotoxicological framework. With continued development and extended use, life tables are widely used in evaluating pest control through natural enemies, insect resistance and biological pesticides (Gao and Yang 2015). For example, Schneider et al. (2009) used the life table method for evaluating the effect of the herbicide glyphosate on the predatory insect Chrysoperla externa whereas Biondi et al. (2013) used this approach to estimate the population dynamics of a generalist parasitoid (Bracon nigricans) exposed to some bioinsecticides. Recently, the age-stage, two-sex life table has been used to describe the population characteristics of Cheilomenes sexmaculata under different conditions (Zhao et al. 2015).
The side effects of pesticides on natural enemies associated to crop pests in ecotoxicological field have hinted the need of more ecological relevant endpoints as measures of the impact of pesticides on beneficial species (Stark et al. 2004; Desneux et al. 2007). Thus, they should include not only the individual mortality and field performance of a given natural enemy, but also its population dynamics. Demographic parameters are particularly useful in assessing sublethal effects at the population level and are also essential in estimating population growth in ecological studies (Stark and Banks 2003; Biondi et al. 2013). In the present study, the imidacloprid effects on fertility had drastic consequences on the estimated demographic parameters, such as the net reproductive rate (R 0 ), the intrinsic rate of increase (r m ), and the finite rate of increase (λ) that tended to be lower in the populations exposed to imidacloprid at 0.484 and 4.837 mg L–1 compared to those values in the control population whereas the mean generation time (T) tended to be higher. The reduction in population growth rates indicated an intense reduction in the next generation population. Using demographic approach, our results demonstrate that imidacloprid at sublethal concentrations can suppress the growth of C. septempunctata populations by lowering both survival and reproduction.
In some cropping systems, employing natural enemies alone to control pests carries some economic risk in modern agriculture system (e.g. see Ragsdale et al. 2011), due to their potential inefficiency to keep the pest populations below the economic threshold. Strengthening the research on natural enemies of insect pests can help to the development of more precise combination strategies to improve the efficacy and the stability of IPM programmes. The results of the present study show that imidacloprid would markedly lower the populations of C. septempunctata by lowering their fecundity. Such a decrease could limit the efficacy of using predators as biocontrol agents in crops where imidacloprid is widely used, with negative implications for IPM programmes. The results of the present experiment, together with recent evidences about potential stimulatory effects of neonicotinoid sublethal concentrations on prey reproduction, namely on aphids (Guedes et al. 2016; Tan et al. 2012; Wang et al. 2017), on the side effects on nontarget populations of various natural enemies (He et al. 2012; Fogel et al. 2013; Malaquias et al. 2013; Zotti et al. 2013) and pollinators (Desneux et al. 2007; Blacquiere et al. 2012; Cresswell et al. 2012; Decourtye et al. 2013; Sanchez-Bayo 2014), suggest that more selective insecticides should be employed when natural enemies are present.
References
Abbes K, Biondi A, Kurtulus A, Ricupero M, Russo A, Siscaro G, Chermiti B, Zappalà L (2015) Combined non-target effects of insecticide and high temperatures on the parasitoid Bracon nigricans. PLoS One 10(9):e0138411
Abbott WS (1925) A method for computing the effectiveness of an insecticide. J Econ Entomol 18:265–267
Ali A, Ahmad F, Biondi A, Wang Y, Desneux N (2012) Potential for using Datura alba leaf extracts against two major stored grain pests, the khapra beetle Trogoderma granarium and the rice weevil Sitophillus oryzae. J Pest Sci 85:359–366
Ahmad M, Ossiewatsch HR, Basedow T (2003) Effects of neem treated aphids as food/hosts on their predators and parasitoids. J Appl Entomol 127:458–464
Baas J, Jager T, Kooijman B (2010) A review of DEB theory in assessing toxic effects of mixtures. Sci Total Environ 408:3740–3745
Bengochea P, Budia F, Viñuela E, Medina P (2014) Are kaolin and copper treatments safe to the olive fruit fly parasitoid Psyttalia concolor? J Pest Sci 87:351–359
Biondi A, Desneux N, Siscaro G, Zappalà L (2012a) Using organic-certified rather than synthetic pesticides may not be safer for biological control agents: selectivity and side effects of 14 pesticides on the predator Orius laevigatus. Chemosphere 87:803–812
Biondi A, Mommaerts V, Smagghe G, Vinuela E, Zappalà L, Desneux N (2012b) The non-target impact of spinosyns on beneficial arthropods. Pest Manag Sci 68:1523–1536
Biondi A, Zappalà L, Stark JD, Desneux N (2013) Do biopesticides affect the demographic traits of a parasitoid wasp and its biocontrol services through sublethal effects? PLoS One 8:e76548
Biondi A, Campolo O, Desneux N, Siscaro G, Palmeri V, Zappalà L (2015) Life stage-dependent susceptibility of Aphytis melinus DeBach (Hymenoptera: Aphelinidae) to two pesticides commonly used in citrus orchards. Chemosphere 128:142–147
Biondi A, Zappalà L, Di Mauro A, Tropea Garzia G, Russo A, Desneux N, Siscaro G (2016) Can alternative host plant and prey affect phytophagy and biological control by the zoophytophagous mirid Nesidiocoris tenuis?. BioControl 61:79–90
Bompard A, Jaworski CC, Bearez P, Desneux N (2013) Sharing a predator: can an invasive alien pest affect the predation on a local pest? Pop Ecol 55:433–440
Blacquiere T, Smagghe G, Van Gestel CA, Mommaerts V (2012) Neonicotinoids in bees: a review on concentrations, side-effects and risk assessment. Ecotoxicology 21:973–992
Bozsik A (2006) Susceptibility of adult Coccinella septempunctata (Coleoptera: Coccinellidae) to insecticides with different modes of action. Pest Manag Sci 62:651–654
Cabral S, Soares AO, Garcia P (2011) Voracity of Coccinella undecimpunctata: effects of insecticides when foraging in a prey/plant system. J Pest Sci 84:373–379
Chen Z, Qu Y, Xiao D, Song L, Zhang S, Gao X, Desneux N, Song D (2015) Lethal and social-mediated effects of ten insecticides on the subterranean termite Reticulitermes speratus. J Pest Sci 88:741–751
Chi H (1988) Life-table analysis incorporating both sexes and variable development rates among individuals. Environ Entomol 17:26–31
Chi H (2012) TWOSEX-MS Chart: a computer program for the age-stage, two-sex life table analysis. National Chung Hsing University, Taichung Taiwan
Chi H, Liu H (1985) Two new methods for the study of insect population ecology. Bull Inst Zool 24:225–240
Chi H, Su HY (2006) Age-stage, two-sex life tables of Aphidius gifuensis (Ashmead) (Hymenoptera: Braconidae) and its host Myzus persicae (Sulzer) (Homoptera: Aphididae) with mathematical proof of the relationship between female fecundity and the net reproductive rate. Environ Entomol 35:10–21
Cresswell JE, Desneux N, vanEngelsdorp D (2012) Dietary traces of neonicotinoid pesticides as a cause of population declines in honey bees: an evaluation by Hill’s epidemiological criteria. Pest Manag Sci 68:819–827
Desneux N, Wajnberg E, Fauvergue X, Privet S, Kaiser L (2004) Oviposition behaviour and patch‐time allocation in two aphid parasitoids exposed to deltamethrin residues. Entomol Exp Appl 112:227–235
Desneux N, Ramirez‐Romero R, Kaiser L (2006) Multistep bioassay to predict recolonization potential of emerging parasitoids after a pesticide treatment. Environ Toxicol Chem 25:2675–2682
Desneux N, Decourtye A, Delpuech JM (2007) The sublethal effects of pesticides on beneficial arthropods. Annu Rev Entomol 52:81–106
Decourtye A, Henry M, Desneux N (2013) Environment: overhaul pesticide testing on bees. Nature 497:188
Eisenback BM, Salom SM, Kok LT, Lagalante AF (2010) Lethal and sublethal effects of imidacloprid on Hemlock woolly Adelgid (Hemiptera: Adelgidae) and two introduced predator species. J Econ Entomol 103:1222–1234
Finney DJ (1971) Probit analysis. Cambridge University Press, Cambridge
Fogel MN, Schneider MI, Desneux N, Gonzalez B, Ronco AE (2013) Impact of the neonicotinoid acetamiprid on immature stages of the predator Eriopis connexa (Coleoptera: Coccinellidae). Ecotoxicology 22:1063–1071
Gao SK, Yang ZQ (2015) Application of life table in pest biological control. Chinese. J Biol Control 31:256–263
Grafton-Cardwell EE, Gu P (2003) Conserving vedalia beetle, Rodolia cardinalis (Mulsant) (Coleoptera: Coccinellidae), in citrus: a continuing challenge as new insecticides gain registration. J Econ Entomol 96:1388–1398
Gerami S, Jahromi KT, Ashouri A, Rasoulian G, Heidari A (2005) Sublethal effects of imidacloprid and pymetrozine on the life table parameters of Aphis gossypii Glover (Homoptera: Aphididae). Comm Appl Biol Sci 70:779–785
Gontijo PC, Moscardini VF, Michaud JP, Carvalho GA (2014) Non-target effects of chlorantraniliprole and thiamethoxam on Chrysoperla carnea when employed as sunflower seed treatments. J Pest Sci 87:711–719
Guedes RNC, Smagghe G, Stark JD, Desneux N (2016) Pesticide-induced stress in arthropod pests for optimized integrated pest management programs. Annu Rev Entomol 61:43–62
Han P, Niu CY, Lei CL, Cui JJ, Desneux N (2010) Quantification of toxins in a Cry1Ac+CpTI cotton cultivar and its potential effects on the honey bee Apis mellifera L. Ecotoxicology 19:1452–1459
Han P, Niu CY, Biondi A, Desneux N (2012) Does transgenic Cry1Ac+CpTI cotton pollen affect hypopharyngeal gland development and midgut proteolytic enzyme activity in the honey bee Apis mellifera L. (Hymenoptera, Apidae)? Ecotoxicology 21:2214–2221
He YX, Zhao J, Zheng Y, Zhan Z, Desneux N, Wu KM (2012) Lethal effect of imidacloprid on the coccinellid predator Serangium japonicum and sublethal effects on predator voracity and on functional response to the whitefly Bemisia tabaci. Ecotoxicology 21:1291–1300
He YX, Zhao JW, Zheng Y, Weng QY, Biondi A, Desneux N, Wu KM (2013) Assessment of potential sublethal effects of various insecticides on key biological traits of the tobacco whitefly, Bemisia tabaci. Int J Biol Sci 9:246–255
Jager T, Zimmer EI (2012) Simplified dynamic energy budget model for analysing ecotoxicity data. Ecol Model 225:74–81
Ji LL, Dong YK, Li ZH (2011) The indoor toxicity effect of four pesticides to the adult of Coccinella septempunctata. Shangdong Agric Sci 5:74–75. (in Chinese)
Liang P, Tian YA, Biondi A, Desneux N, Gao XW (2012) Short-term and transgenerational effects of the neonicotinoid nitenpyram on susceptibility to insecticides in two whitefly species. Ecotoxicology 21:1889–1898
Lu YH, Wu KM, Jiang YY, Guo YY, Desneux N (2012) Widespread adoption of Bt cotton and insecticide decrease promotes biocontrol services. Nature 487:362–365
Lucas E, Giroux S, Demougeo S, Duchesne RM, Coderre D (2004) Compatibility of a natural enemy, Coleomegilla maculata lengi (Col.,Coccinellidae) and four insecticides used against the Colorado potato beetle (Col.,Chrysomelidae). J Appl Entomol 128:233–239
Malaquias JB, Ramalho FS, Omoto C, Godoy WAC, Silveira RF (2013) Imidacloprid affects the functional response of predator Podisus nigrispinus (Dallas) (Heteroptera: Pentatomidae) to strains of Spodoptera frugiperda (JE Smith) on Bt cotton. Ecotoxicology 23:192–200
Mirande L, Desneux N, Haramboure M, Schneider MI (2015) Intraguild predation between an exotic and a native coccinellid in Argentina: the role of prey density. J Pest Sci 88:155–162
Moscardini VF, Gontijo PC, Michaud JP, Carvalho GA (2015) Sublethal effects of insecticide seed treatments on two nearctic lady beetles (Coleoptera: Coccinellidae). Ecotoxicology 5:1152–1161
Moser SE, Obrycki JJ (2009) Non-target effects of neonicotinoid seed treatments: mortality of coccinellid larvae related to zoophytophagy. Biol Control 51:487–492
Nauen R, Koob B, Elbert A (1998) Antifeedant effects of sublethal dosages of imidacloprid on Bemisia tabaci. Entomol Exp Appl 88:287–293
Palumbo JC, Horowitz AR, Prabhaker N (2001) Insecticidal control and resistance management for Bemisia tabaci. Crop Prot 20:739–765
Pan H, Liu Y, Liu B, Lu Y, Xu X, Qian X et al. (2014) Lethal and sublethal effects of cycloxaprid, a novel cis-nitromethylene neonicotinoid insecticide, on the mirid bug Apolygus lucorum. J Pest Sci 87:731–738
Papachristos DP, Milonas PG (2008) Adverse effects of soil applied insecticides on the predatory coccinellid Hippodamia undecimnotata (Coleoptera: Coccinellidae). Biol Control 47:77–81
Pérez-Hedo M, Urbaneja A (2015) Prospects for predatory mirid bugs as biocontrol agents of aphids in sweet peppers. J Pest Sci 88:65–73
Qu YY, Xiao D, Li JY, Chen Z, Biondi A, Desneux N, Gao XW, Song DL (2015) Sublethal and hornesis effects of imidacloprid on the soybean aphid Aphis glycines. Ecotoxicology 24:479–487
Ragsdale DW, Landis DA, Brodeur J, Heimpel GE, Desneux N (2011) Ecology and management of the soybean aphid in North America. Annu Rev Entomol 56:375–399
Ramirez-Romero R, Desneux N, Decourtye A, Chaffiol A, Pham-Delègue MH (2008) Does Cry1Ab protein affect learning performances of the honeybee Apis mellifera L. Ecotoxicol Environ Saf 70:327–333
Saeed R, Razaq M, Hardy IC (2015) The importance of alternative host plants as reservoirs of the cotton leaf hopper, Amrasca devastans, and its natural enemies. J Pest Sci 88:517–531
Sanchez-Bayo F (2014) The trouble with neonicotinoids. Science 6211:806–807
Sanchez-Bayo F, Goulson D, Pennacchio F, Nazzi F, Goka K, Desneux N (2016) Are bee diseases linked to pesticides? – A brief review. Environ Int 89-90:7–11
Schneider MI, Sanchez N, Pineda S, Chi H, Ronco A (2009) Impact of glyphosate on the development, fertility and demography of Chrysoperla externa (Neuroptera: Chrysopidae): ecological approach. Chemosphere 76:1451–1455
Singh SR, Walters KFA, Port GR, Northing P (2004) Consumption rates and predatory activity of adult and forth instar larvae of the seven spot ladybird, Coccinella septempunctata (L.), following contact with dimethoate residue and contaminated prey in laboratory arenas. Biol Control 30:127–133
Smith SF, Krischik VA (1999) Effects of systemic imidacloprid on Coleomegilla maculata (Coleoptera: Coccinellidae). Biol control 28:1189–1195
Sohrabi F, Shishehbor P, Saber M, Mosaddegh MS (2012) Lethal and sublethal effects of buprofezin and imidacloprid on the whitefly parasitoid Encarsia inaron (Hymenoptera: Aphelinidae). Crop Prot 32:83–89
Sohrabi F, Shishehbor P, Saber M, Mosaddegh MS (2013) Lethal and sublethal effects of imidcaloprid and buprofezin on the sweetpotato white parasitoid Eretmocerus mundus (Hymenoptera: Aphelinidae). Crop Prot 45:98–103
Stark JD, Banks JE (2003) Population-level effects of pesticides and other toxicants on arthropods. Annu Rev Entomol 48:505–519
Stark JD, Banks JE, Vargas R (2004) How risky is risk assessment: the role that life history strategies play in susceptibility of species to stress. Proc Natl Acad Sci USA 101:732–736
Stark JD, Vargas RI, Banks JE (2007) Incorporating ecologically relevant measures of pesticide effect for estimating the compatibility of pesticides and biocontrol agents. J Econ Entomol 100:1027–1032
Tan XL, Zhao J, Zhang F, Wang S (2014) Light dependency of life trails, reproduction, locomotion, and predaion in the polyphagous ladybird Hippodamia variegata. Entomol Exp Appl 152:200–208
Tan Y, Biondi A, Desneux N, Gao XW (2012) Assessment of physiological sublethal effects of imidacloprid on the mirid bug Apolygus lucorum (Meyer-Dür). Ecotoxicology 21:1989–1997
Tomizawa M, Casida JE (2001) Structure and diversity of insect nicotinic acetylcholine receptors. Pest Manag Sci 57:914–922
Torres JB, Ruberson JR (2004) Toxicity of thiamethoxam and imidacloprid to Podisus nigrispinus (Dallas) (Heteroptera: Pentatomidae) nymphs associated to aphid and whitefly control in cotton. Neotrop Entomol 33:99–106
van Herk WG, Vernon RS, Vojtko B, Snow S, Fortier J, Fortin C (2015) Contact behaviour and mortality of wireworms exposed to six classes of insecticide applied to wheat seed. J Pest Sci 88:717–739
Vincent C, Ferran A, Guige L, Gambier J, Brun J (2000) Effects of imidacloprid on Harmonia axyridis (Coleoptera: Coccinellidae) larval biological and locomotorybehavior. Eur J Entomol 97:501–506
Wang S, Qi Y, Desneux N, Shi X, Biondi A, Gao X (2017) Sublethal and transgenerational effects of short-term and chronic exposures to the neonicotinoid nitenpyram in Aphis gossypii. J Pest Sci. doi: 10.1007/s10340-016-0770-7
Youn YN, Seo MJ, Shin JG, Jang C, Yub YM (2003) Toxicity of greenhouse pesticides to multicolored Asian ladybeetles, Harmonia axyridis (Coleoptera: Coccinellidae). Biol Control 28:164–170
Yu CH, Fu, MR, LinRH, ZhangY, LiuYQ, JiangH, Brock, T.CM (2014a) Toxic effects of hexaflumuron on the development of Coccinella septempunctata. Environ Sci Pollut Res. 21:1418–1424
Yu CH, Lin RH, Fu MR, Zhou YM, Zong FL, Jiang H, Lv N, Piao XY, Zhang J, Liu YQ, Brock TCM (2014b) Impact of imidacloprid on life-cycle development of Coccinella septempunctata in laboratory microcosms. Ecotoxicol Environ Safe 110:168–173
Zotti MJ, Grutzmacher AD, Lopes IH, Smagghe G (2013) Comparative effects of insecticides with different mechanisms of action on Chrysoperla externa (Neuroptera: Chrysopidae): Lethal, sublethal and dose-response effects. Insect Sci 20:743–752
Zhang DW, Dong M, Du XG (2011) Effects of organic management on predator densities of Myzus persicae (Sulzer) during transition to organic agriculture in peach fields in China. J Sustain Agri 35:826–839
Zhang X, Xu Q, Lu W, Liu F (2015) Sublethal effects of four synthetic insecticides on the generalist predator Cyrtorhinus lividipennis. J Pest Sci 88:383–392
Zhao J, Li S, Gao XW, Zhang F, Wang S (2015) Comparison of life tables of Cheilomenes sexmaculata (Coleoptera: Coccinellidae) under laboratory and greenhouse conditions. J Econ Entomol 108:1700–1707
Acknowledgements
This research was supported by the National Key Basic Research and Development Program (973 program 2013CB127605), the Special Fund for Agro-scientific Research in the Public Interest (201303108), and the Beijing Novo Program (z121105002512039). The authors are grateful to Professor Hsin Chi (Laboratory of Theoretical and Applied Ecology, Department of Entomology, National Chung Hsing University, Taiwan) for providing the age-stage two-sex life table software.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Statement of Human and Animal Rights
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Informed consent
There were no human subjects, thus informed consent is not applicable for this study.
Additional information
Da Xiao and Jing Zhao contributed equally to the study
Rights and permissions
About this article
Cite this article
Xiao, D., Zhao, J., Guo, X. et al. Sublethal effects of imidacloprid on the predatory seven-spot ladybird beetle Coccinella septempunctata . Ecotoxicology 25, 1782–1793 (2016). https://doi.org/10.1007/s10646-016-1721-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-016-1721-z