Abstract
Background
The incidence rate of hepatocellular carcinoma (HCC) in the USA is rapidly rising. Surveillance using biannual ultrasound (US) is recommended, but actual practices are unknown.
Aim
To determine current HCC surveillance practices among gastroenterology and hepatology providers in the USA.
Methods
A 21-question electronic survey was emailed to 12,485 gastroenterology and hepatology providers in the USA. The survey contained questions concerning provider background, surveillance practices, and opinions. Pearson chi-square and multivariate logistic regression tests were used to analyze the data.
Results
We received 777 responses (6.2 % response rate); 656 were eligible for analysis. 92 % place cirrhotic patients under surveillance independent of etiology, 79 % exclusively use a 6-month interval, and 77 % use alpha-fetoprotein. While 93 % use US, only 36 % use US exclusively and 60 % use two or more imaging modalities. Providers from transplant centers favor using additional imaging modalities, instead of only US. Multivariate analysis showed transplant center providers who allocate more time to patient care (OR 1.96, p = 0.004) and see more cirrhotic patients (OR 2.07, p = 0.033) have increased odds of using additional imaging modalities.
Conclusions
Participating providers reported very high rates of surveillance utilization and use of a biannual interval. It is likely that the sample is composed of providers who are very engaged and informed about HCC surveillance. However, their surveillance imaging practices largely deviated from practice guidelines, which all recommend only using US. Providers affiliated with transplant centers tend to use additional imaging modalities such as computed tomography and MRI, instead of US only.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Worldwide, hepatocellular carcinoma (HCC) is the sixth most common cancer with 782,000 new cases and the second leading cause of death by cancer with 746,000 cases in 2012 (GLOBOCAN http://globocan.iarc.fr). Its incidence rate is rapidly rising in Western countries; between 1975 and 2005, the age-adjusted incidence rate of HCC in the USA increased threefold [1]. In fact, HCC is regarded as the fastest growing cause of cancer-related death in the USA [2].
In 1998, before the establishment of clinical practice guidelines, Chalasani et al. [3] conducted a national survey of members of the AASLD (American Association for the Study of Liver Diseases) to understand their surveillance practices. Since 2005, practice guidelines for the management of HCC have been issued by multiple liver and cancer societies, recommending the use of biannual ultrasound for any patient with cirrhosis [4–6].
The aim of our study was to understand current HCC surveillance practices among gastroenterologists and hepatologists in the USA.
Methods
The survey was self-administered, anonymous, and electronic; it was designed and hosted using REDCap electronic data tools [7]. YK and KJ developed the survey questions using the Chalasani et al. study as a model. Questions were asked to ascertain provider type, number of years of experience, type of medical practice, whether the facility is a transplant center, and number of new cirrhotic patients seen each month. The survey asked which patients were placed under surveillance, interval, tests utilized, cost-effectiveness, and whether failure to place an at-risk patient under surveillance poses a malpractice liability. Our survey was unique in asking how providers administer ultrasound (US), via specific protocols and sonographers. These questions were created based on the authors’ experience and personal communications.
Using the authors’ personal network and membership roster of the American Medical Association (AMA), gastroenterology and hepatology providers were asked to participate in a brief online survey. The entire study was approved by the Institutional Review Board of UC San Diego Health System.
A participant was excluded from further analysis if their survey was incomplete, if they failed to provide their consent, or if they do not actively see adult patients. An incomplete response was defined as failure to submit the questionnaire on the last page.
Statistical Analysis
To test differences between categorical variables, the Pearson chi-square test was used. p values < 0.05 were deemed statistically significant. Multivariate logistic regression was used to identify independent factors associated with the use of additional imaging modalities. All data analysis was completed using SPSS version 21 (IBM Corp, Armonk, NY, USA).
Results
Survey Participants
An email invitation was sent to 12,485 gastroenterology and hepatology providers. There were 777 responses (6.2 %); 119 were excluded because they did not complete the survey (n = 45), did not actively see adult patients (n = 12), did not provide their consent (n = 3), or a combination of the above factors (n = 59), resulting in 656 eligible for analysis.
Table 1 contains background information on the entire participating population, including provider type and attributes of medical practices. Thirty-seven percent (n = 150) of gastroenterologists and 77 % (n = 141) of hepatologists are affiliated with transplant centers.
Surveillance Practices and Opinions
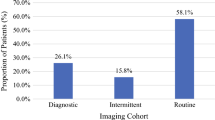
Ninety-seven percent (n = 638) reported placing cirrhotic patients under HCC surveillance, and 92 % (n = 604) did so independent of etiology. Ninety-three percent (n = 609) use US in some capacity, but only 36 % (n = 234) exclusively use US. Sixty-percent of providers (n = 390) use two or more imaging modalities. Seventy-seven percent (n = 507) use AFP (alpha-fetoprotein) for surveillance, 9 % (n = 58) use AFP-L3, 3 % (n = 19) use DCP (Des-gamma carboxyprothrombin), and 6 % (n = 42) use two or more of the three biomarkers for HCC surveillance (Table 2).
Forty-five percent (n = 297) implement specific US protocols for HCC surveillance, which might include additional time, better documentation accounting for factors like quality, or other specific instructions. Additionally, 16 % (n = 104) indicated that they use specific sonographers for surveillance.
Provider opinions on the use of surveillance varied, and 87 % (n = 569) believe that a failure to place an at-risk patient under HCC surveillance poses a malpractice liability. Approximately 50 % (n = 329) think that surveillance is cost-effective for all cirrhotics, including Child-Pugh C patients who are ineligible for liver transplantation.
Providers Who Use Additional Imaging Modalities for Surveillance
All provider types affiliated with transplant centers (n = 337) were compared with those practicing at non-transplant centers (n = 310). Relative to non-transplant center providers, transplant center providers favor using additional imaging modalities such as CT and MRI for surveillance, instead of US alone (p = 0.046).
The variables associated with transplant center providers using additional imaging modalities for surveillance were analyzed univariately. A provider having 10–15 years of experience (p = 0.029), allocating more than 75 % of professional time to patient care (p = 0.008), and seeing more than 20 new cirrhotic patients per month (p = 0.027), were statistically significant.
In multivariate analysis, transplant center providers allocating more than 75 % of their time to patient care (p = 0.004) and transplant center providers seeing more than 20 new cirrhotic patients per month (p = 0.033) remained statistically significant (Table 3).
Discussion
This is the first national survey of gastroenterologists and hepatologists about HCC surveillance since the issuance of clinical practice guidelines. Because of the sample size, the results cannot be generalized broadly to all providers who utilize HCC surveillance. However, we found some interesting results from the 656 gastroenterologists and hepatologists whose responses were analyzed. These providers reported very high rates of general surveillance utilization (97 %), ordering of surveillance tests for cirrhotic patients independent of etiology (92 %), and the use of a biannual surveillance interval (79 %). These findings are not only highly congruent with practice guidelines [4–6], but are significantly higher than previously published studies [8, 9]. This suggests that these participating providers represent a group that is most likely more engaged and informed about HCC surveillance.
Conversely, the same providers strongly deviated from the guidelines in their utilization of surveillance tests, particularly imaging modalities. All major liver societies recommend using US for HCC surveillance [4–6]. Furthermore, biannual US and AFP were effective in reducing HCC-related mortality in the seminal randomized control trial [10]. Multiple analyses have shown US to be the only cost-effective surveillance modality [11, 12]. Yet, only 36 % of respondents exclusively use US; 60 % of participating providers use additional imaging modalities such as CT and/or MRI. We observed that transplant center providers favor using additional imaging modalities such as CT and MRI, instead of US alone. This practice difference was not observed when comparing gastroenterology to hepatology providers. While the performance of US in detecting early HCC lesions is limited [13] and worse than CT and MRI [14], no data exist to support the use of CT and MRI in the surveillance context. We did not ask why responding providers favored using additional imaging modalities. It might be that a provider at a transplant center is more cognizant of the need to detect HCC lesions as early as possible to keep a patient eligible for curative treatment such as liver transplantation. Chalasani et al. [15] found that CT has an 88 % sensitivity compared with 59 % for US among cirrhotic, transplant candidates undergoing surveillance. A provider’s concern with the sensitivity of US in detection of early HCC during surveillance is not unjustified. In their retrospective study of patients from the HALT-C cohort, Singal et al. [16] found that inadequate sensitivity of US explained 70 % of all late HCC cases detected beyond the Milan criteria as opposed to gaps in follow-up or compliance.
In their meta-analysis, Singal et al. [17] found that the sensitivity values of US for detecting early HCC (less than 5 cm in diameter) during surveillance varies from 23 to 98 %, with a pooled sensitivity of 69 %. All of the included studies were conducted in Europe and Japan and none were from the USA, where more than one-third of all adults are obese [18]. While it is suspected that the operator, equipment, patient characteristics, and potentially other factors contribute to US quality, no evidence exists to support these claims. Although not proven, in cases where US quality is poor, it is highly likely that sensitivity may also be poor. More studies are needed to demonstrate this link, and a cost-effective alternative imaging modality may be needed.
Some providers implement additional measures to improve ultrasound’s effectiveness for surveillance, like specific protocols and sonographers. Nearly half of all providers claimed to use specific protocols, and 16 % utilize specific sonographers for surveillance. EASL-EORTC [5] is the only major society to recommend specialized training for sonographers.
In their recent meta-analysis, Singal et al. [19] found that surveillance in cirrhotics was significantly associated with the improved detection of early HCC, increased use of curative treatments, and improved survival. Yet, in the USA, HCC surveillance is plagued by a very low rate. In their literature review, Singal et al. [9] reported a utilization rate of 18.4 % for cirrhotic patients in the USA, which might even be an overestimation. Furthermore, they observed a striking disparity in utilization when comparing gastroenterologists, 51.7 %, to primary care physicians (PCP), 16.9 %. Davila et al. [8] found that cirrhotic patients followed by both gastroenterologists and PCP were 4.5 times more likely to be placed under regular surveillance compared with those followed exclusively by PCP. This finding is particularly relevant since many cirrhotic patients are only followed by PCP. Moreover, when these findings are coupled with the rising incidence of HCC in the USA [20, 21], it creates an added sense of urgency for greater engagement with PCP about HCC and surveillance.
The principal limitation of our study is that it does not reflect all providers who utilize HCC surveillance. First, the study does not contain data from PCPs. Also, not all gastroenterologists and hepatologists in the USA are members of the AMA. Our response rate, 6.2 %, was lower than other recently published surveys of gastroenterologists [22, 23]. Additionally, we were unable to obtain the demographic information of non-respondents and compare it with respondents, positing a non-response bias.
In summary, our study shows that the surveillance practices from this sample, providers who are likely well engaged and informed about HCC surveillance, are not uniform and deviate from clinical practice guidelines most conspicuously when it comes to the utilization of surveillance imaging modalities. The use of additional imaging modalities is highly influenced by a provider’s affiliation with transplant centers. More studies are needed to define the underlying factors that mediate US’s wide-ranging sensitivity, which seems to cause providers to use alternative tests and imaging modalities, even if they are not recommended by clinical practice guidelines [4–6].
References
Altekruse SF, McGlynn KA, Dickie LA, Kleiner DE. Hepatocellular carcinoma confirmation, treatment, and survival in surveillance, epidemiology, and end results registries, 1992–2008. Hepatology. 2012;55:476–482.
El-Serag HB. Hepatocellular carcinoma. N Engl J Med. 2011;365:1118–1127.
Chalasani N, Said A, Ness R, Hoen H, Lumeng L. Screening for hepatocellular carcinoma in patients with cirrhosis in the United States: results of a national survey. Am J Gastroenterol. 1999;94:2224–2229.
Bruix J, Sherman M. Management of hepatocellular carcinoma. Hepatology. 2005;42:1208–1236.
EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol 2012;56:908–943.
Benson AB 3rd, Abrams TA, Ben-Josef E, et al. NCCN clinical practice guidelines in oncology: hepatobiliary cancers. J Natl Compr Canc Netw. 2009;7:350–391.
Harris PA, Taylor R, Thielke R, et al. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377–381.
Davila JA, Morgan RO, Richardson PA, et al. Use of surveillance for hepatocellular carcinoma among patients with cirrhosis in the United States. Hepatology. 2010;52:132–141.
Singal AG, Yopp A, C SS et al. Utilization of hepatocellular carcinoma surveillance among American patients: a systematic review. J Gen Int Med 2012.
Zhang BH, Yang BH, Tang ZY. Randomized controlled trial of screening for hepatocellular carcinoma. J Cancer Res Clin Oncol. 2004;130:417–422.
Andersson KL, Salomon JA, Goldie SJ, Chung RT. Cost effectiveness of alternative surveillance strategies for hepatocellular carcinoma in patients with cirrhosis. Clin Gastroenterol Hepatol. 2008;6:1418–1424.
Pocha C, Dieperink E, McMaken KA, et al. Surveillance for hepatocellular cancer with ultrasonography vs. computed tomography—a randomised study. Aliment Pharmacol Ther. 2013;38:303–312.
Kim CK, Lim JH, Lee WJ. Detection of hepatocellular carcinomas and dysplastic nodules in cirrhotic liver: accuracy of ultrasonography in transplant patients. J Ultrasound Med. 2001;20:99–104.
Yu NC, Chaudhari V, Raman SS, et al. CT and MRI improve detection of hepatocellular carcinoma, compared with ultrasound alone, in patients with cirrhosis. Clin Gastroenterol Hepatol. 2011;9:161–167.
Chalasani N, Horlander JC Sr, Said A, et al. Screening for hepatocellular carcinoma in patients with advanced cirrhosis. Am J Gastroenterol. 1999;94:2988–2993.
Singal AG, Nehra M, Adams-Huet B, et al. Detection of hepatocellular carcinoma at advanced stages among patients in the HALT-C trial: Where did surveillance fail? Am J Gastroenterol. 2013;108:425–432.
Singal A, Volk ML, Waljee A, et al. Meta-analysis: surveillance with ultrasound for early-stage hepatocellular carcinoma in patients with cirrhosis. Aliment Pharmacol Ther. 2009;30:37–47.
Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of obesity in the United States, 2009–2010. NCHS data brief 2012:1–8.
Singal AG, Pillai A, Tiro J. Early detection, curative treatment, and survival rates for hepatocellular carcinoma surveillance in patients with cirrhosis: a meta-analysis. PLoS Med. 2014;11:e1001624.
Altekruse SF, McGlynn KA, Reichman ME. Hepatocellular carcinoma incidence, mortality, and survival trends in the United States from 1975 to 2005. J Clin Oncol. 2009;27:1485–1491.
El-Serag HB. Hepatocellular carcinoma: recent trends in the United States. Gastroenterology. 2004;127:S27–S34.
Saab S, Nguyen S, Ibrahim A, Vierling JM, Tong MJ. Management of patients with cirrhosis in Southern California: results of a practitioner survey. J Clin Gastroenterol. 2006;40:156–161.
Khalili M, Guy J, Yu A, et al. Hepatitis B and hepatocellular carcinoma screening among Asian Americans: survey of safety net healthcare providers. Dig Dis Sci. 2011;56:1516–1523.
Acknowledgments
Wako Diagnostics provided grant support for our data collection through the American Medical Association. Wako was not involved in any research activities up to and including study design, data analysis, data interpretation, and manuscript preparation and manuscript revisions.
Confilct of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Joshi, K., Mendler, M., Gish, R. et al. Hepatocellular Carcinoma Surveillance: A National Survey of Current Practices in the USA. Dig Dis Sci 59, 3073–3077 (2014). https://doi.org/10.1007/s10620-014-3256-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-014-3256-6