Summary
Introduction New therapeutic strategies combining axitinib and immune checkpoint blockers are ongoing in metastatic renal cell carcinoma (mRCC). These strategies do not consider the pharmacokinetic variability of axitinib. We aimed to describe the risk of axitinib under-exposure using routine pharmacologic therapeutic monitoring (PTM). Methods We analyzed axitinib dosage in nine patients with mRCC. Routine axitinib concentration measurements were centralized at Henri Mondor University Hospital (Créteil, France) using a validated method. The primary objective was to describe the evolution of Cmax dosages (1 to 6 h after oral intake) during routine axitinib titration. Results Nine patients with available Cmax axitinib dosages were included. Four out of the nine patients had axitinib titration and Cmax dosages were performed before and after titration. All but one corrected their plasma axitinib exposure after titration, suggesting of a titration success. The last patient was monitored in the Henri Mondor Hospital routine PTM program and a pharmacokinetic profiling was performed after controlled oral intake. Results suggested a poor axitinib absorption. This patient experienced early tumor progression as best response. Conclusion We report a patient with significant axitinib under-exposure, possibly due to a poor absorption. PTM should be evaluated and considered in drug developments evaluating combination therapies based on axitinib.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Axitinib is a tyrosine kinase inhibitor approved in the second-line treatment of metastatic renal cell carcinoma (mRCC). Its pharmacokinetics is characterized by rapid absorption (peak plasma concentrations at 2–6 h after intake), short half-life (t1/2 = 2–5 h), and metabolic elimination through cytochrome metabolism. Moreover, axitinib presents an important inter-individual pharmacokinetic variability, including the risk of under-exposure. Therefore, its dosing may be titrated according to clinical tolerance, mainly hypertension which correlates with clinical outcomes [1]. Importantly, this drug is currently investigated in combination with anti-PD1/anti-PDL1 antibodies in mRCC with promising results [2]. Pharmacologic therapeutic monitoring (PTM) could support dose adjustments and drug exposure in such combinations.
Fifteen patients treated in 3 University Hospital in Paris between 2014 and 2016 had available routine axitinib dosages. Median age was 65 years [range: 42.1–86.2]. All dosages were centralized in the Pharmacology Laboratory of Henri Mondor University Hospital. Axitinib quantification used a validated liquid chromatography-tandem mass spectrometry method. Internal quality controls were regularly performed. Overall, concentrations ranged between 1 to 500 ng/mL (lower limit of quantification: 1 ng/mL). Six patients had very low/undetectable trough concentrations. In view of the short half-life of axitinib, our pharmacokinetic analysis focused on Cmax dosages which is usually performed 1 to 6 h after oral intake. Nine patients had Cmax measurements.
According to published data in patients exposed to axitinib titration, mean Cmax was 28.6 ng/ml (CI95% [20.5–39.9]) [2]. Five patients had Cmax concentration ≥ 20.5 ng/mL at 5 mg bid while 4 patients had Cmax concentrations below 20.5 ng/mL. Patients with Cmax < 20.5 ng/mL had no axitinib-related hypertension and were subsequently managed by axitinib titration on the basis of clinical tolerance, according to French and international routine guidelines. Cmax measurements were performed at each axitinib dose modification.
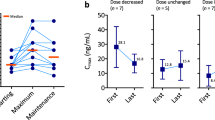
Three out of the four patients with Cmax < 20.5 ng/mL had successful Cmax increase within range after titration. The last patient (Patient D, Fig. 1a) remained at undetectable plasma axitinib Cmax concentration.
Axitinib exposure at baseline and after titration. a. Titration efficiency after dose escalation. For each patient (A to D), axitinib maximal concentration (Cmax) at each dose level before (white bars) and after (grey bars) dose increase. N + N indicates axitinib bid intakes for each dose level. b. Pharmacokinetic analysis for patient D. Axitinib doses were 10 mg bid for black dots (H12 (trough concentration), H1, H2), and 5 mg and 7 mg bid for grey dots (H4:45 and H6, respectively)
This 43 years-old woman, with ECOG performance status 0, without any previous medical or surgical condition, was diagnosed of mRCC and was initially treated with frontline sunitinib. At progression seven months later, she received axitinib 5 mg bid and was subsequently monitored in the Henri Mondor Hospital routine PTM program. Good compliance was confirmed by interview and pill count. No potential drug interaction was found, with the exception of active tobacco use (one pack per day). She did not use estroprogestative contraception. Figure 1b summarizes all dosages performed for this patient. Two dosages were performed during titration, revealing undetectable plasma axitinib concentrations. In-hospital controlled oral intake was realized at 10 mg bid (maximum recommended dose). Dosages were performed in fasting condition before oral intake, and after one and two hours. We observed only a very slight concentration increase, which remained out of the published range ([20.5–39.9] ng/mL [2]). This pattern suggested a poor absorption of axitinib and/or rapid metabolizing phenotype. This patient had early progressive disease as best response.
We report here one patient with titration failure, implying concerns about a potential subsequent lack of antitumor efficacy. Axitinib is metabolized primarily in the liver by cytochrome P450 (CYP) 3A4/5 and, to a lesser extent (<10%), by CYP1A2, CYP2C19, and uridine diphosphate glucuronosyltransferase (UGT) 1A1. One previous case of poor axitinib exposure was reported and suggested involvement of intestinal CYP3A4 into axitinib poor absorption, corrected with the use of CYP3A4 inhibitor [3]. Poor absorption related to intestinal metabolism is probably the main cause of low exposure in the present case but active smoking, as a CYP1A2 inductor may also be involved.
Pharmacokinetic considerations appear to be of interest in axitinib-treated mRCC patients and should be considered in further development of combination therapies with anti-PD1/PD-L1. Of note, axitinib doses are for now fixed at 5 mg bid in combination with avelumab [2], which may result in a higher risk of axitinib under-exposure, and a potential lack of efficacy. Further pharmacokinetic studies are warranted to confirm these data and prospectively validate PTM for axitinib.
References
Rini BI, Melichar B, Fishman MN et al (2015) Axitinib dose titration: analyses of exposure, blood pressure and clinical response from a randomized phase II study in metastatic renal cell carcinoma. Ann Oncol 26:1372–1377
Choueiri TK, Larkin J, Oya M et al (2018) Preliminary results for avelumab plus axitinib as first-line therapy in patients with advanced clear-cell renal-cell carcinoma (JAVELIN renal 100): an open-label, dose-finding and dose-expansion, phase 1b trial. Lancet Oncol 19:451–460
Lubberman FJE, van Erp NP, Ter Heine R et al (2017) Boosting axitinib exposure with a CYP3A4 inhibitor, making axitinib treatment personal. Acta Oncol 56(9):1238–1240
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No individual author has conflict of interest in the setting of this article.
Ethical approval
Drug dosage is a standard procedure in France, approved by National Health Institute. No drug dosing modification were performed on the basis of drug dosage. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. All patients’ data have been managed in compliance with French law on protection of personal data.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A. Hulin and Benoit Rousseau participated equally to the work.
Rights and permissions
About this article
Cite this article
Beinse, G., Hulin, A. & Rousseau, B. Axitinib pharmacologic therapeutic monitoring reveals severe under-exposure despite titration in patients with metastatic renal cell carcinoma. Invest New Drugs 37, 1289–1291 (2019). https://doi.org/10.1007/s10637-019-00743-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-019-00743-1