Summary
Standard therapy for advanced or metastatic non-small cell lung cancer (NSCLC) has primarily consisted of traditional cytotoxic chemotherapy, although use of targeted therapies has been approved in specific settings. Antiangiogenic agents represent a promising therapeutic strategy for treatment of advanced NSCLC. Bevacizumab is currently approved when given in combination with first-line platinum-based therapy in selected patients with nonsquamous NSCLC. Bevacizumab may also provide benefit in other clinical settings, as a part of a combination or maintenance strategy. Other antiangiogenic agents under development, including multi-targeted kinase inhibitors (MTKIs) and antibody-based agents, have exhibited mixed results in the NSCLC population. Published efficacy and safety data from clinical trials for antiangiogenic agents are reviewed, with an emphasis on novel agents and novel settings for established agents. Identification of biomarkers associated with improved efficacy may help select patients likely to receive the most benefit from these agents and may improve outcomes through development of personalized therapeutic strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Non-small cell lung cancer (NSCLC) accounts for approximately 85% of lung cancer diagnoses [1] and the majority of lung cancer deaths worldwide [1, 2]. Recommended systemic treatment options for patients with advanced or metastatic NSCLC have historically included cytotoxic chemotherapy, with targeted agents directed against the vascular endothelial growth factor (VEGF) and the epidermal growth factor receptor (EGFR) representing more recent therapeutic additions for use in specific treatment settings or populations. Maintenance monotherapy with a chemotherapeutic or targeted agent has also emerged as an important consideration following the use of first-line chemotherapy [3].
With respect to cytotoxic chemotherapy, various platinum-based doublets have demonstrated comparable efficacy in terms of palliative benefits and improvements in survival when used in the treatment of patients with advanced NSCLC [3, 4]. Although no single platinum-based doublet has been considered superior in the overall advanced unselected NSCLC population, histology has emerged as an important consideration for individualizing regimen selection. The benefit of histological screening is particularly evident in the improved activity seen with the antifolate pemetrexed (Alimta®, Eli Lilly and Company) when used in patients with adenocarcinoma and large-cell carcinoma versus squamous cell carcinoma [5].
Over the past decade, targeted agents have emerged as novel therapeutic options for NSCLC, both as a part of combination therapies and as single agents. Bevacizumab (Avastin®, Genentech), a monoclonal antibody that targets VEGF, is the only United States (US) Food and Drug Administration (FDA)-approved adjunct to first-line platinum-based chemotherapy for NSCLC. Bevacizumab is specifically indicated for patients with nonsquamous tumors [6] and is recommended for patients with a good performance status and no recent hemoptysis [3]. The EGFR tyrosine kinase inhibitors (TKIs) erlotinib (Tarceva®, Genentech) and gefitinib (Iressa™, AstraZeneca) also have FDA-approved indications for use as monotherapy in patients with chemotherapy-pretreated advanced NSCLC [7, 8]. However, due to inconsistent benefit observed with gefitinib, its indication in the United States is limited to patients who are or have previously benefited from gefitinib therapy [7]. This article provides an overview of the rationale for selection of VEGF, platelet-derived growth factor (PDGF), and fibroblast growth factor (FGF) pathway signaling as a therapeutic target. Past and ongoing trials of bevacizumab and other developmental antiangiogenic agents for the treatment of advanced NSCLC are summarized.
Rationale for antiangiogenic VEGF-targeted therapy
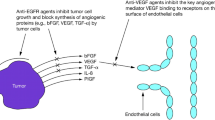
Angiogenesis, the development of new blood vessels, is a critical component of tumor development and progression, as this process provides access to the nutrients and oxygen needed for tumor growth [9, 10]. This is a key therapeutic target because in order to grow larger than a few millimeters in diameter, solid tumors must develop a vascular supply [11]. The process of angiogenesis relies on a multitude of signaling cascades and angiogenic factors [12–15]. Of the many proangiogenic signaling molecules, VEGF (or VEGF-A) is the most widely studied to date [14, 15]. VEGF-A belongs to a larger family of 6 secreted glycoproteins that also includes VEGF-B, -C, and -D, and placenta growth factor (PlGF)-1 and −2 [16, 17]. Although the spatiotemporal expression of these isoforms varies, all are involved in angiogenesis and/or lymphangiogenesis in normal human development and in pathological states [16].
Binding of VEGF family ligands to 3 cognate receptor tyrosine kinases (VEGF receptor-1 [VEGFR-1], VEGFR-2, and VEGFR-3) results in the activation of numerous signaling factors that ultimately promote cellular migration, survival, and proliferation (Fig. 1) [12, 18]. VEGF-A has been shown to activate both VEGFR-1 and VEGFR-2, two highly similar receptors that have distinct roles in angiogenesis. VEGFR-1 is required for proper formation of the vasculature during early embryonic development and can also bind VEGF-B and PlGF [16, 17]. This receptor is expressed in the vasculature and in other cell types and can serve as both a positive and negative regulator of angiogenesis [17]. VEGFR-2 is primarily expressed on the vascular endothelium, where it plays a substantial role in VEGF–A-mediated angiogenesis, although VEGF-C and VEGF-D can also serve as ligands. VEGFR-2 signaling mediates microvascular permeability and proliferation, migration, and survival of endothelial cells [16, 17]. VEGFR-3 preferentially binds VEGF-C and VEGF-D. VEGFR-3 is expressed in the embryonic vasculature, but throughout development and in the adult, its expression is limited to lymphatic endothelial cells. VEGFR-3 is thought to be involved in remodeling of primary vascular networks during embryogenesis and in lymphangiogenesis in the adult [16, 17]. Coordination of VEGFR activity with other signaling pathways, such as the PDGF and FGF pathways, may be required to support tumor angiogenesis. These pathways appear to function in a synergistic manner to promote the formation of new vessels that provide nutrients and oxygen to tumors, in addition to promoting dissemination of tumor cells [19].
The VEGF pathway and sites of action for inhibitors. Binding of VEGF family ligands to their cognate receptors, VEGFR tyrosine kinases (VEGFR-1, VEGFR-2, and VEGFR-3), results in the activation of numerous signaling factors that ultimately promote cellular migration, survival, and proliferation. The sites of action for the antiangiogenic inhibitors discussed herein are also illustrated. The VEGF pathway and sites of action for inhibitors. Used with permission of Society for Experimental Biology and Medicine, from Antiangiogenic therapy in lung cancer: focus on vascular endothelial growth factor pathway by Korpanty G, Smyth E, Sullivan LA, Brekken RA, Carney DN. Exp Biol Med (Maywood) 235:3–9 (2010); permission conveyed through Copyright Clearance Center, Inc
The VEGF signaling axis is now a validated therapeutic target in NSCLC. Early preclinical models have illustrated the ability of VEGF inhibition to slow tumor growth and angiogenesis [20]. Observations supporting high levels of VEGF and VEGFR expression or upregulation in human malignancies have further supported research efforts to develop anti-VEGF/VEGFR therapies [21–23]. Inhibitors targeting the VEGF pathway, as well as other pathways involved in angiogenesis could potentially lead to substantial improvements in outcomes for patients with NSCLC.
Bevacizumab
Bevacizumab is currently the best characterized and only approved VEGF inhibitor in clinical use for NSCLC. Approval of bevacizumab for the treatment of NSCLC was based on the results of 2 phase III trials combining bevacizumab with first-line chemotherapy. In a phase II trial the combination of bevacizumab with cytotoxic chemotherapy increased response rate (RR), time to progression (TTP), and overall survival (OS) compared with chemotherapy alone in patients with advanced nonsquamous NSCLC [24].
In the phase III E4599 registration trial (N = 878) in patients with advanced NSCLC receiving front-line therapy, a significant OS benefit was shown for bevacizumab (15 mg/kg) plus paclitaxel/carboplatin followed by maintenance bevacizumab until disease progression versus standard chemotherapy alone [25]. Significant improvements in OS (12.3 vs 10.3 months; hazard ratio [HR], 0.79; 95% confidence interval [CI], 0.67–0.92; P = 0.003) and progression-free survival (PFS, 6.2 vs 4.5 months; HR, 0.66; 95% CI, 0.57–0.77; P < 0.001) were observed for the bevacizumab arm, as well as an objective RR that was more than twice that of chemotherapy alone (35% vs 15%; P < 0.001) [25]. However, bevacizumab treatment was associated with significant increases in grade ≥3 hematologic toxicities, including neutropenia (25.5% vs 16.8%), febrile neutropenia (5.2% vs 2.0%), and thrombocytopenia (1.6% vs 0.2%), as well as grade ≥3 hypertension (7.0% vs 0.7%), proteinuria (3.1% vs 0%), bleeding (4.4% vs 0.7%), hyponatremia (3.5% vs 1.1%), rash (2.3% vs 0.5%), and headache (3.0% vs 0.5%) over chemotherapy alone (P < 0.05). In addition, 5 of 15 treatment-related deaths in the bevacizumab arm were attributed to pulmonary hemorrhage [25].
In the subsequent AVAiL trial (N = 1,043), bevacizumab was administered at 1 of 2 doses (7.5 mg/kg or 15 mg/kg) and added to a first-line doublet of gemcitabine/cisplatin in chemotherapy-naive patients with advanced NSCLC [26]. Following first-line therapy, patients in the bevacizumab arms continued to receive bevacizumab monotherapy and patients in the placebo arm continued to receive placebo until disease progression, with no crossover to bevacizumab allowed. In addition to patients with squamous tumors, patients with tumors invading or adjacent to major blood vessels were excluded from the trial. The AVAiL trial found that, independent of dose, bevacizumab was associated with significant improvements in the objective RR (20.1%; 34.1%, P < 0.0001; and 30.4%, P = 0.0023 for placebo and bevacizumab 7.5 mg/kg and 15 mg/kg, respectively) and PFS (HR, 0.75; 95% CI, 0.62–0.91; P = 0.003 for 7.5 mg/kg and HR, 0.82; 95% CI, 0.68–0.98; P = 0.03 for 15 mg/kg) [26]. However, in contrast to E4599, no OS benefit was evident in either of the bevacizumab groups [27]. Addition of bevacizumab to chemotherapy in the AVAiL trial modestly increased the incidences of grade ≥3 neutropenia (placebo, 32%; 7.5 mg/kg, 40%; 15 mg/kg, 36%), vomiting (4%; 7%; 9%), hypertension (2%; 6%; 9%), proteinuria (0%; <1%; 1%), and bleeding (2%; 4%; 4%) over chemotherapy alone [26]. The incidences of fatal pulmonary hemorrhage were approximately 1% in each bevacizumab arm and 0.3% in the placebo arm [26].
The phase IV SAiL trial (MO19390, N = 2,212) assessed the efficacy and safety of the addition of bevacizumab to standard chemotherapy regimens, primarily carboplatin doublets (49%) and cisplatin doublets (38%) [28]. Similar efficacy was generally observed across chemotherapy regimens, with the exception of a lower median OS rate for patients who received non-platinum doublets (8.1 months; 95% CI, 5.7–13.0) or monotherapy (9.4 months; 95% CI, 5.3–14.7), who had slightly lower median OS than patients receiving other carboplatin doublet (14.3 months; 95% CI, 13.2–15.6) or cisplatin doublet (14.7 months; 95% CI, 13.7–16.0) regimens. No new safety signals were identified, and grade ≥3 toxicities included thromboembolism (8%), hypertension (6%), bleeding (4%), proteinuria (3%), pulmonary hemorrhage, pulmonary embolism, epistaxis, neutropenia, febrile neutropenia, and deep vein thrombosis (1% each). Thromboembolism (1%) and bleeding (1%) represented the most common cause of treatment-associated deaths [28]. Another observational phase IV clinical trial (ARIES) evaluated the safety and efficacy of bevacizumab plus chemotherapy as first-line therapy in patients with locally advanced nonsquamous NSCLC [29]. Median PFS and OS were 6.7 months and 13.6 months overall (N = 1,970); 6.8 months and 12.6 months among patients ≥70 years of age (n = 650), 5.8 months and 8.1 months among patients with performance status ≥2 (n = 182). Incidence of severe pulmonary hemorrhage (observed in 0.8% overall, 3% of those ≥70 years of age, and 1% of those with performance status ≥2, respectively), grade ≥3 bleeding excluding pulmonary hemorrhage (3%; 3%; 4%), arterial thromboembolism (2%; 3%; 3%), and grade ≥3 central nervous system (CNS) bleeding (0.1%; 0%; 0.5%) were reported. Among patients with CNS metastases (n = 150), median PFS and OS were 6.0 months and 11.7 months, respectively, and no grade ≥3 CNS bleeding was reported in this subgroup.
In addition to these findings, results are also available from a prospective biomarker study of the E4599 trial [30]. Patients with high plasma levels of VEGF were shown to have a higher probability of response to chemotherapy plus bevacizumab compared with chemotherapy alone (33% vs 7%; P = 0.01). However, baseline VEGF levels were not predictive of the OS benefit observed with bevacizumab. In addition, intracellular adhesion molecule-1 (ICAM-1) levels were predictive of response to chemotherapy alone or with bevacizumab and were also prognostic for survival [30].
A number of additional phase II and III clinical trials of bevacizumab have been completed or are ongoing. Emerging results from these trials may support the use of bevacizumab in previously excluded NSCLC patient populations. For example, although bevacizumab is not recommended for patients with treated CNS metastasis due to the risk of CNS bleeding [6], the phase II PASSPORT trial reported no grade ≥2 CNS hemorrhage events among 106 evaluable patients with treated brain metastases [31].
Combination of bevacizumab with other targeted therapies may also have clinical benefit. In the placebo-controlled BeTa phase III trial of bevacizumab plus erlotinib as second-line therapy, no benefit in median OS was observed with the combination versus erlotinib alone (9.3 vs 9.2 months; HR, 0.97; 95% CI, 0.80–1.18; P = 0.76) [32]. The combination numerically improved the objective RR (13% vs 6%) and median PFS (3.4 vs 1.7 months; HR, 0.62; 95% CI, 0.52–0.75), but statistical significance could not be determined because the study prespecified that the primary endpoint be significant before testing secondary endpoints..
Bevacizumab is used as maintenance therapy based on the E4599 and AVAiL trials, and there are combinations with bevacizumab also being evaluated in this setting. The placebo-controlled ATLAS trial (NCT00257608) is currently evaluating bevacizumab plus erlotinib as maintenance therapy following bevacizumab/chemotherapy for advanced NSCLC without progression. Preliminary data from this trial (n = 768) support a significant PFS benefit with bevacizumab and erlotinib versus bevacizumab and placebo (median, 4.8 vs 3.7 months; HR, 0.722; 95% CI, 0.592–0.881; P = 0.0012) [33]. The phase III POINTBREAK study [34], which will evaluate bevacizumab in combination with paclitaxel/carboplatin as induction treatment, followed by bevacizumab/pemetrexed versus bevacizumab as maintenance therapy in patients with advanced NSCLC, is currently recruiting patients. A phase III trial is currently evaluating adjuvant chemotherapy with and without bevacizumab for surgically resected stage IB-IIA NSCLC [35]. Interim safety data showed no unexpected toxicities; however, the addition of bevacizumab to chemotherapy resulted in significantly increased grade 3/4 hypertension (19.6% vs 2.0% with chemotherapy alone; P < 0.001), proteinuria (3.2% vs 0.7%; P = 0.03), abdominal pain (4.6% vs 0.3%; P = 0.001), and overall grade 3/4 toxicity (84.0% vs 68.0%; P < 0.001) [35]. Together, these studies indicate that bevacizumab may represent an effective therapeutic agent for NSCLC in multiple settings and support the rationale for further study of antiangiogenic therapy in this tumor type.
Investigational antiangiogenic agents for NSCLC
In addition to the VEGF pathway, other signaling pathways, such as those mediated by PDGF and FGF, have also been implicated in angiogenesis [36, 37]. These pathways may also play a role in resistance to VEGF-directed treatment [38–41], suggesting that multitargeted TKIs may potentially offer therapeutic benefit in the treatment of advanced NSCLC. Completed and ongoing trials of investigational antiangiogenic agents under evaluation for their therapeutic potential in NSCLC, including various multitargeted TKIs and developmental antibody-based agents, are discussed herein.
Multitargeted antiangiogenic TKIs
Multitargeted TKIs have emerged as potential therapeutics for NSCLC due to the ability of these agents to target multiple pathways involved in angiogenesis. However, efficacy and safety outcomes for these agents vary substantially, and identification of patients most likely to benefit from these agents remains under investigation.
Sorafenib (Nexavar®, Bayer), inhibits VEGFR-2 and −3 and the PDGF receptor (PDGFR)-β and possesses additional antiproliferative potential through inhibition of (v-raf 1 murine leukemia viral oncogene homolog 1) Raf, stem cell factor receptor (c-kit), and fms-like tyrosine kinase 3 (flt-3) [42]. Despite early promise, phase III trials of sorafenib as an adjunct to first-line chemotherapy for advanced NSCLC have failed to demonstrate prolongation of OS relative to chemotherapy alone. The phase III ESCAPE trial (N = 926) of paclitaxel/carboplatin alone or with sorafenib was terminated at interim analysis due to low likelihood of reaching the primary endpoint and revealed an adverse safety signal among patients with squamous tumors [43]. Per protocol amendment, patients with squamous tumors were subsequently excluded from the phase III NExUS trial of gemcitabine/cisplatin alone or with sorafenib (NCT00449033), but efficacy results were consistent with those of ESCAPE, with no OS benefit in the sorafenib arm [44].
Clinical development of sorafenib for advanced NSCLC is currently focused on patients with chemotherapy-refractory disease, a setting in which activity was suggested in prior clinical studies. In a placebo-controlled phase II discontinuation trial in a population of heavily pretreated patients with NSCLC, sorafenib monotherapy was associated with significant prolongation of PFS (3.6 vs 1.9 months with placebo; P = 0.01) and a significantly higher rate of stable disease (29% vs 5% with placebo; P = 0.002). Grade 3 to 4 toxicities in all patients receiving sorafenib prior to randomization to further sorafenib or placebo included rash/hand-foot syndrome (15%) and fatigue (11%) [45]. In addition, a phase III trial (NCT00863746) of sorafenib monotherapy as third-line or fourth-line therapy for advanced NSCLC is currently underway. Phase II randomized studies have also evaluated the potential benefit of adding sorafenib to pemetrexed [46] or erlotinib [47] but have not met prespecified efficacy measures.
Clearly, the development of a predictive biomarker to identify patients likely to benefit or not benefit from antiangiogenic therapy is the next step. Results of a biomarker analysis from a phase II trial of sorafenib plus erlotinib as first-line treatment for advanced NSCLC suggest a potential role for CD133-positive (CD133+) circulating hematopoietic progenitor cells (HPCs) for predicting response and TTP [48]. This study found that baseline levels of CD133+ HPCs were significantly lower among responders versus nonresponders (P = 0.01), and values lower than the median were correlated with prolonged TTP (P = 0.037). However, baseline CD133+ HPCs were not associated with baseline VEGF levels [48].
Biomarkers predictive of sorafenib response were also addressed in the MD Anderson Cancer Center BATTLE (Biomarker-integrated Approaches of Targeted Therapy for Lung Cancer Elimination) program, which was designed to evaluate the role of predictive biomarkers in guiding the selection of targeted therapy for advanced NSCLC [49, 50]. This adapted randomized study screened 11 biomarkers relevant to the EGFR, Ras/Raf, VEGF, and cyclin D1/retinoid X receptor (RXR) pathways in core biopsy specimens. Biomarker results from patients initially enrolled into the umbrella study determined whether patients would enroll in a clinical trial of sorafenib, vandetanib (Zactima™, AstraZeneca), erlotinib, or erlotinib/bexarotene [49]. The disease control rate at 8 weeks (primary endpoint) was 46% across all 4 regimens, ranging from 33% with vandetanib to 58% with sorafenib [49]. These biomarker results will be further evaluated in the upcoming BATTLE-2 trial (NCT01248247). Sorafenib has also been shown to be associated with a high disease control rate in patients with Kirsten ras sarcoma viral oncogene homolog (KRAS) mutation-positive tumors (61% disease control rate at 8 weeks), but significantly lower disease control rates in patients with EGFR mutation-positive tumors (23% vs 64% for EGFR mutation-negative tumors; P = 0.012) or high EGFR polysomy tumors (27% vs 62% for low-polysomy; P = 0.048) [50]. These results suggest that biomarker-based selection of patients most likely to benefit may lead to improved efficacy of sorafenib.
Sunitinib (Sutent®, Pfizer) is a multikinase inhibitor that targets VEGFR-1, -2, and −3 and PDGFR-α/β, as well as c-kit, flt-3, and the rearranged during transfection (RET) receptor. Data from phase II studies support activity of sunitinib monotherapy in patients with chemotherapy-pretreated advanced NSCLC. Socinski and colleagues evaluated 4-weeks-on/2-weeks-off sunitinib dosing in 63 patients pretreated with 1 or 2 prior chemotherapy regimens (which may have included an EGFR TKI). Sunitinib monotherapy was associated with an objective RR of 11.1% (7/63), a rate of stable disease for ≥8 weeks of 28.6% (18/63), and median PFS and OS of 12.0 weeks and 23.4 weeks, respectively. In this study, which excluded patients at high bleeding risk, the most common toxicities were fatigue/asthenia (29%), pain/myalgia (17%), dyspnea (11%), and nausea/vomiting (10%) [51]. Novello and colleagues evaluated continuous dosing of sunitinib in 47 patients pretreated with 1 or 2 prior chemotherapy regimens, with an objective RR of 2.1% (1/47), a rate of stable disease for ≥8 weeks of 23.4% (11/47), and median PFS and OS of 11.9 weeks and 37.1 weeks, respectively. The most common grade 3 to 4 toxicities in this study were fatigue/asthenia (17%), hypertension (9%), and dyspnea (6%) [52]. Sunitinib monotherapy has also been studied in the first-line setting in 63 elderly patients with advanced nonsquamous NSCLC, reporting an overall RR of 5%, disease control rate of 58%, and median PFS of 3.0 months. The most common treatment-related grade 3/4 toxicity was fatigue (18%) and quality of life worsened during the study [53].
A phase III trial (N = 960) has investigated sunitinib as second-line therapy in combination with erlotinib [54]. Addition of sunitinib to erlotinib resulted in a significant increase in PFS (15.5 vs 8.7 weeks for placebo; HR, 0.807; 95% CI, 0.695–0.937; P = 0.0023), but not OS (9.0 vs 8.5 months; HR, 0.922; 95% CI, 0.797–1.067; P = 0.1388). Treatment-related grade ≥3 toxicities included diarrhea (10.4% vs 1.7% for placebo), rash (8.2% vs 2.5%), decreased appetite (3.4% vs 1.3%), fatigue (3.8% vs 1.9%), and nausea (1.3% vs 0.6%). Interim results from a subset analysis of East Asian patients (n = 103) showed significantly longer OS (not reached vs 9.4 months; P = 0.0042). significantly higher RR (38.5% vs 13.7%; P = 0.0083), and numerically longer PFS (31.2 vs 15.2 weeks; HR, 0.723; 95% CI, 0.451–1.161; P = 0.0889) with sunitinib plus erlotinib versus erlotinib alone, with similar toxicity to that reported in the primary analysis [55]. Additional phase III trials are ongoing in patients with advanced NSCLC to determine the role of sunitinib as maintenance therapy (NCT00693992 [CALGB 30607] and NCT01210053).
BIBF 1120 (Boehringer Ingelheim) targets VEGFR-1, -2, and −3, PDGFR-α/β, FGFR-1, -2, and −3, as well as members of the v-src sarcoma viral oncogene homolog (Src) family and flt-3 [56, 57]. BIBF 1120 has been evaluated in a phase II study involving 73 patients with advanced NSCLC, including patients with an Eastern Cooperative Oncology Group Performance Status (ECOG PS) of 0 to 2 [58]. Median OS was 21.9 weeks overall and 37.7 weeks for the 56 patients with an ECOG PS 0–1, with corresponding median PFS of 6.9 weeks and 11.6 weeks, respectively. Grade 3 to 4 toxicities included gastrointestinal complaints (eg, diarrhea [8.2%], nausea [6.8%], abdominal pain [2.7%], vomiting [2.7%]), anorexia (1.4%), fatigue (1.4%), alanine aminotransferase elevation (9.6%), aspartate aminotransferase elevation (1.4%), and gamma glutamyl transferase elevation (4.1%) [58]. Phase III trials have been initiated to evaluate BIBF 1120 as a component of second-line chemotherapy with docetaxel (LUME-Lung 1; NCT00805194) or pemetrexed (LUME-Lung 2; NCT00806819).
Cediranib (Recentin™, AstraZeneca) is a multikinase targeted agent that inhibits VEGFR-1, -2, and −3, c-kit, and PDGFR-β [59]. A phase II/III trial (BR24; N = 296) has evaluated cediranib with paclitaxel/carboplatin as first-line therapy in patients with advanced NSCLC [60]. Interim results from this trial demonstrated a significantly higher response rate with cediranib versus placebo (38% vs 16%; P < 0.0001). During the trial, the original cediranib dose of 45 mg was reduced to 30 mg because of toxicities, but the 30-mg dose was still associated with excessive adverse events, including grade ≥3 hypertension (19% versus 2% with placebo-chemotherapy), grade ≥3 diarrhea (15% versus 2%, respectively). Based on these toxicities, the study was halted to review imbalances in assigned causes of death. In light of the results of BR24, the ongoing phase III BR29 trial was designed to evaluate cediranib 20 mg plus paclitaxel/carboplatin (NCT00795340) but was recently reported to have closed [61].
Cediranib is also under evaluation in combination with pemetrexed for treatment of NSCLC [62]. Results from this study revealed a RR of 29% (95% CI, 17%–45%) and a disease control rate of 74% (95% CI, 58%–85%) for 38 evaluable patients. Grade 3/4 toxicities for this regimen included neutropenia (14%), febrile neutropenia (5%), fatigue (22%), diarrhea (14%), and infection (8%).
Motesanib (Amgen) inhibits VEGFR-1, -2, and −3, PDGFR, c-kit, and RET [63]. A phase II trial (N = 186) has evaluated motesanib (125 mg once daily in arm A or 75 mg twice daily in arm B) plus paclitaxel/carboplatin versus bevacizumab plus paclitaxel/carboplatin (arm C) as first-line therapy for advanced nonsquamous NSCLC [64]. The objective RR was 30%, 23%, and 37%, median PFS was 7.7 months, 5.8 months, and 8.3 months, and median OS was 14.0 months, 12.8 months, and 14.0 months for arms A, B, and C, respectively. Incidence of grade ≥3 adverse events was higher in arms A (71%) and B (79%) than in arm C (63%). The most common grade 3/4 toxicities occurring in ≥20% of patients and in more than 1 treatment arm included diarrhea (19%, 11%, and 3% in arms A, B, and C, respectively), dehydration (17%, 11%, and 3%), and fatigue (17%, 5%, and 8%). Clinical development of motesanib in advanced NSCLC has followed a pattern similar to that for bevacizumab and sorafenib with respect to histology, with temporary suspension of the phase III MONET1 trial of paclitaxel/carboplatin alone or with motesanib in light of increased hemoptysis and mortality among the squamous histology subset [65]. The trial reopened and completed accrual (N = 1,090), though enrollment was restricted to patients with nonsquamous tumors. Preliminary results reported significantly longer PFS (5.6 vs 5.4 months; HR, 0.785; 95% CI, 0.684–0.901; P = 0.0006) and higher RR (40% vs 26%; P < 0.0001) with chemotherapy plus motesanib versus chemotherapy plus placebo; however, the study did not meet its primary endpoint of improving OS (13.0 vs 11.0 months; HR, 0.897; 95% CI, 0.776–1.035; P = 0.137) [66, 67].
Axitinib (Pfizer) inhibits VEGFR-1, -2, and −3, PDGFR-β, and c-kit [68]. A phase II trial of axitinib monotherapy has been conducted in 32 patients with advanced NSCLC, including chemotherapy-naive or chemotherapy-pretreated patients [69]. In this trial, 3 patients (9%) had a partial response and 10 (31%) had stable disease for ≥16 weeks. Median PFS was 4.9 months overall and 9.2 months in the first-line axitinib subset, with median OS of 14.8 months for all patients (14.8 months for previously untreated patients; 15.5 months for previously treated patients). Grade 3 fatigue and hypertension were reported in 22% and 9% of patients, respectively. Current early phase trials are investigating the combination of axitinib with chemotherapy for advanced NSCLC, including a phase I/II trial in nonsquamous tumors (NCT00768755) and a phase II trial in squamous tumors (NCT00735904).
Pazopanib (GlaxoSmithKline) is a multikinase inhibitor that targets VEGFR-1, -2, and −3, PDGFR-α/β, and c-kit [70, 71]. A phase II trial of pazopanib monotherapy as preoperative therapy for stage I/II NSCLC revealed that 3 of 35 patients (9%) had a partial response, and the most common grade ≥3 toxicity was alanine transaminase elevation (6%) [72]. A biomarker analysis of this trial showed that 11 cytokine and angiogenic factors were significantly impacted (reduced or increased) by pazopanib [73]. Serum VEGFR-2 levels showed the most pronounced reduction, and changes in VEGFR-2 and interleukin-4 correlated with tumor shrinkage. A phase II/III trial is currently evaluating pazopanib as adjuvant monotherapy for surgically resected stage I NSCLC (NCT00775307) and a second phase II/III trial is evaluating pazopanib monotherapy in patients with advanced NSCLC as second-line therapy (NCT01208064). Additional phase II trials of pazopanib as monotherapy (NCT01049776) and as a component of combination regimens (with chemotherapy [NCT00866528] or an EGFR inhibitor [NCT01027598]) for advanced NSCLC are also underway.
Antiangiogenic antibody-based agents
In addition to multitargeted TKIs, novel antibody-based therapies have also been studied as antiangiogenic agents with mixed results. Aflibercept or AV0005 (VEGF Trap, Regeneron) is a peptide-antibody fusion containing portions of human VEGFR-1 and −2 [74]. A phase II study (N = 98) that evaluated aflibercept in patients with platinum- and erlotinib-resistant adenocarcinoma of the lung demonstrated an overall RR of 2% and a median PFS and OS of 2.7 months and 6.2 months, respectively [75]. The most common grade ≥3 toxicities included proteinuria (10%), hypertension (23%), and dyspnea (21%). Two treatment-related fatal adverse events, both grade 5 hemoptysis, occurred on study. In a recently-completed phase III trial (NCT00532155), aflibercept was evaluated in combination with docetaxel as second-line therapy for advanced NSCLC. Initial results suggested that this trial did not meet its primary endpoint of improving OS versus docetaxel (HR, 1.01; CI, 0.868–1.174). However, both PFS (HR, 0.82; CI, 0.716–0.937) and ORR (23.3% for aflibercept vs 8.9% for placebo) were improved in patients treated with aflibercept versus placebo. The most common grade 3/4 toxicities in patients treated with aflibercept were fatigue, stomatitis, disease progression, hypertension, febrile neutropenia, dyspnea, neutropenia, and asthenia [76]. In a phase III clinical trial (VITAL), aflibercept/docetaxel and placebo/docetaxel were evaluated as second-line therapy (1 prior platinum-based regimen) in advanced, nonsquamous NSCLC [77]. Aflibercept was associated with improvement in PFS (HR, 0.82; 95% CI, 0.72–0.94; P = 0.0035) and RR (23.3% vs 8.9%; P < 0.0001); however, the primary endpoint of improved OS was not met (HR, 1.01; 95.1% CI, 0.87–1.17; P = 0.898). Stomatitis, weight decrease, hypertension, epistaxis, and dysphonia were reported as occurring at greater than 10% higher incidence with aflibercept compared with placebo.
Ramucirumab or IMC-1121B (ImClone Systems, Inc.) is an anti-VEGFR-2 monoclonal antibody [78]. Preliminary results are available from an open-label phase II study of ramucirumab plus first-line paclitaxel/carboplatin in patients with advanced NSCLC [79]. For 15 evaluable patients, the preliminary objective RR was 67%, including 1 complete response, and median PFS was 5.7 months. Two potentially ramucirumab-related serious adverse events were reported, grade 4 febrile neutropenia and grade 2 pneumothorax. A phase II trial (NCT01160744) of platinum-based chemotherapy (pemetrexed plus carboplatin/cisplatin or gemcitabine plus carboplatin/cisplatin) with or without ramucirumab in patients with previously untreated advanced NSCLC is currently recruiting patients, and a phase III trial (NCT01168973) of docetaxel plus ramucirumab or placebo as second-line therapy for stage IV NSCLC is planned.
Conclusions
The VEGF axis continues to be a key therapeutic target for NSCLC-directed treatments, with substantial interest in simultaneous targeting of other signaling pathways in an effort to improve clinical outcomes. The anti-VEGF monoclonal antibody bevacizumab is currently the only approved and recommended antiangiogenic agent for use in patients with advanced NSCLC, although a number of other antiangiogenic strategies including antibodies and multitargeted TKIs are under clinical evaluation in various settings in NSCLC. Selection of patients most likely to benefit from antiangiogenic therapies may lead to improved outcomes, and toward this end biomarker evaluations are becoming a more prevalent component of clinical trials of antiangiogenic agents. Advances are being made in this regard (as illustrated by the BATTLE program) with the ultimate goal of identifying predictive factors to truly individualize therapy for patients.
References
American Cancer Society (2009) Cancer facts & figures, 2009. American Cancer Society, Atlanta
World Health Organization (2008) Fact sheet No. 310: the top ten causes of death. November 2008. http://www.who.int/mediacentre/factsheets/fs310_2008.pdf. Accessed 8 October 2009
National Comprehensive Cancer Network (2011) NCCN clinical practice guidelines in oncology™. Non-Small Cell Lung Cancer. V.3.2011. http://www.nccn.org/professionals/physician_gls/PDF/nscl.pdf. Accessed 21 May 11 A.D.
Schiller JH, Harrington D, Belani CP, Langer C, Sandler A, Krook J, Zhu J, Johnson DH (2002) Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N Engl J Med 346:92–98
Scagliotti GV, Parikh P, von Pawel J, Biesma B, Vansteenkiste J, Manegold C, Serwatowski P, Gatzemeier U, Digumarti R, Zukin M, Lee JS, Mellemgaard A, Park K, Patil S, Rolski J, Goksel T, de Marinis F, Simms L, Sugarman KP, Gandara D (2008) Phase III study comparing cisplatin plus gemcitabine with cisplatin plus pemetrexed in chemotherapy-naive patients with advanced-stage non-small-cell lung cancer. J Clin Oncol 26:3543–3551
AVASTIN® (bevacizumab) (2011) Solution for intravenous infusion [package insert]. South San Francisco, CA: Genentech, Inc
Iressa® (gefitinib tablets) [package insert]. Wilmington, DE: AstraZeneca Pharmaceuticals LP
Tarceva® (erlotinib tablets) [package insert]. South San Franscisco, CA: Genentech, Inc
Kerbel RS (2006) Antiangiogenic therapy: a universal chemosensitization strategy for cancer? Science 312:1171–1175
Carmeliet P (2005) Angiogenesis in life, disease and medicine. Nature 438:932–936
Folkman J (1971) Tumor angiogenesis: therapeutic implications. N Engl J Med 285:1182–1186
Ferrara N, Gerber HP, LeCouter J (2003) The biology of VEGF and its receptors. Nat Med 9:669–676
Carmeliet P, Jain RK (2000) Angiogenesis in cancer and other diseases. Nature 407:249–257
Folkman J, Klagsbrun M (1987) Vascular physiology. A family of angiogenic peptides. Nature 329:671–672
Folkman J, Shing Y (1992) Angiogenesis. J Biol Chem 267:10931–10934
Hicklin DJ, Ellis LM (2005) Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J Clin Oncol 23:1011–27
Ellis LM, Hicklin DJ (2008) VEGF-targeted therapy: mechanisms of anti-tumour activity. Nat Rev Cancer 8:579–591
Korpanty G, Smyth E, Sullivan LA, Brekken RA, Carney DN (2010) Antiangiogenic therapy in lung cancer: focus on vascular endothelial growth factor pathway. Exp Biol Med (Maywood) 235:3–9
Jain RK, Booth MF (2003) What brings pericytes to tumor vessels? J Clin Invest 112:1134–1136
Ferrara N, Davis-Smyth T (1997) The biology of vascular endothelial growth factor. Endocr Rev 18:4–25
Toi M, Matsumoto T, Bando H (2001) Vascular endothelial growth factor: its prognostic, predictive, and therapeutic implications. Lancet Oncol 2:667–673
Yuan A, Yu C-J, Kuo S-H, Chen W-J, Lin F-Y, Luh K-T, Yang P-C, Lee Y-C (2001) Vascular endothelial growth factor 189 mRNA isoform expression specifically correlates with tumor angiogenesis, patient survival, and postoperative relapse in non-small-cell lung cancer. J Clin Oncol 19:432–441
Millauer B, Longhi MP, Plate KH, Shawver LK, Risau W, Ullrich A, Strawn LM (1996) Dominant-negative inhibition of Flk-1 suppresses the growth of many tumor types in vivo. Cancer Res 56:1615–1620
Johnson DH, Fehrenbacher L, Novotny WF, Herbst RS, Nemunaitis JJ, Jablons DM, Langer CJ, DeVore RF III, Gaudreault J, Damico LA, Holmgren E, Kabbinavar F (2004) Randomized phase II trial comparing bevacizumab plus carboplatin and paclitaxel with carboplatin and paclitaxel alone in previously untreated locally advanced or metastatic non-small-cell lung cancer. J Clin Oncol 22:2184–2191
Sandler A, Gray R, Perry MC, Brahmer J, Schiller JH, Dowlati A, Lilenbaum R, Johnson DH (2006) Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med 355:2542–2550
Reck M, von Pawel J, Zatloukal P, Ramlau R, Gorbounova V, Hirsh V, Leighl N, Mezger J, Archer V, Moore N, Manegold C (2009) Phase III trial of cisplatin plus gemcitabine with either placebo or bevacizumab as first-line therapy for nonsquamous non-small-cell lung cancer: AVAiL. J Clin Oncol 27:1227–1234
Reck M, von Pawel J, Zatloukal P, Ramlau R, Gorbounova V, Hirsh V, Leighl N, Mezger J, Archer V, Moore N, Manegold C (2010) Overall survival with cisplatin-gemcitabine and bevacizumab or placebo as first-line therapy for nonsquamous non-small-cell lung cancer: results from a randomised phase III trial (AVAiL). Ann Oncol 21:1804–1809
Crino L, Dansin E, Garrido P, Griesinger F, Laskin J, Pavlakis N, Stroiakovski D, Thatcher N, Tsai CM, Wu YL, Zhou C (2010) Safety and efficacy of first-line bevacizumab-based therapy in advanced non-squamous non-small-cell lung cancer (SAiL, MO19390): a phase 4 study. Lancet Oncol 11:733–740
Wozniak AJ, Garst J, Jahanzeb M, Kosty MP, Vidaver R, Beatty S, Teng S, Flick ED, Sing A, Lynch TJ, for the ARIES Investigators (2010) Clinical outcomes (CO) for special populations of patients (pts) with advanced non-small cell lung cancer (NSCLC): results from ARIES, a bevacizumab (BV) observational cohort study (OCS). J Clin Oncol 28: Abstract 7618
Dowlati A, Gray R, Sandler AB, Schiller JH, Johnson DH (2008) Cell adhesion molecules, vascular endothelial growth factor, and basic fibroblast growth factor in patients with non-small cell lung cancer treated with chemotherapy with or without bevacizumab-an Eastern Cooperative Oncology Group Study. Clin Cancer Res 14:1407–1412
Socinski MA, Langer CJ, Huang JE, Kolb MM, Compton P, Wang L, Akerley W (2009) Safety of bevacizumab in patients with non-small-cell lung cancer and brain metastases. J Clin Oncol 27:5255–5261
Herbst RS, Ansari R, Bustin F, Flynn P, Hart L, Otterson GA, Vlahovic G, Soh CH, O’Connor P, Hainsworth J (2011) Efficacy of bevacizumab plus erlotinib versus erlotinib alone in advanced non-small-cell lung cancer after failure of standard first-line chemotherapy (BeTa): a double-blind, placebo-controlled, phase 3 trial. Lancet 377:1846–1854
Kabbinavar FF, Miller VA, Johnson BE, O’Connor PG, Soh C, TLAS I (2010) Overall survival (OS) in ATLAS, a phase IIIb trial comparing bevacizumab (B) therapy with or without erlotinib (E) after completion of chemotherapy (chemo) with B for first-line treatment of locally advanced, recurrent, or metastatic non-small cell lung cancer (NSCLC). J Clin Oncol 28: Abstract 7526
Patel JD, Bonomi P, Socinski MA, Govindan R, Hong S, Obasaju C, Pennella EJ, Girvan AC, Guba SC (2009) Treatment rationale and study design for the pointbreak study: a randomized, open-label phase III study of pemetrexed/carboplatin/bevacizumab followed by maintenance pemetrexed/bevacizumab versus paclitaxel/carboplatin/bevacizumab followed by maintenance bevacizumab in patients with stage IIIB or IV nonsquamous non-small-cell lung cancer. Clin Lung Cancer 10:252–256
Wakelee HA, Dahlberg SE, Keller SM, Gandara DR, Graziano S, Leighl NB, Adjei AA, Schiller J (2011) Interim report of on-study demographics and toxicity from Eastern Cooperative Oncology Group (ECOG) E1505, a phase III randomized trial of adjuvant chemotherapy with or without bevacizumab for completely resected early stage non-small cell lung cancer. J Thorac Oncol 6: Abstract O42.03
Levitzki A (2004) PDGF receptor kinase inhibitors for the treatment of PDGF driven diseases. Cytokine Growth Factor Rev 15:229–235
Murakami M, Zheng Y, Hirashima M, Suda T, Morita Y, Ooehara J, Ema H, Fong GH, Shibuya M (2008) VEGFR1 tyrosine kinase signaling promotes lymphangiogenesis as well as angiogenesis indirectly via macrophage recruitment. Arterioscler Thromb Vasc Biol 28:658–664
Bergers G, Song S, Meyer-Morse N, Bergsland E, Hanahan D (2003) Benefits of targeting both pericytes and endothelial cells in the tumor vasculature with kinase inhibitors. J Clin Invest 111:1287–1295
Erber R, Thurnher A, Katsen AD, Groth G, Kerger H, Hammes HP, Menger MD, Ullrich A, Vajkoczy P (2004) Combined inhibition of VEGF and PDGF signaling enforces tumor vessel regression by interfering with pericyte-mediated endothelial cell survival mechanisms. FASEB J 18:338–340
Cabebe E, Wakelee H (2007) Role of anti-angiogenesis agents in treating NSCLC: focus on bevacizumab and VEGFR tyrosine kinase inhibitors. Curr Treat Options Oncol 8:15–27
Casanovas O, Hicklin DJ, Bergers G, Hanahan D (2005) Drug resistance by evasion of antiangiogenic targeting of VEGF signaling in late-stage pancreatic islet tumors. Cancer Cell 8:299–309
Wilhelm SM, Carter C, Tang L, Wilkie D, McNabola A, Rong H, Chen C, Zhang X, Vincent P, McHugh M, Cao Y, Shujath J, Gawlak S, Eveleigh D, Rowley B, Liu L, Adnane L, Lynch M, Auclair D, Taylor I, Gedrich R, Voznesensky A, Riedl B, Post LE, Bollag G, Trail PA (2004) BAY 43–9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis. Cancer Res 64:7099–7109
Scagliotti G, Novello S, von Pawel J, Reck M, Pereira JR, Thomas M, brao Miziara JE, Balint B, de Marinis F, Keller A, Aren O, Csollak M, Albert I, Barrios CH, Grossi F, Krzakowski M, Cupit L, Cihon F, DiMatteo S, Hanna N (2010) Phase III study of carboplatin and paclitaxel alone or with sorafenib in advanced non-small-cell lung cancer. J Clin Oncol 28:1835–1842
Gatzemeier U, Eisen T, Santoro A, Paz-Ares L, Bennouna J, Liao M, Strauss UP, Montegriffo E, Ong TJ, Biesma B (2010) Sorafenib (S) + gemcitabine/cisplatin (GC) vs GC alone in the first-line treatment of advanced non-small cell lung cancer (NSCLC): Phase III NSCLC research experience utilizing sorafenib (NEXUS) trial. Ann Oncol 21:viii7.
Schiller JH, Lee JW, Hanna NH, Traynor AM, Carbone DP (2008) A randomized discontinuation phase II study of sorafenib versus placebo in patients with non-small cell lung cancer who have failed at least two prior chemotherapy regimens: E2501. J Clin Oncol 26: Abstract 8014
Molina JR, Dy GK, Foster NR, Allen Ziegler KL, Adjei A, Rowland KM, Aubry M, Flynn PJ, Mandrekar SJ, Schild SE, Adjei AA (2011) A randomized phase II study of pemetrexed (PEM) with or without sorafenib (S) as second-line therapy in advanced non-small cell lung cancer (NSCLC) of nonsquamous histology: NCCTG N0626 study. J Clin Oncol 29: Abstract 7513
Spigel DR, Burris HA III, Greco FA, Shipley DL, Friedman EK, Waterhouse DM, Whorf RC, Mitchell RB, Daniel DB, Zangmeister J, Bass JD, Hainsworth JD (2011) Randomized, double-blind, placebo-controlled, phase II trial of sorafenib and erlotinib or erlotinib alone in previously treated advanced non-small-cell lung cancer. J Clin Oncol 29:2582–2589
Vroling L, Lind JS, de Haas RR, Verheul HM, van Hinsbergh V, Broxterman HJ, Smit EF (2010) CD133+ circulating haematopoietic progenitor cells predict for response to sorafenib plus erlotinib in non-small cell lung cancer patients. Br J Cancer 102:268–275
Kim ES, Herbst RS, Lee JJ, Blumenschein GR, Jr., Tsao A, Alden CM, Tang X, Liu S, Stewart DJ, Heymach JV, Tran HT, Hicks ME, Erasmus J, Jr., Gupta S, Powis G, Lippman SM, Wistuba II, Hong WK (2010) The BATTLE trial (Biomarker-integrated Approaches of Targeted Therapy for Lung Cancer Elimination): personalizing therapy for lung cancer. Presented at: the 101st Annual Meeting of the American Association for Cancer Research; April 17–21, 2010; Washington, DC
Herbst RS, Blumenschein GR, Jr., Kim ES, Lee J, Tsao AS, Alden CM, Liu S, Stewart DJ, Wistuba II, Hong WK (2010) Sorafenib treatment efficacy and KRAS biomarker status in the Biomarker-Integrated Approaches of Targeted Therapy for Lung Cancer Elimination (BATTLE) trial. J Clin Oncol (Meeting Abstracts) 28:7609.
Socinski MA, Novello S, Brahmer JR, Rosell R, Sanchez JM, Belani CP, Govindan R, Atkins JN, Gillenwater HH, Pallares C, Tye L, Selaru P, Chao RC, Scagliotti GV (2008) Multicenter, phase II trial of sunitinib in previously treated, advanced non-small-cell lung cancer. J Clin Oncol 26:650–656
Novello S, Scagliotti GV, Rosell R, Socinski MA, Brahmer J, Atkins J, Pallares C, Burgess R, Tye L, Selaru P, Wang E, Chao R, Govindan R (2009) Phase II study of continuous daily sunitinib dosing in patients with previously treated advanced non-small cell lung cancer. Br J Cancer 101:1543–1548
Reynolds C, Spira AI, Gluck WL, Pluenneke RE, Boehm KA, Zhan F, Asmar L (2011) Results of a phase II study of single-agent sunitinib in elderly non-small cell lung cancer patients. J Clin Oncol 29: Abstract 7549
Scagliotti GV, Krzakowski M, Szczesna A, Strausz J, Makhson A, Reck M, Tye L, Selaru P, Chao RC, Govindan R (2010) Sunitinib (SU) in combination with erlotinib (E) for the treatment of advanced/metastatic non-small cell lung cancer (NSCLC): a phase III study. Ann Oncol 21:viii3. Abstract LBA6
Thongprasert S, Tung Y, Kim J-H, Chang G-C, Park K, Su W-C, Kim S-W, Mok TSK, Sriuranpong V, Thall A, Tye L, Zhang K, Chao RC, Scagliotti GV, Govindan R (2011) Sunitinib plus erlotinib for the treatment of advanced NSCLC: subset analysis of East Asian patients participating in a phase III trial. J Thorac Oncol 6: Abstract MO09.02
Roth GJ, Heckel A, Colbatzky F, Handschuh S, Kley J, Lehmann-Lintz T, Lotz R, Tontsch-Grunt U, Walter R, Hilberg F (2009) Design, synthesis, and evaluation of indolinones as triple angiokinase inhibitors and the discovery of a highly specific 6-methoxycarbonyl-substituted indolinone (BIBF 1120). J Med Chem 52:4466–4480
Hilberg F, Roth GJ, Krssak M, Kautschitsch S, Sommergruber W, Tontsch-Grunt U, Garin-Chesa P, Bader G, Zoephel A, Quant J, Heckel A, Rettig WJ (2008) BIBF 1120: triple angiokinase inhibitor with sustained receptor blockade and good antitumor efficacy. Cancer Res 68:4774–4782
Reck M, Kaiser R, Eschbach C, Stefanic M, Love J, Gatzemeier U, Stopfer P, von Pawel J (2011) A phase II double-blind study to investigate efficacy and safety of two doses of the triple angiokinase inhibitor BIBF 1120 in patients with relapsed advanced non-small-cell lung cancer. Ann Oncol 22:1374–1381
Wedge SR, Kendrew J, Hennequin LF, Valentine PJ, Barry ST, Brave SR, Smith NR, James NH, Dukes M, Curwen JO, Chester R, Jackson JA, Boffey SJ, Kilburn LL, Barnett S, Richmond GH, Wadsworth PF, Walker M, Bigley AL, Taylor ST, Cooper L, Beck S, Jurgensmeier JM, Ogilvie DJ (2005) AZD2171: a highly potent, orally bioavailable, vascular endothelial growth factor receptor-2 tyrosine kinase inhibitor for the treatment of cancer. Cancer Res 65:4389–4400
Goss GD, Arnold A, Shepherd FA, Dediu M, Ciuleanu TE, Fenton D, Zukin M, Walde D, Laberge F, Vincent MD, Ellis PM, Laurie SA, Ding K, Frymire E, Gauthier I, Leighl NB, Ho C, Noble J, Lee CW, Seymour L (2010) Randomized, double-blind trial of carboplatin and paclitaxel with either daily oral cediranib or placebo in advanced non-small-cell lung cancer: NCIC Clinical Trials Group BR24 study. J Clin Oncol 28:49–55
NCIC Clinical Trials Group (2011) Site committee open/closed/planned/on hold/withdrawn studies. http://www.ctg.queensu.ca/public/Clinical_Trials/ph3_trial_accrual_closed.htm. Accessed 11 August 2011
Gadgeel SM, Ruckdeschel JC, Wozniak AJ, Chen W, Hackstock D, Galasso C, Burger A, Ivy SP, LoRusso P, Edelman MJ (2011) Cediranib, a VEGF receptor 1, 2, and 3 inhibitor, and pemetrexed in patients (pts) with recurrent non-small cell lung cancer (NSCLC). J Clin Oncol 29: Abstract 7564
Polverino A, Coxon A, Starnes C, Diaz Z, DeMelfi T, Wang L, Bready J, Estrada J, Cattley R, Kaufman S, Chen D, Gan Y, Kumar G, Meyer J, Neervannan S, Alva G, Talvenheimo J, Montestruque S, Tasker A, Patel V, Radinsky R, Kendall R (2006) AMG 706, an oral, multikinase inhibitor that selectively targets vascular endothelial growth factor, platelet-derived growth factor, and kit receptors, potently inhibits angiogenesis and induces regression in tumor xenografts. Cancer Res 66:8715–8721
Blumenschein GR Jr, Kabbinavar F, Menon H, Mok TS, Stephenson J, Beck JT, Lakshmaiah K, Reckamp K, Hei YJ, Kracht K, Sun YN, Sikorski R, Schwartzberg L (2011) A phase II, multicenter, open-label randomized study of motesanib or bevacizumab in combination with paclitaxel and carboplatin for advanced nonsquamous non-small-cell lung cancer. Ann Oncol. doi:10.1093
Amgen (2009) Independent data monitoring committee recommends resuming enrollment of non-squamous NSCLC patients in the motesanib MONET1 trial. http://www.amgen.com/media/media_pr_detail.jsp?year=2009&releaseID=1255738. Accessed 12 May 2010
Amgen Inc, Millenium Pharmaceuticals, Takeda Pharmaceutical Company Limited (2011) Top-line results announced of pivotal phase 3 motesanib trial in advanced non-squamous non-small cell lung cancer patients. http://www.takeda.com/press/article_40445.html. Accessed 18 April 2011
Scagliotti G, Vynnychenko I, Ichinose Y, Park K, Kubota K, Blackhall FH, Pirker R, Galiulin R, Ciuleanu T, Sydorenko O, Dediu M, Papai-Szekely Z, Martinez Banaclocha N, McCoy S, Yao B, Hei Y, Spigel DR (2011) An international, randomized, placebo-controlled, double-blind phase III study (MONET1) of motesanib plus carboplatin/paclitaxel (C/P) in patients with advanced nonsquamous non-small cell lung cancer (NSCLC). J Clin Oncol 29: Abstract LBA7512
Choueiri TK (2008) Axitinib, a novel anti-angiogenic drug with promising activity in various solid tumors. Curr Opin Investig Drugs 9:658–671
Schiller JH, Larson T, Ou SH, Limentani S, Sandler A, Vokes E, Kim S, Liau K, Bycott P, Olszanski AJ, von Pawel J (2009) Efficacy and safety of axitinib in patients with advanced non-small-cell lung cancer: results from a phase II study. J Clin Oncol 27:3836–3841
Kumar R, Knick VB, Rudolph SK, Johnson JH, Crosby RM, Crouthamel MC, Hopper TM, Miller CG, Harrington LE, Onori JA, Mullin RJ, Gilmer TM, Truesdale AT, Epperly AH, Boloor A, Stafford JA, Luttrell DK, Cheung M (2007) Pharmacokinetic-pharmacodynamic correlation from mouse to human with pazopanib, a multikinase angiogenesis inhibitor with potent antitumor and antiangiogenic activity. Mol Cancer Ther 6:2012–2021
Podar K, Tonon G, Sattler M, Tai YT, Legouill S, Yasui H, Ishitsuka K, Kumar S, Kumar R, Pandite LN, Hideshima T, Chauhan D, Anderson KC (2006) The small-molecule VEGF receptor inhibitor pazopanib (GW786034B) targets both tumor and endothelial cells in multiple myeloma. Proc Natl Acad Sci U S A 103:19478–19483
Altorki N, Lane ME, Bauer T, Lee PC, Guarino MJ, Pass H, Felip E, Peylan-Ramu N, Gurpide A, Grannis FW, Mitchell JD, Tachdjian S, Swann RS, Huff A, Roychowdhury DF, Reeves A, Ottesen LH, Yankelevitz DF (2010) Phase II proof-of-concept study of pazopanib monotherapy in treatment-naive patients with stage I/II resectable non-small-cell lung cancer. J Clin Oncol 28:3131–3137
Nikolinakos PG, Altorki N, Yankelevitz D, Tran HT, Yan S, Rajagopalan D, Bordogna W, Ottesen LH, Heymach JV (2010) Plasma cytokine and angiogenic factor profiling identifies markers associated with tumor shrinkage in early-stage non-small cell lung cancer patients treated with pazopanib. Cancer Res 70:2171–2179
Lockhart AC, Rothenberg ML, Dupont J, Cooper W, Chevalier P, Sternas L, Buzenet G, Koehler E, Sosman JA, Schwartz LH, Gultekin DH, Koutcher JA, Donnelly EF, Andal R, Dancy I, Spriggs DR, Tew WP (2010) Phase I study of intravenous vascular endothelial growth factor trap, aflibercept, in patients with advanced solid tumors. J Clin Oncol 28:207–214
Leighl NB, Raez LE, Besse B, Rosen PJ, Barlesi F, Massarelli E, Gabrail N, Hart LL, Albain KS, Berkowitz L, Melnyk O, Shepherd FA, Sternas L, Ackerman J, Shun Z, Miller VA, Herbst RS (2010) A multicenter, phase 2 study of vascular endothelial growth factor trap (Aflibercept) in platinum- and erlotinib-resistant adenocarcinoma of the lung. J Thorac Oncol 5:1054–1059
(2011) Regeneron Pharmaceuticals, Inc. Sanofi-aventis and Regeneron report top-line results from phase III study with aflibercept (VEGF Trap) in second-line non-small cell lung cancer. Press release. March 10, 2011. http://newsroom.regeneron.com/releasedetail.cfm?ReleaseID=556162. Accessed 25 March 2011
Novello S, Ramlau R, Gorbunova VA, Ciuleanu TE, Ozguroglu M, Goksel T, Baldotto C, Bennouna J, Shepherd FA, Scagliotti G (2011) Aflibercept in combination with docetaxel for second-line treatment of locally advanced or metastatic non-small-cell lung cancer (NSCLC): Final results of a multinational placebo-controlled phase III trial (EFC10261-VITAL). Abstract associated with oral presentation at: the 14th Biennial World Conference on Lung Cancer; July 3–7, 2011; Amsterdam, Netherlands Abstract O43.06
Spratlin JL, Cohen RB, Eadens M, Gore L, Camidge DR, Diab S, Leong S, O’Bryant C, Chow LQ, Serkova NJ, Meropol NJ, Lewis NL, Chiorean EG, Fox F, Youssoufian H, Rowinsky EK, Eckhardt SG (2010) Phase I pharmacologic and biologic study of ramucirumab (IMC-1121B), a fully human immunoglobulin G1 monoclonal antibody targeting the vascular endothelial growth factor receptor-2. J Clin Oncol 28:780–787
Camidge DR, Ballas MS, Dubey S, Haigentz M, Rosen PJ, Spicer JF, West HJ, Shah GD, Youssoufian H, Mita AC (2010) A phase II, open-label study of ramucirumab (IMC-1121B), an IgG1 fully human monoclonal antibody (MAb) targeting VEGFR-2, in combination with paclitaxel and carboplatin as first-line therapy in patients (pts) with stage IIIb/IV non-small cell lung cancer (NSCLC). J Clin Oncol 28: Abstract 7588
Acknowledgments
This work was supported by Boehringer Ingelheim Pharmaceuticals, Inc (BIPI). Writing and editorial assistance was provided by Staci Heise, PhD, of MedErgy, which was contracted by BIPI for these services. The author meets criteria for authorship as recommended by the International Committee of Medical Journal Editors (ICMJE), was fully responsible for all content and editorial decisions, and was involved at all stages of manuscript development. The author received no compensation related to the development of the manuscript.
Conflicts of Interest
The author declares that he has no conflicts of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Blumenschein, G.R. Developmental antiangiogenic agents for the treatment of Non-Small Cell Lung Cancer (NSCLC). Invest New Drugs 30, 1802–1811 (2012). https://doi.org/10.1007/s10637-011-9750-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-011-9750-1