Abstract
Background
Vonoprazan is a novel potassium-competitive acid blocker (P-CAB) recently approved for Helicobacter pylori eradication therapy in Japan.
Aims
To compare PPI- and P-CAP-containing triple therapy and vonoprazan-based triple therapy.
Methods
Two hundred ninety-five initial subjects received a PPI-containing triple therapy; the next 125 subjects received vonoprazan-containing triple therapy. Two sequential groups received 7-day eradication regimens consisting of amoxicillin 750 mg, clarithromycin 200 mg both twice a day with standard dose PPI or vonoprazan (20 mg) each twice daily. H. pylori eradication was confirmed by a 13C-UBT. Clarithromycin susceptibility was evaluated by 23S rRNA PCR.
Results
Population cure rates with clarithromycin susceptible strains were 89.6 versus 100 % for PPI and vonoprazan therapies, respectively. Cure rates with resistant strains were 40.2 % with PPI therapy versus 76.1 % with vonoprazan triple therapy. There was no difference in side effects.
Conclusions
Although 7-day P-CAB triple therapy was superior to 7-day PPI triple therapy, neither was highly effective, or can be recommended, in the presence of clarithromycin-resistant infections.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In February 2013, the Japanese Minister of Health, Labour and Welfare approved medical insurance payments for Helicobacter pylori eradication in patients with chronic gastritis diagnosed by endoscopy. This approval was based on the strategy that H. pylori eradication would reduce gastric cancer deaths [1]. However, only clarithromycin triple therapy was approved for first eradication regimen despite the fact that clarithromycin’s effectiveness had steadily declined worldwide due to increasing clarithromycin resistance [2–6]. Vonoprazan is a potassium-competitive acid blocker (P-CAB) discovered and developed by Takeda Pharmaceutical Company Ltd., Japan [7]. Like PPIs, P-CABs inhibit gastric H+, K+-ATPase, the enzyme that catalyzes the final step in the gastric acid secretion pathway. However, unlike PPIs, inhibition is competitive, reversible but longer lasting due to the increased half-life of the drug [8]. The inhibitory effect of vonoprazan (pKa 9.4) is also largely unaffected by ambient pH, and it accumulates in parietal cells under both secreting and resting conditions [9].
Vonoprazan was recently approved in Japan for first-line H. pylori eradication with clarithromycin-containing triple therapy and for second-line therapy with metronidazole and amoxicillin [10]. This study investigated the effectiveness and safety of H. pylori eradication using vonoprazan, amoxicillin, and clarithromycin for 1 week as part of an outpatient cancer screening clinic.
Methods
The study was performed at Kawasaki Medical School hospital in Japan. The protocol was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. The ethics committee approved the study. Written informed consent was obtained from each patient.
Study Design
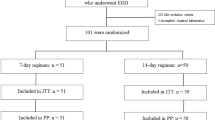
The study was designed as a study of two sequential groups. Both groups consisted of H. pylori-infected patients seen in a cancer screening clinic and diagnosed as having active H. pylori infections. The first group received the then insurance approved first-line H. pylori eradication therapy consisting of a 7-day PPI-containing clarithromycin triple therapy. After vonoprazan was approved, a second consecutive group received the newly approved vonoprazan-containing triple H. pylori eradication therapy. Results from the two groups were analyzed and compared retrospectively.
Patients
Consecutive H. pylori-positive patients for whom H. pylori eradication therapy was planned were enrolled between August 2013 and March 2016. The first group consisted of patients seen from August 2013 to March 2015 during which PPI-containing triple therapy was administered as allowed by the Japanese insurance system. Between April 2015 and March 2016, vonoprazan was substituted for the PPI in the triple therapy.
Demographic data collected at study entry included age, gender, smoking habit, alcohol consumption, underlying disease, drug treatments, and eradication history.
Helicobacter pylori Eradication Therapies
Subjects received 7-day regimens consisting of a PPI (esomeprazole 20 mg, lansoprazole 30 mg, or rabeprazole 10 mg) or vonoprazan 20 mg in combination with amoxicillin 750 mg plus clarithromycin 200 mg, all given twice a day after meals. Patients who failed initial H. pylori eradication received the approved second-line therapy in which clarithromycin was changed to metronidazole (250 mg three times a day) for 7 days.
Helicobacter pylori Diagnosis
Venous blood samples were analyzed for IgG H. pylori antibodies using an enzyme-linked immunosorbent assay (ELISA) kit (the E plate test, Eiken Kagaku, Inc., Tokyo, Japan). Gastric mucous obtained by gently scraping gastric mucosa from antrum greater curvature to corpus greater curvature using biopsy forceps was used for a rapid urease test (RUT), as previously reported [11]. The mucous samples were inoculated into a RUT tube (Helico check® Otsuka Pharmaceutical, Tokyo, Japan) [12]. A positive result was recorded when the color changed from yellow to pink within 2 h.
Helicobacter pylori positivity was defined as at least two of the three tests (serum antibody, histology, and RUT) being positive. H. pylori eradication was confirmed by a negative 13C-UBT 6–8 weeks following completion of H. pylori eradication therapy.
Clarithromycin Susceptibility
DNA was extracted from the mucous sample in the RUT tube using a commercial kit (QIAamp DNA mini kit, QIAGEN, Hilden, Germany) according to the manufacture’s protocol and subjected to PCR as previously reported [2–4]. For PCR detection of H. pylori infection and clarithromycin susceptibility, we used the following primers: 5′-ATGAATGGCGTAACGAGAT-3′ corresponding to H. pylori 23S rRNA position 2051–2070 and 5′-ACACTCAACTTGCGATTTCC-3′ position 2410-2391. PCR was performed in a thermal cycler using Ex Taq HS (Takara Biomedicals, Kusatsu city, Japan). The size of final PCR products was confirmed by electrophoresis in 2.5 % agarose gels. Point mutations of 23S rRNA at A 2142G and A 2143G for clarithromycin susceptibility were evaluated by DNA direct sequencing performed using an ABI PRISM 3100 DNA sequencer (Applied Biosystems, Foster City, CA, USA) with a BigDye Terminator version 3.1 cycle sequencing kit (Applied Biosystems).
Statistical Analyses
Parametric values were expressed as the mean ± SD. Mantel–Haenszel Chi-square analysis and the unpaired t test were performed to measure differences in demographic and clinical characteristics. A two-sided p value of less than 0.05 was considered statistically significant. All statistical computations were performed using SPSS (SPSS Inc. Chicago, IL, USA).
Results
A total of 420 patients were candidates for the study; the PPI group included 295 subjects and the vonoprazan group 125 subjects. There were no significant differences in clinical characteristics between the two groups (Table 1).
Eradication rate by intention-to-treat (ITT) analysis with PPI-containing clarithromycin triple therapy was 71.9 % (212/295) (95 % CI 67–77) which was lower than that achieved with the vonoprazan-containing clarithromycin triple therapy: 89.6 % (112/125) (95 % CI 84–95) (p < 0.001). The eradication rates per protocol (PP) were 73.1 % (212/290) (95 % CI 68–78) versus 89.6 % (112/125) (95 % CI 84–95) (p < 0.001) for PPI and vonoprazan triple therapies, respectively.
Importantly, the eradication rates with clarithromycin susceptible strains were 100 % (57/57) with vonoprazan and 87.8 % (173/212) with PPI-containing therapies (p < 0.001). The eradication rates with clarithromycin-resistant strains were 40.2 % (39/97) versus 76.1 % (35/46) for PPI and vonoprazan, respectively (p < 0.001) (Fig. 1).
Clarithromycin resistance strains (23S rRNA A2143G or A2142G) were detected in 97 of 294 (33 %) in those receiving PPI-containing therapy. A2143G was detected most often; A2142G was detected in only 5 subjects. Clarithromycin-resistant strains (23S rRNA A2143G or A2142G) were detected in 46 of 103 (44.7 %) in those receiving vonoprazan. Again, A2143G was detected in all but one strain which was A2142G (Table 2).
All 18 patients (100 %) who failed vonoprazan eradication were then successfully eradicated by a 7-day regimen consisting of vonoprazan in combination with amoxicillin plus metronidazole (200 mg b.i.d.).
Side effects resulted in 5 dropouts in the PPI group versus none in the vonoprazan group. There was no significant difference in side effects between those receiving PPI- or vonoprazan-containing triple therapies (Table 3).
Discussion
Similar to the pivotal trial with vonoprazan, this study showed very high cure rates with 7-day vonoprazan-containing therapy with clarithromycin susceptible infections (100 vs. 97 %) as well as a higher cure rate than with PPIs in the presence of clarithromycin resistance (82 and 76 vs. 40 % for vonoprazan vs. PPI, respectively) [10]. Clarithromycin resistance effectively removes clarithromycin from the triple therapy and transforms the antisecretory drug, amoxicillin, clarithromycin triple therapy into an antisecretory drug plus amoxicillin dual therapy (Fig. 1). In the original vonoprazan study, both the PPI and vonoprazan clarithromycin-containing therapies produced cure rates of 97 % with clarithromycin susceptible infections [10, 13]. In this study, the vonoprazan-containing therapy produced a similar high cure rate (e.g., 100 %) but the PPI-containing therapy resulted in only 89 % treatment success. Our study used three different PPIs which were similar when assessed in terms of maintaining the intragastric pH ≥ pH 4 (i.e., when considered as omeprazole equivalents, the esomeprazole 20 mg equivalent to 32 mg of omeprazole, rabeprazole 10 mg equivalent to 18 mg, and lansoprazole 30 mg equivalent to approximately 27 mg) [14]. It is unclear why traditional triple therapy produced unexpectedly low cure rates in this study compared to the previous study. We both used standard PPIs and not generic products. Possibly, the different results related to differences in the populations. The original study was restricted to subjects with a history of gastric or duodenal ulcer, whereas this was study included a wide variety of pathology as was more representative of clinical practice. Possibly also real-life situations require longer duration of therapy with PPI-containing triple therapy (e.g., 10 or 14 days) as is the case in western societies [15]. For example, prior studies in Japan have shown that PPI, metronidazole, amoxicillin triple therapy provides the best result when given for 14 days [16, 17].
Nonetheless, the major difference in both studies was the marked increased in effectiveness of the dual antisecretory–amoxicillin therapy received by those with clarithromycin-resistant infections. PPI amoxicillin dual therapy is sensitive to the effectiveness of the antisecretory drug in increasing and maintaining the intragastric pH at 6 or greater [4, 18–24]. Vonoprazan differs from PPIs in that it is able to achieve maximal and suppression from the first day, whereas PPIs require three to five days to reach maximal acid suppression [25, 26]. The increase in potency and the rapidity of onset may both be important in vonoprazan being able to reliably achievement of highly successful 7-day H. pylori eradication regimens.
In both the original study and this study, the cure rate of vonoprazan-containing therapy in those with resistant infections acceptable was only approximately 80 % (76 % in this study vs. 82 % in the original study). The population cure rates represent the results of combining of the results with two different populations (i.e., susceptible and resistant), and the differences increase in proportion to the prevalence of resistance [27] (Fig. 2). However, the results of combining the two populations obscure the fact that one population (susceptible infections) achieved excellent results, whereas all those with resistant infections experienced an unacceptably low cure rate (40 % with PPI-containing therapy and 76 % with vonoprazan. Looked at another way, treatment with vonoprazan resulted in a cure rate of only approximately 80 % in the presence of clarithromycin resistance. Thus, only one in five patients potentially benefited from the presence of a third drug (i.e., in this case clarithromycin) as 4 of 5 would have been cured by the dual therapy alone. Because of the high prevalence of clarithromycin resistance in Japan and in most countries, clarithromycin has become a poor choice for the third drug in triple therapy [4–6]. As illustrated by the 100 % cure rates with vonoprazan amoxicillin and metronidazole in this and in the original study, metronidazole would be preferred because of the low prevalence of resistance in Japan.
Hp-normogram [35]. This normogram plots the cure rate with susceptible infections on the left vertical axis and those with resistant infections on the right vertical axis. The proportion with resistance is shown on the horizontal axis. The cure rates are connected with a line allowing one to visualize the population cure rates for any prevalence of resistance [35]. This plot shows the cure rates with vonoprazan (circle)- and PPI (triangle)-containing clarithromycin triple therapies. Also plotted are the cure rates for the two groups which differed in the prevalence of resistance (e.g., 33 vs. 44.7 % for PPI and vonoprazan, respectively)
Another, and possibly preferred, option would be to improve the outcome with vonoprazan amoxicillin dual therapy. Dual antisecretory drug–amoxicillin therapy has a long history [28], and studies from Asia have shown that high cure rates can be obtained if the intragastric pH can be maintained at 6 or greater [24, 28, 29]. This has proven more difficult in western countries where rapid PPI metabolism is prevalent [28]. For example, Miehlke et al. [30] gave omeprazole 40 mg four times daily before meals and amoxicillin 750 mg every 6 h for 2 weeks and reported a PP cure rate of 83.8 % (95 % CI 68–94) and ITT cure rate of only 75.6 % (95 % CI 60–88). However, because vonoprazan is more effective for acid suppression, it may be able to be used to achieve a reliably dual therapy [31]. The failure to achieve a cure rate greater than 82 % in the presence of clarithromycin resistance (the surrogate for dual PPI amoxicillin therapy) suggests that (1) the pH was not consistently above 6, (2) that the duration of therapy was insufficient, (3) that the dose or frequency of administration of amoxicillin was insufficient, or (4) a combination of these factors. It is thought that the bactericidal activity of beta-lactam antibiotics is likely time rather than concentration dependent [32] such that more frequent administration such as every 6 h may be required to maintain the minimal inhibitory concentration throughout the entire day. However, Asian several studies have shown that every 8 h dosing may also be effective [33, 34]. Nonetheless, it appears that a reliably effective dual vonoprazan amoxicillin therapy is a distinct possibility and that the third drug (clarithromycin, metronidazole, or a fluoroquinolone) may become unnecessary.
Although increasing clarithromycin resistance has made empiric therapy undesirable in many areas, clarithromycin triple therapy remains useful when clarithromycin susceptibility has been confirmed. Ideally, traditional culture and susceptibility testing are preferred, but as our results show clarithromycin susceptibility can be assessed by molecular methods such as PCR of gastric mucus, or biopsy even using material used for RUT, or in stools [11, 35]. Molecular methods are not without limitations, and their validity should likely be confirmed locally. Because metronidazole resistance is rare in Japan, a better choice for first-line empiric therapy might be metronidazole containing triple therapy for 7 days with vonoprazan or for 14 days with a PPI.
References
Asaka M, Mabe K. Strategies for eliminating death from gastric cancer in Japan. Proc Jpn Acad Ser B Phys Biol Sci. 2014;90:251–258.
Maeda S, Yoshida H, Ogura K, Kanai F, Shiratori Y, Omata M. Helicobacter pylori specific nested PCR assay for the detection of 23S rRNA mutation associated with clarithromycin resistance. Gut. 1998;43:317–321.
Melake NA, Shaker GH, Salama MA. Incidence of Helicobacter pylori infection and their clarithromycin-resistant strains in otitis media with effusion regarding phenotypic and genotypic studies. Saudi Pharm J: SPJ.. 2012;20:345–353.
Graham DY, Fischbach L. Helicobacter pylori treatment in the era of increasing antibiotic resistance. Gut. 2010;59:1143–1153.
Kim SY, Choi DJ, Chung JW. Antibiotic treatment for Helicobacter pylori: is the end coming? World J Gastrointest Pharmacol Ther. 2015;6:183–198.
Megraud F, Benejat L, Benejat L, Ontsira Ngoyi EN, Lehours P. Molecular approaches to identify Helicobacter pylori antimicrobial resistance. Gastroenterol Clin North Am. 2015;44:577–596.
Shin JM, Inatomi N, Munson K, et al. Characterization of a novel potassium-competitive acid blocker of the gastric H,K-ATPase, 1-[5-(2-fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamine monofumarate (TAK-438). J Pharmacol Exp Ther. 2011;339:412–420.
Hori Y, Imanishi A, Matsukawa J, et al. 1-[5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamine monofumarate (TAK-438), a novel and potent potassium-competitive acid blocker for the treatment of acid-related diseases. J Pharmacol Exp Ther. 2010;335:231–238.
Matsukawa J, Hori Y, Nishida H, Kajino M, Inatomi N. A comparative study on the modes of action of TAK-438, a novel potassium-competitive acid blocker, and lansoprazole in primary cultured rabbit gastric glands. Biochem Pharmacol. 2011;81:1145–1151.
Murakami K, Sakurai Y, Shiino M, Funao N, Nishimura A, Asaka M. Vonoprazan, a novel potassium-competitive acid blocker, as a component of first-line and second-line triple therapy for Helicobacter pylori eradication: a phase III, randomised, double-blind study. Gut. 2016;65:1439–1446.
Matsumoto H, Shiotani A, Nishibayashi H, et al. Molecular detection of H. pylori using adherent gastric mucous to biopsy forceps. Helicobacter. 2016. doi:10.1111/hel.12310.
Uotani T, Graham DY. Diagnosis of Helicobacter pylori using the rapid urease test. Ann Transl Med. 2015;3:9.
Graham DY. Vonoprazan Helicobacter pylori eradication therapy: ethical and interpretation issues. Gut 2016. doi:10.1136/gutjnl-2016-311796.
Kirchheiner J, Glatt S, Fuhr U, et al. Relative potency of proton-pump inhibitors-comparison of effects on intragastric pH. Eur J Clin Pharmacol. 2009;65:19–31.
Yuan Y, Ford AC, Khan KJ, et al. Optimum duration of regimens for Helicobacter pylori eradication. Cochrane Database Syst Rev. 2013;12:Cd008337.
Nishizawa T, Maekawa T, Watanabe N, et al. Clarithromycin versus metronidazole as first-line Helicobacter pylori eradication: a multicenter, prospective, randomized controlled study in Japan. J Clin Gastroenterol. 2015;49:468–471.
Hori K, Miwa H, Matsumoto T. Efficacy of 2-week, second-line Helicobacter pylori eradication therapy using rabeprazole, amoxicillin, and metronidazole for the Japanese population. Helicobacter. 2011;16:234–240.
Savarino V, Sandro Mela G, Zentilin P, Vigneri S, Celle G. Acid inhibition and amoxicillin activity against Helicobacter pylori. Am J Gastroenterol. 1993;88:1975–1976.
Furuta T, Shirai N, Kodaira M, et al. Pharmacogenomics-based tailored versus standard therapeutic regimen for eradication of H. pylori. Clin Pharmacol Ther. 2007;81:521–528.
Marcus EA, Inatomi N, Nagami GT, Sachs G, Scott DR. The effects of varying acidity on Helicobacter pylori growth and the bactericidal efficacy of ampicillin. Aliment Pharmacol Ther. 2012;36:972–979.
Sachs G, Shin JM, Munson K, et al. Review article: the control of gastric acid and Helicobacter pylori eradication. Aliment Pharmacol Ther. 2000;14:1383–1401.
Scott D, Weeks D, Melchers K, Sachs G. The life and death of Helicobacter pylori. Gut. 1998;43:S56–S60.
Graham DY, Shiotani A. New concepts of resistance in the treatment of Helicobacter pylori infections. Nat Clin Pract Gastroenterol Hepatol. 2008;5:321–331.
Yang JC, Lin CJ, Wang HL, et al. High-dose dual therapy is superior to standard first-line or rescue therapy for Helicobacter pylori infection. Clin Gastroenterol Hepatol. 2015;13:895.e895–905.e895.
Shin JM, Sachs G. Long lasting inhibitors of the gastric H,K-ATPase. Expert Rev Clin Pharmacol. 2009;2:461–468.
Hori Y, Matsukawa J, Takeuchi T, et al. A study comparing the antisecretory effect of TAK-438, a novel potassium-competitive acid blocker, with lansoprazole in animals. J Pharmacol Exp Ther. 2011;337:797–804.
Graham DY. Hp-normogram (normo-graham) for assessing the outcome of H. pylori therapy: effect of resistance, duration, and CYP2C19 genotype. Helicobacter. 2016;21:85–90.
Dore MP, Lu H, Graham DY. Role of bismuth in improving Helicobacter pylori eradication with triple therapy. Gut. 2016;65:870–888.
Furuta T, Graham DY. Pharmacologic aspects of eradication therapy for Helicobacter pylori infection. Gastroenterol Clin North Am. 2010;39:465–480.
Miehlke S, Kirsch C, Schneider-Brachert W, et al. A prospective, randomized study of quadruple therapy and high-dose dual therapy for treatment of Helicobacter pylori resistant to both metronidazole and clarithromycin. Helicobacter. 2003;8:310–319.
Graham DY, Dore MP. Helicobacter pylori therapy: a paradigm shift. Expert Rev Anti Infect Ther. 2016;14:577–585.
Midolo PD, Turnidge JD, Munckhof WJ. Is bactericidal activity of amoxicillin against Helicobacter pylori concentration dependent? Antimicrob Agents Chemother. 1996;40:1327–1328.
Ren L, Lu H, Li HY, et al. New dual therapy for primary treatment of Helicobacter pylori infection: a prospective randomized study in Shanghai, China. J Dig Dis. 2014;15:622–627.
Yang JC, Wang HL, Chern HD, et al. Role of omeprazole dosage and cytochrome P450 2C19 genotype in patients receiving omeprazole-amoxicillin dual therapy for Helicobacter pylori eradication. Pharmacotherapy. 2011;31:227–238.
Smith SI, Fowora MA, Lesi OA, et al. Application of stool-PCR for the diagnosis of Helicobacter pylori from stool in Nigeria—a pilot study. SpringerPlus. 2012;1:78.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors state that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Matsumoto, H., Shiotani, A., Katsumata, R. et al. Helicobacter pylori Eradication with Proton Pump Inhibitors or Potassium-Competitive Acid Blockers: The Effect of Clarithromycin Resistance. Dig Dis Sci 61, 3215–3220 (2016). https://doi.org/10.1007/s10620-016-4305-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-016-4305-0