Abstract
Background
Gastric emptying scintigraphy (GES) using a solid meal is often recommended for the evaluation of gastroparesis. However, some patients cannot tolerate the standardized egg-white sandwich (EWS) solid meal and an alternative meal is needed.
Aim
The aim of this study was to compare GES, regional gastric emptying, and gastric contractility using a liquid nutrient meal (LNM; Ensure Plus®) to those using EWS.
Methods
Twenty healthy volunteers underwent GES using EWS and LNM on separate days. Gastric emptying was measured using scintigraphy and with a wireless motility capsule (WMC; SmartPill®).
Results
The gastric emptying half-time with LNM was similar to that with EWS (1.41 ± 0.11 vs 1.52 ± 0.08 h; P = 0.28) and the two were significantly correlated (r = 0.53; P = 0.017). There were time-related differences in gastric emptying of the LNM compared to EWS: in the first hour, gastric retention of EWS was slightly greater than that of LNM, whereas at 3 and 4 h, gastric retention of EWS was slightly less than that of LNM. Regionally, the percentage retention of the meal in the proximal stomach was greater for EWS than for LNM at 0.5 h. WMC gastric emptying times and gastric contractility for the two meals were not significantly different.
Conclusions
Overall gastric emptying of the LNM was similar to that of the EWS meal. The LNM empties without a lag phase and takes slightly longer to empty from the distal stomach, likely due to its higher fat content. These differences are likely due to early accommodation with retention of solids in the proximal stomach and the need for trituration of solids. We conclude that this LNM can serve as an alternative to the conventional solid EWS for GES.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gastroparesis is a chronic motility disorder characterized by delayed gastric emptying in the absence of mechanical gastric outlet obstruction [1]. The most common categories of gastroparesis are diabetic, idiopathic, and post-surgical. The diagnosis of gastroparesis is based on dyspeptic symptoms compatible with gastroparesis, no abnormalities visualized by upper endoscopy, and evidence of delayed gastric emptying.
Traditional assessment of gastric emptying in patients with dyspeptic symptoms involves the use of a radiolabeled solid meal. However, various technical and patient-related factors can affect the results of gastric emptying scintigraphy (GES) [2]. A 4-h GES test using a standard low-fat, egg-white sandwich (EWS) meal, as proposed in the consensus guideline by the American Neurogastroenterology and Motility Society and the Society of Nuclear Medicine, is considered the standard for measuring gastric emptying [3, 4], but some patients are unable to tolerate eggs or have celiac disease and are unable to consume the bread. An alternative meal is therefore needed for such patients.
A high-caloric, high-fat liquid nutrient meal (LNM), such as Ensure® (Abbott Nutrition, Columbus, OH), has been used to assess gastric emptying in patients who are unable to tolerate a solid meal. Ensure® has also been used as a test meal in other tests of gastric function, such as electrogastrography, satiety testing, and antroduodenal manometry [5]. Ensure® has the advantage of requiring no preparation and can be used in patients who cannot tolerate or have allergies to eggs and/or bread. To date, normative data for the gastric emptying of Ensure® are not available.
The aim of our study was to compare GES using a LNM (Ensure Plus®; Abbot Nutrition) to that using a low-fat, liquid EWS meal in healthy male and female volunteers. We also wanted to compare the regional distribution of the two meals during gastric emptying using scintigraphy and to assess any changes in gastric contractility using a wireless motility capsule (WMC) during emptying of the two different meals from the stomach.
Methods
Overview
Using a protocol that was approved by our Institutional Review Board and after obtaining informed consent from the subjects, 20 healthy volunteers (10 males, 10 females) were recruited for participation in this study. Each normal subject had two gastric emptying studies: one using the EWS meal and another using a LNM (Ensure Plus®) on separate days in a randomized fashion. A WMC (SmartPill®; SmartPill Corp, Buffalo, NY) was used to assess gastric contractility during each gastric emptying test. Ensure Plus® was used in this study rather than Ensure® since the caloric content of the former better approximates the caloric content of the EWS meal.
Study Subjects
To qualify as a normal healthy subject for this study, volunteers were required to have a standard physical examination and be assessed as having none of the following: gastrointestinal symptoms, prior history of peptic ulcer disease, symptoms of functional dyspepsia or irritable bowel syndrome, or prior surgery on the esophagus, stomach, or small intestine. Subjects were excluded if they were taking medications for gastrointestinal disorders, opioid narcotic analgesic medication, intravenous recreational drugs, anti-cholinergic agents, prokinetic compounds, or non-steroidal anti-inflammatory agents (except for low-dose aspirin). In addition, subjects with egg allergy or a known diagnosis of celiac sprue were excluded. Female subjects were also excluded if they were pregnant, trying to become pregnant, or breast feeding. Women were studied in the luteal phase of their menstrual cycle. All pre-menopausal women had a urine pregnancy test prior to participation to exclude pregnancy.
Gastric Emptying Scintigraphy
Subjects were studied twice in random order, once using the EWS meal and then again using the LNM. Each subject was studied following an overnight fast. The low-fat, EWS meal consisted of 120 ml liquid egg white (Egg Beaters®; ConAgra Foods, Omaha, NE) mixed with 0.5 mCi Tc-99m–sulfur colloid and cooked in a microwave oven for 2 min on high power until completely solid. This was served with two slices of white bread, 1 ounce of jam and 120 ml water [4]. The high-caloric LNM consisted of 237 ml Ensure Plus® mixed with 0.5 mCi Tc-99m–sulfur colloid. The low-fat, EWS meal has a caloric value of 255 kcal and contained 2 % fat, 24 % protein, 72 % carbohydrate, and 2 % fiber, while the LNM had a caloric value of 350 kcal and contained 28 % fat, 15 % protein, 57 % carbohydrate, and no fiber (http://abbottnutrition.com/products/ensure-plus). The tests were done 2 days apart to allow for complete decay of the technetium-99m for the second study and to help assure that the pre-menopausal women were evaluated in the same luteal phase of their menstrual cycle for the two tests. Images, both anterior and posterior views, with the subject standing, were obtained immediately after meal ingestion and every 30 min thereafter for 4 h using a large field-of-view gamma camera with the energy window set to the 140-keV photopeak of Tc-99m ± 10 % [3]. In between images, subjects were generally sitting, but they could remain standing. Regions were manually defined on each image using a cursor. The geometric mean of the decay-corrected anterior and posterior counts was calculated for each time point. Gastric emptying was analyzed as the percentage of radioactivity retained in the stomach over time [6]. The percentage gastric retention at each time interval was determined for the proximal, distal, and whole stomach separately by drawing separate regions and calculating the geometric mean counts for each. The regions for the proximal and distal stomach were defined as previously reported [7, 8]. The lag phase and gastric emptying half-time (t1/2) were calculated for each meal by fitting the percentage retention versus time to a modified power exponential function [7].
Wireless Motility Capsule
With each meal, subjects also ingested the WMC (SmartPill®) with 100 ml water, immediately after consumption of the radiolabeled meal. Gastric emptying time using the wireless capsule was defined as the time taken from ingestion until the pH changed from acidic to alkaline (marked by a sudden rise in pH of >3.0) as the capsule entered the duodenum [9]. In addition, a motility index (MI; mmHg × s)/min) [10] was calculated from the pressure data for 15-min intervals prior to WMC gastric emptying using the GIMS Data Viewer v2.2 (SmartPill Corp).
Statistical Analysis
Results are expressed as mean ± standard error of the mean. In some instances, the standard deviation (SD) is used, where noted. Student’s t test for continuous variables and χ2 analysis for proportions were used to compare the two meals (EWS vs. LNM). Pearson’s correlation coefficient was used to calculate correlations between the two meals. Data were analyzed using SPSS v17.0 statistical software (SPSS, Chicago, NY).
Results
Study Subjects
Twenty healthy subjects were recruited to the study, of whom ten were males [mean age 39.8 ± 3.1 years; mean weight 193.8 ± 9.3 lbs; body mass index (BMI) 26.5 ± 1.3)] and ten were females (mean age 42.6 ± 3.4 years; mean weight 138 ± 5.6 lbs; BMI 23.3 ± 1.0). All subjects met the inclusion criteria and none had a history of diabetes.
Total Gastric Emptying
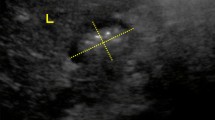
Figure 1 shows representative gastric emptying scintigraphy images for each of the two meals. Emptying of the radioactive meal from the stomach into the small intestine was observed over time.
Sample images from a normal subject undergoing the two gastric emptying tests—one with liquid nutrient meal (Ensure Plus®) and one with egg-white sandwich. Emptying of the radioactive meal from the stomach into the small intestine is seen over time. The two different test meals are associated with regional and time-related differences in meal emptying
Figure 2 shows the gastric emptying of each meal expressed as the percentage retention in the stomach over the 4-h period following meal ingestion. The mathematically calculated lag phase was significantly less for the LNM meal than for the EWS meal (0.74 ± 0.11 vs. 1.19 ± 0.08 h, respectively; P < 0.01). The t½ for the LNM was similar to that for the EWS meal (1.41 ± 0.11 vs 1.52 ± 0.08 h, respectively; P = 0.28), and the two were significantly correlated (r = 0.53, P = 0.017). For the LNM, there was 64.45 ± 13.61 % (SD) gastric retention at 1 h; 31.95 ± 18.55 % gastric retention at 2 h; 6.08 ± 8.25 % gastric retention at 4 h. There were slight, but significant, time-related differences in the emptying of the high-fat LNM meal compared to the EWS meal. In the first hour, gastric retention of the EWS meal was greater than that of the LNM meal (74 ± 3 vs. 64 ± 3 %, respectively; P = 0.014). At 1.5, 2, and 2.5 h after meal ingestion, there was no difference between the EWS and LNM meals in terms of retention. In the later time points of the study, retention of the low-fat, EWS meal was less than that of the LNM meal (8 ± 2 vs. 15 ± 2 %, respectively at 3 h; P = 0.022; 2 ± 1 vs. 6 ± 2 %, respectively, at 4 h; p = 0.012).
Gastric emptying in our male and female subjects was compared (Fig. 3a, b). Gastric emptying of the solid EWS meal at 2 h was slower in females than males (38.1 ± 4.7 vs 22.1 ± 2.6 % retention, respectively; P = 0.01) but not different at 4 h (2.34 ± 1.0 vs. 1.07 ± 0.1 % retention, respectively; P = 0.25). There were no significant gender differences in gastric emptying of the LNM at 2 h (36.5 ± 7.7 vs 27.4 ± 3.0 % retention; P = 0.29) or at 4 h (8.4 ± 3.5 vs. 3.7 ± 0.9 % retention; P = 0.22). For each gender, there was an increase in gastric retention in the early phase of gastric emptying (30 and 60 min) for the EWS and less retention at later stages of gastric emptying (3 and 4 h). The t1/2 values for females were 1.73 ± 0.11 h for EWS and 1.59 ± 0.20 for LNM (P = 0.430), and those for males were 1.31 ± 0.06 for EWS and 1.24 ± 0.08 for LNM (P = 0.449).
Regional Gastric Emptying
Regional gastric emptying was expressed as the percentage of radioactivity in both the proximal and distal stomach over time (Fig. 4). Regionally, movement of the meal from the proximal to the distal stomach differed between the meals. The percentage of meal in the proximal stomach for the low-fat, EWS meal was greater than that for LNM at 0.5 h (61 ± 3 vs. 34 ± 4 %, respectively; p = 0.001). Thereafter, there was similar retention of the meal in each gastric region for each meal.
Wireless Motility Capsule
The gastric emptying time of the WMC was not significantly different between the two meals [209 ± 13 (LMN) vs. 236 ± 13 (EWS) min; p = 0.12]. The WMC MI was similar for the EWS and LNM. With each meal, there was an increase in the MI in the 30 down to 15 min prior to gastric emptying (EWS: from 64 ± 15 to 116 ± 19; p = 0.007; LNM: from 79 ± 15 to 134 ± 24; P = 0.035. There were no differences in the motility indices between the meals prior to WMC emptying into the duodenum [30 min: 64 ± 19 (EWS) vs. 79 ± 15 (LMN); P = 0.76; 15 min: 116 ± 19 vs. 134 ± 24; P = 0.56).
Discussion
In this study we compared the gastric emptying characteristics of two meals, namely, the conventional low-fat, EWS meal and a LNM (Ensure Plus®), to determine if the LNM might be a suitable alternative to the EWS meal. Our results show that the gastric emptying of the two meals was correlated and that the overall gastric emptying time for these two meals was similar, as demonstrated by the t½ for each meal. Hence, in patients who cannot tolerate the solid EWS, a LNM can be used as an alternative.
In addition to measuring total gastric emptying, we also assessed regional gastric emptying from the proximal and distal stomach. There were temporal and regional differences in gastric emptying of the low-fat, EWS meal and the higher fat-containing LNM. In the early stages of gastric emptying (60 min), proximal gastric retention and overall gastric retention of the EWS were increased compared to the LNM. These differences are likely due to early retention of the solids of the EWS meal in the proximal stomach and the need for trituration of solids following initial accommodation. The LNM, on the other hand, leaves the proximal stomach with a shorter lag phase, likely due to its liquid characteristics. There was a slight increased gastric retention of the LNM at the later stages of gastric emptying, likely due to its higher fat content compared to the EWS. Nutrient liquids have been shown not to empty exponentially, although the rate of emptying of liquids is influenced by the presence of a solid meal in the stomach [11]. A LNM empties at about the same rate as an equicaloric solid meal after the lag phase [11, 12]. The longer lag time with the EWS than with the LNM meal is consistent with the necessity for trituration of solids [13].
We also found that both meals underwent a similar near-complete gastric emptying at 4 h in both men and women. This similar gastric emptying for the EWS meal for males and females at 4 h was also observed in the initial studies on the EWS meal by Tougas et al. [4]. There was, however, slower gastric emptying with an increase in gastric retention at 2 h for the EWS meal in females compared to males. Various studies have suggested that gastric emptying can be affected by gender [14–19]. Gender-related differences have also been reported to be present in the proximal stomach, affecting motility and perception [20]. Although gastric emptying can also be affected by the phase of the menstrual cycle in pre-menopausal women [14, 16], we minimized this possibility by performing the two gastric emptying tests within 2 days of each other.
Our results suggest that an Ensure®-based LNM may be an acceptable meal for GES in patients unable to tolerate an egg sandwich meal due to an inability to ingest solid food, food allergies, gluten sensitivity, and/or dietary or religious preferences. To date, our study is the largest to study LNM in normal subjects. Previous studies have suggested that gastric emptying time does not correlate well with dyspeptic symptoms in patients with delayed gastric emptying [21]. Differences in regional emptying have been suggested to play a role in causing gastroparesis systems, even though the overall gastric emptying time may be normal [22]. The LNM empties without a lag phase and does not need trituration, as does the solid meal. Differences in fat content of the test meals may also play a role in dyspeptic symptoms [23]. The use of a high-caloric, high-fat LNM may help the clinician to recognize a separate sub-group of patients who present with dyspeptic symptoms and delayed gastric emptying. Further studies comparing the differences in gastric emptying in patients with dyspeptic symptoms are needed to characterize the clinical utility of this LNM. The availability of an alternative meal opens a new window on potentially understanding the complexities of gastric motility and will require further studies to understand the clinical implications for management and treatment of patients with dyspepsia.
In this study we also used the WMC to continuously record pH, pressure, and temperature as it traversed the gastrointestinal tract. Gastric emptying time using this device is measured by the time it takes for the capsule pH to change from acidic (in the stomach) to alkaline (when it enters the duodenum). The WMC has been shown in earlier studies to have a good correlation with results of a gastric emptying scintigraphy with a standard low-fat, EWS meal [9]. We also used the WMC used to record gastric contractions for measurement purposes. Previous studies have shown that emptying of a non-digestible solid occurs with the return of the phase III migrating motor complex (MMC), which occurs after the emptying of the solid meal; a good correlation between time to first phase III MMC and WMC gastric residence time was found [24]. For both the EWS and the LNM, there was an increase in the MI in the time period immediately preceding the emptying of the capsule, suggesting that the capsule empties from the stomach with the return of the phase III MMC after converting back from the postprandial fed state to the fasting state. Both EWS and LNM induced the postprandial fed state, which may be an important finding when using the LNM compared with a non-nutrient water meal which may not challenge the gastroparetic patient’s stomach to simulate their problem after meal ingestion. The reliability of the capsule in detecting contractions at different times may be influenced by the location of the capsule in the stomach (e.g., fundus, body, or antrum) which is not precisely known during the recording phase.
Other studies have looked at the gastric emptying of a similar LNM. The aim of the study of Kwiatek et al. [25] was to gain a better understanding of the mechanism of gastric emptying as it relates to the fundic pressure-pump theory of liquid emptying. Kwiatek et al. used magnetic resonance imaging (MRI), which enables the imaging of changes in gastric volume, but is not quantitative in terms of tracking the emptying of the meal. A study by Fruehauf et al. [26] also used MRI and a LNM, but again focused on gastric volume changes rather than gastric emptying. The focus of these other studies differed from that of our study in that our aim was to determine whether a radiolabeled LNM might be a suitable replacement for the current standard radiolabeled low-fat, egg-white sandwich test meal in patient evaluations.
In conclusion, we found that the overall gastric emptying time was similar for a high-calorie LNM and a low-fat, EWS meal. This result suggests that a high-calorie LNM can be used as an alternative meal in patients who cannot tolerate the conventional egg white sandwich meal for GES.
References
Parkman HP, Hasler WL, Fisher RS. American Gastroenterological Association technical review on the diagnosis and treatment of gastroparesis. Gastroenterology. 2004;127:1592–1622.
Christian PE, Datz FL, Sorenson JA, et al. Technical factors in gastric emptying studies. J Nucl Med. 1983;24:264–268.
Abell TL, Camilleri M, Donohoe K, et al. Consensus recommendations for gastric emptying scintigraphy: a joint report of the American Neurogastroenterology and Motility Society and the Society of Nuclear Medicine. Am J Gastroenterol. 2008;103:753–763.
Tougas G, Eaker EY, Abell TL, et al. Assessment of gastric emptying using a low-fat meal: establishment of international control values. Am J Gastroenterol. 2000;95:1456–1462.
Parkman HP, Jones MP. Tests of gastric neuromuscular function. Gastroenterology. 2009;136:1526–1543.
Guo J-P, Maurer AH, Urbain J-L, Fisher RS, Parkman HP. Extending gastric emptying scintigraphy from two to four hours detects more patients with gastroparesis. Dig Dis Sci. 2001;46:24–29.
Knight LC, Parkman HP, Brown KL, et al. Delayed gastric emptying and decreased antral contractility in normal premenopausal women compared with men. Am J Gastroenterol. 1997;92:968–975.
Urbain JL, Siegel JA, Charkes ND, Maurer AH, Malmud LS, Fisher RS. The two-component stomach: effects of meal particle size on fundal and antral emptying. Eur J Nucl Med. 1989;15:254–259.
Kuo B, McCallum RW, Koch K, et al. Comparison of gastric emptying of a non-digestible capsule to a radiolabeled meal in healthy and gastroparetic subjects. Aliment Pharmacol Ther. 2008;27:186–196.
Ouyang A, Sunshine AG, Reynolds JC. Caloric content of a meal affects duration but not contractile pattern of duodenal motility in man. Dig Dis Sci. 1989;34:528–536.
Horowitz M, Maddox A, Bochner M, et al. Relationships between gastric emptying of solid and caloric liquid meals and alcohol absorption. Am J Physiol. 1989;257:G291–G298.
Siegel JA, Krevsky B, Maurer AH, Charkes ND, Fisher RS, Malmud LS. Scintigraphic evaluation of gastric emptying: are radiolabeled solids necessary? Clin Nucl Med. 1989;14:40–46.
Meyer JH, MacGregor IL, Gueller R, Martin P, Cavalieri R. 99mTc-tagged chicken liver as a marker of solid food in the human stomach. Am J Dig Dis. 1976;21:296–304.
Datz FL, Christian PE, Moore J. Gender-related differences in gastric emptying. J Nucl Med. 1987;28:1204–1207.
Hutson WR, Roehrkasse RL, Wald A. Influence of gender and menopause on gastric emptying and motility. Gastroenterology. 1989;93:934–940.
Gill RC, Murphey PD, Hooper HR, et al. Effect of the menstrual cycle on gastric emptying. Digestion. 1987;36:168–174.
Mones J, Carrio I, Calabuig R, et al. Influence of the menstrual cycle and of menopause on the gastric emptying rate of solids in female volunteers. Eur J Nucl Med. 1993;20:600–602.
Horowitz M, Maddern GJ, Chatterton BE, et al. The normal menstrual cycle has no effect on gastric emptying. Br J Obstet Gynecol. 1985;92:743–746.
Camilleri M, Iturrino J, Bharucha AE, et al. Performance characteristics of scintigraphic measurement of gastric emptying of solids in healthy participants. Neurogastroenterol Motil. 2012;24:1076–e562.
Mearadji B, Penning C, Vu MK, et al. Influence of gender on proximal gastric motor and sensory function. Am J Gastroenterol. 2001;96:2066–2073.
Pasricha PJ, Colvin R, Yates K, et al. Characteristics of patients with chronic unexplained nausea and vomiting and normal gastric emptying. Clin Gastroenterol Hepatol. 2011;9:567–576.
Troncon LE, Bennett RJ, Ahluwalia NK, et al. Abnormal intragastric distribution of food during gastric emptying in functional dyspepsia patients. Gut. 1994;35:327–332.
Lin HC, Van Citters GW, Zhao XT, et al. Fat intolerance depends on rapid gastric emptying. Dig Dis Sci. 1999;44:330–335.
Cassilly D, Kantor S, Knight L, Maurer A, Fisher RS, Parkman HP. Gastric emptying of a nondigestible solid: assessment with simultaneous SmartPill pH and pressure capsule, antroduodenal manometry, gastric emptying scintigraphy. Neurogastroenterol Motility. 2008;20:311–319.
Kwiatek MA, Menne D, Steingoetter A, et al. Effect of meal volume and calorie load on postprandial gastric function and emptying: studies under physiological conditions by combined fiber-optic pressure measurement and MRI. Am J Physiol Gastrointest Liver Physiol. 2009;297:G894–G901.
Fruehauf H, Menne D, Kwiatek MA, et al. Inter-observer reproducibility and analysis of gastric volume measurements and gastric emptying assessed with magnetic resonance imaging. Neurogastroenterol Motil. 2011;23:854–861.
Acknowledgments
This study was supported in part by a NIH Midcareer Investigator Award in Patient-Oriented Research to H.P. Parkman (NIH DK02921).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sachdeva, P., Kantor, S., Knight, L.C. et al. Use of a High Caloric Liquid Meal as an Alternative to a Solid Meal for Gastric Emptying Scintigraphy. Dig Dis Sci 58, 2001–2006 (2013). https://doi.org/10.1007/s10620-013-2665-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-013-2665-2