Abstract
Catch-and-release angling is growing as a tool for the conservation of fish stocks because it assumes that the impacts of angling are negligible. However, many studies have shown that catch-and-release can be stressful to the fish and even result in mortality. Bonefishing represents a popular catch-and-release fishery in the tropics and subtropics, with most anglers spending 6+ hours per day in full sunlight. To protect themselves, anglers typically employ sun protection in the form of liquid sunscreen and UV-blocking clothing. Exposure to chemicals contained in sunscreens may impose additional stressors on fish that are handled and subsequently released. In this study we conducted two separate experiments in the lab facilities in Cape Eleuthera, Bahamas. The first examined bonefish feeding behaviors in response to bait handled with zinc-based sunscreen, oxybenzone-based sunscreen, and no coating on the researcher’s hands. The second experiment quantified the effects of sunscreens and UV blocking gloves on the removal of fish’s protective mucus layer as a result of handling. We did not observe evidence of a change in feeding behavior when bait was handled with hands covered in sunscreen compared to wet hands. However, there was an increase in removal of protective mucus of bonefish when researcher’s hands were coated in oxybenzone containing sunscreen compared to researchers handling fish with wet hands. The results of this study indicate wet hands are the best way to handle fish when participating in catch-and-release angling.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Bonefishing is a popular recreational fishery in the Bahamas, and anglers voluntarily partake in catch-and-release practices. Catch-and-release angling is often practiced in an effort to increase the longevity and sustainability of a fishery. This type of angling assumes the costs of landing a fish are negligible and that fish survival will not be negatively impacted by capture (Muoneke and Childress 1994; Cooke and Suski 2005; Cooke and Schramm 2007; Danylchuk et al. 2007b); however, recent research has shown that catch-and-release practices can result in greater susceptibility to predation and thus can lead to mortality (Broadhurst et al. 2005; Danylchuk et al. 2007a). Sub-lethal consequences of catch-and-release angling such as changes in physiology, reproductive behavior, swimming performance, and physical injury due to hooking can also result in delayed mortality or reduced fitness (Suski et al. 2003; Schreer et al. 2005; Arlinghaus et al. 2007; Stein et al. 2012).
The physiological and behavioral consequences of catch-and-release angling have been attributed to stressors such as exhaustive exercise, injury, air exposure, and handling (Suski et al. 2007; Danylchuk et al. 2007a). In particular, injuries incurred as a result of handling (e.g., mucus removal, abrasions, scale loss, and fin fraying) increase a fish’s susceptibility to diseases/pathogens, cause dehydration due to passive water loss, and increase the energetic demand of swimming due to reduced lubrication, all of which can lead to delayed mortality (Bernadsky et al. 1993; Barthel et al. 2003). Kostecki et al. (1987), for example, found that mucus and scale removal in Atlantic salmon smolts resulted in mortality directly from the injuries, but more often from secondary problems such as predation or disease. Typically, handling fish with dry hands results in the greatest loss of mucus and scales with most catch-and-release guidelines indicating it is best to handle fish with wet hands or wet gloves (Pelletier et al. 2007), yet very little is known about how sunscreen applied to hands might influence mucus loss.
Anglers are often concerned with the negative health issues associated with prolonged exposure to the sun (Kricker et al. 1994). This is particularly true in the tropics, where the glare/reflection of the sun off the surface of the water increases sun exposure (Woolley et al. 2002). To protect themselves, anglers typically apply sun protection in the form of conventional cream or spray sunscreen and/or wear UV blocking clothing (e.g., gloves, shirts, buffs) (Neale et al. 2002). UV blocking apparel has become more popular due to the fact that sunscreen can wash off over time. There are many different types of sunscreens available to the average angler; however, these can generally be broken up into oxybenzone containing sunscreens, and zinc-based sunscreens (Cross et al. 2007).
Many anglers believe that fish can detect certain odors on artificial flies such as, gasoline, insect repellent and sunscreen (Brown 2008), which will make them less likely to consume bait. A common response to environmental contaminants is reduced feeding which can lead to reduced energy intake (Maltby 1999). Research by Brown et al. (1987) found that for largemouth bass the addition of the chemical pentachlorophenol to the water caused fish to perform fewer strikes. Additionally, Morgan and Kiceniuk (1990) found that low levels of fenitrothion decrease the efficiency of Atlantic salmon’s attack sequence as well as the number of prey ingested. However, Little et al. (1990) tested 6 different agricultural chemicals, carbaryl, chlordane, dimethylamine salt of 2,4-dichlorophenoxyacetic acid (2,4-DMA), tributyl phosphorotrithioate (DEF1), methyl parathion, and pentachlorophenol, and found that for rainbow trout the frequency of strikes is less sensitive to certain toxicants compared to others. These studies have focused on what adding chemicals to the water will do to feeding behavior, and there is a lack of literature focusing on how directly contaminated bait will affect feeding response.
The efficacy of catch-and-release angling in preserving fish stocks may be reduced if sun sunscreens result in injury or altered feeding behaviors. The goals of this study were two-fold. First, we quantified the effects of oxybenzone sunscreen and zinc-oxide sunblock on the feeding behavior of bonefish. Second, we assessed how sunscreens, UV-blocking gloves, and wet hands affected mucus layer removal and recovery time of bonefish following a simulated angling event. The outcomes of this study will improve best handling practices for catch-and-release angling of bonefish.
Materials and methods
Bonefish collection and husbandry
This study was conducted at the Cape Eleuthera Institute (CEI), Eleuthera, the Bahamas between January 24th and July 18th, 2014. Adult bonefish N = 77 [average size = 396.3 mm fork length, standard error (SE) = 5.8 mm, range = 326–505 mm] were collected from nearby saltwater tidal creeks (see Danylchuk et al. 2007a for a description of sampling sites) by seine net, then transported in 142 L plastic coolers by boat to CEI’s wetlab. The duration of transport was typically 15–30 min and water was exchanged every 5 min (Murchie et al. 2009). Prior to experimentation, bonefish were acclimated to continuously aerated 13,180 L flow-through (1800 L/h) seawater holding tanks for at least 3 days or until they resumed feeding. During acclimation, water temperature (range 21–28.1 °C), dissolved oxygen (range 5.16–8.3 mg/L), and salinity (range 34.7–39.4 ppt) were monitored on a daily basis. Bonefish were fed standard sized, 1–2 g, pieces of commercially available raw shrimp (Sea Best, Beaver Street Fisheries Inc., Jacksonville, FL, USA).
Feeding preference
Following acclimation, fish (N = 31) were transferred to 53 L totes filled with seawater and transported to an individual 13,180 L tank. Bonefish were acclimated to their tank for 24 h (Murchie et al. 2009), then continually fed raw shrimp for a 10 min observation period, or until fish became satiated (Grove et al. 1978). Any shrimp remaining in the tank following the 10-min period was collected and weighed. During this observational period, feeding behaviors of the bonefish were recorded, and the mass of shrimp consumed was calculated as the difference between the wet weight of shrimp placed in the tank, and the wet weight of shrimp left uneaten. Four different feeding behaviors were recorded along with the time they were first exhibited: investigation, pick-up, expel, and mouth (Bardach et al. 1980). Investigation was classified as visual examination of the shrimp, indicated by bonefish coming into close proximity to the raw shrimp. Pick-up was recorded when a bonefish drew a piece of shrimp into its mouth, and expel was noted when a fish dropped a piece of shrimp. Mouthing was recorded when the bonefish mechanically processed (i.e., broke the food item into smaller pieces) the shrimp, exhibited by simultaneous rapid opening and closing of the mouth, head thrusts, and operculum flares.
The influence of sunscreen on the feeding behavior of bonefish was assessed after fasting the fish for 24 h. Researchers applied oxybenzone based sunscreen (Banana Boat, broad spectrum SPF 30 2012 Energizer Personal Care, LLC, Shelton, CT, U.S.A.) (N = 10) or zinc-based sunscreen (Solar Sense, broad spectrum SPF 50, CCA Industries, Inc., Rutherford, NJ, U.S.A.) (N = 11) to their hands before handling the same quantity of shrimp consumed on the previous day. Bonefish were continually fed raw shrimp for a 10 min observation period, or until fish became satiated. Feeding behavior during this period was recorded as described above. Shrimp handled with wet, clean hands were fed to bonefish (N = 10) and served as controls for this experiment. All researcher’s hands were washed prior to handling food or applying sunscreen. The suggested amount of sunscreen by the U.S. FDA is 2 mg for every cm2 of skin, so 0.25–0.30 g of sunscreen was applied (Danovaro et al. 2008).
Handling and recovery
Throughout acclimation bonefish (N = 46) were fed Spanish sardines (Sardinella aurita, McRoberts Sales Co., Gainesville, FL, USA). Once acclimated, bonefish were transferred using rubber dip nets to individual 1400 L tanks (Casselman 2005). Anglers typically fight a fish to exhaustion (4 min) and then handle the fish to remove the hook (Cooke and Philipp 2004). To simulate a typical angling event, bonefish were exercised by lightly grabbing the tail in an effort to make the fish swim continuously for 4 min, (Cooke and Philipp 2004; Danylchuk et al. 2007a). Following exercise, bonefish were handled with wet hands (N = 9), wet hands coated with 0.25–0.30 g oxybenzone based sunscreen (N = 9), wet hands coated with 0.25–0.30 g zinc based sunscreen (N = 10), and wet fishing gloves (N = 9) (Abaco Bay Fingerless Sun Glove, Glacier Glove, Reno, NV, U.S.A.). These fish were also air exposed for 30 s to simulate the typical amount of time needed for anglers to take a photo of their fish (Cooke and Philipp 2004; Danylchuk et al. 2007a). Control fish (N = 9) were exercised and transferred into a holding tank using a rubber dip net without being handled. All fish were transferred to tanks using a rubber dip net, in an attempt to standardize treatments. Mucus lost by bonefish as a result of handling was collected using a plastic scraper to remove mucus from the experimenters’ hands, and a sieve (No. 35, 32 mesh, 0.500 mm, #3076 Hubbard) was used to collect any additional mucus remaining in the water of the 53L handling tote. Bonefish mucus is generally easily identifiable on the experimenters’ hands and the surface of the water due to its “clumping” nature (Fig. 1). Mucus was air dried for 48 h and the dry weight measured to the nearest 0.001 g (Gemini-20, Portable milligram scale, AWS, Norcross, GA).
Following the 30 s handling protocol, bonefish were transferred to a 53 L tote filled with seawater, dart tagged for identification (80 mm, PDS small plastic tipped dart tag, Hallprint, Hindmarsh Valley, South Australia, Australia), and then transferred to a separate 13,180 L flow-through tank for recovery. Prior to tag insertion, the ends of the dart tags were dipped in different colored Plasti Dip (Performix, Plasti Dip International, Blaine, MN) to aid in visual identification of individual fish in tanks. Injury caused by handling treatment was quantified daily by visually estimating the percent of the body covered by bacterial growth and bruising. Recovery was monitored daily for 2–3 weeks after handling, or until the bonefish exhibited normal feeding behavior and showed no sign of bacterial growth or bruising.
Analyses
A visual analysis of fitted residuals, using a normal probability plot (Anscombe and Tukey 1963), was used to assess normality, while Hartley’s Fmax test (Hartley 1950), combined with visual inspection of the distribution of fitted residuals, were used to assess homogeneity of variance for all tests. Ranked data were used if either normality or homogeneity of variance assumptions were violated (Siegel and Castellan 1988). Time until initial pickup in the control, zinc and oxybenzone treatments was compared with a one-way ANOVA. The difference in the amount of shrimp consumed between uncoated and sunscreen coated treatments was quantified with a one-way, repeated-measures ANOVA, where the main effects were mass of shrimp consumed, sunscreen treatment (control, zinc, or oxybenzone), and their interaction, with fish ID as a repeated random variable. A repeated measures design was necessary because multiple measurements were taken from each animal on sequential days meaning that each measurement might not be independent and could potentially be correlated within an individual (Laird and Ware 1982; Lindstrom and Bates 1990). Time until first expulsion minus time until first pick-up of shrimp in control, zinc and oxybenzone treatments as well as weight of mucus removed across treatments were assessed using one-way analysis of variance (ANOVA) followed by a Tukey-Kramer HSD test when appropriate. All analyses were performed using JMP 7.0.1 (SAS Institute Inc., Cary, NC, USA), all means reported as ± SE where appropriate. Significance was accepted at probabilities of 0.05 or less.
Results
Feeding preference
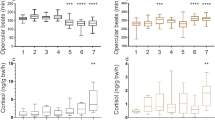
Fork length (346.5 ± 3.7 mm; mean ± SE) of bonefish did not vary across treatments (ANOVA, F = 1.45, P = 0.27). The time it took for a fish to pick-up shrimp did not vary for the zinc-based, oxybenzone-based or control treatments (repeated measures ANOVA, F = 1.49, P = 0.24) (Fig. 2a). Similarly, bonefish did not expel shrimp covered in oxybenzone or zinc-based sunscreen faster relative to uncoated shrimp (repeated measures ANOVA, F = 0.34, P = 0.71) (Fig. 2b). The amount of uncoated shrimp consumed compared to the amount of shrimp consumed the following day (when shrimp was coated in sunscreen) did not vary across treatments (RMANOVA, F = 2.07, P = 0.15) (Fig. 3).
Handling and recovery
The fork length did not vary across handling treatments (380.2 ± 5.0 mm, mean ± SE) (F = 0.94, P = 0.45). No mucus loss was observed when transporting bonefish in the rubber-bagged dip net. Bonefish handled with wet hands lost 50 % less mucus (0.05 ± 0.01 SE g) than the bonefish that were handled with oxybenzone-based sunscreen (0.10 ± 0.01 SE g) (ANOVA, F = 3.38, P 0.03; Fig. 4). The zinc and glove treatments resulted in an intermediate loss of mucous, but no significant difference between these treatments and the mass of mucuos lost in either the wet hands or oxybenzone-coated hands treatments was observed (Fig. 4). Despite this, recovery time did not vary across treatments (ANOVA, F = 1.6, P = 0.19). Two of the bonefish handled with zinc sunscreen-coated hands developed a bacterial infection, which lasted the duration of the 2-week recovery period. Bacterial infection was not observed in the control, oxybenzone-based sunscreen, or UV glove handling treatments.
Discussion
A handling event is stressful for fish because it often involves air exposure, the removal of protective mucus, abrasions, loss of scales, and fin fraying (Ashley 2007; Cooke and Schramm 2007). This study shows that the amount of protective mucus layer removed increased by 50 % when fish were handled with hands coated in oxybenzone-based sunscreen relative to fish handled with wet hands. The zinc and gloves treatments removed intermediate amounts of mucus, but were not significantly different from either wet hands or the oxybenzone treatment. Mucus acts as a physical barrier between a fish and its environment, and contains chemical properties to minimize viral, bacterial, or parasitic infection (Hellio et al. 2002; Ashley 2007). Removal of the mucus layer due to handling may result in delayed mortality, and heightened energetic demands to cope with infection and physiological stress (Kostecki et al. 1987). Dry hands were not chosen as a treatment in this study because it is well documented that dry hands are the most detrimental way to handle a fish due to the removal of the slime layer (Ashley 2007; Butcher et al. 2009; Thomson 2011; Schwabe et al. 2014). Not handling a fish removed the least amount of mucus followed by handling fish with wet hands, where as handling bonefish with hands coated in oxybenzone-based sunscreen resulted in the greatest loss of mucus. Thus, if possible fish should not be handled, but if handling is unavoidable, then fish should be handled with wet uncontaminated hands.
Although recovery time did not vary across treatments, the only two fish to exhibit bacterial infections after handling were those in the zinc-sunscreen treatment group. Zinc-based sunscreens are thicker and can clog pores (Moloney et al. 2002). Although clogging pores has only been observed in humans, it may have contributed to the infection on the fish handled with zinc. The risk of infection may be exacerbated because of the additional stressors (e.g., exhaustion, air exposure, injury) that occur during a catch-and-release angling event (Meka 2004). Even though a difference in mucus removal was observed, all the fish were able to recover after 2 weeks, and most showed no visible signs of bruising or abrasions. Stable laboratory conditions may have contributed to an underestimation of recovery time as bonefish were not exposed to typical stressors after release such as predators and increased energetic cost of foraging. Thus, the fish could have used their energy to heal rather than spend energy foraging or avoiding predators.
Although time to pick-up was slightly shorter for uncoated shrimp than for shrimp exposed to sunblock, there was no significant difference across treatments, nor was there a difference between the time it took for a fish to expel the shrimp across treatments. Similarly, bonefish did not consume less zinc or oxybenzone-based sunscreen-coated shrimp when compared to uncoated shrimp. In teleosts, the identification of a forage item via olfactory, visual, or vibratory cues elicits a feeding response indicated by changes in fish behavior (e.g., gill flaring, initiating a search pattern) to best maximize the likelihood of encountering the prey item (Jones 1992; Hara 2006). Primary foraging habitat of bonefish is characterized by shallow (<2 m) sand, mud, or sea grass flats, with generally good visibility (Crabtree et al. 1998). The non-discriminate foraging by bonefish of food contaminated by sunscreen in this experiment suggests that other cues such as sight may play a more important role in identifying prey than olfactory cues.
Catch-and-release angling is often promoted as a way conserve fish stocks and a wealth of information exists on the best practices when handling bonefish (Cooke and Philipp 2004; Danylchuk et al. 2007b). Recommendations include limiting fight time, avoiding air exposure, using barbless hooks, avoiding locations with high predator burdens, and handling fish with wet hands (Cooke et al. 2006). Here we show that some, but not all UV protection techniques have the potential to injure bonefish by removing mucus. However, UV protection remains a concern for anglers, a population of people that spend a disproportionate amount of time exposed to the elements (Kricker et al. 1994). To limit mucus loss and provide sun protection for anglers, we recommend the following options; remove hooks without handling the fish or, if the previous option is not feasible, then handle the fish with wet hands that have not been coated with sunscreen. By applying these recommendations in conjunction with other best handling practices, anglers will minimize disturbances associated with catch-and-release angling.
References
Anscombe FJ, Tukey JW (1963) The examination and analysis of residuals. Technometrics 5:141–160
Arlinghaus R, Cooke SJ, Lyman J, Policansky D, Schwab A, Suski CD, Sutton SG, Thorstad EB (2007) Understanding the complexity of catch-and-release in recreational fishing: an integrative synthesis of global knowledge from historical, ethical, social, and biological perspectives. Rev Fish Sci 15:75–167
Ashley PJ (2007) Fish welfare: current issues in aquaculture. Appl Anim Behav Sci 104:199–235
Bardach JE, Manguson JJ, May RC, Reinhart JM (1980) Fish behavior and its use in the capture and culture of fishes. International Center for Living Aquatic Resources Management, Manila
Barthel BL, Cooke SJ, Suski CD, Philipp DP (2003) Effects of landing net mesh type on injury and mortality in a freshwater recreational fishery. Fish Res 63:275–282
Bernadsky G, Sar N, Rosenberg E (1993) Drag reduction of fish skin mucus: relationship to mode of swimming and size. J Fish Biol 42:797–800
Broadhurst MK, Gray CA, Reid DD, Wooden MEL, Young DJ, Haddy JA, Damiano C (2005) Mortality of key fish species released by recreational anglers in an Australian estuary. J Exp Mar Biol Ecol 321:171–179
Brown D (2008) Fly fishing for bonefish, 3rd edn. The Lyons Press, Gulford
Brown JA, Johansen PH, Colgan PW, Mathers RA (1987) Impairment of early feeding behavior of largemouth bass by pentachlorophenol exposure: a preliminary assessment. Trans Am Fish Soc 116:71–78
Butcher PA, Broadhurst MK, Hall KC, Cullis BR, Nicoll RG (2009) Scale loss and mortality in angled-and-released eastern sea garfish (Hyporhamphus australis). ICES Journal of Marine Science: Journal Du Conseil
Casselman S (2005) Catch-and-release angling: a review with guidelines for proper fish handling practices. Fisheries Section, Fish and Wildlife Branch, Ontario Ministry of Natural Resources, Ontario, Canada
Cooke SJ, Philipp DP (2004) Behavior and mortality of caught-and-released bonefish (Albula spp.) in Bahamian waters with implications for a sustainable recreational fishery. Biol Conserv 118:599–607
Cooke SJ, Schramm HL (2007) Catch-and-release science and its application to conservation and management of recreational fisheries. Fish Manag Ecol 14:73–79
Cooke S, Suski C (2005) Do we need species-specific guidelines for catch-and-release recreational angling to effectively conserve diverse fishery resources? Biodivers Conserv 14:1195–1209
Cooke SJ, Danylchuk AJ, Danylchuk SE, Suski CD, Goldberg TL (2006) Is catch-and-release recreational angling compatible with no-take marine protected areas? Ocean Coast Manag 49:342–354
Crabtree RE, Stevens C, Snodgrass D, Stengard FJ (1998) Feeding habits of bonefish, Albula vulpes, from the waters of the Florida Keys. Fish Bull 96:754–766
Cross SE, Innes B, Roberts ME, Tsuzuki T, Robertson TA, McCormik P (2007) Human skin penetration of sunscreen nanoparticles: In-vitro assessment of a novel micronized zinc oxide formulation. Skin Pharmacol Physiol 20:148–154
Danovaro R, Bongiorni L, Corinaldesi C, Giovannelli D, Damiani E, Astolfi P, Greci L, Pusceddu A (2008) Sunscreens cause coral bleaching by promoting viral infections. Environ Health Perspect 116:441–447
Danylchuk AJ, Danylchuk SE, Cooke SJ, Goldberg TL, Koppelman JB, Philipp DP (2007a) Post-release mortality of bonefish, Albula vulpes, exposed to different handling practices during catch-and-release angling in Eleuthera, the Bahamas. Fish Manag Ecol 14:149–154
Danylchuk SE, Danylchuk AJ, Cooke SJ, Goldberg TL, Koppelman J, Philipp DP (2007b) Effects of recreational angling on the post-release behavior and predation of bonefish (Albula vulpes): the role of equilibrium status at the time of release. J Exp Mar Biol Ecol 346:127–133
Grove D, Loizides L, Nott J (1978) Satiation amount, frequency of feeding and gastric emptying rate in Salmo gairdneri. J Fish Biol 12:507–516
Hara TJ (2006) Feeding behaviour in some teleosts is triggered by single amino acids primarily through olfaction. J Fish Biol 68:810–825
Hartley HO (1950) The maximum F-ratio as a short-cut test for heterogeneity of variance. Biometrika 37:308–312
Hellio C, FAU PA, Beaupoil CF, Bourgougnon NF, Gal YL (2002) Antibacterial, antifungal and cytotoxic activities of extracts from fish epidermis and epidermal mucus. Int J Antimicrob Agents 20:214–219
Jones KA (1992) Food search behaviour in fish and the use of chemical lures in commercial and sports fishing. Fish Chemoreception 6:288–320
Kostecki PT, Clifford P, Gloss SP, Carlisle JC (1987) Scale loss and survival in smolts of Atlantic salmon (Salmo-salar) after turbine passage. Can J Fish Aquat Sci 44:210–214
Kricker A, Armstrong BK, English DR (1994) Sun exposure and non-melanocytic skin cancer. Cancer Causes Control 4:367–392
Laird NM, Ware JH (1982) Random-effects models for longitudinal data. Biometrics 963-974
Lindstrom MJ, Bates DM (1990) Nonlinear mixed effects models for repeated measures data. Biometrics 673-687
Little RD, Archeski BA, Flerov VI, Kozlovskaya (1990) Behavioral indicators of sublethal toxicity in rainbow trout. Arch Environ Contam Toxicol 19:380–385
Maltby L (1999) Studying stress: the importance of organism-level responses. Ecol Appl 9:431–440
Meka JM (2004) The influence of hook type, angler experience, and fish size on injury rates and the duration of capture in an Alaskan catch-and-release rainbow trout fishery. N Am J Fish Manag 24:1309–1321
Moloney FJ, Collins S, Murphy GM (2002) Sunscreens: safety, efficacy and appropriate use. Am J Clin Dermatol 3:185–191
Morgan MJ, Kiceniuk JW (1990) Effect of fenitrothion on the foraging behavior of juvenile Atlantic salmon. Environ Toxicol Chem 9:489–495
Muoneke MI, Childress WM (1994) Hooking mortality: a review for recreational fisheries. Rev Fish Sci 2:123–156
Murchie KJ, Danylchuk SE, Pullen CE, Brooks E, Shultz AD, Suski CD, Danylchuk AJ, Cooke SJ (2009) Strategies for the capture and transport of bonefish, Albula vulpes, from tidal creeks to a marine research laboratory for long-term holding. Aquacult Res 40:1538–1550
Neale R, Williams G, Green A (2002) Application patters among participants randomized to daily sunscreen use in a skin cancer prevention trial. Am Med Assoc 138:1319–1325
Pelletier C, Hanson KC, Cooke SJ (2007) Do catch-and-release guidelines from state and provincial fisheries agencies in North America conform to scientifically based best practices? Environ Manage 39:760–773
Rollin BE, Kessel ML (2012) Guidelines for the treatment of animals in behavioural research and teaching. Anim Behav 83:301–309
Schreer JF, Resch DM, Gately ML, Cooke SJ (2005) Swimming performance of brook trout after simulated catch-and-release angling: looking for air exposure thresholds. N Am J Fish Manag 25:1513–1517
Schwabe M, Meinelt T, Phan TM, Cooke SJ, Arlinghaus R (2014) Absence of handling-induced Saprolenia infection in juvenile rainbow trout with implications for catch-and-release angling. N Am J Fish Manag 34:1221–1226
Siegel S, Castellan NJ (1988) Nonparametric statistics for the behavioral sciences. McGraw-Hill Book Company, New York
Stein JA, Shultz AD, Cooke SJ, Danylchuk AJ, Hayward K, Suski CD (2012) The influence of hook size, type, and location on hook retention and survival of angled bonefish (Albula vulpes). Fish Res 113:147–152
Suski CD, Svec JH, Ludden JB, Phelan FJS, Philipp DP (2003) The effect of catch-and-release angling on the parental care behavior of male smallmouth bass. Trans Am Fish Soc 132:210–218
Suski CD, Cooke S, Danylchuk A, O’Connor C et al (2007) Physiological disturbance and recovery dynamics of bonefish (Albula vulpes), a tropical marine fish, in response to variable exercise and exposure to air. Comp Biochem Physiol A Mol Integr Physiol 148:664–673
Thomson GH (2011) Protection of the undersized fish. Trans Am Fish Soc 42:171–178
Woolley T, Buettner P, Lowe J (2002) Sun-related behaviors of outdoor working men with a history of non-melanoma skin cancer. J Occup Environ Med 44:847–854
Acknowledgments
Special thanks go to the interns at the Cape Eleuthera Institute for the long days in the field and laboratory. Jessica Clarke, Matti McAlpin and Kevin Delaney played key roles in maintaining fish stocks in the laboratory. Eric Schneider was instrumental in the collection of bonefish from tidal creeks in South Eleuthera. Thanks to the Cape Eleuthera Institute for providing lab space. All research was carried out under research permit (MAF/FIS/17 and MAF/FIS/34) issued by the Bahamian Department of Marine Resources and according to the Cape Eleuthera Institute’s animal care protocols developed using the guidelines of the Association for the Study of Animal Behaviour and the Animal Behaviour Society (Rollin and Kessel 2012).
Compliance with Ethical Standards
These authors would like to report no conflicts on interest. This article does not contain any studies with human participants performed by any of the authors. All applicable international and institutional guidelines for the care and use of animals were followed.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hannan, K.D., Zuckerman, Z.C., Haak, C.R. et al. Impacts of sun protection on feeding behavior and mucus removal of bonefish, Albula vulpes . Environ Biol Fish 98, 2297–2304 (2015). https://doi.org/10.1007/s10641-015-0457-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-015-0457-6