Abstract
The outcome of Helicobacter pylori infection has been related to specific virulence-associated bacterial genotypes. The best known genotypic virulence factors of H. pylori are cytotoxin-associated gene A (cagA) and vacuolating cytotoxin gene A (vacA). The objective of this study was to assess the relationship between H. pylori cagA and vacA status and histopathological findings. Esophagogastrodoedonoscopy was performed in 80 dyspeptic patients. Antrum and corpus biopsies were obtained for isolation of H. pylori and for histopathological assessment. The polymerase chain reaction was used to detect cagA and vacA genes of H. pylori using specific primers. Biopsy samples were stained with hematoxylin and eosin, and histopathological findings were graded using the “updated Sydney system”. H. pylori from 57 of the 80 patients was incubated. Of the 57 patients, 44 were cagA positive. In the corpus biopsy specimens there was a significant relationship between the density of H. pylori colonization (P = 0.02) and chronic inflammation (P = 0.02) and cagA-positive genotypes. In the antrum specimens there was a significant relationship between cagA positivity and neutrophil activity (P = 0.003) and glandular atrophy (P = 0.002), but not with H. pylori density, chronic inflammation, and intestinal metaplasia. The odds ratio of cagA-positive vs. cagA-negative strains for the presence of glandular atrophy, irrespective of grading and of gastric localization, was 4.62 (95% CI, 1.18–18.08, P = 0.041). No significant relationships were observed between vacA s1 and s2 genotypes and histopathological parameters. Corpus neutrophil infiltration was found to be more severe in the m1 group than in the m2 group (P = 0.004). Other histopathological features showed no difference between m1 and m2 genotypes. In conclusion H. pylori strains showing cagA positivity are associated with more severe gastritis in some histological features but virulence factors of H. pylori do not appear to determine the overall pattern of gastritis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Helicobacter pylori is a spiral, microaerophilic, Gram-negative bacterium that permanently colonizes gastric epithelial cells in approximately 25% of the population in developed countries and 70–90% in developing countries [1]. Whereas most infected individuals are asymptomatic, chronic H. pylori infection in susceptible individuals is associated with a variable degree of mucosal damage ranging from mild gastritis and ulcer disease to gastric carcinoma and mucosa-associated lymphoid tissue (MALT) lymphoma [2, 3]. The clinical outcome of H. pylori infection has been associated with bacterial virulence factors, host gastric mucosal factors, and the environment [4].
The two best studied bacterial determinants of H. pylori infection are the presence of cytotoxin-associated gene A and vacuolating cytotoxin genotype. Cytotoxin-associated gene A encodes a high-molecular-weight immunodominant protein. This cagA gene product is not itself a virulence factor but is part of a 40 kb cluster of genes (cag pathogenicity island), some of which contribute to pathogenicity [5]. A number of studies in Western countries have confirmed that infection with cagA-positive strains is associated with more severe gastritis and higher prevalence of peptic ulcer and gastric cancer [6, 7]. However, studies in Far Eastern countries demonstrate equally high prevalence of cagA-positive strains in patients with peptic ulcer, gastric cancer, and nonulcer dyspepsia and in control subjects [8]. In Turkey, cagA positivity and the clinical outcome of H. pylori infection has been studied. In these studies, a relationship has been demonstrated between peptic ulcer and gastric cancer and cagA positivity, similar to Western countries [9, 10].
The vacuolating cytotoxin A gene, which is another important virulence factor of H. pylori, encodes an 87 kD protein that induces vacuolation of epithelial cells [11]. The vacA gene is present in all strains of H. pylori and comprises two variable parts. The H. pylori strains have one of two types of vacA signal sequence (s1 and s2) and two types of mid region (m1 and m2) [12, 13]. The mosaic combination of s and m region allelic types determines the production of the cytotoxin and is associated with pathogenicity of the bacteria [3, 14]. As with cagA status, there are geographic differences between vacA status and the H. pylori-related diseases. In Western countries infection with vacA s1 strain is more common in patients with peptic ulcer than in those with chronic gastritis. However in Asian populations, the association between vacA diversity and clinical outcome is not established [15, 16]. In a study from Turkey, the vacA s1a genotype was detected in 66.7, 96.4, and 87.9% of isolates from patients with functional dyspepsia, duodenal ulcer, and gastric cancer, respectively [17]. The objective of this study was to research gastric histopathology in Turkish dyspeptic patients infected with H. pylori and to assess its relationship with bacterial virulence-associated cagA and vacA genotypes.
Materials and Methods
Patients
A total of 57 H. pylori isolates were obtained from gastric biopsies from Turkish patients who underwent upper gastrointestinal endoscopy. Patients with a history of gastric surgery, active gastrointestinal bleeding, or use of steroids, immunosuppressive drugs, antibiotics, bismuth compounds, or proton pump inhibitors were excluded. Patients with duodenal ulcer, gastric ulcer and gastric cancer were also excluded from the study. All patients provided written informed consent for this study. The ethical committee of Trakya University approved the study protocol.
H. pylori Culture, Preparation of Genomic DNA and Polymerase Chain Reaction (PCR)-Based Genotyping
The biopsy specimens were spread with an applicator and were cultured under microaerophilic conditions (GenBox Microaer; Biomerieux, France) on special culture media (Pyloriagar; Biomerieux) containing horse serum, antibiotic mixture, and polyvitex for up to 5 days. The organisms were identified as H. pylori by Gram staining, colony morphology, and positive oxidase, catalase, and urease reactions.
For H. pylori chromosomal DNA extraction, bacteria were harvested in phosphate buffered saline solution and centrifuged. The pelleted cells were resuspended in 200 μl proteinase K solution containing 5 mmol/l ethylenediaminetetraacetic acid, 0.5% sodium dodecylsulfate, and 10 mg/ml proteinase K in 10 mmol/l Tris–HCl and subsequently incubated at 50°C for 2 h. The DNA was extracted with phenol–chloroform–isoamyl alcohol by the standard procedure, and was finally precipitated by adding 1/10 vol 3 mol/l sodium acetate and 2.5 vol ethanol. After centrifugation, the pellet was washed with 70% ethanol and then dissolved in 50 μl TE buffer (10 mmol/l Tris–HCl, pH 8.0; 5 mmol/l EDTA, pH 8.0).
PCR reactions were performed in a volume of 100 μl containing 71.5 μl distilled water, 10 μl PCR buffer, 2 μl deoxynucleoside triphosphate, 0.5 μl Taq polymerase, 1 μl BSA, and 5 μl of both forward and reverse primers. CAGAF and CAGAR primers were used to amplify a 349-bp product from the middle conservative region of the cagA gene. VAG-F and VAG-R primers were used to amplify a 570-bp product for m1 and 645-bp products for m2 from the middle of the vacA gene. VA1-F and VA1-R primers were used to amplify a 259-bp product for s1 and 286-bp products for s2 from the signal sequence of the vacA gene [6, 16]. PCR primers used in this study are listed in Table 1. Thermal cycling for each set of primers was performed as previously described [16].
Histopathology
Two gastric biopsy specimens, one from the antrum and one from the corpus, were immersed in 10% formalin and embedded in paraffin. Sections were stained with hematoxylin and eosin. Only cases with adequate sized biopsy specimens of both antral and corpus mucosa were accepted for histological assessment by an experienced pathologist. The pathologists were unaware of the clinical information of each patient. H. pylori density, chronic inflammation, neutrophil activity, glandular atrophy, and intestinal metaplasia were scored on an ordinal scale (0–3) using the criteria described in the updated Sydney classification system [18].
Statistical Analysis
Data were analyzed in Minitap release 13 (Licence Number: wcp1331.00197). Chi-square test and Fisher’s exact test were used to assess the relationship between individual genotypes. Histopathological parameters were scored on ordinal scales (from 0 to 3) and analyzed by the Mann–Whitney test. Differences were regarded as statistically significant when the P-value was ≤0.05.
Results
Antral biopsies were obtained for H. pylori culture from 80 functional dyspeptic patients who had undergone upper gastrointestinal endoscopy with positive rapid urease test. H. pylori was isolated successfully from 57 of the 80 patients’ specimens (71.2%). H. pylori was histopathologically demonstrated in just 53 of the 57 culture-positive patients. A total of 57 culture positive patients (30 men and 27 women), with a mean age 44.9 years (range 20–72 years) were included in our study.
A total of 44 of the 57 H. pylori isolates were cagA positive (77.2%). There was no association between cagA status and the mean ages or genders of the patients. In typing of the vacA gene m region, 23 H. pylori isolates were vacA m1 and 30 were vacA m2. In three isolates, genotyping could not be performed and in one isolate, both m1 and m2 genotypes were observed. These four isolates were excluded from the statistical analysis. Evaluation of the vacA signal region revealed that 32 isolates were s1 and 18 were s2, while seven showed more than one signal region. These seven isolates with multiple signal regions were excluded from the analysis.
H. pylori Genotypes and Histopathological Findings
Relationships between histological parameters determined in gastric biopsy specimens and H. pylori cagA status, and vacA s and m genotypes are shown in Tables 2–4, respectively.
H. pylori Density
The density of H. pylori was scored in antral and corpus biopsy specimens. The density scores of H. pylori in the corpus biopsies of the cagA-positive isolates were higher than in the cagA-negative isolates (P = 0.02). There was no significant relationship between cagA positivity and H. pylori density in the antrum. No relationships were found between vacA s or m regions and H. pylori density in either the antrum or corpus.
Neutrophil Activity
cagA-positive genotypes were strongly associated with higher activity in the antrum (P = 0.003) but not in the corpus. The activity was higher only in the corpus with the vacA m1 genotype (P = 0.004). No relationships were found between activity and vacA s genotypes in the antrum or corpus.
Chronic Inflammation
There was an association between cagA positivity and high inflammation scores in both corpus and antrum biopsy specimens, but only in corpus biopsies did statistical analysis show significance (P = 0.017). There was no relationship between vacA s or m genotypes and chronic inflammation in either corpus nor antrum biopsies.
Glandular Atrophy
There was a strong association between the presence of glandular atrophy in the antrum and individual cagA-positive genotypes, compared to the cagA-negative genotypes (P = 0.02). The same relationship could not be observed in the corpus biopsy specimens. No association was found between vacA genotypes and the presence of glandular atrophy.
We investigated the relationship between cagA positivity and atrophy in any localization and degree. Atrophy was observed in any localization and degree in 69.8% of the cagA-positive and 33.3% of the cagA-negative cases. The odds ratio of cagA-positive vs. cagA-negative strains for the presence of glandular atrophy, irrespective of grading and of gastric localization, was 4.62 (95% CI, 1.18–18.08, P = 0.041).
Intestinal Metaplasia
The presence of intestinal metaplasia was not associated with cagA or vacA genotypes in the corpus or antrum biopsies.
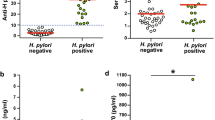
All histological parameters determined in antral and corpus biopsy specimens according to the updated Sydney system and H. pylori cagA status are shown in Figs. 1 and 2.
Discussion
H. pylori infection results in chronic gastritis and, eventually, diseases, such as peptic ulcer, gastric cancer, and MALT lymphoma [1, 19, 20]. Genotypic alterations of H. pylori are thought to be responsible for the various clinical manifestations and for infection without symptoms or with symptoms of gastric carcinoma and MALT lymphoma. In our study, the presence of cagA, which is thought to be associated with severe diseases, was investigated in patients with functional dyspepsia and compared with histological findings. The prevalence of cagA expression in H. pylori strains is reported as 60% with geographic variation. The positivity of cagA shows variation within nationalities and states. cagA positivity is reported as 61.7–80.4% in our country; in our studies cagA positivity was 77.1% in patients with functional dyspepsia [21, 22].
Although an association between H. pylori infection and chronic gastritis is clear, development of severe gastric diseases is rare. These variations in the clinical consequences are because of factors such as duration of the infection, inflammatory response of the patient, virulence of H. pylori strains, etc. Infection with less virulent strains is associated with mild symptoms whereas infection with more virulent strains is thought to be associated with more severe gastric inflammation and, eventually, peptic ulcer, gastric adenoma, and MALT lymphoma. Gastric gland atrophy, intestinal metaplasia, and dysplasia may develop after prolonged H. pylori infection. Compared with the rest of the population, the risk of development of gastric cancer in patients with duodenal ulcer is low. Two different pathways—duodenal ulcer or gastric ulcer with atrophy, intestinal metaplasia and cancer—may be associated with H. pylori infection. The relationship between H. pylori genotypes and infection with gastric inflammatory response varies within nationalities. In Western countries it has been demonstrated that more severe gastric inflammation develops after infection with cagA-positive strains; in Asian countries there is no such difference. Studies from Turkey on cagA positivity and the histopathological findings of gastritis led to conflicting results. Demirturk et al. suggested that cagA positivity is associated with more severe glandular atrophy, inflammation, and activity, whereas Saruc et al. demonstrated a relationship between cagA positivity with inflammation, H. pylori density, and intestinal metaplasia but not with glandular atrophy [21, 23]. In our study, cagA-positive patients showed more severe neutrophil infiltration and chronic inflammation in both the antrum and the corpus. Nevertheless, only the activity of the gastritis in the antrum and the chronic inflammation in the corpus were statistically significant. The rate of the glandular atrophy and the H. pylori density were found to be significant in cagA-positive subjects.
Atrophic gastritis and intestinal metaplasia are important aspects of the development of gastric cancer. The association between H. pylori and chronic superficial gastritis, atrophic gastritis, and intestinal metaplasia is already known. Because prolonged H. pylori colonization is related to distal gastric adenocancer and H. pylori is accepted as a carcinogen type-1, it is reported in the Maastricht-III Concensus that patients with H. pylori infection with atrophic gastritis should definitely receive therapy [24]. Furthermore, despite the fact that most of the population is infected with H. pylori, the development of gastric adenocancer is rare.
Modes of treatment of H. pylori vary, because the infection is frequently asymptomatic yet is also related to severe outcomes, such as gastric cancer. Especially in patients with functional dyspepsia, the decision to eradicate H. pylori remains unclear. According to the Maastricht-2 report, patients with functional dyspepsia should certainly not receive eradication therapy. Nevertheless, patients with severe gastritis (atrophia, etc.) should receive medication. The willingness of the patient to undergo medication is accepted as an indication, because H. pylori type-1 is known to be a carcinogen and the prevalence is increasing within the population [25]. There are no proven data that eradication therapy is effective for the symptoms of the patients with functional dyspepsia. As a result, more objective data are needed for the decision to give medication to patients with functional dyspepsia.
In our study, relation with cagA positivity and atrophy in the gastric antrum or corpus is demonstrated in patients with functional dyspepsia and H. pylori infection (odds ratio: 4.62; 95% CI, 1.18–18.08, P = 0.041). It has recently become possible to detect antibodies for cagA proteins with ELISA methods [8, 26]. Along with isolation of H. pylori, demonstration of cagA positivity with non-invasive methods could be helpful in some patient groups. Studies and data are needed for evaluation of sensitivity and specificity of the non-invasive methods for showing functional cag-PAI.
The variability of the vacA s and m regions is thought to have effects over the secretion of vacuolating toxin. s1/m1 strains produce huge amounts of toxins and are highly detrimental. s2 strains do not produce such toxins. In many studies, the variability of the s genotypes is associated with disease formation but not proven. In our study, we did not find any statistically significant relationships between vacA signal region genotype and H. pylori density in the mucosa of the antrum and the corpus, neutrophil infiltration, chronic inflammation, glandular atrophy, and intestinal metaplasia. vacA signal region genotype and gastric inflammatory alterations show regional variations. Warburton et al. reported no relationship between vac s1 and s2 regions and histological findings, similar to our results [27]. This finding is controversial, because it is known that strains with s1 genotype produce much greater amounts of toxins. Subtypes of vacA s1 region (s1a, s1b) may be closely related to histological parameters. Nevertheless, Atherton et al. suggested that s1a genotype is associated with peptic ulcer and more severe gastritis [28].
There are limitations to our study. For example, genotyping was performed only on H. pylori strains obtained from antrum biopsies. For this reason, there may be no absolute relationship between the histopathological changes of the corpus mucosa and H. pylori genotypes, for there may be different strains in the corpus and the antrum. However, the false-negative rates are lower in the biopsies obtained from the antrum, because the antrum is accepted as the major region of settlement of H. pylori.
Besides the cagA and vacA genotypes, variations of acid production, the genetics of the infected subject, tobacco and alcohol intake, and other virulence factors of H. pylori may affect both clinical outcomes and the histopathological findings. More studies are needed to evaluate other factors besides the H. pylori genotypes, because H. pylori is thought to be a cause of serious diseases.
References
Dunn BE, Cohen H, Blaser MJ (1997) Helicobacter pylori. Clin Microbiol Rev 10:720–741
Parsonnet J, Hansen S, Rodriguez L, Gelb AB, Warnke RA, Jellum E, Orentreich N, Vogelman JH, Friedman GD (1994) Helicobacter pylori infection and gastric lymphoma. N Engl J Med 330:1267–1271. doi:10.1056/NEJM199405053301803
Israel DA, Peek RM (2001) Pathogenesis of Helicobacter pylori-induced gastric inflammation. Aliment Pharmacol Ther 15:1271–1290. doi:10.1046/j.1365-2036.2001.01052.x
McGee DJ, Mobley HLT (2000) Pathogenesis of Helicobacter pylori infection. Curr Opin Gastroenterol 16:24–31. doi:10.1097/00001574-200001000-00005
Jenks PJ, Megraud F, Labigne A (1998) Clinical outcome after infection with Helicobacter pylori does not appear to be reliably predicted by the presence of any of the genes of the cag pathogenicity island. Gut 43:752–758
van Doorn LJ, Figueiredo C, Sanna R, Plaisier A, Schneeberger P, de Boer W, Quint W (1998) Clinical relevance of the cagA, vacA, and iceA status of Helicobacter pylori. Gastroenterology 115:58–66. doi:10.1016/S0016-5085(98)70365-8
Yamaoka Y, Souchek J, Odenbreit S, Haas R, Arnqvist A, Boren T, Kodama T, Osato MS, Gutierrez O, Kim JG, Graham DY (2002) Discrimination between cases of duodenal ulcer and gastritis on the basis of putative virulence factors of Helicobacter pylori. J Clin Microbiol 40:2244–2246. doi:10.1128/JCM.40.6.2244-2246.2002
Palli D, Menegatti M, Masala G, Ricci C, Saieva C, Holton J, Gatta L, Miglioli M, Vaira D (2002) Helicobacter pylori infection, anti-cagA antibodies and peptic ulcer: a case-control study in Italy. Aliment Pharmacol Ther 16:1015–1020. doi:10.1046/j.1365-2036.2002.01253.x
Aydin F, Kaklikkaya N, Ozgur O, Cubukcu K, Kilic AO, Tosun I, Erturk M (2004) Distribution of vacA alleles and cagA status of Helicobacter pylori in peptic ulcer disease and non-ulcer dyspepsia. Clin Microbiol Infect 10:1102–1104. doi:10.1111/j.1469-0691.2004.00989.x
Sezikli M, Guliter S, Apan TZ, Aksoy A, Keles H, Ozkurt ZN (2006) Frequencies of serum antibodies to Helicobacter pylori CagA and VacA in a Turkish population with various gastroduodenal diseases. Int J Clin Pract 60:1239–1243. doi:10.1111/j.1742-1241.2005.00778.x
Ito Y, Azuma T, Ito S, Miyaji H, Hirai M, Yamazaki Y, Sato F, Kato T, Kohli Y, Kuriyama M (1997) Analysis and typing of the vacA gene from cagA-positive strains of Helicobacter pylori isolated in Japan. J Clin Microbiol 35:1710–1714
van Doorn LJ, Figueiredo C, Sanna R, Pena S, Midolo P, Ng EK, Atherton JC, Blaser MJ, Quint WG (1998) Expanding allelic diversity of Helicobacter pylori vacA. J Clin Microbiol 36:2597–2603
Atherton JC, Cao P, Peek RM Jr, Tummuru MK, Blaser MJ, Cover TL (1995) Mosaicism in vacuolating cytotoxin alleles of Helicobacter pylori. Association of specific vacA types with cytotoxin production and peptic ulceration. J Biol Chem 270:17771–17777. doi:10.1074/jbc.270.30.17771
Pagliaccia C, de Bernard M, Lupetti P, Ji X, Burroni D, Cover TL, Papini E, Rappuoli R, Telford JL, Reyrat JM (1998) The m2 form of the Helicobacter pylori cytotoxin has cell type-specific vacuolating activity. Proc Natl Acad Sci USA 95:10212–10217. doi:10.1073/pnas.95.17.10212
Maeda S, Ogura K, Yoshida H, Kanai F, Ikenoue T, Kato N, Shiratori Y, Omata M (1998) Major virulence factors, VacA and CagA, are commonly positive in Helicobacter pylori isolates in Japan. Gut 42:338–343
Yamaoka Y, Kodama T, Kita M, Imanishi J, Kashima K, Graham DY (1998) Relationship of vacA genotypes of Helicobacter pylori to cagA status, cytotoxin production, and clinical outcome. Helicobacter 3:241–253. doi:10.1046/j.1523-5378.1998.08056.x
Erzin Y, Koksal V, Altun S, Dobrucali A, Aslan M, Erdamar S, Dirican A, Kocazeybek B (2006) Prevalence of Helicobacter pylori vacA, cagA, cagE, iceA, babA2 genotypes, and correlation with clinical outcome in Turkish patients with dyspepsia. Helicobacter 11:574–580. doi:10.1111/j.1523-5378.2006.00461.x
Dixon MF, Genta RM, Yardley JH, Correa P (1996) Classification and grading of gastritis. The updated Sydney system. International workshop on the histopathology of gastritis, Houston 1994. Am J Surg Pathol 20:1161–1181
Spencer J, Wotherspoon AC (1997) Gastric MALT lymphoma and Helicobacter pylori. Cancer Surv 30:213–231
Graham DY (2000) Helicobacter pylori infection is the primary cause of gastric cancer. J Gastroenterol 35(Suppl 12):90–97
Demirturk L, Ozel AM, Yazgan Y, Solmazgul E, Yildirim S, Gultepe M, Gurbuz AK (2001) CagA status in dyspeptic patients with and without peptic ulcer disease in Turkey: association with histopathologic findings. Helicobacter 6:163–168. doi:10.1046/j.1523-5378.2001.00024.x
Bulent K, Murat A, Esin A, Fatih K, MMMurat H, Hakan H, Melih K, Mehmet A, Bulent Y, Fatih H (2003) Association of CagA and VacA presence with ulcer and non-ulcer dyspepsia in a Turkish population. World J Gastroenterol 9:1580–1583
Saruc M, Demir MA, Kucukmetin N, Kandiloglu AR, Akarca US, Yuceyar H (2002) Histological and clinical predictive value of determination of tissue CagA status by PCR in Helicobacter pylori infected patients; results of the large population based study in western Turkey. Hepatogastroenterology 49:878–881
Malfertheiner P, Megraud F, O’Morain C, Bazzoli F, El-Omar E, Graham D, Hunt R, Rokkas T, Vakil N, Kuipers EJ (2007) Current concepts in the management of Helicobacter pylori infection: the Maastricht III consensus report. Gut 56:772–781. doi:10.1136/gut.2006.101634
Malfertheiner P, Megraud F, O’Morain C, Hungin AP, Jones R, Axon A, Graham DY, Tytgat G (2002) Current concepts in the management of Helicobacter pylori infection–the Maastricht 2–2000 consensus report. Aliment Pharmacol Ther 16:167–180. doi:10.1046/j.1365-2036.2002.01169.x
Beales IL, Crabtree JE, Scunes D, Covacci A, Calam J (1996) Antibodies to CagA protein are associated with gastric atrophy in Helicobacter pylori infection. Eur J Gastroenterol Hepatol 8:645–649
Warburton VJ, Everett S, Mapstone NP, Axon AT, Hawkey P, Dixon MF (1998) Clinical and histological associations of cagA and vacA genotypes in Helicobacter pylori gastritis. J Clin Pathol 51:55–61
Atherton JC, Peek RM Jr, Tham KT, Cover TL, Blaser MJ (1997) Clinical and pathological importance of heterogeneity in vacA, the vacuolating cytotoxin gene of Helicobacter pylori. Gastroenterology 112:92–99. doi:10.1016/S0016-5085(97)70223-3
Acknowledgments
This study was supported financially by the Trakya University Scientific Research Fund (TUBAP-594) and has received a grant from the United European Gastroenterology Federation in the 13th UEGW, Copenhagen, Denmark, 2005.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Umit, H., Tezel, A., Bukavaz, S. et al. The Relationship Between Virulence Factors of Helicobacter pylori and Severity of Gastritis in Infected Patients. Dig Dis Sci 54, 103–110 (2009). https://doi.org/10.1007/s10620-008-0316-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-008-0316-9