Abstract
Background and Study Aims
Among cases of difficult biliary cannulation, alternatives include use of a pancreatic duct stent (PDS) or guidewire (PDW) to facilitate access. We compared the effectiveness of a PDS versus a PDW to facilitate common bile duct (CBD) cannulation.
Patients and Methods
We conducted a randomized, crossover trial at two endoscopy referral centers, limited to patients undergoing ERCP without a history of biliary sphincterotomy. After meeting predefined criteria for difficult cannulation, patients were randomized to using a PDS or PDW to facilitate CBD cannulation. Outcomes included cannulation rate within 6 min, overall cannulation rate, frequency of precut, and complication rates.
Results
Among 442 eligible patients, 87 (19.7 %) met criteria for difficult cannulation. Forty two were randomized to PDW, 54 to PDS (including 9 PDW patients crossed over to PDS). The rate of CBD cannulation within 6 min was similar in the PDW (38.1 %) and PDS (51.9 %) groups (p = 0.18). In a secondary analysis limited to patients who successfully underwent PDW or PDS deployment, the rate was also comparable (PDW 59.3 %, PDS 65.1 %; p = 0.62). The overall frequency of CBD cannulation was 66.7 % in PDW and 90.7 % in PDS patients. Precut was required in 9.5 % of PDW and 25.9 % of PDS patients. Complication rates were similar, with 4 (4.6 %) patients having post-ERCP pancreatitis and 1 (1.1 %) having post-ERCP pain without confirmation of pancreatitis.
Conclusions
Use of a PDS or PDW facilitates CBD cannulation while maintaining a low complication rate and reducing the need for precut sphincterotomy in the majority of cases.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Selective biliary cannulation while minimizing manipulation of the papilla reduces the probability of ERCP-associated complications of pancreatitis and hemorrhage [1]. There is conclusive evidence that a precut sphincterotomy and one or more injections of the pancreatic duct (PD) significantly increase the rate of post-ERCP pancreatitis, and complications of precut are highest among less experienced physicians who perform this intervention less than once per week [2]. Endoscopists will inevitably encounter the challenging papilla where PD cannulation can be achieved but deep access to the common bile duct (CBD) cannot. Although there is no consensus definition for “difficult” biliary cannulation, inadvertent manipulation of the PD and more than ten attempts to cannulate have been associated with higher rates of post-ERCP pancreatitis [1, 3]. In cases of challenging access to the bile duct, options to facilitate cannulation include persistence using standard techniques, precut/needle knife sphincterotomy or placement of a PD guidewire [4–7] or stent [8–10]. Use of a PD stent is an attractive option since multiple cannulation attempts is an independent risk factor of post-ERCP pancreatitis [1], and placement of a PD stent reduces the odds of this complication by nearly half (odds ratio 0.44) [11–16].
Some authors have advocated the use of earlier precut sphincterotomy compared to persistence using standard techniques, but these observations were limited to experienced endoscopists that perform ERCP at a consistently high volume [3, 17, 18]. Given the known risks of precut sphincterotomy [2, 19, 20], particularly among lower volume endoscopists, a less invasive technique (e.g., use of a PD guidewire or stent) with comparable efficacy may be more easily generalizable to less experienced ERCP practitioners. We have previously reported that using a PD stent facilitated bile duct cannulation without the need for precut in 79 % of cases, while achieving a favorable post-ERCP pancreatitis rate of 5.3 % [8]. However, this study was limited by its retrospective design and lack of a comparison group. The primary objective of this randomized clinical trial was to compare the efficacy and safety of using a PD stent versus a PD guidewire to facilitate CBD cannulation in cases of challenging access to the biliary tree. We hypothesized that while both approaches would improve biliary cannulation rates, the PD stent technique would be superior since we had anecdotally observed that managing two guidewires (one in the PD and one being used for biliary cannulation) was somewhat more cumbersome than leaving a stent deployed in the PD.
Methods
Patient Selection and Randomization
We conducted a randomized, clinical trial at two endoscopic referral centers (Washington University in St. Louis, St. Louis, MO, USA; Northwestern University, Chicago, IL, USA) involving six endoscopists, each having >1,000 ERCPs of experience. All endoscopists had experience using the PDS and PDW techniques to facilitate biliary cannulation prior to initiating this study. Prior to patient recruitment, the protocol was approved by the Institutional Review Board of each participating center and registered at www.ClinicalTrials.gov (NCT00919074). Eligible patients signed a written, informed consent prior to their ERCP. Patients who had never undergone a biliary or pancreatic sphincterotomy were included; we excluded those undergoing ERCP for suspected sphincter of Oddi dysfunction, having an indication of endoscopic pancreatic therapeutics, or unwilling or unable to provide informed consent. We also excluded patients with postoperative anatomy (e.g., Roux-en-Y, Billroth).
“Difficult” biliary cannulation was defined as failure by the attending physician to achieve deep biliary cannulation within 6 min or inadvertent cannulation (wire, injection, or both) of the PD three consecutive times. The clock began when the first cannulation attempt commenced. If an advanced endoscopy fellow was participating in the procedure, the attending was given an additional 6 min (or three inadvertent manipulations of the PD) to attempt cannulation prior to enrollment. We chose this interval based on a previous clinical trial studying cases of difficult biliary cannulation at an expert medical center [21]. In addition, we elected to randomize after three inadvertent manipulations of the PD since this event increases the risk of post-ERCP pancreatitis [1]. During these initial efforts, we did not mandate a specific cannulation technique, but a precut sphincterotomy was not permitted and wire-guided cannulation (either physician or assistant-controlled) using a short-tip traction sphincterotome and a straight tip, hydrophilic guidewire was usually the preferred primary approach [22].
We employed a single, blind, stratified randomization protocol based on participating institutions to assure equal representation of both study groups at each facility. When a patient met enrollment criteria during the procedure, we used concealed envelopes to randomize subjects in a 1:1 fashion to the PD guidewire (PDW) or PD stent (PDS) group. Patients were blinded to their assignment group but not the treating endoscopist. A randomization list was created using a computer-based number generator (Microsoft Excel, Microsoft Corp., Redmond, WA, USA). After randomization, further cannulation efforts were performed by the attending endoscopist.
PD Stent (PDS) Group
We have previously reported the technique of using a PDS to facilitate CBD cannulation [8]. Briefly, a guidewire (0.025 or 0.035″) is left in the mid-body of the pancreas to facilitate stent placement. The type of stent is left to the discretion of the endoscopist: either a 4 or 5Fr stent (2–9 cm in length) with an external pigtail and single internal flange (Freeman pancreatic stent® Hobbs Medical Inc., Stafford Springs, CT, USA) or a 5Fr stent with a double external and single internal flange (Geenen pancreatic stent®, Cook Medical, Bloomington, IN, USA). The internal flange is occasionally removed to promote spontaneous passage of the stent after the procedure. However, the internal flange was usually left in place to minimize the likelihood of spontaneous migration during efforts to cannulate the CBD. A pancreatic sphincterotomy is not performed. After stent deployment, the endoscopist uses a sphincterotome and guidewire to cannulate the CBD. In most cases, physician- or assistant-controlled wire-guided cannulation is employed [22]. If deep cannulation is not achieved after 6 min of effort (starting from the time of first attempt after the PDS was fully deployed), the endoscopist was allowed to persist in their efforts with or without performing a precut sphincterotomy. After 6 min, the decision to terminate the procedure is left to the treating physician.
PD Wire Group
The technique of PD guidewire-assisted cannulation has also been described previously [4–7]. To summarize, the endoscopist leaves a 0.025 or 0.035″ guidewire in the PD; the depth of wire insertion is not pre-specified, but ideally left beyond the genu whenever possible. Alongside the guidewire, the endoscopist uses a sphincterotome pre-loaded with a second guidewire (0.025 or 0.035″) to cannulate the bile duct. Similar to the PDS group, wire-guided cannulation is the preferred method to selectively cannulate the CBD.
If deep cannulation is not achieved after 6 min of effort (starting from the time of first attempt after the PD wire is locked in position and efforts to cannulate the CBD resume), the patient is crossed over to the PDS group. The technique for PDS deployment and further efforts at CBD cannulation are summarized above. If deep CBD cannulation is not achieved after cross-over and an additional 6 min of effort, the endoscopists could persist in their efforts with or without performing a precut sphincterotomy. The decision to terminate the procedure is left to the treating physician.
If deep PD cannulation could not be achieved after a minimum of 6 min of effort after randomization, the attending physician was allowed to proceed with freehand needle knife sphincterotomy to gain access. These patients were included in the intent-to-treat analysis but excluded from a descriptive, per protocol analysis as outlined in the statistical analysis section.
Outcome Measures
The primary outcome was “successful cannulation,” defined as deep cannulation with selective injection of radiographic contrast into the CBD within 6 min after PDS or PDW deployment. We chose this time interval to minimize futile efforts when a specific technique is ineffective and to allow crossover from PDW to PDS. In addition, standard practice by the participating endoscopists in this study is to try an alternate cannulation technique after a reasonable effort is ineffective. Secondary outcome measures included overall cannulation rate, frequency of precut sphincterotomy, cannulation time and complication rates. Specific complications of interest were post-ERCP pancreatitis, GI bleeding and perforation. We assessed for complications by telephone contact 1 week after the procedure or by review of hospital records if patients were admitted following the procedure.
Statistical Analysis
We used descriptive statistics (mean with standard deviation, and simple proportions) to present relevant patient and procedure characteristics. Comparative statistics (Pearson’s χ2 test) were employed to measure categorical differences between the two study groups. Since we allowed crossover from PDW to PDS if the primary outcome of CBD cannulation within 6 min was not reached, we did not conduct a formal comparison of overall cannulation times, precut sphincterotomy rates and overall cannulation rates. These results should be considered descriptive.
As there are limited published data on the PDW and PDS techniques, sample size estimates for the primary outcome of successful cannulation within 6 min are imperfect. However, based on our previous study utilizing PDS [8] and other trials of using a PDW to facilitate cannulation [23, 24], we predicted a 60 % success rate for the PDS group with a relative improvement in success of at least 30 % compared to the PDW group for the primary outcome of CBD cannulation within 6 min [4]. Any smaller difference between the two techniques for the primary outcome measure was felt to be clinically irrelevant. Therefore, we planned for a sample size of 108 cases (40 assigned to PDW and 68 assigned PDS group, including crossovers from the PDW group), to achieve 80 % statistical power to detect a difference in the primary outcome, with a two-sided alpha error of 5 %. The primary analysis included all patients who were randomized to the PDW and PDS groups, including patients who crossed over from PDW to PDS since this was planned a priori. We also conduct a secondary, exploratory analysis limited to patients in whom guidewire access to the PD was unsuccessful. The secondary analysis reflects the efficacy of each technique since it is limited to those patients in whom a PDW with or without a PDS could be placed. Since the protocol directed a crossover from the PDW group to the PDS group, those individuals that crossed were included in the primary analysis. We terminated patient recruitment after the targeted sample size was achieved in the PDW group but not the PDS group since the rate of crossover from PDW to PDS was lower than anticipated. An interim analysis revealed marginal differences for the primary outcome between the study groups, i.e., completing enrollment to the target sample size would not have impacted interpretation of the results.
Results
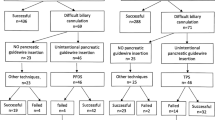
The study was conducted between September 2008 and January 2011. Of 442 potential candidates who provided consent prior to ERCP, 87 (19.7 %) met our criteria for difficult biliary cannulation and underwent randomization to the PDW (n = 42) and PDS (n = 45) groups. Among PDW subjects, 9 (21.4 %) were crossed over to the PDS group per protocol, increasing the PDS sample to 54 (Fig. 1). Baseline characteristics were similar between the study groups in terms of age, indication for ERCP, and relationship of the papilla to a diverticulum (if any) (Table 1). For patients in whom a PDS was deployed (n = 61), 58 (95.1 %) were 5Fr and ≥5 cm in length. There was no difference in the frequency of PDS placement between PDW (40.5 %) and PDS (42.6 %) patients (p = 0.84).
For the primary outcome of CBD cannulation within 6 min, the success rate of PDS (28/54, 51.9 %) and PDW (16/42, 38.1 %) patients was not significantly different (p = 0.18) (Table 2). When crossover patients are excluded, the success rate of the PDS group (n = 45) was 26/45, 57.8 % (p = 0.07 vs. PDW group). There was no difference in the frequency of successfully advancing a guidewire into the PD between the PDW (64.3 %) and PDS (79.6 %) groups (p = 0.09). In a secondary, exploratory analysis limited to patients in whom the PDW (n = 27) and PDS (n = 45) technique could be utilized, the cannulation rate within 6 min was 59.3 and 65.1 %, respectively (p = 0.62). Among nine patients who crossed over from the PDW to PDS group, two achieved cannulation within 6 min after PDS deployment, one after 6 min but without precut sphincterotomy, and all others (6) after precut.
Among patients randomized, 77 of 87 (88.5 %) achieved biliary cannulation during the first ERCP, 66.7 % in the PDW and 90.7 % in the PDS group. The higher rate in the PDS group is largely explained by the crossover of PDW patients who did not meet the primary outcome measure. A freehand needle knife sphincterotomy was performed in four of 42 (9.5 %) patients randomized to PDW, all of who failed PD cannulation (2 of whom subsequently achieved CBD cannulation) and 14 of 54 (25.9 %) patients in the PDS group (13 of whom subsequently achieved CBD cannulation). The average cannulation time was 17.0 ± 10.8 min in the PDW group and 23.6 ± 15.96 min in the PDS group; we did not perform a statistical comparison of overall cannulation rates and times between groups since PDW patients were allowed to cross over to the PDS group after 6 min.
There were no observed differences in post-procedure complication rates between the two groups. These included mild post-ERCP pancreatitis in 4 (4.6 %), three of whom were randomized to the PDS group (p = 0.34), and one case of post-ERCP abdominal pain without biochemical evidence of pancreatitis. A PDS had been successfully placed in all four cases of post-ERCP pancreatitis. There were no cases of perforation or hemorrhage.
Discussion
When standard approaches using a cannula or sphincterotome with and without a guidewire are unsuccessful in achieving deep biliary cannulation, options include precut sphincterotomy, transpancreatic sphincterotomy [25–29], and use of a PDS or guidewire. The latter two techniques minimize the need for electrocautery, thereby minimizing risks of hemorrhage and perforation while also protecting the pancreatic orifice from undue trauma. There are no previous studies randomizing patients to using a PDS to facilitate CBD cannulation, despite convincing evidence that this maneuver lowers the rate of post-ERCP pancreatitis [1, 12–15]. The current results are consistent with our previous retrospective observation that utilizing a PDS can facilitate CBD cannulation without the need for precut sphincterotomy in >74 % of cases. The rate of CBD cannulation within 6 min was higher among patients randomized to PDS (51.9 %) versus PDW (39.8 %), but this did not achieve statistical significance; PP analysis revealed an even smaller difference (PDS 65.1 % vs. PDW 59.3 %) between the two groups. These results illustrate that both techniques can lead to efficient (<6 min) CBD cannulation with minimal need for precut sphincterotomy.
There is no consensus definition of “difficult biliary cannulation.” We chose a 6-min threshold based on a previous clinical trial [21] whereas others have used a longer time period (10 min) [24] or “number of attempts” (authors have suggested from 5 to 15) [2, 4]. We chose a time measure to quantify difficulty since an “attempt” at cannulation is susceptible to greater subjectivity. Our randomization rate (19.7 %) among patients with a native papilla who provided informed consent a priori, along with the low rate of inadvertent CBD cannulation (11.5 %) during attempts to advance a PDW, are reasonable for experienced endoscopists. Furthermore, these numbers are similar to a recent randomized trial comparing the PDW technique with persistence where 22 % of patients met criteria for difficult cannulation and 17.5 % randomized to the PDW subgroup subsequently had unintentional CBD cannulation during attempts to advance a PDW [4]. The overall cannulation rate among patients who provided consent (97.7 %) further reflects the proficiency of the endoscopists participating in this trial.
The majority of less experienced/lower volume endoscopists are uncomfortable with placing PDSs and do not utilize them for pancreatitis prophylaxis [30, 31]. This is problematic since difficult biliary cannulation is an independent predictor of post-ERCP pancreatitis [17, 32] and endoscopists cannot predict difficult cases by visual inspection of the papilla alone in the majority of cases [33]. In addition to facilitating bile duct cannulation, a pancreatic stent offers two distinct advantages. First, the rate of ERCP-associated pancreatitis is lower among high risk patients who receive a prophylactic PDS [12–15, 30]. Cases of difficult biliary cannulation are at higher risk, particularly in the setting of repeated PD injection [34, 35]. Second, while precut sphincterotomy is an effective salvage technique when performed by an experienced endoscopist in challenging cases, a PDS should be deployed first to minimize the risk of pancreatitis [36, 37]. The low rate of complications in our study reflects the minimally invasive nature of the PDS and PDW techniques. These techniques require further study among less experienced, lower volume providers where their benefits as compared to merely persisting with usual techniques could be magnified.
Strengths of this study include its randomized trial design, standard definitions for difficult CBD cannulation, and incorporation of two medical centers/six endoscopists to improve generalizability. A crude analysis of participating endoscopists did not reveal significant differences in the primary outcome measure (data not shown). There are no previous prospective trials evaluating the efficacy of using a PDS to facilitate biliary cannulation. Our protocol is limited by lacking a “persistence” study arm; we justify our study design since our standard practice is to try alternative techniques after a reasonable effort is unsuccessful and we routinely utilize PDSs in all cases of difficult cannulation. This practice is based on evidence that multiple unsuccessful cannulation attempts are an independent predictor of post-ERCP pancreatitis [17, 32]. Furthermore, the purpose of this study was to compare alternative (PDS and PDW) techniques since both have been described but never compared against each other. The study design allowing crossover from PDW to PDS but not vice versa introduces a potential bias favoring the PDS technique. This is justified since we decided it would be unethical to cross from PDS to PDW since this would require removing the PDS, advancing a guidewire into the PD a second time, and then having to deploy a second PDS. We adjusted for this bias by focusing on the primary outcome of “cannulation within 6 min,” which represents an equal amount of time to achieve cannulation in either study arm and represents an acceptable cut-off for any particular technique in our cohort. Further, there was a higher prevalence of “anticipated difficult cannulation” papillae in the PDW (48.1 %) versus PDS (38 %) group based on the endoscopist’s visual inspection of the papilla. The interobserver and intraobserver variability of visual inspection is unknown, but if a greater number of “more difficult” papillae were randomized to the PDW group, this may introduce another bias favoring the PDS technique. We chose to terminate patient enrollment after reaching the enrollment goal for the PDW group but short of the PDS group (since the rate of crossover was lower than anticipated) because the observed difference in efficacy (13.8 %) for the primary endpoint would have required >400 patients to have adequate statistical power to detect a significant difference. However, we believe our sample size provides sufficient data demonstrating the feasibility of both techniques while minimizing the need for precut sphincterotomy.
In experienced hands, CBD cannulation can be achieved in <6 min using the PDS or PDW technique in the majority of patients in whom standard cannulation techniques are unsuccessful. While both are feasible, the PDS technique may be preferable since (1) most patients with difficult biliary cannulation require a PDS for pancreatitis prophylaxis anyway, (2) the PDW technique requires use of a second guidewire, thereby increasing procedural costs, and (3) the PDW technique is somewhat more cumbersome since locking the wire in the PD can be tenuous, especially when focusing on CBD cannulation thereafter. Importantly, the efficacy and safety of using the PDW and PDS techniques, along with precut sphincterotomy, need to be studied among lower volume ERCP practitioners, where the reported risks of precutting are highest [2]. Nevertheless, in a patient requiring biliary cannulation in who PD access is inadvertently attained, use of a PDW or PDS facilitates biliary cannulation while reducing the need for precut sphincterotomy in the majority of cases and maintains a low rate of post-procedure complications.
References
Dumonceau JM, Andriulli A, Deviere J, et al. European society of gastrointestinal endoscopy (ESGE) guideline: prophylaxis of post-ERCP pancreatitis. Endoscopy. 2010;42:503–515.
Freeman ML, Nelson DB, Sherman S, et al. Complications of endoscopic biliary sphincterotomy. N Engl J Med. 1996;335:909–918.
Manes G, Di Giorgio P, Repici A, et al. An analysis of the factors associated with the development of complications in patients undergoing precut sphincterotomy: a prospective, controlled, randomized, multicenter study. Am J Gastroenterol. 2009;104:2412–2417.
Herreros de Tejada A, Calleja JL, Diaz G, et al. Double-guidewire technique for difficult bile duct cannulation: a multicenter randomized, controlled trial. Gastrointest Endosc. 2009;70:700–709.
Kramer RE, Azuaje RE, Martinez JM, et al. The double-wire technique as an aid to selective cannulation of the common bile duct during pediatric endoscopic retrograde cholangiopancreatography. J Pediatr Gastroenterol Nutr. 2007;45:438–442.
Draganov P, Devonshire DA, Cunningham JT. A new technique to assist in difficult bile duct cannulation at the time of endoscopic retrograde cholangiopancreatography. JSLS. 2005;9:218–221.
Ito K, Fujita N, Noda Y, et al. Pancreatic guidewire placement for achieving selective biliary cannulation during endoscopic retrograde cholangio-pancreatography. World J Gastroenterol. 2008;14:5595–5600; discussion 5599.
Cote GA, Ansstas M, Pawa R, et al. Difficult biliary cannulation: use of physician-controlled wire-guided cannulation over a pancreatic duct stent to reduce the rate of precut sphincterotomy (with video). Gastrointest Endosc. 2010;71:275–279.
Goldberg E, Titus M, Haluszka O, et al. Pancreatic-duct stent placement facilitates difficult common bile duct cannulation. Gastrointest Endosc. 2005;62:592–596.
Slivka A. A new technique to assist in bile duct cannulation. Gastrointest Endosc. 1996;44:636.
Andriulli A, Forlano R, Napolitano G, et al. Pancreatic duct stents in the prophylaxis of pancreatic damage after endoscopic retrograde cholangiopancreatography: a systematic analysis of benefits and associated risks. Digestion. 2007;75:156–163.
Fazel A, Quadri A, Catalano MF, et al. Does a pancreatic duct stent prevent post-ERCP pancreatitis? A prospective randomized study. Gastrointest Endosc. 2003;57:291–294.
Sherman S, Earle DT, Bucksot L, et al. Does leaving a main pancreatic duct stent in place reduce the incidence of precut biliary sphincterotomy (ES)-induced pancreatitis? A final analysis of a randomized prospective study. Abstract. Gastrointest Endosc. 1996;43:A489.
Smithline A, Silverman W, Rogers D, et al. Effect of prophylactic main pancreatic duct stenting on the incidence of biliary endoscopic sphincterotomy-induced pancreatitis in high-risk patients. Gastrointest Endosc. 1993;39:652–657.
Saad AM, Fogel EL, Mchenry L, et al. Pancreatic duct stent placement prevents post-ERCP pancreatitis in patients with suspected sphincter of Oddi dysfunction but normal manometry results. Gastrointest Endosc. 2008;67:255–261.
Ito K, Fujita N, Noda Y, et al. Can pancreatic duct stenting prevent post-ERCP pancreatitis in patients who undergo pancreatic duct guidewire placement for achieving selective biliary cannulation? A prospective randomized controlled trial. J Gastroenterol. 2010;45:1183–1191.
Bailey AA, Bourke MJ, Kaffes AJ, et al. Needle-knife sphincterotomy: factors predicting its use and the relationship with post-ERCP pancreatitis (with video). Gastrointest Endosc. 2010;71:266–271.
Cennamo V, Fuccio L, Zagari RM, et al. Can early precut implementation reduce endoscopic retrograde cholangiopancreatography-related complication risk? Meta-analysis of randomized controlled trials. Endoscopy. 2010;42:381–388.
Sherman S, Ruffolo TA, Hawes RH, et al. Complications of endoscopic sphincterotomy. A prospective series with emphasis on the increased risk associated with sphincter of Oddi dysfunction and nondilated bile ducts. Gastroenterology. 1991;101:1068–1075.
Barthet M, Lesavre N, Desjeux A, et al. Complications of endoscopic sphincterotomy: results from a single tertiary referral center. Endoscopy. 2002;34:991–997.
Tang SJ, Haber GB, Kortan P, et al. Precut papillotomy versus persistence in difficult biliary cannulation: a prospective randomized trial. Endoscopy. 2005;37:58–65.
Cheung J, Tsoi KK, Quan WL, et al. Guidewire versus conventional contrast cannulation of the common bile duct for the prevention of post-ERCP pancreatitis: a systematic review and meta-analysis. Gastrointest Endosc. 2009;70:1211–1219.
Cennamo V, Fuccio L, Zagari RM, et al. Can a wire-guided cannulation technique increase bile duct cannulation rate and prevent post-ERCP pancreatitis? A meta-analysis of randomized controlled trials. Am J Gastroenterol. 2009;104:2343–2350.
Maeda S, Hayashi H, Hosokawa O, et al. Prospective randomized pilot trial of selective biliary cannulation using pancreatic guide-wire placement. Endoscopy. 2003;35:721–724.
Kahaleh M, Tokar J, Mullick T, et al. Prospective evaluation of pancreatic sphincterotomy as a precut technique for biliary cannulation. Clin Gastroenterol Hepatol. 2004;2:971–977.
Goff JS. Common bile duct pre-cut sphincterotomy: transpancreatic sphincter approach. Gastrointest Endosc. 1995;41:502–505.
Goff JS. Long-term experience with the transpancreatic sphincter pre-cut approach to biliary sphincterotomy. Gastrointest Endosc. 1999;50:642–645.
Akashi R, Kiyozumi T, Jinnouchi K, et al. Pancreatic sphincter precutting to gain selective access to the common bile duct: a series of 172 patients. Endoscopy. 2004;36:405–410.
Catalano MF, Linder JD, Geenen JE. Endoscopic transpancreatic papillary septotomy for inaccessible obstructed bile ducts: comparison with standard pre-cut papillotomy. Gastrointest Endosc. 2004;60:557–561.
Dumonceau JM, Rigaux J, Kahaleh M, et al. Prophylaxis of post-ERCP pancreatitis: a practice survey. Gastrointest Endosc. 2010;71:934–939.
Cote GA, Keswani RN, Jackson T, et al. Individual and practice differences among physicians who perform ERCP at varying frequency: a national survey. Gastrointest Endosc. 2011;74:e12.
Bailey AA, Bourke MJ, Williams SJ, et al. A prospective randomized trial of cannulation technique in ERCP: effects on technical success and post-ERCP pancreatitis. Endoscopy. 2008;40:296–301.
Cote GA, Hovis CE, Edmundowicz SA, et al. Predicting difficult bile duct cannulation: accuracy of endoscopic visualization of the papilla to predict cannulation difficulty. Abstract. Gastrointest Endosc. 2010;71:AB164.
Cheng CL, Sherman S, Watkins JL, et al. Risk factors for post-ERCP pancreatitis: a prospective multicenter study. Am J Gastroenterol. 2006;101:139–147.
Freeman ML, Disario JA, Nelson DB, et al. Risk factors for post-ERCP pancreatitis: a prospective, multicenter study. Gastrointest Endosc. 2001;54:425–434.
Madacsy L, Kurucsai G, Fejes R, et al. Prophylactic pancreas stenting followed by needle-knife fistulotomy in patients with sphincter of Oddi dysfunction and difficult cannulation: new method to prevent post-ERCP pancreatitis. Dig Endosc. 2009;21:8–13.
Abu-Hamda EM, Baron TH, Simmons DT, et al. A retrospective comparison of outcomes using three different precut needle knife techniques for biliary cannulation. J Clin Gastroenterol. 2005;39:717–721.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Coté, G.A., Mullady, D.K., Jonnalagadda, S.S. et al. Use of a Pancreatic Duct Stent or Guidewire Facilitates Bile Duct Access with Low Rates of Precut Sphincterotomy: A Randomized Clinical Trial. Dig Dis Sci 57, 3271–3278 (2012). https://doi.org/10.1007/s10620-012-2269-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-012-2269-2