Abstract
There are relatively few studies that have examined in detail the effects of long term exploitation on population characteristics of Arctic charr, Salvelinus alpinus, coincident with natural changes in environmental conditions. This is particularly the case for anadromous charr despite their widespread use and importance in areas such as northern Canada. Here, the response of north Labrador charr to major reductions in commercial exploitation following long periods of intense fishing is described for three stock complexes with emphasis on changes in size, growth, and age configurations. Changes in age and size distribution, specifically long-term declines in weight, are linked with periods of intensive exploitation and possibly with variability in the climate of the northwest Atlantic that resulted in identifiable diet shifts during the early 1990s. Despite observed changes in some metrics, populations have maintained relatively stable length compositions with variations in weight showing resilience to return to earlier configurations as exploitation was reduced and environmental conditions ameliorated. Results of the long-term investigations challenge traditional beliefs associated with the inability of all anadromous charr to withstand periods of intense exploitation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In northern lakes, unexploited populations of freshwater resident Arctic charr, Salvelinus alpinus, are frequently characterized by maintaining constant size distributions despite being subject to environmental variability over the course of their recent history (Parker and Johnson 1991; Johnson 1994). When subject to perturbation, such as exploitation, population structure can vary. However, the original configurations often persist for a number of years before changes are manifest despite substantive removals from the population (Johnson 1976, 1983, 1994). In terms of long-term stability and resilience of charr in Arctic lakes, populations frequently return to their original configurations when left unperturbed, as noted by Johnson (1976, 1994, 2001, 2002).
Long-term investigations on the capacity of anadromous Arctic charr to yield a harvest under exploitation are generally lacking (Johnson 1980). Exceptions include the work at Nauyak Lake, Nunavut (Johnson 1989), north Labrador (Dempson 1995), and more recently at Cambridge Bay, Nunavut (Day and de March 2005). Early reports consistently indicated that owing to their relatively slow growth and late sexual maturity, anadromous Arctic charr were unable to withstand periods of intense exploitation and under these circumstances populations were frequently overfished or collapsed often resulting in fishery closures (e.g. Yessipov 1935; Dunbar and Hildebrand 1952; Bigelow 1963; Mattox 1973; Hunter 1976; Kristofferson and Sopuck 1983; Kristofferson et al. 1984; Crawford 1989; Johnson 1989). North Labrador charr were no exception, indeed, at one time populations were deemed to be in imminent danger of collapse (Coady and Best 1976). In the latter case interpretation was based largely on the rapid decline in catches over several years as had been observed in other areas (e.g. Andrews and Lear 1956; Mattox 1973; Hunter 1976).
Previously, Dempson (1995) examined the effects of exploitation on one north Labrador stock complex harvested primarily by Inuit fishers and subject to intense commercial fishing and questioned the notion that all anadromous charr populations collapse when moderately exploited. The period of analysis extended into the early 1990s when anomalous environmental conditions occurred throughout the Northwest Atlantic (Colbourne et al. 1997; Rose et al. 2000; Colbourne 2004). Since 1993, there have been substantive reductions in the amount of directed effort to the extent that commercial harvesting of north Labrador charr has declined by 70–75% over the past 12 years. Thus in general the situation with north Labrador anadromous Arctic charr parallels the controlled perturbation experiments carried out by Johnson (1994) on two Arctic lakes where periods of moderate to high fishing were subsequently followed by periods of little or no exploitation. Accordingly, the intent of this paper is to examine the response of anadromous Arctic charr to major reductions in commercial exploitation following long periods of intense fishing with emphasis on changes in size, growth, and age configurations. Analyses incorporate two additional stock complexes from that originally examined (Dempson 1995), with investigations extending over a period of time when ocean climate conditions in the Northwest Atlantic have returned to more normal conditions.
Materials and methods
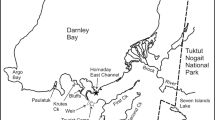
Since 1974, almost 80% of the commercial catch of anadromous Arctic charr has originated from three stock complexes, Voisey, Nain, and Okak that were previously defined and described by Dempson and Kristofferson (1987) and Dempson (1995), that extend along a limited 140 km stretch of the north Labrador coast in the Labrador Inuit Settlement Area of Nunatsiavut. Commercial fisheries for charr can occur at sea from mid-June to early September using 114 and 127 mm mesh multifilament gill nets, although most of the catches (>70%) are usually taken over a six week period in July-August. Arctic charr were landed in the gutted head-on form and converted to round (whole) weight using a conversion factor of 1.22 (Dempson 1984). Since fish were gutted when landed, it precludes the examination of differences in growth between sexes as well as trends in age at maturity and changes in fecundity over time. Catch data were derived from sales slips that are issued at the fish plant when catches were landed.
Arctic charr from all three stock complexes were sampled continuously from 1977 whenever fisheries occurred. Details pertaining to sampling programs and commercial landings information are provided by Dempson (1995) and Power et al. (2000). Previously, limited biological sampling was carried out in 1953 (Andrews and Lear 1956) and again in 1973 and 1974 (Coady and Best 1976). Briefly, information on length (L), weight (W), and age of charr caught in the commercial fishery was obtained as charr were processed at the fish plant in the community of Nain (56°32′ N, 61°41′ W). Ages were interpreted from otoliths following Nordeng (1961) and as described previously by Grainger (1953) and Andrews and Lear (1956). In a supplementary study Frost (1978) concluded that at least for young charr of known age and history, otoliths accurately recorded the age of the fish. Baker and Timmons (1991) also reported that surface-examined otoliths were appropriate for precise estimation of the age of Arctic charr.
From 1977 to 1979, Arctic charr were collected from as many samples as practical. Beginning in 1980, a two-stage sampling program was implemented, where larger numbers of charr were measured for length and subsamples stratified by length taken for age information. Length-frequencies were usually derived by measuring all fish from an individual catch and subsamples taken to ensure representation in 2 cm length strata. Coefficients of variation associated with the estimated total catch numbers at age were calculated following Baird (1983) and were generally less than 25% for the dominant ages in the catch for the Voisey and Okak stock complexes and usually less than 15% for the more heavily fished and sampled Nain stock complex. Collectively over all years the numbers of charr sampled for length frequency (NL) and subsampled for age (NA) were as follows: Voisey: N L = 62783; N A = 8846; Nain: N L = 122328; N A = 20950; Okak: N L = 67240; N A = 9766. Average age was determined from the numbers of fish at age weighted by the estimated catch numbers at age while mean weights were derived from weight-length relationships where the length parameter was estimated from length-frequency distributions. Length-frequency distributions were compared by likelihood ratio G-Tests (Sokal and Rohlf 1998) while trends in mean weights were simply calculated from the regression of mean weight against year as applied to various species of Pacific salmon (Ricker 1981). Condition was estimated as W/L3 (Ricker 1975) and summarized as the annual mean determined from individual specimens from within the fork length range 450–599 mm. Johnson (1980) recommended this approach for a predetermined number of length groups around the modal size rather than estimating condition for the entire size range of charr available. Differences in condition among years were compared by analysis of variance.
Changes in length-at-age were analysed by comparing the nonlinear von Bertalanffy growth equations fitted to length-at-age data:
where Lt = the estimated length-at-age, L∞ = the asymptotic maximum length, K is a growth coefficient, and to is the hypothetical age when length is zero (Ricker 1975). The non-linear equations were estimated using the SAS NLIN procedure (SAS 2000). Sexes were combined because charr were landed in a gutted form, as noted above. In addition, a previous study of Arctic charr from one specific river within the Nain stock complex found sexes were generally similar in terms of lengths at age (Dempson and Green 1985). For ease of analysis and clarity, Voisey and Nain charr were grouped into 5 year intervals from 1977 to 2001, with the last interval (2002–2005) consisting of four years. Okak charr were also grouped into 5 year intervals from 1977 to 1996, but owing to the termination of fishing after 2002, the last year grouping was set to cover six years (1997–2002). Growth curves were compared using the residual sum of squares analysis described by Chen et al. (1992) and as outlined by Haddon (2001).
Stomach samples of Arctic charr from the Nain stock complex, 1997–2005, were analysed following methods and procedures described in Dempson et al. (2002). Data were examined in relation to the response of charr to changes in prey availability, specifically capelin, Mallotus villosus, coincident with improved oceanographic conditions in the Northwest Atlantic that began during the late 1990s.
Results
Commercial catch
Since 1974, 2789 tonnes of anadromous Arctic charr were caught in commercial fisheries along the north Labrador coast. Of this amount, 15% (427 t) and 23% (646 t) originated from the Voisey and Okak stock complexes with by far the greatest catches (39% – 1083 t) taken directly from the Nain complex (Fig. 1). In terms of numbers of fish, over 1.2 million charr have been caught from the three stock complexes with approximately half (640 thousand charr) removed from the Nain stock complex. Peak removals of 53- to 88-thousand fish occurred annually during the late 1970s and early 1980s. There were no commercial harvests of charr on the Voisey stock complex in 1995 and 2005, minimal catches ( <1 t) at Okak in 1992–93, and no fisheries in the latter area since 2002. Values represent minimum removals as they pertain to commercial harvests only; subsistence or food fishery catches, or charr caught in recreational fisheries from these areas are largely unknown. By comparison with the earlier years, landings declined dramatically in the early 1990s with the average for each of the three stock complex areas having declined by 70–75% by comparison with the prior 20 year interval (1974–1993). The reduced harvests, however, still represent annual removals of 10- to 20-thousand adult charr.
Age structure
Arctic charr are first recruited into the commercial fishery at 6 years of age. For each stock complex, commercial harvests have been dominated by Arctic charr 7–10 years with no apparent tendency for any consistent change to either older or younger modal ages (Fig. 2). Despite 9–12 age classes being represented in the catch, charr age eight and nine frequently made up 45–50% of the harvest with relatively few fish greater than age 12. While modal ages have not shown any consistent change, mean age of the catch declined by about 1.2–1.5 years in both Nain and Okak stocks, respectively, during the period 1982–2000 before increasing in latter years (Fig. 3a). Mean age of Voisey charr increased from the late 1970s–1991, declined to 1993 but has increased since the late 1990s. As noted above, more than 640 thousand charr have been harvested from the Nain stock complex itself since 1974, and fisheries continue to be supported on the basis of relatively few age classes.
Size structure
Length-frequency distributions were unimodal with modal lengths remarkably stable for the Voisey stock complex over all time periods (Fig. 4a) although owing to large sample sizes, there were significant differences among length-frequency distributions (P < 0.0001). There was a shift to a smaller modal length at Nain following the initial time period (Fig. 4b; 1980–1985), while at Okak, largest modal sizes occurred during the first (Fig. 4c; 1980–1985) and last (2001–2005) time intervals. Again owing to very large sample sizes, statistically significant differences in the length distributions (P < 0.0001) were evident for both Nain and Okak stock complexes. For all stocks, 85–90% of the commercial catch consisted of charr from the 44 to 56 cm length intervals. Subtle changes were also evident. Fish ≥60 cm declined over the first four intervals before showing a slight increase during the most recent time period. Overall, however, charr of this size contributed less than 8% of the total catch.
There were substantive changes in mean weight over years (Fig. 3b). Indeed, all stocks showed a significant decline (P < 0.001) in overall weight during the period 1977–1997, reaching minimum values in 1997 (Fig. 3b). Negative regression coefficients indicated that Arctic charr caught at Nain and Okak declined by about 30 g y−1 for the period 1977 to 1997 while Voisey charr declined by about 40 g y−1. Coincident with reduced commercial harvests since the late 1990s, mean weights have increased in recent years (Fig. 3b).
Condition varied significantly across years (Voisey: F = 71.42, P < 0.001; Nain: F = 78.29, P < 0.001; Okak: F = 51.11, P < 0.001) but with an overall tendency for a decline beginning in the early 1980s, followed by an increase in the late 1980s but were then variable with somewhat of an increasing trend beginning in the latter 1990s (Fig. 3c). This was followed by a brief period of increasing condition and then another decline in 1997 before showing a moderate increase in recent years as observed with changes in weight.
Growth
Changes in length-at-age over time determined from comparisons of the von Bertalanffy growth equations were evident in all three stock complexes, but particularly in charr from the Voisey and Nain stocks (Table 1; Fig. 5a, b). Analyses of the residual sums of squares indicated significant differences for each complex (Voisey: F = 20.72, P < 0.001; Nain: F = 54.25, P < 0.001; Okak: F = 10.56, P < 0.001). The largest charr from the Voisey and Nain stocks, and those for which the asymptotic length (L∞) was the greatest, occurred in fish sampled from the initial 5 year time period (1977–1981) (Fig. 5a, b). The smallest fish at the dominant ages in the catch (ages 7–10) were noted for the 1992–1996 period. In both Voisey and Nain charr the length-at-age increased in recent years, with the highest growth coefficients (K) occurring during the 1997–2001 period. Growth of Okak charr varied the least among the three stocks (Fig. 5c) with charr aged 8 and above somewhat larger in length during the last two time periods by comparison with the earlier years.
Food and feeding
Various species of fish were the dominant items in the diet of charr from all three stock complexes (Table 2) with sand lance, Ammodytes spp., and various sculpin species (Cottidae: Triglops spp. and Myoxocephalus spp.) being the most important taxa for charr caught in the Voisey stock complex along with hyperiid amphipods. For charr caught in the inshore zone of the Nain stock complex, the percentage contribution of sand lance and hyperiidae amphipods, by weight, declined in recent years, while capelin, Mallotus villosus, increased substantially averaging almost 40% of the biomass of all food items consumed during the 2003–2005 period (Table 2). In contrast, sculpins were by far the most important contribution in the diet of charr caught in the offshore zone of the Nain complex (island area outside of Nain and Tikkoatokak bays) with capelin and sand lance showing little or no change in recent years (Table 2).
Discussion
Historically, it was commonly believed that anadromous Arctic charr were highly susceptible to over fishing with populations reportedly collapsing in relatively short periods of time when heavily exploited. This scenario does not seem to apply to the situation regarding north Labrador charr. Following peak catches of 100 to almost 190 t y−1, representing 50 to almost 90 thousand charr per year during the late 1970s and early 1980s from the Voisey, Nain, and Okak stock complexes, harvest levels declined primarily as a result of reduced effort. Regardless, over the past three decades more than 1.2 million charr were caught in commercial fisheries in a limited geographic area with populations maintaining remarkably stable length and age distributions. Despite these removals, catches were continually dominated by four age groups (charr aged 7 to 10 years) with modal lengths frequently in the range of 48–52 cm. Ages and lengths were also similar to those reported by Andrews and Lear (1956) for charr sampled at Nain in 1953 while Okak charr, historically, were somewhat smaller but older. Thus, over a period of time extending approximately 50 years, the age and length composition of charr caught in north Labrador commercial fisheries has changed relatively little. Collectively, the results point to the capacity of the north Labrador region to produce as well as sustain fisheries on anadromous Arctic charr over long periods of time.
Part of the stability in length distributions of north Labrador charr likely relates to the influence of gill net selectivity and the large degree of overlap observed in length at age as commonly reported in previous investigations on anadromous Arctic charr (see Johnson 1980). However, Johnson (1989) reported similar results regarding stability in age and size coincident with intensive exploitation and declining abundance when monitoring Nauyak Lake anadromous charr by a largely non-selective fish weir. Only in the final stages of population decline was there any notable change in modal size and age (Johnson 1989). Results from north Labrador also parallel those reported by Johnson (1994) regarding the stability of freshwater resident populations of Arctic charr when subject to controlled exploitation experiments. When exploitation was reduced, or terminated, Johnson found that populations returned to their original configurations. This phenomenon, however, differed with results of controlled mass removals of resident charr from Takvatan, Norway (Klemetsen et al. 2002) where intense fishing changed the population structure of charr in the lake. The mechanisms by which populations are believed to be regulated have previously been discussed at length (Johnson 1976, 1983, 1994, 2001, 2002) and are thought to apply equally to resident freshwater as well as anadromous populations (Johnson 1989), although alternative explanations have been suggested (e.g. Power 1978; Finstad et al. 2000). It is noted that investigations on other species, such as orange roughy, Hoplostethus atlanticus, have also reported remarkable stability in size structure despite major reductions in stock size (Clark et al. 2000 and references therein).
While age and length distributions remained remarkably stable in the case of north Labrador charr, dramatic changes were observed in mean weight with more modest changes occurring in length-at-age. This pattern was observed in all three stock complexes but with the rate of change differing more in charr caught in the Voisey stock complex by comparison with either Nain or Okak charr. Jensen (1995) reported that fatter fish are more efficiently captured than thinner fish of the same length. Thus, in recent years, possibly as a result of reduced fishing since the early 1990s, mean weights as well as length-at-age, have begun to return to values not recorded since the mid-to-late 1980s. It is uncertain, however, whether the observed changes in population structure are simply a direct result of reduced exploitation on the anadromous stocks, or a combination of reduced fishing plus the influence of environmental conditions that were previously shown also to have affected measured stock characteristics of Nain charr (Power et al. 2000). In the latter study summer sea-surface temperatures, and summer precipitation and temperature lagged 4 years, were shown to have a strong influence on patterns observed in trends in mean weight. During the 1990s environmental conditions in the northwest Atlantic fluctuated from among the coldest to warmest conditions recorded (Colbourne 2004) often with consequences linked to dramatic changes in abundance, distribution and biology of various marine species (e.g. Rose et al. 2000; Vilhjálmsson and Hoel 2005). Capelin, once a dominant component of the diet of charr from the inshore component of the Nain stock complex (Dempson et al. 2002), all but disappeared from the diet by 1991 while in recent years it has once again become the single most important prey item utilized by these charr. But, the contribution of capelin in the diet of Voisey charr was minor by comparison with sand lance and scuplins, yet substantive increases in mean weight have also occurred in recent years coincident with reduced harvests in commercial fisheries.
The stability observed in age and length distributions that has persisted over decades of high fishery removals preclude their utility to infer when stocks are being over exploited whereas changes in mean weight, or mean weight-at-age, may yield more useful information at least in the context of north Labrador Arctic charr. Currently, mean weights are on the increase coincident with commercial harvests that are now a fraction of historical removals. Nevertheless, in the absence of actual changes in abundance coupled with less selective means by which biological data can be obtained, it may be difficult to quantify sustainable harvest levels. Johnson (1980) found that harvesting at a rate of 11% of the standing stock was excessive for anadromous charr in the Nauyak Lake system while Kristofferson et al. (1991) reported that an annual harvest rate of 5% was sustainable at Cambridge Bay but recommended a safe harvest level of about 10% on the Koukdjuak River, Nettling Lake stock. Harvest rates of 16–18% were estimated for Hornaday River, Northwest Territories, charr (Harwood 1999). Harwood (1999) acknowledged that harvest rates of around 11% may be somewhat conservative when applied to stocks with faster growth rates. By comparison, about 16% of the total estimated stock size, or 40% of the exploitable component of the population when selectivity of the gill net fishery was considered, was estimated to have been harvested from the Nain stock complex in north Labrador during the late 1970s and 1980s (Dempson 1995). Harvesting at these rates yielded results consistent with growth overfishing (Dempson 1995), but no evidence of overall recruitment failure or stock collapse. These rates are also considered to be minimum values as the amount of charr taken in subsistence or recreational fisheries is largely unknown. Given the substantive reductions in harvests, it is unknown how long the Nain charr stock complex could have sustained the continued high levels of fishery removals recorded in the late 1970s and 1980s before more dramatic changes in stock characteristics became apparent.
Management of anadromous Arctic charr populations is challenging owing to factors such as incomplete harvest information and dramatic fluctuations in abundance of stocks (Crawford 1989), coupled with variable year class strength (e.g. Day and de March 2004), the lack of directional change in length or age structure (Johnson 1989), the absence of established biological reference points or conservation requirements, and the added complexity of their migratory patterns and tendency for fish to interchange among rivers in local areas (Dempson and Kristofferson 1987; Beddow et al. 1998). Nevertheless, with the potential for increased demands associated with the process by which Inuit domestic harvest levels are established under land claim responsibilities (e.g. Canada 2004), and the likely impacts of climate change on northern fish populations including species such as Arctic charr (Wrona et al. 2005; Reist et al. 2006a, 2006b, 2006c), long term investigations of the dynamics of northern populations will become even more valuable to maintain or initiate in order to assist in the sustainable conservation of the resource. To this end, adaptive approaches such as that suggested by Kristofferson (2002) for the management of the Cambridge Bay, Nunavut, Arctic charr resource have merit and could be implemented in the north Labrador region. For example, Kristofferson’s (2002) approach would involve community consultations to review past results and trends, and identify management goals as well as data deficiencies, incorporate traditional ecological knowledge along with alternative fishing and management options, such as pulse or rotational fisheries, and then contrast results with outcomes of past approaches. By way of continued consultation decisions could be made as to where changes are needed. An approach such as this would allow users of the resource to participate fully in the development of sound management options.
References
Andrews CW, Lear E (1956) The biology of Arctic char (Salvelinus alpinus L.) in northern Labrador. J Fish Res Board Can 13:843–860
Baird JW (1983) A method to select optimum numbers for aging in a stratified approach. Can Spec Publ Fish Aquat Sci 66:161–164
Baker TT, Timmons LS (1991) Precision of ages estimated from five bony structures of Arctic char (Salvelinus alpinus) from the Wood River system, Alaska. Can J Fish Aquat Sci 48:1007–1014
Beddow TA, Deary C, McKinley RS (1998) Migratory and reproductive activity of radio-tagged Arctic char (Salvelinus alpinus L.) in northern Labrador. Hydrobiologia 371/372:249–262
Bigelow HB (1963) Fishes of the western North Atlantic. Arctic charr. Memoir Sears Foundation for Marine Research. New Haven, Connecticut, pp. 507–524
Canada. (2004) Land claims agreement between the Inuit of Labrador and Her Majesty the Queen in right of Newfoundland and Labrador and Her Majesty the Queen in right of Canada. Department of Indian Affairs & Northern Development, Ottawa, Canada
Chen Y, Jackson DA, Harvey HH (1992) A comparison of von Bertalanffy and polynomial functions in modelling fish growth data. Can J Fish Aquat Sci 49:1228–1235
Clark MR, Anderson OF, Francis RICC, Tracey DM (2000) The effects of commercial exploitation on orange roughy (Hoplostethus atlanticus) from the continental slope of the Chatham Rise, New Zealand, from 1979 to 1997. Fish Res 45:217–238
Coady LW, Best CW (1976) Biological and management investigations of the Arctic char fishery at Nain, Labrador. Can Fish Mar Serv Res Dev Tech Rep 624, 103 pp
Colbourne EB (2004) Decadal changes in the ocean climate in Newfoundland and Labrador waters from the 1950s to the 1990s. J Northw Atl Fish Sci 34:43–61
Colbourne E, deYoung B, Narayanan S, Helbig J (1997) Comparison of hydrology and circulation on the Newfoundland shelf during 1990–1993 with the long-term mean. Can J Fish Aquat Sci 54(Suppl. 1):68–80
Crawford R (1989) Exploitation of Arctic fishes. Can Manuscr Rep Fish Aquat Sci 2002, 43 pp
Day AC, de March B (2004) Status of Cambridge Bay anadromous Arctic char stocks. Fisheries and Oceans Canada, Can Sci Adv Sec Res Doc 2004/052, 78 pp
Dempson JB (1984) Conversion factors for northern Labrador Arctic charr landings statistics. Can Atl Fish Scientific Advisory Committee Res Doc 84/6, 8p
Dempson JB (1995) Trends in population characteristics of an exploited anadromous Arctic charr, Salvelinus alpinus, stock in northern Labrador. Nordic J Freshw Res 71:197–216
Dempson JB, Green JM (1985) Life history of anadromous Arctic charr, Salvelinus alpinus, in the Fraser River, northern Labrador. Can J Zool 63:315–324
Dempson JB, Kristofferson AH (1987) Spatial and temporal aspects of the ocean migration of anadromous Arctic char. Am Fish Soc Symp 1:340–357
Dempson JB, Shears M, Bloom M (2002) Spatial and temporal variability in the diet of anadromous Arctic charr, Salvelinus alpinus, in northern Labrador. Env Biol Fish 64:49–62
Dunbar MJ, Hildebrand HH (1952) Contribution to the study of fishes of Ungava Bay. J Fish Res Board Can 9:83–128
Finstad AG, Jansen PA, Langeland A (2000) Gillnet selectivity and size and age structure of an alpine char (Salvelinus alpinus) population. Can J Fish Aquat Sci 57:1718–1727
Frost WE (1978) The scales of the charr, (Salvelinus willuughbii Günther), in Windermere, and their use for the determination of age and growth. J du Conseil Int pour l’Exploration de la Mer 38:208–215
Grainger EH (1953) On the age, growth, migration, reproductive potential and feeding habitats of the Arctic char (Salvelinus alpinus) of Frobisher Bay, Baffin Island. J Fish Res Board Can 10:326–370
Haddon M (2001) Modelling and quantitative methods in fisheries. Chapman & Hall/CRC. Boca Raton, Florida
Harwood LA (1999) Status of anadromous Arctic charr (Salvelinus alpinus) of the Hornaday River, Northwest Territories, as assessed through community-based sampling of the subsistence fishery, August–September 1990–1998. Fisheries and Oceans Canada, Canadian Stock Assessment Secretariat Research Document 99/182
Hunter JG (1976). Arctic char and hydroelectric power in the Sylvia Grinnell River. Fish Res Board Can Manuscr Rep 1376, 21 pp
Jensen JW (1995) Evaluating catches of salmonids taken in gillnets. J Fish Biol 46:862–871
Johnson L (1976) Ecology of arctic populations of lake trout, Salvelinus namaycush, lake whitefish, Coregonus clupeaformis, Arctic char, S. alpinus, and associated species in unexploited lakes of the Canadian Northwest Territories. J Fish Res Board of Can 33:2459–2488
Johnson L (1980) The Arctic charr, Salvelinus alpinus. In: Balon EK (ed) Charrs: Salmonid Fishes of the Genus Salvelinus. Dr. Junk Publishers, The Hague, pp. 15–98
Johnson L (1983) Homeostatic characteristics of single species fish stocks in Arctic lakes. Can J Fish Aquat Sci 40:987–1024
Johnson L (1989) The anadromous Arctic charr, Salvelinus alpinus of Nauyuk Lake, N.W.T., Canada. Physiol Ecol Japan Spec 1:201–227
Johnson L (1994) Long-term experiments on the stability of two fish populations in previously unexploited arctic lakes. Can J Fish Aquat Sci 51:209–225
Johnson L (2001) Imperfect symmetry: action principles in ecology and evolution. In: Jørgensen SE (ed) Thermodynamics and ecological modelling. Lewis Publishers, Boca Raton, Florida, pp. 231–285
Johnson L (2002) Imperfect symmetry: thermodynamics in ecology and evolution. Torgoch Publishing, Sidney, British Columbia. 221 pp
Klemetsen A, Amundsen P-A, Grotnes PE, Knudsen R, Krustoffersen R, Svenning MA (2002) Takvatn through 20 years: long-term effects of an experimental mass removal of Arctic charr, Salvelinus alpinus, from a subarctic lake. Env Biol Fish 64:39–47
Kristofferson AH (2002) Identification of Arctic char stocks in the Cambridge Bay area, Nunavut Territory, and evidence of stock mixing during overwintering. PhD Dissertation, University of Manitoba, Winnipeg, Manitoba
Kristofferson AH, Sopuck RD (1983) The effects of exploitation on the Arctic charr population of Sylvia Grinnell River, Northwest Territories. Can Manuscr Rep Fish Aquat Sci 1721, 35pp
Kristofferson AH, McGowan DK, Carder GW (1984) Management of the commercial fishery for anadromous Arctic charr in the Cambridge Bay Area, Northwest Territories, Canada. In: Johnson L, Burns BL (eds) Biology of the Arctic charr. Proceedings of the international symposium on arctic charr, Winnipeg, Manitoba, May 1981, Univ Manitoba Press, Winnipeg, pp. 447–461
Kristofferson AH, Sopuck RD, McGowan DK (1991) Commercial fishing potential for searun Arctic charr, Koukdjuak River and Nettling Lake, Northwest Territories. Can Manuscr Rep Fish Aquat Sci 2120
Mattox WG (1973) Fishing in west Greenland 1910–1966 – the development of a new native industry. Medd Om Grönland Bd 197(1)
Nordeng H (1961) On the biology of char (Salmo alpinus L.) in Salangen, North Norway. Part I. Age and spawning frequency determined from scales and otoliths. Nytt Mag Zool 10:67–123
Parker HH, Johnson L (1991) Population structure, ecological segregation and reproduction in non-anadromous Arctic charr, Salvelinus alpinus, in a series of unexploited lakes in the Canadian Arctic. J Fish Biol 38:123–147
Power G (1978) Fish population structure in Arctic lakes. J Fish Res Board Can 35:53–59
Power M, Dempson JB, Power G, Reist JD (2000) Environmental influences on an exploited anadromous Arctic charr stock in Labrador. J Fish Biol 57:82–98
Reist JD, Wrona FJ, Prowse TD, Power M, Dempson JB, Beamish RJ, King JR, Carmichael TJ, Sawatzky CD (2006a) General effects of climate change on Arctic fishes and fish populations. Ambio 35:370–380
Reist JD, Wrona FJ, Prowse TD, Power M, Dempson JB, King JR, Beamish RJ (2006b) An overview of effects of climate change on selected Arctic freshwater and anadromous fishes. Ambio 35:381–387
Reist JD, Wrona FJ, Prowse TD, Dempson JB, Power M, Köck G, Carmichael TJ, Sawatzky CD, Lehtonen H, Tallman RF (2006c) Effects of climate change and UV radiation on fisheries for Arctic freshwater and anadromous species. Ambio 35:402–410
Ricker, WE (1975) Computation and interpretation of biological statistics of fish populations. Bull Fish Res Board Can 191, 382 pp
Ricker WE (1981) Changes in the average size and average age of Pacific salmon. Can J Fish Aquat Sci 38:1636–1656
Rose GA, deYoung B, Kulka DW, Goddard SV, Fletcher GL (2000) Distribution shifts and overfishing the northern cod (Gadus morhua): a view from the ocean. Can J Fish Aquat Sci 57:644–663
SAS Institute Inc (2000) SAS/STAT User’s guide. Version 8., SAS Institute Inc, Cary, North Carolina
Sokal RR, James Rohlf F (1998) Biometry: the principles and practice of statistics in biological research. W. H. Freeman & Company, New York
Vilhjálmsson HA, Hoel H (2005) Fisheries and aquaculture. In: ACIA - Arctic Climate Impact Assessment. Cambridge University Press, Cambridge, pp. 691–780
Wrona FJ, Prowse TD, Reist JD (2005) Freshwater ecosystems and fisheries. In: ACIA – Arctic Climate Impact Assessment. Cambridge University Press, Cambridge, pp. 353–452
Yessipov VK (1935) Materials on the biology and fishery of the Arctic char on Novaya Zemlya (Salvelinus alpinus). Transactions of the Arctic Institue 17:5–71 (in Russian, with translation from Russian: Canada Fish Mar Serv Transl No. 4436, 1978)
Acknowledgements
Mike O’Connell, Geoff Veinnott, and Mike Power critically reviewed earlier drafts of this manuscript and provided editorial advice. I also wish to acknowledge the encouragement and interest in our north Labrador Arctic charr work by the late Lionel Johnson whose friendship extended over a period of 28 years and included a number of “fanatical” trips during which time frequent questions and suggestions were often mixed with a few ‘libations’. These will long be remembered.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brian Dempson, J., Shears, M., Furey, G. et al. Resilience and stability of north Labrador Arctic charr, Salvelinus alpinus, subject to exploitation and environmental variability. Environ Biol Fish 83, 57–67 (2008). https://doi.org/10.1007/s10641-007-9261-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-007-9261-2