Abstract
Background
The number of patients with ulcerative colitis (UC) in China has increased in the past 10 years. Thus, it is anticipated that the incidence of UC-associated colorectal cancer (UC-CRC) will also increase. However, the risk of CRC in UC patients is still unknown in Chinese. The aim of this study was to identify the risk and risk factors of UC-CRC in Chinese.
Methods
A total of 3,922 patients with UC were retrospectively collected from five central teaching hospitals in China, in which high-quality endoscopic and histological diagnoses were available from 1998 to 2009. The database of the UC and UC-associated CRC patients was evaluated.
Results
CRC was diagnosed 34 in patients, and the overall prevalence of CRC in patients with UC was 0.87%. The cumulative risk of developing CRC after a disease duration of 10 years was 1.15% (95% confidence interval [CI] 0.71–1.84%); 20 years, 3.56% (95% CI 2.14–5.89%); and 30 years, 14.36% (95% CI 7.57–26.3%). Longer disease duration, extensive colitis, and dysplasia found in the biopsy specimen were identified as risk factors for developing CRC. 5-ASA use was identified as a protective factor of UC-CRC.
Conclusions
The period prevalence of CRC was lower than that reported from the West. However, the cumulative risk was found to be comparable to that of Western countries, which suggests that the period prevalence of UC-CRC in China may be growing in the future.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Colorectal cancer (CRC) has been recognized as a severe complication of ulcerative colitis (UC). Although UC-associated colorectal cancer (UC-CRC) accounts for only 1–2% of all cases of all CRC in the general population, it is considered a serious complication of the disease and accounts for approximately 10–15% of all deaths in patients with inflammatory bowel disease (IBD) [1]. Compared to sporadic CRC, UC-CRC is 15–20 years earlier at age when diagnosed [2]. Since Crohn and Rosenberg [3] reported the first case of IBD-associated CRC, numerous studies have been carried out, especially in Western countries, to elucidate the association between CRC and UC. Nowadays, the increased risk of CRC in UC patients is widely accepted, and there is a general consensus that longer-duration and extensive UC have been regarded as the risk factors for developing CRC [2, 4].
Although it is widely accepted that there is increased risk of CRC in UC patients, the risk of CRC in UC reported varied a lot, which can be contributed to many different factors, such as the study design and geographic differences [4–9]. Eaden et al. [4] published a meta-analysis on the prevalence and risk factors of UC-CRC. The cumulative Incidence of CRC in UC was 2% at 10 years, 8% at 20 years, and 18% at 30 years of follow-up. Moreover, the study also suggests that CRC incidence rates in UC patients varied geographically, with higher risk among residents of the USA and UK than among Scandinavians and other countries. Studies based on patients at a tertiary care center or hospitals tend to demonstrate a higher cancer risk and worse prognosis. Yet a series of population-based studies published within the past 5 years suggests that the risk of CRC had decreased over time [10]. Limited data available from other countries, for example, Hungary [11], South Korea [12], and India [13], showed lower risk than that reported from the West.
In China, the number of patients with ulcerative colitis (UC) in China has increased threefold in the past 10 years [14, 15]. Therefore, the number of patients with colorectal cancer (CRC) associated with UC might also increase since there is an increased risk of CRC in UC. However, there is no data available on UC-CRC from China so far. Our aim was to address the epidemiology and risk factors for UC-associated CRC in a multi-center retrospective study.
Materials and Methods
The present study was a retrospective cohort analysis of all patients with a confirmed diagnosis of UC seen in five central teaching hospitals located in south, east, north, and central China, respectively (Table 1). Diagnosis was based on appropriate clinical, endoscopic, histopathological, and radiologic findings that satisfy the internationally accepted Lennard-Jones criteria [16]. In the five centrals, the majority of patients were advised a mapping colonoscopy at first visit and yearly follow-up colonoscopies after 7 years of disease. Therefore, patients were included in the study only if they had undergone colonoscopy. From 1998 to 2009, 3,922 patients were reviewed. During colonoscopy, 0.4% indigo carmine was sprayed to dye the field which inflammation involved. Biopsies were taken where there was an abnormal pit pattern according to Kudo’s classification [17]. Dysplasia was diagnosed by one experienced gastrointestinal pathologist and then confirmed by another. Data including gender, age, disease location, disease duration, colonoscopic follow-up, presence of dysplasia in biopsy before CRC, 5-ASA, and steroid therapy were collected from all of the patients for analysis.
Statistical Analysis
Statistical analysis was performed with SPSS 14.0 software with the help of a statistician. Variables were tested for normality by χ2 test. Logistic regression was used to test differences in disease phenotype between subgroups of patients with UC. Odds ratios (OR) were calculated. Life table survival analysis was performed to determine the cumulative risk of CRC in UC patients. p < 0.05 was considered significant.
Results
Period Prevalence and Cumulative Incidence of CRC in UC Patients
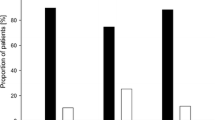
Of the 3,922 patients (male/female, 2,117/1,805), CRC was diagnosed in 34 patients (male 14; female 20) during follow-up. The period prevalence of CRC in patients with UC was 0.87% from the period between 1998 and 2009. In addition, the cumulative risk of CRC in China was estimated to be 1.15% (95% confidence interval [CI] 0.71–1.84%) for patients that had UC for 10 years, 3.56% (95% CI 2.14–5.89%) for patients that had UC for 20 years, and 14.36% (95% CI 7.57–26.3%) for patients that had UC for 30 years (Fig. 1).
Characteristics of Patients with UC-CRC
The overall duration of UC in patients with UC-associated CRC was a median of 12.8 years (range 8–32 years), significantly longer when compared to UC patients without CRC (p < 0.01). The median age at the time of diagnosis with colorectal cancer was 57.5 years (range 25–82 years) compared to 40.0 years (10–82 years) in UC patients (p < 0.01). Twenty-eight (82.3%) of the UC-CRC patients were at an advanced stage when diagnosed. Of these 34 tumors, seven (20.5%) were located in the rectum, 11 (32.4%) in the sigmoid colon, one (2.9%) in the descending colon, and 15 (44.2%) in the transverse and right colon. Fourteen (41.2%) of the patients with UC-CRC had been regularly examined with colonoscopy and ten (29.4%) of the patients had dysphasia in follow-up colonoscopy. Of the 34 patients, ten (29.4%) patients had extensive colitis and 12 (35.3%) patients had pancolitis.
Risk Factor for CRC in UC Patients
As shown in Table 2, age, disease duration, extent of colitis and the presence of dysplasia in the biopsy sample, 5-ASA/sulfasalazine (5-ASA: ≥1.5 g/day; sulfasalazine: ≥2 g/day for maintenance therapy) use each were found statistically significant between UC and UC-CRC patients. We then further analyzed these factors by logistic regression. As shown in Table 3, disease duration, extensive colitis, and the presence of dysplasia in the biopsy sample were identified as risk factors of UC-CRC. 5-ASA/sulfasalazine was identified as a protective factor of UC-CRC (Table 3).
Discussion
UC has been widely accepted as one of the risk factors of CRC. The risk of developing CRC within IBD individuals increases by 0.5–1.0% annually 8–10 years after the diagnosis [4]. However, the risk reported varied greatly due to the difference of geographic environment, ethnicity, and study design. Studies from the USA and Western Europe showed that the period prevalence ranged from 1.4 to 34% [18, 19]. There was limited data from Asian countries due to lower UC incidence. In South Korea [12], a population-based nationwide study showed that the period prevalence of CRC in patients with UC was 0.37%. The cumulative risk of UC-CRCs was 0.7% by 10 years, 7.9% by 20 years, and 33.2% by 30 years, which was comparable to that of Western countries. A single-center retrospective showed the period prevalence was 0.94% in India [13], much lower than that in Western countries. In our multi-center retrospective study, our data showed that the period prevalence of CRC in Chinese UC patients was 0.87%. The cumulative risk of UC-CRCs was 1.15% by 10 years, 3.56% by 20 years, and 14.36% by 30 years. The period prevalence was higher than that in South Korea (0.37%); the possible explanation for this might be that the study in Korea was population-based while ours was hospital-based. In accordance with previous reports in Asian countries, the period prevalence of UC-CRCs determined in the present study was lower when compared to the results of the global meta-analysis conducted by Eaden et al. [14]. Several factors may have contributed to the low incidence. First, the incidence of UC in China was still lower than the Western countries, although it increased in recent years [14, 15]. Second, the severity of inflammation was proven to be a risk factor for colorectal neoplasia in UC [20, 21]. Most UC patients in China have mild inflammation and can maintain remission by 5-aminosalicylates [14, 15]. Third, regarding the study design, most studies in the Western countries were prospective studies in which patients were regularly followed-up and enough biopsy specimens, usually four biopsies in every 10 cm, were taken. However, our study was a retrospective one and some of patients were not followed-up regularly. Even in regularly followed-up patients, fewer specimens from fewer parts were taken, significantly lower than that in the prospective studies. Of note, the cumulative risk was found to be comparable to those of Western countries, which suggested that the period prevalence of UC-CRC in China might be growing. Therefore, we may conclude that the accuracy period prevalence of CRC in Chinese UC patients may be higher in future prospective studies.
Disease duration and extent are frequently identified as two independent risk factors for CRC in patients with UC [4]. In accordance with previous studies, of the 34 UC-CRC patients in our cohort, 24 have disease durations of more than 8 years and 64.7% of the patients have extensive colitis or pancolitis. Statistical analysis also revealed that a disease duration longer than 10 years and extensive colitis were risk factors of CRC in UC patients.
Given the theory that chronic inflammation plays the most important role in malignant transformation, it can be reasonably concluded that anti-inflammatory therapy such as 5-ASA or steroid use may potentially act as a protective factor during the malignant transformation in UC patients. However, there was some conflict as to whether the chemoprevention can prevent CRC progression in UC patients. Theoretically, Brown et al. [22] confirmed that Mesalamine can inhibit epithelial beta-catenin activation in chronic ulcerative colitis, thereby preventing CRC progression in UC. In clinical studies, although Terdiman et al. [23] proved that treating IBD patients with 5-ASA medications was not found to have a protective effect against colitis-related CRC when assessed over a short period of exposure, another large epidemiological study showed that regular 5-ASA use is associated with some reduction in the risk of CRC developing in UC [24]. In agreement with previous studies, we have demonstrated a negative association between 5-ASA use and UC-CRC, suggesting that this may be a protective role of UC-CRC. However, we do not have exact data on patient compliance as for the duration or dose change. So the result that 5-ASA may be a protective role of UC-CRC in our study is not so reliable.
Endoscopic surveillance remains to be an important method to detect UC-CRC [25]. Surveillance colonoscopy may permit earlier detection of CRC, with a correspondingly improved prognosis. Guideline and consensus on screening and surveillance UC-CRC from the Europe and USA both paid much attention on the endoscopic surveillance in UC [26, 27]. However, confirmed evidence is lacking that surveillance colonoscopy prolongs survival in patients with UC. In our study, 14 patients with UC-CRC received endoscopic follow-up and 10 was found dysplasia before diagnosed CRC. In UC patients who underwent regular endoscopic follow-up, once dysplasia is found, there will indicate a high risk of CRC in these patients. Our data proved that there was an increased risk in patients who were found to have dysplasia during surveillance, which indicated that more careful follow-up should be performed in these patients.
Apart from the risk factors mentioned above, primary sclerosing cholangitis, diabetes, smoking, and appendectomy [28, 29] are also regarded as risk factors for UC-CRC. However, we failed to collect these data because our study was retrospective, which was major limitation of our study. Despite this, our study provided the risk and risk factors of CRC in UC patients for the fist time in China. In conclusion, we found that the period prevalence of CRC was high in our patients with UC but was lower compared to that reported in Western countries. However, the cumulative risk in China was comparable to that in the West, which indicated that the incidence of UC-CRC might not have reached a steady state in China. Thus, a population-based, prospective study should be carried out to evaluate the accurate risk of CRC in UC patients in China.
References
Munkholm P. Review article: the incidence and prevalence of colorectal cancer in inflammatory bowel disease. Aliment Pharmacol Ther. 2003;18:1–5.
Lakatos PL, Lakatos L. Risk for colorectal cancer in ulcerative colitis: changes, causes and management strategies. World J Gastroenterol. 2008;7:3937–3947.
Crohn B, Rosenberg H. The sigmoidoscopic picture of chronic ulcerative colitis (non-specific). Am J Med Sci. 1925;170:220–228.
Eaden JA, Abrams KR, Mayberry JF. The risk of colorectal cancer in ulcerative colitis: a meta-analysis. Gut. 2001;48:526–535.
Lennard-Jones JE, Morson BC, Ritchie JK, et al. Cancer surveillance in ulcerative colitis: experience over 15 years. Lancet. 1983;2:149–152.
Eaden J. Review article: colorectal carcinoma and inflammatory bowel disease. Aliment Pharmacol Ther. 2004;20:24–30.
Ekbom A, Helmick C, Zack M, et al. Ulcerative colitis and colorectal cancer. A population-based study. N Engl J Med. 1990;323:1228–1233.
Karlen P, Lofberg R, Brostrom O, et al. Increased risk of cancer in ulcerative colitis: a population-based cohort study. Am J Gastroenterol. 1999;94:1047–1052.
Maratka Z, Nedbal J, Kocianova J, et al. Incidence of colorectal cancer in proctocolitis: a retrospective study of 959 cases over 40 years. Gut. 1985;26:43–49.
Loftus EV Jr. Epidemiology and risk factors for colorectal dysplasia and cancer in ulcerative colitis. Gastroenterol Clin North Am. 2006;35:517–531.
Lakatos L, Mester G, Erdelyi Z, et al. Risk factors for ulcerative colitis-associated colorectal cancer in a Hungarian cohort of patients with ulcerative colitis: results of a population-based study. Inflamm Bowel Dis. 2006;12:205–211.
Kim BJ, Yang SK, Kim JS, et al. Trends of ulcerative colitis-associated colorectal cancer in Korea: a KASID study. J Gastroenterol Hepatol. 2009;24:667–671.
Venkataraman S, Mohan V, Ramakrishna BS, et al. Risk of colorectal cancer in ulcerative colitis in India. J Gastroenterol Hepatol. 2005;20:705–709.
Jiang L, Xia B, Li J, et al. Retrospective survey of 452 patients with inflammatory bowel disease in Wuhan city, central China. Inflamm Bowel Dis. 2006;12:212–217.
Yun J, Xu CT, Pan BR. Epidemiology and gene markers of ulcerative colitis in the Chinese. World J Gastroenterol. 2009;15:788–803.
Lennard-Jones JE. Classification of inflammatory bowel disease. Scand J Gastroenterol. 1989;24:2–6.
Kudo S, Tamura S, Nakajima T, et al. Diagnosis of colorectal tumorous lesions by magnifying endoscopy. Gastrointest Endosc. 1996;44:8–14.
Hendriksen C, Kreiner S, Binder V. Long-term prognosis in ulcerative colitis based on results from a regional patient group from the county of Copenhagen. Gut. 1985;26:158–163.
Kewenter J, Ahlman H, Hulten L. Cancer risk in extensive ulcerative colitis. Ann Surg. 1978;188:824–828.
Rutter M, Saunders B, Wilkinson K, et al. Severity of inflammation is a risk factor for colorectal neoplasia in ulcerative colitis. Gastroenterology. 2004;126:451–459.
Gupta RB, Harpaz N, Itzkowitz S, et al. Histologic inflammation is a risk factor for progression to colorectal neoplasia in ulcerative colitis: a cohort study. Gastroenterology. 2007;133:1099–1105. quiz 1340-1341.
Brown JB, Lee G, Managlia E, et al. Mesalamine inhibits epithelial beta-catenin activation in chronic ulcerative colitis. Gastroenterology. 2010;138:595–605.
Terdiman JP, Steinbuch M, Blumentals WA, Ullman TA, Rubin DT. 5-Aminosalicylic acid therapy and the risk of colorectal cancer among patients with inflammatory bowel disease. Inflamm Bowel Dis. 2007;13(4):367–371.
Bernstein CN, Blanchard JF, Metge C, et al. Does the use of 5-aminosalicylates in inflammatory bowel disease prevent the development of colorectal cancer? Am J Gastroenterol. 2003;98:2784–2788.
Shanahan F. Review article: colitis-associated cancer—time for new strategies. Aliment Pharmacol Ther. 2003;18:6–9.
Eaden JA, Mayberry JF, British Society for Gastroenterology, Association of Coloproctology for Great Britain, Ireland. Guidelines for screening and surveillance of asymptomatic colorectal cancer in patients with inflammatory bowel disease. Gut. 2002;51:V10–V12.
Itzkowitz SH, Present DH, Crohn’s and Colitis Foundation of America Colon Cancer in IBD Study Group. Consensus conference: Colorectal cancer screening and surveillance in inflammatory bowel disease. Inflamm Bowel Dis. 2005;11:314–321.
Loftus EV Jr, Harewood GC, Loftus CG, et al. PSC-IBD: a unique form of inflammatory bowel disease associated with primary sclerosing cholangitis. Gut. 2005;54:91–96.
Pinczowski D, Ekbom A, Baron J, et al. Risk factors for colorectal cancer in patients with ulcerative colitis: a case control study. Gastroenterology. 1994;107:117–120.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gong, W., Lv, N., Wang, B. et al. Risk of Ulcerative Colitis-Associated Colorectal Cancer in China: A Multi-Center Retrospective Study. Dig Dis Sci 57, 503–507 (2012). https://doi.org/10.1007/s10620-011-1890-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-011-1890-9