Abstract
We examined the effects of river environment on the timing of spawning migrations by anadromous Pacific lamprey, Lampetra tridentata, in the Columbia River (U.S.A.). In a 41-year time series of adult lamprey counts, migration timing was earliest in warm, low-discharge years and latest in cold, high-flow years. Threshold temperatures associated with run timing were similar throughout the dataset despite significant impoundment-related warming, suggesting that temperature-dependent migration cues have been temporally stable. Within each year, migration rates of PIT-tagged lampreys were positively correlated with temperature and negatively correlated with discharge through multiple river reaches, offering additional evidence for environmental control of upstream movement. Both visual count and PIT-tag data indicated that there may be population-based differences in migration timing within the aggregate Columbia River lamprey run. These life history and behavioral results have potentially far-reaching implications for management of lamprey species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lampreys, jawless fish of the Family Petromyzontidae, are among the oldest existing vertebrates, having changed little since emerging more than 350 million years ago (e.g. Gess et al. 2006). Despite their persistence through time, many lamprey populations are now declining throughout much of their distribution (e.g., Renaud 1997). Attempts to conserve and restore lampreys are hindered by a lack of basic information on their natural history and habitat requirements. In the USA, lamprey managers are also confronted by negative perceptions of lampreys as parasites, largely as a result of damage to fisheries following invasion of sea lampreys (Petromyzon marinus) into the Laurentian Great Lakes (e.g., Bryan et al. 2005). The research described here was undertaken to improve our understanding of the life history and migration behaviors of Pacific lamprey (Lampetra tridentata), an imperiled population in the Pacific Northwest, USA.
In recent decades, anadromous Pacific lampreys along the west coast of North America, have experienced broad-based population declines and regional extirpations (Beamish and Northcote 1989; Kostow 2002; Moser and Close 2003). These declines parallel those of Pacific salmonids (Oncorhynchus spp.), perhaps because the two groups share widely sympatric distributions (Scott and Crossman 1973; Simpson and Wallace 1978; Moyle 2002) and similar anadromous life histories (McDowall 2001; Quinn and Myers 2004). Pacific lamprey adults feed and mature in the ocean, generally enter freshwater and migrate towards spawning areas during summer, and then overwinter and spawn the following spring. Adults do not feed while in freshwater. Larval lamprey (ammocoetes) rear in freshwater as filter feeders for three to seven years and then transform to migrating juveniles (macrophthalmia), develop an oral disc and eyes, and migrate to the ocean to begin their parasitic life stage. Threats to anadromous taxa in the Pacific region include habitat degradation and loss, dam-related mortality, increases in non-native predators, and environmental changes in freshwater and marine habitats (e.g., Close et al. 2002; Ruckelshaus et al. 2002; Battin et al. 2007).
In 2003, the U.S. Fish and Wildlife Service (USFWS) considered listing Pacific lamprey under the U.S. Endangered Species Act. The ensuing review concluded that there were insufficient data on Pacific lamprey abundance (historic and current), population trends, stock structure, and basic biology to support federal protection for the species (USFWS 2004). The findings reflected the historically low priority of native lamprey management (Renaud 1997; Close et al. 2002; Moser et al. 2007).
A central unresolved question in the 2004 USFWS Pacific lamprey decision was whether the species exhibits stock structure resulting from natal homing. Are Pacific lampreys primarily panmictic, or are there genetically distinct, geographically-separated populations? Recent genetic analyses have provided evidence for both hypotheses. A mitochondrial DNA study by Goodman et al. (2008) supported Pacific lamprey panmixia, with high levels of gene flow among geographic regions. Similar results have been reported for anadromous sea lamprey (Petromyzon marinus) populations in North American and Spanish river systems (Rodríguez-Muñoz et al. 2004; Bryan et al. 2005; Waldman et al. 2008). In contrast, a Pacific lamprey study that examined 180 polymorphic loci (Lin et al. 2008) found clear genetic differentiation among populations around the Pacific Rim and in the Pacific Northwest. The latter finding suggests that Pacific lamprey population structure may be analogous at some level to that of Pacific salmonids, with multiple locally- or regionally-adapted stocks (e.g., Ricker 1972; Waples et al. 2001). If confirmed, evidence for genetically-based Pacific lamprey population structure will significantly affect conservation and management strategies for this species and for other migratory lamprey species worldwide.
A shift towards stock-based management of declining lamprey populations could benefit from some of the approaches used to protect Pacific salmonids. For example, data on stock-specific migration timing are widely used to limit harvest of at-risk salmon populations in mixed-stock fisheries (e.g., Boatright et al. 2004; Parken et al. 2008). Similarly, juvenile outmigration timing data are used to reduce outmigration mortality for a variety of species at dams and other passage barriers (e.g., Benstead et al. 1999; Achord et al. 2007). To date, however, there has been little systematic study of the factors that affect Pacific lamprey migration timing, with the exception of broad descriptions of migration seasons (e.g., Beamish 1980; Beamish and Levings 1991; Moser and Close 2003). Moreover, no attempt has been made to test for population-based timing differences within Pacific lamprey aggregations.
In this study, we used a combination of historic adult Pacific lamprey count data at dams and individual migration histories of tagged fish to: (1) test for correlations between river flow, water temperature, and annual lamprey counts at dams; (2) evaluate the effects of river environment and fish size on the upstream progress of tagged lampreys; and (3) assess whether migration timing provides evidence for stock-based population structure.
Materials and methods
Study site
The main stem Columbia River and its largest tributary, the Snake River, drain approximately 673 000 km2 of the north-western United States and south-eastern British Columbia, Canada (Fig. 1). Most of the main stem has been developed for hydroelectric power generation, and dams without passage facilities block anadromous fish at Chief Joseph Dam on the Columbia River (rkm ~877) and Hells Canyon Dam (rkm ~919) on the Snake River. As a result of impoundment, the Columbia River has lower peak flows during snowmelt events, higher fall and winter flows, earlier warming in spring, and later cooling in fall compared to pre-dam conditions (Quinn and Adams 1996; Quinn et al. 1997).
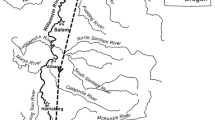
Map of the lower Columbia and lower Snake rivers showing main stem dams and major tributaries. HDX-PIT tagged adult Pacific lamprey were monitored in fish ladders at Bonneville (BO), The Dalles (TD), John Day (JD), McNary (MN), and Ice Harbor (IH) dams in 2006–2007. Adult lamprey were counted by the U.S. Army Corps of Engineers during daylight from 1939–1969 and 1998–2007 at Bonneville Dam and from 2000–2007 at the other dams shown. PR = Priest Rapids Dam. Tributaries with notable Pacific lamprey populations in the study area and/or included in the genetic study of Lin et al. (2008): (1) Willamette River; (2) Toutle River; (3) Deschutes River; (4) John Day River; (5) Umatilla River
Pacific lamprey were widely distributed in the Columbia system historically, migrating as far as 1,500 rkm upstream to Shoshone Falls on the Snake River and spawning in most of the Columbia and Snake River tributaries used by anadromous salmonids (Simpson and Wallace 1978; Wydoski and Whitney 2003; Cohnauer and Claire 2004). Lampreys pass through the lower main stem rivers from late spring through early fall, mostly after the snowmelt runoff peak in May–June. Overall, the adult lamprey migration coincides with decreasing river flow and increasing water temperature. In most years, Columbia River water temperature and flow are negatively correlated during the Pacific lamprey migration (i.e., low-flow conditions also tend to have higher water temperatures [Caudill et al. 2007]). Mean adult lamprey daytime counts at Bonneville Dam (~235 river kilometers from the Pacific Ocean) have dropped from ~100 000 in the 1940s–1960s (Starke and Dalen 1995) to ~50 000 in the last decade. In the Snake River, the largest Columbia River tributary, adult counts have dropped from tens of thousands in the 1960s (Close et al. 2002) to several hundred in recent years (U.S. Army Corps of Engineers 2007).
Lamprey counts and river environment data
Visual counts of adult lampreys at Bonneville Dam were made daily (05:00–21:00 h) by the U.S. Army Corps of Engineers in 1939–1969 (Starke and Dalen 1995) and again from 1998-present. Data were also intermittently collected at other main stem Columbia and Snake River dams, with consistent counts at most sites starting in 2000 (archived at http://www.cbr.washington.edu/dart/adult). Included in this evaluation were data from Bonneville, The Dalles, John Day, and McNary dams on the lower Columbia River and Ice Harbor Dam on the lower Snake River. At all dams, lampreys were counted during the day as they passed through lighted constrictions in fishways designed for counting adult salmonids. The count data are considered relative indices of adult population size because Pacific lampreys are primarily nocturnal migrants (Moser and Close 2003; Robinson and Bayer 2005). However, counting protocols were generally consistent across years and at each site, and the counts are among the best long-term Pacific lamprey migration datasets. Further, in a separate study we used digital video recordings to enumerate night-time (2100–0500 h) lamprey passage at dam count windows. Quartile (25th, 50th [median], and 75th percentile) passage dates differed by ≤2 d between daytime-only counts and 24 h counts (unpublished data), indicating that daytime counts are a good indicator of adult run timing.
Columbia River discharge data (daily means) for the full time series were collected by the U.S. Geological Survey near The Dalles Dam (site 14105700, rkm ~308). River temperature data for recent years (1998–2007) were collected by the U.S. Army Corps of Engineers at a near-surface water quality monitoring (WQM) site at Bonneville Dam. Pre-1970 river temperature data were compiled by the U.S. Environmental Protection Agency from a variety of sources at Bonneville Dam (archived at: http://www.streamnet.org.subbasin/StuTempData.html). The historic data were principally from sites near turbine intakes (12–17 m deep); however, the Bonneville reservoir does not typically have strong thermal stratification and temperatures near turbine intakes are positively correlated with near-surface temperatures. In recent years, differences in daily mean temperatures at Bonneville WQM and turbine intake sites were usually <0.5° C during summer (data available: http://www.cbr.washington.edu/dart).
Lamprey collection, tagging, and monitoring
Adult Pacific lampreys were collected in traps set in the Washington-shore fish ladder at Bonneville Dam in 2005–2007. Fish were anaesthetized using 60 ppm (3 mL·50 L−1) eugenol and then measured (length and girth to the nearest mm), weighed (nearest g), and outfitted with a 4 × 32 mm or 4 × 23 mm glass-encapsulated, half-duplex passive integrated transponder tag (HDX-PIT tag, 134.2 kHz, Texas Instruments, Dallas, TX, USA). Uniquely-coded HDX-PIT tags were surgically implanted through a <1 cm incision in the body cavity along the ventral midline near the anterior insertion of the first dorsal fin. Incisions did not require sutures. After recovery from anesthesia, fish were released near the top of the Bradford Island fish ladder (2005) or ~3 km downstream from Bonneville Dam near Hamilton Island (2006–2007).
In all years, lampreys were tagged opportunistically in approximate proportion to their abundance during the migratory period. Across years, 3598 fish were tagged from late May or early June through late July or mid-August (Table 1). Mandatory collection stoppages occurred in all years when Columbia River water temperatures exceeded 21°C, usually in late July. This resulted in under-representation of later portions of the runs.
In 2005, lamprey movements were monitored using underwater HDX-PIT tag interrogation antennas at Bonneville and McNary (rkm ~470) dams on the lower Columbia River and at Ice Harbor Dam on the lower Snake River (rkm ~538; Fig. 1). In 2006, antennas were installed at The Dalles (rkm ~308) and John Day (rkm ~346) dams. At all dams, antennas were located near tops of fishways where fish exit to the dam forebays (top-of-ladder exits). Additional HDX-PIT antennas were used to help establish final fish locations and were located near lamprey bypass structures (Bonneville Dam) and inside fishways at Bonneville, McNary, and Ice Harbor dams.
Statistical analyses
We calculated annual migration timing statistics (25th, 50th [median], and 75th percentile passage dates) for all years with available count data at the five study dams. Annual pairwise differences in median lamprey passage dates at dams were compared for eight years (2000–2007) using Kruskal-Wallis tests. To examine associations between river environment and Pacific lamprey migration timing, we calculated pairwise correlations between mean monthly water temperature and discharge (April–August) and median and quartile lamprey migration dates from the count data at Bonneville Dam (1939–1969, 1998–2007) and the four upstream dams (2000–2007). To estimate potential migration timing thresholds, we assessed cumulative lamprey passage distributions in relation to water temperature using 0.5°C increments. In each analysis, we emphasized counts at Bonneville Dam because it was the first dam fish encounter after leaving the Pacific Ocean and it had the longest count time series.
We calculated migration times (d) and rates (km·d−1) for HDX-PIT tagged lampreys from the time of release downstream from Bonneville Dam (2006–2007) to top-of-ladder records at all four lower Columbia River dams. Additional times were calculated between top-of-ladder sites at pairs of dams, for a total of ten river reaches. In 2005, times were calculated between top-of-ladder sites at Bonneville and McNary dams only. Relationships between log-transformed lamprey passage times and fish weight, river discharge, and water temperature were evaluated using correlation and multiple regression analyses. Environmental data in these evaluations were mean daily values on either the fish release date or the date each fish passed top-of-ladder exits at the downstream end of each river reach.
Potential stock-specific migration timing differences were assessed by comparing release and dam passage dates for groups of fish based on their last recorded locations. Each fish was assigned to a Columbia or Snake River reach upstream from Bonneville Dam. Because spawning areas were unknown for all fish, the assigned reaches represented only approximate final geographic distributions. Among-group differences in medians (dates of lamprey release and dam passage) were compared using Kruskal-Wallis tests, and mean dates were compared using ANOVA and pairwise Tukey’s tests.
Results
Count data and environmental correlates
In the 8 years (2000–2007) with lamprey count data at all five study dams, median lamprey passage dates were positively correlated among dams in nine of ten comparisons (0.24 ≤ r ≤ 0.77), indicating that run timing was consistent across sites within years; only the correlation for the two most upstream dams (McNary and Ice Harbor ) was significant (r = 0.77; P = 0.027). Notably, annual median dates at Ice Harbor Dam were 2-14 d earlier (mean = 5 d earlier) than median dates 68 rkm downstream at McNary Dam (Fig. 2). This suggests that lampreys entering the Snake River were generally early migrants though it is possible some had overwintered. In all other comparisons, median dates were later at each successive dam upstream in each year. Means of annual median migration dates across the eight years were 7 July (Bonneville), 21 July (The Dalles), 28 July (John Day), 8 August (McNary), and 3 August (Ice Harbor) (Fig. 2). Annual median dates varied by 17–23 d across years at each dam.
Adult Pacific lamprey migration timing distributions at the four lower Columbia River dams (Bonneville, The Dalles, John Day, McNary) and at Ice Harbor Dam on the lower Snake River, 2000–2007. Solid circles = means of median dates; vertical lines = means of 1st and 3rd quartile dates; horizontal lines = means of 10th and 90th percentile dates; open circles = annual median dates
Median passage dates in the 41-year Bonneville Dam time series (1939–1969, 1998–2007) ranged from 15 June to 12 August (mean = 18 July, SD = 12.3 d). Lamprey run timing shifted progressively earlier from 1939 to 2007, coincident with decreasing Columbia River discharge and increasing water temperature. The average median passage date was 21 July for 1939–1969 and was 8 July for 1998–2007, a shift of 13 d. Annual median and quartile passage dates were negatively correlated with year (−0.46 ≤ r ≤ −0.39; 0.002 ≤ P ≤ 0.013). Year was also correlated with mean May, June, and July discharge (−0.58 ≤ r ≤ −0.41; P ≤ 0.007) and mean June and July water temperature (0.51 ≤ r ≤ 0.54; P < 0.001).
Lamprey migration timing at Bonneville Dam was consistently earlier in warm and low-discharge years and later in cold and high-discharge years. The highest correlation coefficients for median passage dates were with mean July discharge (r = 0.79; P < 0.001) and mean June water temperature (r = −0.77; P < 0.001) (Fig. 3). Patterns were similar for first quartile (i.e., June temperature r = −0.67) and third quartile (i.e., July flow r = 0.63) dates. Residuals from linear regressions of median date and year were also significantly (P < 0.05) negatively correlated with temperature and positively correlated with discharge in these months.
Relationships between median adult Pacific lamprey migration dates at Bonneville Dam (○, 1939–1969; ● 1998–2007) and mean Columbia River flow in July (USGS site at The Dalles Dam) and mean water temperature in June (WQM site at Bonneville Dam). Linear regressions: (date) = 4.1●flow + 49.2, r 2 = 0.63; (date) = −8.0●temperature + 199.0, r 2 = 0.59
Median and quartile dates at The Dalles, John Day, McNary, and Ice Harbor dams (2000–2007) were positively correlated with river discharge and negatively correlated with water temperature (Table 2). On average, discharge was more correlated with passage dates at upstream dams (McNary, Ice Harbor) and temperature data were more correlated with dates at downstream dams (The Dalles, John Day).
Cumulative lamprey passage distributions at Bonneville Dam showed that very few fish passed before water temperatures reached 15°C (Fig. 4). On average, about 25% of the run had passed by the time the river warmed to 17°C, and half the run had passed by ~19°C. In most years, about 80% of the run had passed by the time water temperatures reached seasonal peaks of ~21–23°C. Distributions were very similar for the early (1939–1969) and late (1998–2007) time periods (Fig. 4), suggesting that fish responded to similar environmental cues throughout the period of record despite long-term warming in the system.
Behavior of tagged lampreys
HDX-PIT tagged lamprey passage times (d) and rates (km·d−1) were highly variable within year, with some fish taking weeks to months to pass through individual study reaches. In contrast, median passage times and rates were relatively consistent among years. Median passage times between top-of-ladder antennas at Bonneville and McNary dams (~235 rkm) ranged from 17.2–18.9 d (12.4–13.7 km·d−1) in the three study years. Passage times between top-of-ladder sites at pairs of adjacent dams (2006–2007 only) ranged from 4.0–12.8 d (9.6–18.3 km·d−1).
Tagged lampreys migrated faster through almost all reaches as water temperatures increased and river discharge decreased (Fig. 5). In 2006–2007, correlations between environmental variables and passage times (d) were generally highest for the reaches that started at the release site (reaches 1–4, Fig. 5). In 2007, fish size was negatively correlated with passage times through these same reaches. (r = −0.15; P < 0.001). No consistent weight × passage time patterns were observed in 2006. In multiple regression analyses, combinations of environmental variables and lamprey weight had only slightly higher r 2 values than the univariate models.
Correlation coefficients (r) for log-transformed HDX-PIT tagged Pacific lamprey passage times (d) through ten Columbia River reaches in 2006 (black bars) and 2007 (white bars). Predictor variables included Columbia River discharge (panel a), water temperature (panel b), and fish weight (panel c). Weight was collected when fish were tagged. Flow and temperature data were from daily mean values on the dates fish were released or when they passed top-of-ladder sites at dams. Reaches: (1) Release-Bonneville, (2) Release-The Dalles, (3) Release-John Day, (4) Release-McNary, (5) Bonneville-The Dalles, (6) Bonneville-John Day, (7) Bonneville-McNary, (8) The Dalles-John Day, (9) The Dalles-McNary, (10) John Day-McNary. * P < 0.05
Based on the last detection of each fish, lampreys that passed Bonneville Dam were assigned to one of five reaches in 2006–2007: Bonneville-The Dalles, The Dalles-John Day, John Day-McNary, top of McNary, and Snake River. Sample sizes rapidly decreased in successive upstream reaches as fish either entered tributaries or failed to pass dams: 41–52% passed Bonneville Dam, 28–33% passed The Dalles Dam, 17–19% passed John Day Dam, 4–6% passed McNary Dam, and about 1% was recorded at Ice Harbor Dam. Mean lamprey weights differed significantly by final location, with larger fish last recorded in upstream reaches (12.2 ≤ F ≤ 18.5, P < 0.001). The heaviest group, on average, returned to the Snake River.
In 2006, the mean tag date for fish recorded in the Snake River was 8–9 d earlier than mean dates for fish last recorded in the other four reaches (F = 3.52; P = 0.007). Mean passage dates for the Snake River group were also 11–12 d earlier at The Dalles Dam, 10–13 d earlier at John Day Dam, and 13 d earlier at McNary Dam. In each ANOVA, the Snake River group was significantly earlier than the comparison groups (2.82 ≤ F ≤ 6.21; 0.002 ≤ P ≤ 0.043). No other pairwise comparisons were significantly different (P > 0.05). In 2007, when sample sizes were smaller, mean passage dates for the Snake River group were earlier by 1–12 d in all comparisons, but no among-group differences were significant (P > 0.05). In both years, Kruskal-Wallis comparisons of median dates were consistent with these ANOVA results.
Discussion
Migration patterns described here provide evidence that in-river environmental conditions influence the upstream progression of adult Pacific lamprey in the Columbia River system. Aggregate run timing was earliest in warm and low-flow years and later in cold and high-flow years. This closely parallels the migration timing patterns of other anadromous species, including sea lamprey (Stier and Kynard 1986), sockeye salmon (O. nerka; Hodgson et al. 2006), Chinook salmon (O. tshawytscha; Keefer et al. 2008), Atlantic salmon (Salmo salar; Dahl et al. 2004), and American shad (Alosa sapidissima; Quinn and Adams 1996). In these and other studies, environmental effects on run timing and upstream migration rates have generally been attributed to physiological constraints. For example, high flows are energetically demanding, can create velocity barriers, and tend to reduce ground speeds of lampreys and other species (e.g., Hinch and Rand 1998; Moser et al. 2002; Almeida et al. 2002, 2005; Quintella et al. 2004). Similarly, cold temperatures reduce swim speeds, metabolic performance, and overall fish activity (e.g., Beamish 1974; Lee et al. 2003; Binder and McDonald 2008). Each of these effects potentially results in later arrival timing at upstream sites.
Within each year, tagged lampreys migrated more slowly at higher flows and lower temperatures, offering some mechanistic support for how environmental conditions affect lamprey migration. Unfortunately, untangling the relative effects of water temperature and discharge was difficult because these two variables were highly correlated with each other and with photoperiod. Photoperiod may be a critical stimulus for the initiation of lamprey migration given its influence on maturation and reproductive timing (Bromage et al. 2001; Pankhurst and Porter 2003). However, most adult lampreys are in the early stages of gonadal development when they enter freshwater (Beamish 1980). The lamprey’s premature migration strategy decouples migration timing from reproductive timing (e.g., Hodgson and Quinn 2002) and allows considerable flexibility in how they respond to photoperiod and proximate environmental conditions during migration and spawning.
In most rivers, discharge is more variable than water temperature within and across years and consequently temperature may be the more reliable migration cue. The count data at Bonneville Dam indicated that Pacific lamprey timing in relation to temperature was stable through time, despite significant long-term warming of the Columbia River. In both historical (1939–1969) and recent (1998–2007) periods, very few lampreys passed Bonneville Dam before water temperatures reached 15°C and the midpoint of the run typically passed by about 19°C. These temperatures may therefore be useful thresholds for making in-season assessments of run timing and size. The run-timing shift of approximately two weeks between the historic and more recent lamprey counts reflects the earlier spring warming and declining summer discharge in the Columbia River. It is unclear however, how earlier timing may affect lamprey maturation, survival, spawn timing, or other life history traits.
Overall, the relatively strong relationships between river environment and aggregate lamprey run timing in the Columbia Basin indicate a potential for timing-based selection, an important precursor to population divergence. Divergence generally develops in response to long-term environmental conditions along migration routes and in breeding and rearing habitats (Dingle 1996; Hodgson and Quinn 2002). Over time, fitness benefits associated with optimal migration timing produce genetic separation among related populations (e.g., Waples et al. 2004; Dickerson et al. 2005) and this is strongly reinforced by natal homing (Dittman and Quinn 1996; McDowall 2001). Population-level genetic divergence in Pacific lamprey may have occurred in the Columbia River system, as Lin et al. (2008) reported significant genetic variation among five Columbia River samples. However, their results did not follow a pattern of genetic isolation by geographic distance, suggesting that other demographic or phenotypic (i.e., size or timing) mechanisms were responsible. Further testing with larger and more geographically representative samples is needed.
In our study, both timing patterns in the count data and behavior of tagged lampreys indicated that population-based migration timing differences may exist in the Columbia system. The consistently earlier timing by lampreys that returned to the Snake River basin signals that this group may be genetically or phenotypically distinct. The Snake River group was larger, on average, than fish last recorded in any other study reach, which may be an evolutionary response to longer migration distance (e.g., Crespi and Teo 2002). Similarly, the earlier timing for Snake River fish may reflect historically warmer (+1–3°C) summer water temperatures in the Snake River. It is also possible that the timing patterns simply reflect phenotypic plasticity whereby larger fish migrated earlier and further because they were in better initial condition or were more efficient swimmers. Seasonal variation in lamprey size has been recorded in other migratory species (e.g., Maitland et al. 1994), though it is unclear whether these patterns are related to population effects (i.e., geographically-based size differences) or other factors such as sex ratios (e.g., Jang and Lucas 2005).
Speculation about Pacific lamprey population structure hinges on there being some level of natal homing, a behavior not yet documented in this species. However, we are aware of only one field study of lamprey homing. Bergstedt and Seelye (1995) tagged and released sea lamprey juveniles in their natal stream and subsequently examined adults at multiple sites to evaluate homing patterns. They found that sea lampreys had very low site fidelity, and the result is widely cited as evidence that lampreys do not home. Instead, the generally accepted hypothesis is that lampreys select spawning sites by orienting to olfactory cues produced by larval or conspecific lampreys (e.g., Waldman et al. 2008). This has been tested experimentally with several species (Vrieze and Sorenson 2001; Bjerselius et al. 2000; Gaudron and Lucas 2006; Johnson et al. 2006). In almost all cases, tests showed clear adult lamprey preferences for water with pheromones, including pheromones of other lamprey species (e.g., Fine et al. 2004). It is currently unknown, however, if pheromones are essential to find suitable spawning sites or if the imprinting-based homing that is typical among other anadromous species (e.g., Dittman and Quinn 1996; McDowall 2001) occurs at some scale.
If Pacific lampreys do select spawning tributaries based on pheromones only, then increases in larval or adult mortality rates could produce negative feedback effects on the number of returning spawners. This is a plausible explanation for the order-of-magnitude Pacific lamprey decline in the Snake River, where four dams were built on the lower river and several impassable dams were built further upstream between 1959 and 1975. The dams prevented access to many lamprey spawning and rearing areas, likely increased macrophthalmia outmigration mortality (Moser and Close 2003), and may have dramatically reduced pheromone concentrations in the Snake River plume.
Clearly, a better understanding of the range-wide Pacific lamprey declines will require additional research on several fronts. Studies are needed that can clarify relationships between lamprey recruitment and environmental conditions (e.g., Nunn et al. 2008), and between population productivity and a suite of life history, genetic, and phenotypic differences. It will also be critical to address the homing and population structure questions raised in the discussion above, both for Pacific lamprey and other lamprey species. These steps will help resolve whether a stock-based paradigm is appropriate for effective management and conservation of anadromous lampreys worldwide.
References
Achord S, Zabel RW, Sandford BJ (2007) Migration timing, growth, and estimated parr-to-smolt survival rates of wild Snake River spring-summer Chinook salmon from the Salmon River basin, Idaho, to the lower Snake River. Trans Am Fish Soc 136:142–154. doi:10.1577/T05-308.1
Almeida PR, Quintella BR, Dias NM (2002) Movement of radio-tagged anadromous sea lamprey during the spawning migration in the River Mondego (Portugal). Hydrobiologia 483:1–8. doi:10.1023/A:1021383417816
Almeida PR, Quintella BR, Koed A, Andrade NO (2005) Using electromyogram telemetry to study the spawning migration of sea lamprey (Petromyzon marinus L.). In: Spedicato MT, Marmulla G, Lembo G (eds) Aquatic telemetry: Advances and applications. FAO-COISPA, Rome, pp 3–11
Battin J, Wiley MW, Ruckelshaus MH, Palmer RN, Korb E, Bartz KK, Imaki H (2007) Projected impacts of climate change on salmon habitat restoration. Proc Natl Acad Sci USA 104:6720–6725. doi:10.1073/pnas.0701685104
Beamish FWH (1974) Swimming performance of adult sea lamprey, Petromyzon marinus, in relation to weight and temperature. Trans Am Fish Soc 103:355–358. doi:10.1577/1548-8659(1974)103<355:SPOASL>2.0.CO;2
Beamish RJ (1980) Adult biology of the river lamprey (Lampetra ayresi) and the Pacific lamprey (Lampetra tridentata) from the Pacific coast of Canada. Can J Fish Aquat Sci 37:1906–1923. doi:10.1139/f80-232
Beamish RJ, Levings CD (1991) Abundance and freshwater migrations of the anadromous parasitic lamprey, Lampetra tridentata, in a tributary of the Fraser River, British Columbia. Can J Fish Aquat Sci 48:1250–1263
Beamish RJ, Northcote TG (1989) Extinction of a population of anadromous parasitic lamprey, Lampetra tridentata, upstream of an impassable dam. Can J Fish Aquat Sci 46:420–425. doi:10.1139/f89-056
Benstead JP, March JG, Pringle CM, Scatena FN (1999) Effects of a low-head dam and water abstraction on migratory tropical stream biota. Ecol Appl 9:656–668. doi:10.1890/1051-0761(1999)009[0656:EOALHD]2.0.CO;2
Bergstedt RA, Seelye JG (1995) Evidence for lack of homing by sea lampreys. Trans Am Fish Soc 124:235–239. doi:10.1577/1548-8659(1995)124<0235:EFLOHB>2.3.CO;2
Binder TR, McDonald DG (2008) The role of temperature in controlling diel activity in upstream migrant sea lampreys (Petromyzon marinus). Can J Fish Aquat Sci 65:1113–1121. doi:10.1139/F08-070
Bjerselius R, Li W, Teeter JH, Seelye JG, Johnsen PB, Maniak PJ, Grant GC, Polkinghorne CN, Sorensen PW (2000) Direct behavioral evidence that unique bile acids released by larval lamprey (Petromyzon marinus) function as a migratory pheromone. Can J Fish Aquat Sci 57:557–569. doi:10.1139/cjfas-57-3-557
Boatright C, Quinn T, Hilborn R (2004) Timing of adult migration and stock structure for sockeye salmon in Bear Lake, Alaska. Trans Am Fish Soc 133:911–921. doi:10.1577/T03-142.1
Bromage N, Porter M, Randall C (2001) The environmental regulation of maturation in farmed finfish with special reference to the role of photoperiod and melatonin. Aquaculture 197:63–98. doi:10.1016/S0044-8486(01)00583-X
Bryan MB, Zalinski D, Filcek KB, Libants S, Li W, Scribner KT (2005) Patterns of invasion and colonization of the sea lamprey (Petromyzon marinus) in North America as revealed by microsatellite genotypes. Mol Ecol 14:3757–3773. doi:10.1111/j.1365-294X.2005.02716.x
Caudill CC, Daigle WR, Keefer ML, Boggs CT, Jepson MA, Burke BJ, Zabel RW, Bjornn TC, Peery CA (2007) Slow dam passage in Columbia River salmonids associated with unsuccessful migration: delayed negative effects of passage obstacles or condition-dependent mortality? Can J Fish Aquat Sci 64:979–995. doi:10.1139/F07-065
Close DA, Fitzpatrick MS, Li HW (2002) The ecological and cultural importance of a species at risk of extinction, Pacific lamprey. Fisheries 27:19–25. doi:10.1577/1548-8446(2002)027<0019:TEACIO>2.0.CO;2
Cohnauer T, Claire C (2004) Evaluate status of Pacific lamprey in the Clearwater River drainage, Idaho. Bonneville Power Administration report DOE/BP-00004039-4, Portland, Oregon
Crespi BJ, Teo R (2002) Comparative phylogenetic analysis of the evolution of semelparity and life history in salmonid fishes. Evolution Int J Org Evolution 56:1008–1020
Dahl J, Dannewitz J, Karlsson L, Petersson E, Löf A, Ragnarsson B (2004) The timing of spawning migration: implications of environmental variation, life history, and sex. Can J Zool 82:1864–1870. doi:10.1139/z04-184
Dickerson BR, Brinck KW, Willson MF, Bentzen P, Quinn TP (2005) Relative importance of salmon body size and arrival time at breeding grounds to reproductive success. Ecology 86:347–352. doi:10.1890/03-625
Dingle H (1996) Migration - biology of life on the move. Oxford University Press, Oxford
Dittman AH, Quinn TP (1996) Homing in Pacific salmon: mechanisms and ecological basis. J Exp Biol 199:83–91
Fine JM, Vrieze LA, Sorensen PW (2004) Evidence that Petromyzontid lampreys employ a common migratory pheromone that is partially composed of bile acids. Chem Ecol 30:2091–2110. doi:10.1023/B:JOEC.0000048776.16091.b1
Gaudron SM, Lucas MC (2006) First evidence of attraction of adult river lamprey in the migratory phase to larval odour. J Fish Biol 68:640–644. doi:10.1111/j.0022-1112.2006.00937.x
Gess RW, Coates MI, Rubidge BS (2006) A lamprey from the Devonian period of South Africa. Nature 443:981–984. doi:10.1038/nature05150
Goodman DH, Reid SB, Docker MF, Haas GR, Kinziger AP (2008) Mitochondrial DNA evidence for high levels of gene flow among populations of a widely distributed anadromous lamprey Entosphenus tridentatus (Petromyzontidae). J Fish Biol 72:400–417. doi:10.1111/j.1095-8649.2007.01731.x
Hinch SG, Rand PS (1998) Swim speeds and energy use of upriver-migrating sockeye salmon (Oncorhynchus nerka): role of local environment and fish characteristics. Can J Fish Aquat Sci 55:1821–1831. doi:10.1139/cjfas-55-8-1821
Hodgson S, Quinn TP (2002) The timing of adult sockeye salmon migration into fresh water: adaptations by populations to prevailing thermal regimes. Can J Zool-Rev Can Zool 80:542–555. doi:10.1139/z02-030
Hodgson S, Quinn TP, Hilborn R, Francis RC, Rogers DE (2006) Marine and freshwater climatic factors affecting interannual variation in the timing of return migration to fresh water of sockeye salmon (Oncorhynchus nerka). Fish Oceanogr 15:1–24. doi:10.1111/j.1365-2419.2005.00354.x
Jang M-H, Lucas MC (2005) Reproductive ecology of the river lamprey. J Fish Biol 66:499–512. doi:10.1111/j.0022-1112.2005.00618.x
Johnson NS, Luehring MA, Siefkes MJ, Li W (2006) Mating pheromone reception and induced behavior in ovulating femal sea lampreys. N Am J Fish Manage 26:88–96. doi:10.1577/M05-018.1
Keefer ML, Peery CA, Caudill CC (2008) Migration timing of Columbia River spring Chinook salmon: effects of temperature, river discharge, and ocean environment. Trans Am Fish Soc 137:2230–1133. doi:10.1577/T07-008.1
Kostow K (2002) Oregon lampreys: natural history, status, and analysis of management issues, Oregon Department of Fish and Wildlife, Portland
Lee CG, Farrell AP, Lotto A, MacNutt MJ, Hinch SG, Healey MC (2003) The effect of temperature on swimming performance and oxygen consumption in adult sockeye (Oncorhynchus nerka) and coho (O. kisutch) salmon stocks. J Exp Biol 206:3239–3251. doi:10.1242/jeb.00547
Lin B, Zhang Z, Wang Y, Currens KP, Spidle A, Yamazaki Y, Close DA (2008) Amplified fragment length polymorphism assessment of genetic diversity of Pacific lamprey. N Am J Fish Manage 28:1182–1193. doi:10.1577/M07-035.1
Maitland PS, Morris KH, East K (1994) The ecology of lampreys (Petromyzonidae) in the Loch Lomand area. Hydrobiologia 290:105–120. doi:10.1007/BF00008958
McDowall RM (2001) Anadromy and homing: two life-history traits with adaptive synergies in salmonid fishes? Fish Fish 2:78–85. doi:10.1046/j.1467-2979.2001.00036.x
Moser ML, Close DA (2003) Assessing Pacific lamprey status in the Columbia River basin. Northwest Sci 77:116–125
Moser ML, Ocker PA, Stuehrenberg LC, Bjornn TC (2002) Passage efficiency of adult Pacific lampreys at hydropower dams on the lower Columbia River, USA. Trans Am Fish Soc 131:956–965. doi:10.1577/1548-8659(2002)131<0956:PEOAPL>2.0.CO;2
Moser ML, Butzerin JM, Dey DB (2007) Capture and collection of lampreys: the state of the science. Rev Fish Biol Fish 17:45–56. doi:10.1007/s11160-006-9037-3
Moyle PB (2002) Inland fishes of California. University of California Press, Berkeley
Nunn AD, Harvey JP, Noble RAA, Cowx IG (2008) Condition assessment of lamprey populations in the Yorkshire Ouse catchment, north-east England, and the potential influence of physical migration barriers. Aquat Conserv: Mar Freshwat Ecosyst 18:175–189. doi:10.1002/aqc.863
Pankhurst NW, Porter MJR (2003) Cold and dark or warm and light: variations on the theme of environmental control of reproduction. Fish Physiol Biochem 28:385–389. doi:10.1023/B:FISH.0000030602.51939.50
Parken CK, Candy JR, Irvine JR, Beacham TD (2008) Genetic and coded wire tag results combine to allow more-precise management of a complex Chinook salmon aggregate. N Am J Fish Manage 28:328–340. doi:10.1577/M06-110.1
Quinn TP, Adams DJ (1996) Environmental changes affecting the migratory timing of American shad and sockeye salmon. Ecology 77:1151–1162. doi:10.2307/2265584
Quinn TP, Myers KW (2004) Anadromy and the marine migrations of Pacific salmon and trout: Rounsefell revisited. Rev Fish Biol Fish 14:421–442. doi:10.1007/s11160-005-0802-5
Quinn TP, Hodgson S, Peven C (1997) Temperature, flow, and the migration of adult sockeye salmon (Oncorhynchus nerka) in the Columbia River. Can J Fish Aquat Sci 54:1349–1360. doi:10.1139/cjfas-54-6-1349
Quintella BR, Andrade NA, Koed A, Almeida PR (2004) Behavioural patterns of sea lamprey’s spawning migration through difficult passage areas, studied by electromyogram telemetry. J Fish Biol 65:961–972. doi:10.1111/j.0022-1112.2004.00497.x
Renaud CB (1997) Conservation status of northern hemisphere lampreys (Petromyzontidae). J Appl Ichthy 13:143–148. doi:10.1111/j.1439-0426.1997.tb00114.x
Ricker WE (1972) Hereditary and environmental factors affecting certain salmonid populations. pp. 27–160. In: Simon RC, Larkin PA (eds) The stock concept in Pacific salmon. Univeristy of British Columbia, Vancouver
Robinson TC, Bayer JM (2005) Upstream migration of Pacific lampreys in the John Day River, Oregon: behavior, timing, and habitat use. Northwest Sci 79:106–119
Rodríguez-Muñoz R, Waldman JR, Grunwald C, Roy NK, Wirgin I (2004) Absence of shared mitochondrial DNA haplotypes between sea lamprey from North American and Spanish rivers. J Fish Biol 64:783–787. doi:10.1111/j.1095-8649.2004.00334.x
Ruckelshaus MH, Levin P, Johnson JB, Kareiva PM (2002) The Pacific salmon wars: what science brings to the challenge of recovering species. Annu Rev Ecol Syst 33:665–706. doi:10.1146/annurev.ecolsys.33.010802.150504
Scott WB, Crossman EJ (1973) Freshwater Fishes of Canada. Fish Res Board Can, Bull 184, Ottawa
Simpson J, Wallace R (1978) Fishes of Idaho. University of Idaho Press, Moscow
Starke GM, Dalen JT (1995) Pacific lamprey (Lampetra tridentate) passage patterns past Bonneville Dam and incidental observations of lamprey at the Portland District Columbia River dams in 1993. U.S. Army Corps of Engineers, Cascade Locks, Oregon
Stier K, Kynard B (1986) Movement of sea-run sea lampreys, Petromyzon marinus, during the spawning migration in the Connecticut River. Fish Bull (Wash D C) 84:749–753
U.S. Army Corps of Engineers (2007) Annual fish passage report, U.S. Army Engineer Districts, Portland, Oregon and Walla Walla, Washington
U.S. Fish and Wildlife Service (2004) Endangered and threatened wildlife and plants; 90-day finding on a petition to list three species of lampreys as threatened or endangered. Fed Regist 69:77158–77167
Vrieze LA, Sorenson PW (2001) Laboratory assessment of the role of a larval pheromone and natural stream odor in spawning stream localization by migratory sea lamprey (Petromyzon marinus). Can J Fish Aquat Sci 58:2374–2385. doi:10.1139/cjfas-58-12-2374
Waldman J, Grunwald V, Wirgin I (2008) Sea lamprey Petromyzon marinus: an exception to the rule of homing in anadromous fishes. Biol Lett 4:659–662. doi:10.1098/rsbl.2008.0341
Waples RS, Gustafson RG, Weitkamp LA, Myers JM, Johnson OW, Busby PJ, Hard JJ, Bryant GJ, Waknitz FW, Neely K, Teel D, Grant WS, Winans GA, Phelps S, Marshall A, Baker BM (2001) Characterizing diversity in salmon from the Pacific Northwest. J Fish Biol 59(S1):1–41
Waples RS, Teel DJ, Meyer JM, Marshall AR (2004) Life-history divergence in Chinook salmon: historic contingency and parallel evolution. Evolution Int J Org Evolution 58:386–403. doi:10.1554/03-323
Wydoski RS, Whitney RR (2003) Inland fishes of Washington, 2nd edn. American Fisheries Society, Bethesda, Maryland and University of Washington Press, Seattle
Acknowledgements
We thank H. Pennington and J. Roos (Pacific States Marine Fisheries Commission) for capture, PIT-tagging, and monitoring of the lamprey. W. Wassard (NOAA Fisheries), W. Leach (Oregon RFID), and S. Lee (University of Idaho) helped with development and installation of the PIT detection system. K. Tolotti and T. Dick (University of Idaho) helped maintain and monitor PIT-tag detection sites. J. Simonson and J. Moser (NOAA Fisheries) fabricated and installed the traps used to collect lamprey in this study. D. Clugston, T. Mackey, R. Cordie, J. Rerecich, M. Zyndol, and M. Plummer (US Army Corps of Engineers) and D. Dey, P. McAteer, T. Ruehle, and R. Martinson (NOAA Fisheries and PSMFC) provided administrative assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Keefer, M.L., Moser, M.L., Boggs, C.T. et al. Variability in migration timing of adult Pacific lamprey (Lampetra tridentata) in the Columbia River, U.S.A.. Environ Biol Fish 85, 253–264 (2009). https://doi.org/10.1007/s10641-009-9490-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-009-9490-7