Abstract
Skates by virtue of their abundance and widespread occurrence appear to play an influential role in the food webs of demersal marine communities. However, few quantitative dietary studies have been conducted on this elasmobranch group. Therefore, to better understand the ecological role of skates, standardized diet compositions and trophic level (TL) values were calculated from quantitative studies, and compared within and among skate and shark taxa. Prey items were grouped into 11 general categories to facilitate standardized diet composition and TL calculations. Trophic level values were calculated for 60 skate species with TL estimates ranging from 3.48 to 4.22 (mean TL = 3.80 ± 0.02 SE). Standardized diet composition results revealed that decapods and fishes were the main prey taxa of most skate species followed by amphipods and polychaetes. Correspondingly, cluster analysis of diet composition data revealed four major trophic guilds, each dominated by one of these prey groups. Fish and decapod guilds were dominant comprising 39 of 48 species analyzed. Analysis of skate families revealed that the Arhynchobatidae and Rajidae had similar TL values of 3.86 and 3.79 (t-test, P = 0.27), respectively. The Anacanthobatidae were represented by a single species, Cruriraja parcomaculata, with a TL of 3.53. Statistical comparison of TL values calculated for five genera (Bathyraja, Leucoraja, Raja, Rajella, Rhinoraja) revealed a significant difference between Bathyraja and Rajella (t-test, P = 0.03). A positive correlation was observed between TL and total length (L T) with larger skates (e.g. >100 cm L T) tending to have a higher calculated TL value (>3.9). Skates were found to occupy TLs similar to those of several co-occurring demersal shark families including the Scyliorhinidae, Squatinidae, and Triakidae. Results from this study support recent assertions that skates utilize similar resources to those of other upper trophic-level marine predators, e.g. seabirds, marine mammals, and sharks. These preliminary findings will hopefully encourage future research into the trophic relationships and ecological impact of these interesting and important demersal predators.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Skates (Chondrichthyes: Rajiformes: Rajoidei) are the most diverse group of living cartilaginous fishes, with approximately 245 species recognized worldwide (Ebert and Compagno 2007). This total represents nearly 25% of the described species of chondrichthyan fishes (Compagno 2005). In addition, skates appear to exhibit a fairly high degree of endemism, with many species having somewhat localized regional distributions (McEachran and Miyake 1990; Compagno et al. 1991; Menni and Stehmann 2000; Last and Yearsley 2002). A morphologically conservative group, skates are most commonly found along outer continental shelves and upper slopes. They are the only cartilaginous fish taxon to exhibit a great diversity of species at higher latitudes.
Skates, by virtue of their abundance and species diversity, may play influential roles in the food webs of demersal marine communities. It has been hypothesized that skates may negatively impact commercially valuable groundfishes via ecological interactions such as competition and predation (Murawski 1991; Mayo et al. 1992; Fogarty and Murawski 1998; Link et al. 2002; Orlov 2004). Quantifiable studies, however, on the diet composition and trophic relationships of skates are few (Garrison and Link 2000; Bulman et al. 2001; Davenport and Bax 2002). Additionally, although some skate species are considered top predators (Macpherson and Roel 1987; Ebert et al. 1991; Orlov 1998, 2003; Link et al. 2002) surprisingly few quantitative estimates of trophic level values exist to substantiate this perception (Morato et al. 2003; Braccini and Perez 2005). Furthermore, the impact that skates may have on associated demersal marine communities is still relatively unknown.
If skates are upper trophic level predators, as demonstrated for sharks (Cortés 1999; Estrada et al. 2003), marine mammals (Bowen 1997; Pauly et al. 1998), and seabirds (Sanger 1987; Hobson 1993), they may influence the relative abundance and diversity of co-occurring demersal species (Beddington 1984; Rogers et al. 1999). Therefore, to better understand the ecological role of skates, we present standardized diet compositions and trophic level values calculated from quantitative studies. We then compare results within skate taxa and among skate and shark taxa to investigate similarities and differences in these parameters between elasmobranch groups.
Materials and methods
Diet composition data were summarized from peer-reviewed journal articles, graduate theses, and gray literature (see Appendix I). Only sources that included quantitative information in the form of indices or provided sufficient information to facilitate index calculations were utilized. The following search engines were initially used to locate citations: Aquatic Sciences and Fisheries Abstracts, Biosis, Web of Science, and Zoological Record. After relevant articles were obtained, their literature cited sections were perused for additional references. Colleagues were also contacted to obtain supplemental literature and especially unpublished graduate theses that may have been overlooked during previous searches. All pertinent literature was accumulated and examined, regardless of publication language.
Prey items were grouped into 11 general categories to facilitate standardized diet composition and trophic level calculations (Table 1). Trophic levels for generalized prey categories were directly incorporated when available or estimated by calculating a mean value from information contained in the following sources: Fauchauld and Jumars (1979); Hobson and Welch (1992); Hobson (1993); Pauly and Christensen (1995); Pauly et al. (1998); Cortés (1999); and Nyssen et al. (2002). Because multiple diet studies existed for some skates, an index of standardized diet composition, weighted by relative sample size, was calculated after Cortés (1999) to determine the proportion of each prey category in the diet of a species (Pj):
where Pij = the proportion of prey category j from source i, Ni = the number of stomach samples containing food that were used to calculate Pij for source i, n = number of sources, j = total number of prey categories, and ∑Pj = 1.
The proportion of each prey category (Pij) was determined for each source using the following hierarchical criteria. Compound indices (e.g., index of relative importance, geometric index of importance, index of absolute importance) were used if available. If no compound index was available but more than one single index was used, a geometric index of importance was calculated on a percentage basis by averaging all available indices (e.g., (%N + %W)/2), as described by Assis (1996). Single indices were used when multiple indices were not available. Incidentally ingested materials (e.g., plant matter, sediment, detritus) and undeterminable items (e.g., offal, unidentified organic matter) were not incorporated in analyses. In addition, all non-additive indices, such as percent frequency of occurrence (%FO) or the original index of relative importance (Pinkas et al. 1971), were standardized so that the contribution of each index to diet composition calculations was equal. When only the total number of occurrences of each prey item was available, the occurrence index (O) was calculated after Mohan and Sankaran (1988). When only proportions (%FO) were provided, they were summed and standardized in the same manner.
Trophic levels were then calculated for each species using the following equation, after Cortés (1999):
where TLk = trophic level of species k, Pj = proportion of prey category j in the diet of species k, n = total number of prey categories, and TLj = trophic level of prey category j. Mean trophic level (TL) values were also calculated for the order, families, and genera using TL estimates from individual species. When data were normally distributed and of equal variances, TL values were compared between and among groupings with t-tests and ANOVAs (SYSTAT, version 10, SPSS, Inc., Chicago, IL). If either of these assumptions were violated, data were log-transformed and re-evaluated prior to analysis. Because it was not possible to generate precision estimates from the available data (Ferry and Cailliet 1996), a minimum sample size of 20 individuals was arbitrarily chosen as the limit for inclusion of a species in all analyses.
Cluster analysis was utilized to compare standardized diet compositions among skate species. Calculations were preformed in SYSTAT (version 10, SPSS, Inc., Chicago, IL) using the unweighted pairwise group mean average method (UPGMA) with Euclidean distance as a measure of dissimilarity. Dissimilarity values among clusters that were ≥50% of the maximum overall dissimilarity distance were considered to indicate major divisions and used to distinguish trophic guilds of skates (Root 1967; Yoklavich et al. 2000).
Results
Standardized diet compositions and trophic levels were calculated for 60 skate species (Table 2). Overall, trophic level estimates ranged from 3.48 (Rajella caudaspinosa) to 4.22 (Dipturus chilensis). Eleven skate species were estimated to have TL values ≥4, with the genera Bathyraja and Dipturus each having four species represented. Two species, Amblyraja radiata and Raja clavata, had the most quantitative diet studies with nine each, followed by Raja montagui with six studies, and Bathyraja aleutica and Leucoraja naevus with five studies each. Three species had a total of four different diet studies, whereas six species and 14 species had three and two studies each, respectively. Thirty-two of the species included in the present study had only a single diet study. The species with the most stomachs examined was Leucoraja erinacea (n = 19,738), whereas Rajella dissimilis (n = 2) had the fewest. Six species had >1,000 stomachs examined from various combined studies, 31 species had between 134 and 952 stomachs examined, and 23 species had <100 stomachs examined.
Standardized diet composition results revealed that decapod crustaceans and secondarily fishes were the main prey taxa of most skate species with sufficient sample sizes (Table 2). Overall, decapods comprised 36.35 ± 3.06 SE of mean percent diet composition, were present in the diets of all species examined, and ranged from 0.26% of dietary composition in Rhinoraja maclovania to 77.16% for Rioraja agassizii. Fishes contributed 28.61 ± 3.31 to the mean diet composition of the skate species studied and comprised >50% of the diet composition of nine species (Table 2). Fishes were of greatest dietary importance to Dipturus batis (70.76%) and D. chilensis (81.76%), but in contrast were not present in the diets of Leucoraja melitensis or R. maclovania. Polychaetes and amphipods were of supplemental importance in skate diets, only contributing >50% of the diet composition of Bathyraja griseocauda (52.54%), Rhinoraja albomaculata (51.53%), R. maclovania (89.24%), and R. taranetzi (52.71%). Squids (3.36 ± 0.87), euphausids and mysids (6.07 ± 1.55), and other crustaceans (3.68 ± 0.79) were minor prey items. Shelled molluscs, octopi and cuttlefishes, and chondrichthyans were not important prey items for skates, with maximum reported diet composition values of 10.92% (Leucoraja erinacea), 14.01% (Rostroraja alba), and 5.00% (Leucoraja fullonica), respectively. Other invertebrates and unidentified invertebrates were only reported from 35.4% of the species studied and contributed no more than 0.82% to diet composition (A. radiata).
Cluster analysis of calculated diet composition estimates for sufficiently sampled species revealed four major trophic guilds with Euclidean distances (ED) > 13.7, or 50% of maximum dissimilarity (Fig. 1). These guilds were dominated by the following prey taxa: polychaetes, amphipods, fishes, and decapods. The polychaete guild (ED = 18.62) included only two species, both in the genus Rhinoraja; R. albomaculata and R. macloviana. The amphipod guild (ED = 16.86) was comprised of seven relatively small species; four of which are found within two genera, Bathyraja and Rhinoraja. Amphipods were the dominant prey taxon of this guild (41.90 ± 1.66), but decapods were also important (28.74 ± 2.68) and were the main taxon of Bathyraja violacea and L. erinacea. The fish guild (ED = 15.53), which contained many of the largest skate species within this study, had the greatest number of species at 20; half of which were represented by two genera, Bathyraja (n = 6) and Raja (n = 4). All representative species within the genera Amblyraja (n = 2) and Dipturus (n = 3) were found within this guild. Decapods were of substantial supplemental importance within the fish guild and were the primary prey taxon of one species (Raja straeleni). The decapod guild (ED = 13.9) contained 19 species, with the genus Raja represented by eight species. Decapods were dominant in the diets of all species within this guild, with fishes typically of secondary importance. Both the fish and decapod guilds had seven skate genera represented. Although no genera were represented among all four guilds, three genera, Bathyraja, Leucoraja, and Rajella, had representatives among three of the four trophic guilds.
Cluster analysis of standardized diet compositions of skate species (n = 48). Hierarchical clustering was performed using average linkage with Euclidean distance as a measure of dissimilarity. Major trophic guilds are indicated (see Table 2 for description of prey categories)
The mean TL for all skate species combined was 3.80 ± 0.02 SE (n = 60). A breakdown of the three skate families revealed that the Arhynchobatidae and Rajidae had similar trophic levels of 3.86 and 3.79, respectively (Table 3). The Anacanthobatidae were represented by a single species, Cruriraja parcomaculata, with a TL of 3.53. Estimated TL values for the two families (Arhynchobatidae, n = 18 and Rajidae, n = 29) for which multiple samples were available showed normal distributions (Kolmogorov-Smirnov, P > 0.05) and equal variances (F-test, P > 0.05). Comparison of these two families using a two sample t-test revealed no significant difference (t = 1.11; P = 0.27).
The family Rajidae had eight genera represented, followed by the Arhynchobatidae with four and a single genus for the Anacanthobatidae. Of the 13 genera included in this study, five were represented by a single species. The remaining eight genera had at least two or more species studied within the genus. Bathyraja (n = 13) had the most species studied followed by the Raja (n = 12). All genera, except for the monotypic Rostroraja (TL = 4.15), had a mean TL between 3.5 and 3.9. The genera with the lowest TLs were Cruriraja and Neoraja at 3.53 and 3.52, respectively.
Statistical comparison of log-transformed TL values calculated for five genera (Bathyraja, Leucoraja, Raja, Rajella, Rhinoraja) for which sample size was reasonably high (Table 3) revealed a significant difference among samples (ANOVA, F = 2.85; P = 0.04). Bathyraja (n = 12) was found to feed at a higher TL than Rajella (n = 4) (Tukey’s, P = 0.04). There were no significant differences found in comparisons between any of the other genera.
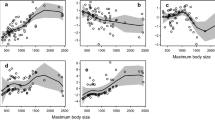
A positive relationship was observed between TL and total length (L T) of skates (Fig. 2). Skates with TL values >3.9 (n = 15) were usually the larger species, e.g. those exceeding 100 cm L T, however, Bathyraja brachyurops (73 cm L Tmax) was an exception (TL = 4.08). Of those skates with TLs between 3.8 and 3.9 (n = 17) most were <100 cm L T; three species (Dipturus pullopunctata, Raja brachyura, R. rhina) grow to between 120 and 137 cm L T and one (Raja binoculata) exceeds 200 cm L T. At the other extreme, Psammobatis extenta, at 31 cm L Tmax, appears to have a much higher TL (3.84) than would be expected for a skate of this size. All skates with TLs <3.8, except for two species (Amblyraja radiata and Raja pulchra), have a maximum L T of <100 cm.
Comparison of TL values at the ordinal level (Table 4) between skates and sharks showed that skates (Rajiformes) had lower trophic levels than all but two shark orders; the Heterodontiformes and Orectolobiformes. Log-transformed trophic level values for skate families (Table 4) relative to values for sympatric demersal shark families (Scyliorhinidae, Squalidae, Squatinidae, and Triakidae) revealed significant differences (ANOVA, F = 10.04; P < 0.001). The combined data for Arhynchobatidae and Rajidae were not significantly different from those of Scyliorhinidae, Squatinidae, and Triakidae (Tukey’s, P < 0.05), but were significantly lower than the TL value calculated for Squalidae (Tukey’s, P < 0.001). The Pristiophoridae, a small group of demersal sharks, were represented by a single species (Pliotrema warreni), that appeared to feed at a higher TL (4.2) than skates.
Discussion
Of the 245 described skate species (Ebert and Compagno 2007) <24% have had any quantitative dietary information reported, with a single study conducted for most of these species (53.3%). Furthermore, only 37 (14.6%) of 253 skate species have had >100 stomachs examined. In contrast to the relatively small percentage of individual skate species that have been studied, coverage at the generic level has been relatively broad, with quantitative diet information reported for 13 of 27 recognized skate genera (Ebert and Compagno 2007). Although significant differences were found between some skate genera, it appears that phylogeny is a less important predictor of TL than the prey categories used to calculate TL values, size, and possibly regional differences.
Determination of prey categories used to calculate standardize diet compositions may influence the TL of individual species. Psammobatis extenta, for example, was found to have a lower TL (3.53) (Braccini and Perez 2005) based on the more general categories established for sharks by Cortés (1999) as compared to our higher estimated TL (3.84) using slightly different prey categories than we determined from a review of published skate diet studies. We attributed this difference mainly to our use of amphipods, a primary prey taxon of this and other skate species, as a distinct prey category. Based on published stable isotope studies (Hobson and Welch 1992; Nyssen et al. 2002), amphipods had a much higher calculated TL value (Table 1) than was estimated using the broad category of marine invertebrates (TL = 2.5; Cortés 1999). Therefore, the actual TL of this species was likely underestimated by Braccini and Perez (2005). Similarly, Morato et al. (2003) collapsed their data for Raja clavata into the same categories used by Cortés (1999) and then compared trophic levels among size classes and regions; probably affecting the accuracy of their results. These examples highlight the importance of calculating and perhaps refining prey categories that are more realistic to the predator in question.
Of the four guilds that characterized the diet of skates, fish and decapod guilds were dominant comprising 39 of 48 species. This finding is consistent with other studies that reveal skates to be primarily benthopelagic piscivores or epibenthic predators specializing on invertebrates and small crustaceans (Garrison and Link 2000; Bulman et al. 2001; Davenport and Bax 2002). It is noteworthy that 13 of 20 species in the fish guild had TL values >3.90, and with no species having a TL < 3.77. Fifteen of 20 species in this guild exceed 100 cm L Tmax, with three species >2 m L Tmax. This is not unexpected as larger fish species, including skates, generally have a higher proportion of fish in their diet (Garrison and Link 2000; Davenport and Bax 2002). By comparison, the decapod guild had a TL range of 3.48–3.87 and was composed of smaller species, with 17 of 19 species <100 cm L Tmax.
Total length and TL were positively correlated, with larger skates tending to have a higher calculated TL value. This positive trend between body length and TL was similar to Cortés’ (1999) findings for sharks, but differed from Pauly et al.’s (1998) who found that body length and TL were inversely related for marine mammals. Many marine mammals, e.g. baleen whales, are plankton feeders and like the two largest shark species, i.e. Cetorhinus maximus and Rhincodon typus, tend to feed at a lower TL; usually within a TL range of 3.2–3.4 (Cortés 1999; Pauly et al. 1998). Cortés (1999) suggested that body mass may be a better predictor of TL, but this variable was not available for many species.
Trophic level comparisons among species are best made at identifiable and comparable life history stages or at a similar maximum body size (Jennings 2005). Although ontogenetic shifts in diet are well documented among individual skate species (e.g., Ajayi 1982; Ellis et al. 1996; Muto et al. 2001; Brickle et al. 2003; Robinson et al. 2007; Treloar et al. 2007), only one study has attempted to calculate TL values at different size classes (Morato et al. 2003). Several studies have shown a shift from a diet primarily consisting of amphipods and crustaceans in smaller individuals to a diet primarily consisting of teleosts in larger skates (Smale and Cowley 1992; Yeon et al. 1999; Robinson et al. 2007). Using a fixed TL for an individual species that does not take into account an increase in body size, coupled with a likely shift in diet, will not appropriately describe the structure of aquatic food webs (Jennings 2005). Different size classes within a species may be considered functionally different species in terms of trophic dynamics (Ross 1986; Garrison and Link 2000). Therefore, calculations and comparisons of diet composition for different size classes or life history stages are more appropriate than those using all available data and result in more accurate estimates of TL and determinations of trophic roles within food webs.
The importance of skates to regional ecosystems in terms of abundance and biomass, and their subsequent impact via predation on commercially important groundfish has been the subject of much debate. On Georges Bank off the northeast coast of the U.S., for example, it has been suggested that predation by skates and other elasmobranchs may negatively influence recruitment of potentially important groundfish species (Murawski 1991; Mayo et al. 1992; Fogarty and Murawski 1998). However, there is little evidence to support this hypothesis, and in fact the predatory impact by skates on commercially valuable groundfish on the Georges Bank is considered by some to be insignificant (Link et al. 2002). Conversely, in the Aleutian Islands and Bering Sea skates are considered to be major predators of commercially important groundfish species (Orlov 2003; Gaichas et al. 2005).
Trophic level analysis of allopatric skate species provides little insight into the true role of these fish as predators or competitors within an ecosystem. Conversely, examination of co-occurring skate species will provide a more accurate understanding of these high TL predators. Although skates are secondary or tertiary consumers, their TL appears to vary between, and within, different ecosystems. Raja clavata, perhaps one of the best studied skate species (Ajayi 1982; Ellis et al. 1996; Holden and Tucker 1974; Quiniou and Andriamirado 1979), had a significantly higher TL in the Azores than in other regions of the northeastern Atlantic. This difference was attributable to a higher proportion of teleosts in the diet of individuals from the Azores (Morato et al. 2003).
Individual skate species within an ecosystem often occupy different TLs. However, when treated as a species complex, skates may occupy different TLs among different ecosystems. For example, in the eastern Bering Sea, skates appear to occupy somewhat higher mean TLs (3.88, n = 9 species) than those in the Benguela Current (3.73, n = 9) and California Current (3.75, n = 4) ecosystems. Reasons for these differences are unclear, but there are very few other demersal elasmobranch competitors, e.g. sharks, in the eastern Bering Sea (Mecklenberg et al. 2002). Since diets of demersal sharks typically include a greater proportion of fishes than skate diets (Cortés 1999) the higher TL observed for skates in the eastern Bering Sea may be reflective of a lack of other demersal sharks and the associated niche expansion of skates in this region. By comparison, the Benguela Current Ecosystem has one of the most diverse elasmobranch faunas with >60 different species (Compagno et al. 1991). Many of the shark families found within the Benguela Current Ecosystem, e.g. the Squalidae and Scyliorhinidae, have higher TL values, as calculated by Cortés (1999), relative to sympatric skates. Likewise, the California Current Ecosystem has several demersal scyliorhinid and squalid species (Ebert 2003) that, at least at the family level, have higher TL values (Cortés 1999) than the four skate species reported here. Conversely, the eastern Bering Sea has no scyliorhinids and only a single squalid species that is considered rare within that ecosystem (Mecklenberg et al. 2002).
Comparison of the skates to the eight major shark taxonomic groups at the ordinal level is somewhat misleading given the variety of external body morphologies, habitats, life-styles, foraging strategies, and prey items consumed by sharks (Compagno 1990). As an example, skates were found to occupy a higher TL than two of the shark orders (Cortés 1999; Table 4). Both of these orders have representatives that differ morphologically and generally occupy distinctly different habitats than most skate species. The Heterodontiformes are small, stout-bodied sharks with one living family and genus. Unlike skates, heterodontids are primarily durophagous, feeding on such hard-bodied prey items as sea urchins and gastropods (Compagno et al. 2005). The Orectolobiformes contain seven families and 14 genera of mostly small benthic, warm temperate to tropical sharks. This order includes the whale shark Rhincodon typus, the largest living fish and a plankton feeder (Compagno et al. 2005). Although the heterodontids somewhat overlap the skates in portions of their distribution, the orectolobids generally do not spatially overlap with skates in terms of habitat (Compagno 1990). It would have been of interest to compare the skates to other batoid groups, but unfortunately no comparable study, to the best of our knowledge, exists.
At the family level, skates were found to occupy TLs similar to those of several co-occurring shark families including the Scyliorhinidae, Squatinidae, and Triakidae (Cortés 1999; Table 4). Of the 23 shark families that Cortés (1999) examined, 14 had TL values ≥4 whereas the remaining 9 had TLs ≤3.9. Six of these nine families occupy distinctly different habitats from that of skates. Three of them (Cetorhinidae, Megachasmidae, Rhincodontidae) are plankton feeders and three (Hemiscyllidae, Sphyrnidae, Stegostomidae) are considered shallow warm temperate to tropical groups (Compagno et al. 2005). Five of these families (Cetorhinidae, Hemiscyllidae, Megachasmidae, Rhincodontidae, Stegostomidae) have TLs <3.6. The similarity of TL between the squatinids and skates is not unexpected as both occupy a similar habitat, co-occur in many areas, and are of similar size. Dietary studies on squatinids have shown them to be consumers of benthic and epibenthic teleosts, cephalopods, and crustaceans (Capape 1975; Ellis et al. 1996; Ebert 2003; Volger et al. 2003). Squatinids, with a mean TL of 4.1, are similar to many of the larger skates (e.g. Dipturus batis, D. chilensis, Rostroraja alba), mainly those with a L T > 100 cm, whose TL is ≥4. Although some larger skate species may occupy similar TLs to that of squatinids, it appears based on limited evidence that these co-occurring species do not overlap in their dietary preferences. In a comparative study of the feeding ecology of six sharks (including members of the families Scyliorhinidae, Squatinidae, and Triakidae) and four skate species, Ellis et al. (1996) found a wide variety of food preferences with little overlap. In fact, only two of the skate species (Raja clavata and R. montagui) in this study exhibited slight dietary overlap with a single shark species, the triakid shark Mustelus asterias (Ellis et al. 1996). In diet studies conducted along the west coast of southern Africa it was determined that most skates preferred crustaceans and small benthic teleosts whereas scyliorhinids preferred myctophids (Ebert et al. 1991, 1996). Therefore, though skates may occupy similar habitats, and have similar TLs, to scyliorhinids, squatinids, and triakids, they appear to exhibit very little dietary overlap with these shark families. It is interesting to note that the Squalidae and Pristiophoridae both had significantly higher TLs at 4.1 and 4.2, respectively, than skates of the families Arhynchobatidae and Rajidae; Anacanthobatidae was represented by one species and not statistically tested. Members of both these aforementioned shark families occupy similar habitats to those of many skate species. Numerous diet studies on squalids have shown that members of this family tend to feed mainly on teleosts and cephalopods (Ebert et al. 1992; Ellis et al. 1996; Cortés 1999; Link et al. 2002). On a species level the most frequently studied squalid, Squalus acanthias, had a calculated TL value of 3.9 (Cortés 1999), similar to many of the studied skates.
Skates are versatile colonizers of bottom habitats, with an abundant and diverse fauna on continental shelves and insular slopes in cool temperate to boreal and deepsea environs (Compagno 1990). They are among the top predators in demersal marine habitats and appear to play important trophic roles (Ebert et al. 1991; Orlov 2003). Results from this study support recent assertions that skates utilize similar resources to those of other upper trophic-level predators, e.g. seabirds, marine mammals, large teleosts, and some sharks (Sanger 1987; Bowen 1997; Cortés 1999; Garrison 2000; Davenport and Bax 2002). However, skates, unlike these other high TL marine vertebrates, have historically often been overlooked as top predators. This is most evident by the lack of quantitative diet studies on skates. All quantitative diet studies used in our analysis, with one exception, have been published since 1972 (see Appendix I). Reasons for the lack of quantitative studies are several and include a lack of adequate systematic knowledge of the group, a lack of resources to study non-target species or those of little economic value, and because skates often live in habitats (e.g. deepsea benthic habitats) that are difficult to study.
Skates as top predators and potential competitors with groundfishes may play an influential role in structuring demersal marine communities in which they occur. It has been well documented that some batoid groups, e.g. myliobatids and rhinobatids, do in fact play an influential role in shaping infaunal communities on soft-bottom substrates (Lasiak 1982; VanBlaircom 1982; Rossuow 1983; Smith and Merriner 1985; Gray et al. 1997; Ebert and Cowley 2003). However, similar studies on skates are still wanting. It is hoped that these preliminary findings will encourage future research, perhaps incorporating the use of stable isotopes, into the trophic relationships and ecological impact of these interesting and important demersal predators.
References
Ajayi TO (1982) Food and feeding habits of Raja Species (Batoidei) in Carmarthen Bay, Bristol Channel. J Mar Biol Assoc UK 62:215–223
Assis CA (1996) A generalized index for stomach contents analysis in fish. Sci Mar 60:385–389
Beddington JR (1984) The response of multispecies systems to perturbations. In: May RM (ed) Exploitation of marine communities. Springer, Berlin, pp 209–225
Bowen WD (1997) Role of marine mammals in aquatic ecosystems. Mar Ecol Prog Ser 158:267–274
Braccini JM, Perez JE (2005). Feeding habits of the sandskate Psammobatis extenta (Garman, 1913): sources of variation in dietary composition. Mar Freshw Res 56:395–403
Brickle P, Laptihovsky V, Pompert J, Bishop A (2003) Ontogenetic changes in the feeding habits and dietary overlap between three abundant rajid species on the Falkland Island’s shelf. J Mar Biol Assoc UK 83:1119–1125
Bulman C, Althaus F, He X, Bax NJ, Williams A (2001) Diets and trophic guilds of demersal fishes of the south-eastern Australian shelf. Mar Freshw Res 52:537–548
Capapé C (1975) Contribution a la biologie des Rajidae des cotes tunisiennes. 4. Raja clavata (Linne 1758): regime alimentaire. Ann Inst Michel Pacha 8:16–32
Compagno LJV (1990) Alternate life history styles of cartilaginous fishes in time and space. Environ Biol Fish 28:33–75
Compagno LJV (2005) Checklist of living chondrichthyes. In: Hamlett WC (ed) Reproductive biology and phylogeny of Chondrichthyes: sharks, batoids, and chimaeras. Science Publishers, Inc., pp 501–548
Compagno LJV, Ebert DA, Cowley PD (1991) Distribution of offshore demersal cartilaginous fishes (Class Chondrichthyes) off the west coast of southern Africa, with notes on their systematics. S Afr J Mar Sci 10:71–81
Compagno LJV, Dando M, Fowler S (2005) A field guide to the sharks of the world. Harper-Collins, London, 416 pp
Cortés E (1999) Standardized diet compositions and trophic levels in sharks. ICES J Mar Sci 56:707–717
Davenport SR, Bax NJ (2002) A trophic study of a marine ecosystem off southeastern Australia using stable isotopes of carbon and nitrogen. Can J Fish Aquat Sci 59:514–530
Ebert DA (2003) The sharks, rays and chimaeras of California. University California Press, Berkeley, California, USA, 285 pp
Ebert DA, Cowley PD (2003) Diet, feeding behaviour and habitat utilization of the blue stingray Dasyatis chrysonota (Smith, 1828) in South African waters. Mar Freshw Res 54:957–965
Ebert DA, Compagno LJV (2007) Biodiversity and systematics of skates (Chondrichthyes: Rajiformes: Rajoidei). Environ Biol Fish (this volume)
Ebert DA, Cowley PD, Compagno LJV (1991) A preliminary investigation of the feeding ecology of skates (Batoidea: Rajidae) off the west coast of southern Africa. S Afr J Mar Sci 10:71–81
Ebert DA, Compagno LJV, Cowley PD (1992) A preliminary investigation of the feeding ecology of squaloid sharks off the west coast of southern Africa. S Afr J Mar Sci 12:601–609
Ebert DA, Cowley PD, Compagno LJV (1996) A preliminary investigation of the feeding ecology of catsharks (Scyliorhinidae) off the west coast of southern Africa. S Afr J Mar Sci 17:233–240
Ellis JR, Pawson MG, Shackley SE (1996) The comparative feeding ecology of six species of shark and four species of ray (Elasmobranchii) in the North-East Atlantic. J Mar Biol Assoc UK 76:89–106
Estrada JA, Rice AN, Lutcavage ME, Skomal GB (2003) Predicting trophic position in sharks of the north-west Atlantic Ocean using stable isotope analysis. J Mar Biol Assoc UK 83:1347–1350
Fauchauld K, Jumars PA (1979) The diet of worms: a study of polychaetes feeding guilds. Oceanogr Mar Biol Annu Rev 17:193–284
Ferry LA, Cailliet GM (1996) Sample size sufficiency and data analysis: are we characterizing and comparing diet properly? In: MacKinlay D, Shearer K (eds) Feeding ecology and nutrition in fish: proceedings of the symposium on the feeding ecology and nutrition in fish. International Congress on the Biology of Fishes, San Francisco, CA, 14–18 July 1996, pp 71–80
Fogarty MJ, Murawski SA (1998) Large-scale disturbance and the structure of marine systems: fishery impacts on Georges Bank. Ecol Appl 8:S6–S22
Gaichas S, Matta B, Stevenson D, Hoff J (2005) Bering Sea and Aleutian Islands skates. In: Stock assessment and fishery evaluation report for the groundfish resources of the Bering Sea/Aleutian Islands regions. North Pacific Fisheries Management Council, Anchorage, AK, Section 16.3:825–857
Garrison LP (2000) Spatial and dietary overlap in the Georges Bank groundfish community. Can J Fish Aquat Sci 57:1679–1691
Garrison LP, Link JS (2000) Fishing effects on spatial distribution and trophic guild structure of the fish community in the Georges Bank region. ICES J Mar Sci 57:723–730
Gray AE, Mulligan TJ, Hannah RW (1997) Food habits, occurrence, and population structure of the bat ray, Myliobatis californica, in Humboldt Bay, California. Environ Biol Fish 49:227–238
Hobson KA (1993) Trophic relationships among high Arctic seabirds: insights from tissue-dependent stable-isotope models. Mar Ecol Prog Ser 95:7–18
Holden MJ, Tucker RN (1974) The food of Raja clavata Linnaeus 1758, Raja montagui Fowler 1910, Raja naevus Mϋller and Henle 1841, and Raja brachyura LaFont 1873 in British waters. J Cons Inst Explor Mer 35:189–193
Hobson KA, Welch HE (1992) Determination of trophic relationships within a high Arctic marine food web using δ13C and δ15N analysis. Mar Ecol Prog Ser 84:9–18
Jennings S (2005) Size-based analyses of aquatic food webs. In: Belgrano A, Scharler UM, Dunne J, Ulanowicz RE (eds) Aquatic food webs. Oxford University Press, Oxford, pp 86–97
Lasiak TA (1982) Structural and functional aspects of the surf zone fish community in the Eastern Cape. PhD dissertation, University of Port Elizabeth, Port Elizabeth, South Africa
Last PR, Yearsley GK (2002) Zoogeography and relationships of Australasian skates (Chondrichthyes: Rajidae). J Biogeogr 29:1627–1641
Link JS, Garrison LP, Almeida FP (2002) Ecological interactions between elasmobranchs and groundfish species on the Northeastern U.S. Continental Shelf. I. Evaluating predation. N Am J Fish Manage 22:550–562
Macpherson E, Roel BA (1987) Trophic relationships in the demersal fish community off Namibia. In: Payne AIL, Gulland JA, Brink KH (eds) The Benguela and comparable ecosystems. S Afr J Mar Sci 5:585–596
Mayo RK, Fogarty MJ, Serchuk FM (1992) Aggregate fish biomass and yield on Georges Bank, 1960–87. J Northwest Atl Fish Sci 14:59–78
McEachran JD, Miyake T (1990) Zoogeography and bathymetry of skates (Chondrichthyes, Rajoidei). In: Pratt HL, Gruber SH, Taniuchi T (eds) Elasmobranchs as living resources: advances in the biology, ecology, systematics, and the status of the fisheries. NOAA Tech Rept 90, pp 305–326
Mecklenberg CW, Mecklenberg TA, Thorsteinson LK (2002) Fishes of Alaska. American Fisheries Society, Bethesda, MD, USA, 1037 pp
Menni RC, Stehmann MFW (2000) Distribution, environment, and biology of batoid fishes off Argentina, Uruguay, and Brazil. A review. Rev Mus Argentino Cienc Nat 2:69–109
Mohan MV, Sankaran TM (1988) Two new indices for stomach content analysis of fishes. J Fish Biol 33:289–292
Morato T, Solà E, Grós MP (2003) Diets of thornback ray (Raja clavata) and tope shark (Galeorhinus galeus) in the bottom longline fishery of the Azores, northeastern Atlantic. Fish Bull 101:590–602
Murawski SA (1991) Can we manage our multispecies fisheries? Fisheries 16:5–13
Muto EY, Soares LSH, Goitein R (2001) Food resource utilization of the skates Rioraja agassizii (Mϋller and Henle, 1841) and Psammobatis extenta (Garman, 1913) on the continental shelf off Ubatuba, south-eastern Brazil. Brazil J Biol 61:217–238
Nyssen F, Brey T, Lepoint G, Bouquegneau JM, De Broyer C, Dauby P (2002) A stable isotope approach to the eastern Weddell Sea trophic web: focus on benthic amphipods. Polar Biol 25:280–287
Orlov AM (1998) The diets and feeding habits of some deep-water benthic skates (Rajidae) in the Pacific waters off the northern Kuril Islands and southeastern Kamchatka. Alaska Fish Res Bull 5:1–17
Orlov AM (2003) Diets, feeding habits, and trophic relations of six deep-benthic skates (Rajidae) in the western Bering Sea. J Ichthyol Aquat Biol 7:45–59
Orlov AM (2004) Trophic interrelationships in predatory fishes of Pacific waters circumambient the northern Kuril Islands and southeastern Kamchatka. Hydrobiol J 40:106–123
Pauly D, Christensen V (1995) Primary production required to sustain global fisheries. Nature 374:255–257
Pauly D, Trites AW, Capuli E, Christensen V (1998) Diet composition and trophic levels of marine mammals. ICES J Mar Sci 55:467–488
Pinkas LM, Oliphant S, Iverson ILK (1971) Food habits of albacore, bluefin tuna and Bonito in Californian waters. Calif Fish Game 152:1–105
Quiniou L, Andriamirado GR (1979) Variations of the diet of three species of rays from Douarnenez Bay (Raja montagui Fowler, 1910; Raja brachyura Lafont, 1873; Raja clavata L., 1758). Cybium 3:27–39
Robinson HJ, Cailliet GM, Ebert DA (2007) Food habits of the longnose skate, Raja rhina (Jordan and Gilbert, 1880), in central California waters. Environ Biol Fish (this volume)
Rogers SI, Clarke KR, Reynolds JD (1999) The taxonomic distinctness of coastal bottom-dwelling fish communities of the North-east Atlantic. J Anim Ecol 68:769–782
Root RB (1967) The niche exploitation pattern of the blue-gray gnatcatcher. Ecol Monogr 37:317–350
Ross ST (1986) Resource partitioning in fish assemblages: a review of field studies. Copeia 2:352–368
Rossuow GJ (1983) The biology of the lesser sandshark. Rhinobatos annulatus in Algoa Bay with notes on other elasmobranchs. PhD dissertation, University of Port Elizabeth, Port Elizabeth, South Africa
Sanger GA (1987) Trophic levels and trophic relationships of seabirds in the Gulf of Alaska. In: Croxall JP (ed) Seabirds: feeding ecology and role in marine ecosystems. Cambridge University Press, Cambridge, pp 229–257
Smale MJ, Cowley PD (1992) The feeding ecology of skates (Batoidea: Rajidae) off the Cape South Coast, South Africa. S Afr J Mar Sci 12:823–834
Smith JW, Merriner JV (1985) Food habits and feeding behavior of the cownose ray, Rhinoptera bonasus, in Lower Chesapeake Bay. Estuaries 8:305–310
Treloar MA, Stevens JD, Laurenson LJB (2007) Dietary comparisons of seven species of skates (Rajidae) in southeastern Australian waters. Environ Biol Fish (this volume)
VanBlaircom GR (1982) Experimental analyses of structural regulation in a marine sand community exposed to oceanic swell. Ecol Monogr 52:283–305
Volger R, Milessi AC, Quinones RA (2003) Trophic ecology of Squatina guggenheim on the continental shelf off Uruguay and Northern Argentina. J Fish Biol 62:1254–1267
Yeon IJ, Hong SH, Cha HK, Kim ST (1999) Feeding habits of Raja pulchra in the Yellow Sea. Bull Nat’l Fish Res Dev Inst Korea 57:1–11
Yoklavich MM, Greene HG, Cailliet GM, Sullivan DE, Lea RN, Love MS et al (2000) Habitat associations of deep-water rockfishes in a submarine canyon: an example of a natural refuge. Fish Bull 98:625–641
Acknowledgments
We thank Simon Brown, Pacific Shark Research Center (PSRC), Moss Landing Marine Laboratories (MLML), for his help in summarizing the articles used in this paper and inputting data, and Joan Parker and the library staff at MLML for their invaluable help in obtaining literature. Funding for this research was provided by NOAA/NMFS to the National Shark Research Consortium and PSRC, and in part by the National Sea Grant College Program of the U.S. Department of Commerce’s National Oceanic and Atmospheric Administration under NOAA Grant no. NA04OAR4170038, project number R/F-199, through the California Sea Grant College Program and in part by the California State Resources Agency.
Author information
Authors and Affiliations
Corresponding author
Appendix I
Appendix I
List of all skate diet references used for this study.
-
Ajayi TO (1982) Food and feeding habits of Raja species (Batoidei) in Carmarthen Bay, Bristol Channel. J Mar Biol Assoc UK 62:215–223
-
Berestovsky EG (1989) Feeding in the Skates, Raja radiata and Raja fyllae, in the Barents and Norwegian Seas. Voprosy Ikhtiologii 6:994–1002
-
Bjelland O, Bergstad OA, Skjaeraasen JE, Meland K (2000) Trophic ecology of deep-water fishes associated with the continental slope of the eastern Norwegian Sea. Sarsia 85:101–117
-
Bowman RE, Stillwell CE, Michaels WL, Grosslein MD (2000) Food of northwest Atlantic fishes and two common species of squid. NOAA Tech Memo NMFS-NE-155
-
Braccini JM, Perez JE (2005) Feeding habits of the sandskate Psammobatis extenta (Garman, 1913): sources of variation in dietary composition. Marine Freshw Res 56:395–403
-
Brickle P, Laptihovsky V, Pompert J, Bishop A (2003) Ontogenetic changes in the feeding habits and dietary overlap between three abundant rajid species on the Falkland Island’s shelf. J Mar Biol Assoc UK 83:1119–1125
-
Capapé C (1975) Contribution a la biologie des Rajidae des cotes Tunisiennes. 4. Raja clavata (Linne 1758): regime alimentaire. Ann Inst Michel Pacha 8:16–32
-
Capapé C (1976) Contribution a la biologie des Rajidae de cotes tunisiennes. 8. Raja melitensis Clark, 1926, Regime alimentaire. Arch Inst Pasteur Tunis 53:39–45
-
Capapé C (1977) Contribution a la biologie des Rajidae des cotes tunisiennes. 12. Raja alba Lacepede, 1803, Regime alimentaire. Arch Inst Pasteur Tunis 54:85–95
-
Capapé C, Azouz A (1975) Etude du regime alimentaire de deux raies communes dans le golfe de Tunis: Raja miraletus Linne, 1758 et R. radula, Delaroche, 1809. Arch Inst Pasteur Tunis 52:233–250
-
Chuchukalo VI, Napazakov VV (2002) Feeding and trophic status of abundant skate species (Rajidae) of the western Bering Sea. Izvestiya TINRO 130:422–428
-
Dolgov A (2005) Feeding and food consumption by the Barents Sea skates. J Northw Atl Fish Sci 35:496–503
-
DuBuit MJ (1972) The role of geographical and seasonal factors in the diet of R. naevus and R. fullonica. Trav Lab Biol Halieutique, Rennes 6:35–50
-
Ebert DA, Cowley PD, Compagno LJV (1991) A preliminary investigation of the feeding ecology of skates (Batoidea: Rajidae) off the west coast of southern Africa. S Afr J Mar Sci 10:71–81
-
Ellis JR, Pawson MG, Shackley SE (1996) The comparative feeding ecology of six species of shark and four species of ray (Elasmobranchii) in the North-East Atlantic. J Mar Biol Assoc UK 76:89–106
-
Ezzat A, Abd El-Aziz SH, El-Charabawy MM, Hussein MD (1987) The food of Raja miraletus Linnaeus 1758 in Mediterranean waters off Alexandria. Bull Inst Oceanogr Fish. Cairo 13:59–74
-
Fitz ES, Daiber FC (1963) An introduction to the biology of Raja eglanteria Bosc 1802 and Raja erinacea Mitchill 1825 as they occur in Delaware Bay. Bull Bingham Oceanogr Collection 18:69–96
-
Glubokov AI, Orlov AM (2000) Some morphophysiological indices and feeding peculiarities of the Aleutian skate Bathyraja aleutica from the western Bering Sea. Russian Federal Research Institute of Fisheries and Oceanography 1:126–149
-
Gordon JDM, Duncan JAR (1989) A note on the distribution and diet of deep-water rays (Rajidae) in an area of the Rockall Trough. J Mar Biol Assoc UK 69:655–658
-
Hacunda JS (1981) Trophic relationships among demersal fishes in a coastal area of the Gulf of Maine. Fish Bull 79:775–788
-
Holden MJ, Tucker RN (1974) The food of Raja clavata Linnaeus 1758, Raja montagui Fowler 1910, Raja naevus Mϋller and Henle 1841, and Raja brachyura LaFont 1873 in British waters. J Cons Inst Explor Mer 35:189–193
-
Koen Alonso M, Crespo EA, Garcia NA, Pedraza SN, Mariotti PA, Beron Vera B, Mora NJ (2001) Food habits of Dipturus chilensis (Pisces: Rajidae) off Patagonia, Argentia. ICES J Mar Sci 58:288–297
-
Link JS, Almeida FP (2000) An overview and history of the food web dynamics program of the Northeast Fisheries Science Center, Woods Hole, Massachusetts. NOAA Tech Memo NMFS-NE-159
-
Lucifora LO, Valero JL, Bremec CS, Lasta ML (2000) Feeding habits and prey selection by the skate Dipturus chilensis (Elasmobranchii: Rajidae) from the south-western Atlantic. J Mar Biol Assoc UK 80:953–954
-
Mabragana E, Giberto DA, Bremec CS (2005) Feeding ecology of Bathyraja macloviana (Rajiformes: Arhynchobatidae): a polychaete-feeding skate from the south-west Atlantic. Sci Mar 69:405–413
-
Monteiro-Marques V, Ré P (1978) Regime alimentaire de quelques Rajidae des cotes Portugaises. Arq Mus Bocage 6:1–8
-
Morato T, Solà E, Grós MP, Menezes G (2003) Diets of thornback ray (Raja clavata) and tope shark (Galeorhinus galeus) in the bottom longline fishery of the Azores, northeastern Atlantic. Fish Bull 101:590–602
-
Muto EY, Soares LSH, Goitein R (2001) Food resource utilization of the skates Rioraja agassizii (Mueller and Henle, 1841) and Psammobatis extenta (Garman, 1913) on the continental shelf off Ubatuba, south-eastern Brazil. Brazilian J Biol 61:217–238
-
Olaso I, Sanchez F, Rodriguez-Cabello C, Velasco F (2002) The feeding behaviour of some demersal fish species in response to artificial discarding. Sci Mar 66:301–311
-
Orlov AM (1998) The diets and feeding habits of some deep-water benthic skates (Rajidae) in the Pacific waters off the northern Kuril Islands and southeastern Kamchatka. Alaska Fish Res Bull 5:1–17
-
Orlov AM (2003) Diets, feeding habits, and trophic relations of six deep-benthic skates (Rajidae) in the western Bering Sea. J Ichthyol Aquat Biol 7:45–59
-
Packer DB, Zetlin CA, Vitaliano JJ (2003) Essential Fish Habitat Source Document: Thorny skate, Amblyraja radiata, life history and habitat characteristics. NOAA Tech Mem NMFS-ME-178:39
-
Pederson SA (1995) Feeding habits of the starry ray (Raja radiata) in West Greenland waters. ICES J Mar Sci 52:43–53
-
Quiniou L, Andriamirado GR (1979) Variations of the diet of three species of rays from Douarnenez Bay (Raja montagui Fowler, 1910; Raja brachyura Lafont, 1873; Raja clavata L., 1758). Cybium 3:27–39
-
Rinewalt CS, Ebert DA, Cailliet GM (2007) The feeding habits of the sandpaper skate, Bathyraja kincaidii (Garman, 1908) in central California: seasonal variation in diet linked to oceanographic conditions. Environ Biol Fish (this volume)
-
Robinson HJ, Cailliet GM, Ebert DA (2007) Food habits of the longnose skate, Raja rhina (Jordan and Gilbert, 1880), in central California waters. Environ Biol Fish (this volume)
-
Rousset J (1987) Feeding of the ray Raja microocellata (Montagu, 1818) in Bertheaume Bight (Brittany). Cah Bio Mar, Paris 28:199–206
-
Serrano A, Velasco F, Olaso I (2003) Macrobenthic crustaceans in the diet of demersal fish in the Bay of Biscay in relation to abundance in the environment. Sarsia 88:36–48
-
Scenna LB, García de la Rosa SB, Díaz de Astarloa JM (2006) Trophic ecology of the Patagonian skate, Bathyraja macloviana, on the Argentine continental shelf. ICES J Mar Sci 63:867–874
-
Skjaeraasen JE, Bergstad OA (2000) Distribution and feeding ecology of Raja radiata in the northeastern North Sea and Skagerrak (Norwegian Deep). ICES J Mar Sci 57:1249–1260
-
Smale MJ, Cowley PD (1992) The feeding ecology of skates (Batoidea: Rajidae) off the Cape South Coast, South Africa. S Afr J Mar Sci 12:823–834
-
Soares, LSH, de Moraes Vazzoler AEA, Roberto Correa A (1999) Diel feeding chronology of the skate Raja agassizii (Mϋller and Henle) (Pisces, Elasmobranchii) on the continental shelf off Ubatuba, southeastern Brazil. Rev Bras Zool 16:201–212
-
Templeman W (1982) Stomach contents of the thorny skate, Raja radiata, from the Northwest Atlantic. J Northw Atl Fish Sci 3:123–126
-
Wakefield WW (1984) Feeding relationships within assemblages of nearshore and mid-continental shelf benthic fishes off Oregon. Unpubl. Masters Thesis, Oregon State University
-
Yang M-S (2003) Food habits of the important groundfishes in the Aleutian Islands in 1994 and 1997. Alaska Fish Sci Cent, Natl Mar Fish Serv AFSC Progress Rept 2003–07:231
-
Yeon IJ, Hong SH, Cha HK, Kim ST (1999) Feeding habits of Raja pulchra in the Yellow Sea. Bull Nat’l Fish Res Dev Inst Korea 57:1–11
Rights and permissions
About this article
Cite this article
Ebert, D.A., Bizzarro, J.J. Standardized diet compositions and trophic levels of skates (Chondrichthyes: Rajiformes: Rajoidei). Environ Biol Fish 80, 221–237 (2007). https://doi.org/10.1007/s10641-007-9227-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-007-9227-4