Abstract
Akt (also known as protein kinase B, PKB) is involved in a variety of biological processes, for example cell development, proliferation, and angiogenesis. Clinical studies in support of the idea that increased activity of Akt could contribute directly to gastric carcinogenesis are rare, however. In this study we discovered that phospho-Akt1 was overexpressed in human gastric cancers and its levels correlated with tumor differentiation and pTNM. Akt1 activation promoted cell survival, because the phosphatidylinositol 3-kinase(PI3K) inhibitor LY294002 inhibited Akt1 phosphorylation and inhibited cell growth, especially in cells with active Akt1. Dominant negative Akt inhibited proliferation of gastric cancer cells and induced G1 cell-cycle arrest whereas constitutively active Akt increased cell proliferation. We have therefore identified Akt1 as an active kinase that contributes to gastric cancer progression and promotes proliferation of gastric cancer cells.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Akt (also known as protein kinase B, PKB) regulates a variety of cellular processes by mediating extracellular (mitogenic growth factor, insulin, and stress) and intracellular (altered tyrosine receptor kinases, Ras and Src) signals. Activation of Akt by these signals is via binding of its pleckstrin homology (PH) domain to products of PI3K. Increases in 3′ phosphoinositides lead to membrane translocation of downstream effectors, for example the serine/threonine protein kinase Akt, the cellular homologue of the viral oncogene v-Akt. On translocation, Akt is phosphorylated and activated, ultimately resulting in stimulation of cell growth and survival [1, 2].
Akt1, Akt2, and Akt3 are three isoforms with high sequence homology encoded by three separate genes [3]. Overexpression of Akt may contribute to tumor development and progression. The work that originally identified Akt as a potential human oncogene detected amplification of Akt1 in a single gastric carcinoma [4]. Akt2 gene amplification and detection at kinase and protein levels was found in some types of human cancer, for example ovarian, pancreatic, and breast tumors [5, 6]. Akt3 has been reported to cause overexpression of mRNA and selective activation of the protein by growth factors in hormone-independent breast and prostate cancer cell lines [7]. Overall, these studies indicate that Akt gene amplification may be a frequent occurrence in several human cancers.
Abnormal proliferation is one of the characteristics of tumors. Recent observations indicate PI3K is involved in cell proliferation and differentiation. Addition of the PI3K inhibitor LY294002 induced G1 cell-cycle arrest. It has also been demonstrated that inactivation of PI3K markedly inhibits proliferation of small lung cancer cells by stimulating apoptosis and promoting cell-cycle delay in G1 [8]. Akt also targets several key cell-cycle regulators, including p21cip1/waf1, p27kip1, and MDM2. Phosphorylation by Akt enhances protein stability of p21, possibly leading to activation of cyclin/CDK and DNA replication. Akt phosphorylation results in exclusion of p27 from the nucleus and inhibits p27 at the transcriptional level, leading to activation of cyclin/Cdk and cell-cycle progression [9–12]. There is, therefore, increasing evidence that deregulated Akt expression may contribute to the various properties of tumor cells. Thus, the PI3K/Akt signaling pathway seems to be involved in cell-cycle regulation.
Gastric cancer is one of the most malignant throughout the world, particularly in the Eastern Asian countries China, Korea, and Japan [13–16]. The molecular mechanisms of gastric carcinogenesis remain unclear, however; in particular, the importance of Akt signaling in cell-cycle progression in gastric cancer cells is not fully understood. To explore whether or not Akt1 expression is related to gastric carcinogenesis we investigated p-Akt1 protein levels in clinical materials and the effect of Akt1 on cell proliferation and cell-cycle progression in gastric cancer cells. The results indicated that p-Akt1 is associated with gastric carcinogenesis and could regulate cell cycles.
Materials and Methods
Cell Lines and Cell Culture
AGS, MKN45, MGC803, and SGC7901 cells are human gastric cancer cell lines. AGS cells were obtained from the American Type Culture Collection; MKN45and MGC803 cells were obtained from RIKEN Cell Bank (The Institute of Physical and Chemical Research), Japan; and SGC7901 cells were obtained from Science Academe of China. GES-1 cells are SV40 transformed immortalized gastric epithelial cells established by the Beijing Cancer Institute, China [17]. These cells were cultured in RPMI1640 medium (Life Technologies) supplemented with 10% FBS (Life Technologies), 100 mg mL−1 streptomycin (Sigma), 100 units mL−1 penicillin (Sigma), and L-glutamine (Life Technologies), at 37°C, in a 5% CO2 incubator.
Specimen Collection
For immunohistochemistry, paraffin-embedded specimens from 124 unselected gastric cancer and normal tissues were obtained from Xijing Hospital, Oncology Center and Central Hospital, Xi’an, China, from 1999 to 2002. Histological classification of the tumors was in accordance with the World Health Organization. A total of 101 of the samples were identified as carcinoma tissues, 13 as normal tissues, and the other 10 as intestinal metaplasia tissues. For Western blot assay fresh surgical gastric cancer specimens and adjacent tissues were obtained from twelve patients in Xijing Hospital at the time of surgery. These samples were snap-frozen with liquid nitrogen and stored at −70°C. The histological types of all the subjects were clinically and pathologically proved. All patients gave informed consent before collection of the specimens, in accordance with the guidelines of our institute.
Western Blot
Frozen specimens and monolayer-cultured cells were homogenized in lysis buffer containing 50 mmol L−1 Tris–HCl (pH 7.6), 150 mmol L−1 NaCl, 0.1% NP-40, 0.1 mol L−1 DTT, 10 mmol L−1 NaF, 2 mmol L−1 Na3VO4, and 2 mmol L−1 phenylmethylsulfonyl fluoride, and 2 μg mL−1 aprotinin and leupeptin. Lysates were centrifuged at 12,000 × g for 15 min at 4°C before Western blotting. The protein concentration of the supernatant was measured by use of Bradford’s assay [18]. Equivalent protein was loaded, and the lysates were separated by SDS-PAGE and then transferred to Hybond-C membrane (Amersham–Pharmacia Biotech, Buckinghamshire, UK). The membranes were blocked for 2 h in blocking buffer (25 mmol L−1 Tris–HCl, 150 mmol L−1 NaCl, 10% milk, and 0.2% Tween 20) at room temperature and placed in primary antibody (25 mmol L−1 Tris–HCl, 150 mmol L−1 NaCl, 5% milk, and 0.10% Tween 20; 1:1000 antibody) overnight at 4°C. Nitrocellulose membranes were washed three times in wash buffer (25 mmol L−1 Tris–HCl, 150 mmol L−1 NaCl, 0.2% Tween 20). Primary antibody was detected by using horseradish peroxidase-linked goat anti-mouse or goat anti-rabbit IgG antibodies and visualized with the enhanced chemiluminescent detection system (Amersham–Pharmacia Biotech). Western blot experiments were performed at least three times.
Immunohistochemistry
Expression of p-Akt1 in gastric tissues was examined by means of immunohistochemistry. A 4-μm section was cut from paraffin blocks of gastric tissues. Each section was mounted on a silane-coated glass slide and then deparaffinized and hydrated by passing through xylene and a graded series of ethanol. Antigen retrieval was performed for 20 min at 98°C in 0.01 mol L−1 sodium citrate buffer (pH 6.4), in a microwave oven. To block endogenous peroxidase activity the sections were incubated with 0.3% hydrogen peroxide for 30 min. After blocking for 30 min in 10% normal goat serum, the sections were incubated overnight, at 4°C, with polyclonal antibody against S473 p-Akt1 (1:200 dilution, Upstate, Cell Signaling Solutions, USA). The sections were washed in phosphate-buffered saline (PBS) then incubated with biotinylated second antibody followed by avidin peroxidase using the Vectastain ABC elite kit (Vector Laboratories, Burlingame, CA, USA). After counterstaining with hematoxylin and mounting, the slides were evaluated under a light microscope. Preimmune serum was used instead of the first antibody as negative control.
Evaluation of Immunostaining
All sections were examined independently by two observers to determine histopathological characteristics and specific immunoreactivity (IR). The staining was semiquantitatively evaluated by assigning a score for the intensity of immunoreactivity and for the proportion of cells positively stained (PROP). The product of these two values was used for calculation of the overall IR score (TS), as described elsewhere [19, 20]. The intensity of the immunoreactivity (intensity score) was classified into four categories: 0, no IR; 1, weak IR; 2, moderate IR; and 3, strong IR. The proportion of positive cells (PROP) was classified into four groups: 0, ≤5% of tumor cells with IR; 1, 5–25% of tumor cells with IR; 2, 26–50% of tumor cells with IR; 3, 51–75% of tumor cells with IR, and 4 >75% of tumor cells with IR. A total score of 0–12 was calculated by multiplying the positive cells score by the staining intensity grade and the totals were finally graded as negative (−, score:0–1); weak (+, 2–4); moderate (++, 5–8); and strong (+++, 9–12).
Reverse Transcription-polymerase Chain Reaction (RT-PCR) Analysis
Total RNAs were isolated from gastric cancer cell lines by use of the TRIzol Reagent in accordance with procedure suggested by the manufacturer (Life Technologies). Cells in exponential phase were collected. RNA pellets were dissolved in nuclease-free sterile water (Sangon, PRC), and their concentrations were determined by measurement of absorbance at 260 nm. Total RNA (10 μg) was incubated with 5 units of DNase I (Life Technologies) at 20°C for 15 min in 50 μL reaction buffer (20 mmol L−1 Tris, pH 8.4, 2 mmol L−1 MgCl2, and 50 mmol L−1 KCl). The reaction was heat-inactivated at 65°C for 15 min in the presence of 2.5 mmol L−1 EDTA. cDNA (10 μL) was mixed with 1 μL oligo(dT)18 primer (MBI, USA) and incubated at 70°C for 5 min. After annealing at 37°C for 5 min, 200 units RevertAid M-Mulv reverseTranscriptase (MBI, USA) was added and further incubation of 20 μL of the reaction mixture was performed at 42°C for 60 min and the reaction was then terminated by incubating at 70°C for 10 min. For PCR, primers for Akt isoforms were described by Okano et al. [21]: Akt1: sense: 5′-GCTGGACGATAGCTTGGA-3′, antisense: 5′-GATGACAGATAGCTGGTG-3′; Akt2: sense: 5′-GGCCCCTGATCAGACTCTA-3′, antisense: 5′-TCCTCAGTCGTGGAGGAGT-3′; Akt3: sense: 5′-GCAAGTGGACGAGAATAAGTCTC-3′, antisense: 5′-ACAATGGTGGGCTCATGACTTCC-3′; β-actin: sense: 5′-AGCGGGAAATCGTGCGTG-3′, antisense: 5′-CAGGGTACATGGTGGTGCC-3′. PCR was performed in a thermal cycler using 24 amplification cycles for β-actin and 28 amplification cycles for Akt (denaturing at 94°C for 45 s, annealing at 55°C for 45 s, and extension at 72°C for 45 s) in 50 μL reaction mixture consisting of 10 mmol L−1 Tris, pH 8.3, 50 mmol L−1 KCl, 1.5 mmol L−1 MgCl2, 200 mmol L−1 dNTPs, 200 nmol L−1 primers, 2.5 units of Taq DNA polymerase (Promega), and 1 μL cDNA. The PCR products were electrophoresed on a 1.5% agarose gel.
Monolayer Growth Rate
Cells in 200 μL medium were seeded in 96-well plates and grown under normal conditions. Cultures were assayed at the times indicated by addition of 20 μL 5 mg mL−1 MTT and incubation for 4 h at 37°C. The MTT-containing medium was aspirated, and 150 μL DMSO (Sigma) was added to lyze the cells and solubilize the formazon. The absorbance of the lysates at 490 nm was determined by use of a BioHit BP800 microplate reader.
Cell-cycle Analysis
Cells were fixed with 70% ice-cold ethanol and stocked at 4°C overnight, washed with PBS, and stained with propidium iodide(PI) (50 μg mL−1) in PBS supplemented with RNase (10 mg mL−1) for 30 min. Cell-cycle histograms were generated after analysis of PI-stained cells by fluorescence-activated cell sorting with a Becton–Dickinson FACScan. For each sample at least 1 × 104 events were recorded. Histograms generated by FACS were analyzed by ModFit Cell Cycle Analysis Software (Verity, Topsham, ME, USA) to determine the percentage of cells in each phase (G1, S, and G2/M).
Anchorage-independent Growth in Soft Agar
Sterile low-melting agarose (0.6%, 1 mL) in 10% FBS-supplemented RPMI1640 medium was added to a 12-well plate as the base layer. The cell layer was then prepared by diluting agarose to 0.3% with 103 cells in 10% FBS- RPMI1640 medium per 1.5 mL per well. Colonies greater than or equal to 100 μm in diameter were counted after incubation for 3 weeks at 37°C in a 5% CO2 incubator.
Statistical Analysis
Results are expressed as mean ± standard deviation (SD). Statistical analysis was performed with SPSS 10.0 statistical software (SPSS, Chicago, lL, USA). Student’s t-test and the nonparametric Mann–Whitney test were adopted. Significance was defined as P < 0.05.
Results
Levels of p-Akt1 Are Increased in Human Gastric Cancer
To examine the role of p-Akt1 in gastric cancer we detected expression of p-Akt1 in gastric cancer by use of Western blot analysis and immunohistochemistry. Fresh, surgical tumor tissue and adjacent tissue were subjected to Western blot analysis. Akt1 activity was assessed with phospho-specific antibodies against phosphorylated S473. It is apparent from Fig. 1 that total Akt1 was highly expressed in both tumor tissue and adjacent tissue, and that levels of p-Akt1 were significantly higher in tumor tissue than in adjacent tissue (P < 0.05). Thus high-level p-Akt1 protein expression correlates with the transformed phenotype, indicating that p-Akt1 may have a role in the tumorigenesis of gastric cancer.
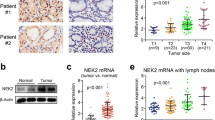
Immunostaining of p-Akt in human gastric cancer. Sections from paraffin-embedded tissues were subjected to antigen retrieval then immunostaining with anti p-Akt antibodies. Gastritis (A, ×100) results in weak and mainly cytoplasm staining. Intestinal metaplasia (B, ×200) results in moderate and diffuse staining. Carcinomas (C and D, ×200) result in intense staining in both membrane cytoplasm and nuclei. p-Akt staining is positive in lymphonodes metastasis (E, ×50, and F, ×400)
Expression and subcellular location of p-Akt1 were studied by immunohistochemistry of 101 gastric cancers, 10 intestinal metaplasias and dysplasias, and 13 normal gastric mucosal specimens. Detectable but weak and restricted p-Akt staining, primarily in cytoplasm, was observed for normal gastric epithelial cells (Fig. 2A) whereas approximately 30% of examined epithelial cells stained positive in intestinal metaplasia and dysplasia specimens, with an average staining score of 2 (Fig. 2B). In contrast, strongly positive p-Akt1 staining was observed for carcinoma cells. In these cells, intense p-Akt1 staining was detected in the membrane, cytoplasm, and, occasionally, in the nuclei (Fig. 2C, D). The results showed that p-Akt1 expression was substantially greater in carcinoma tissue than in normal or intestinal metaplasia and dysplasia tissue (P = 0.036). No association was found between p-Akt1 and sex or age, however (P = 0.687 and P = 0.704, respectively).
Western blot for p-Akt in human gastric cancer. A–C: Fresh surgical gastric cancer specimens (T) and adjacent tissues (N) were obtained from 12 patients at the time of surgery. Tissue homogenates were quantified, equal amounts of protein were subjected to SDS-PAGE, and Western blot was performed as described. Phosphorylation of Akt at S473 is shown in the top panel. Total levels of Akt protein are shown in the bottom panel. D and E: Relative expression of p-Akt and t-Akt. Bands in Western blot were determined by densitometric analysis and compared with β-actin. p-Akt/t-Akt were evaluated and analyzed by use of a paired t test. This showed that t-Akt1 is highly expressed both in tumor tissue and in adjacent tissue, but that p-Akt1 is significantly higher in tumor tissue than in adjacent tissue (P < 0.05)
Further analysis of the clinicopathological characteristics of the 101 gastric cancer specimens revealed a positive association of p-Akt1 staining intensity with the extent of tumor differentiation. In well-differentiated tumor cells average staining was weaker (Fig. 2C) whereas p-Akt1 was detected in most epithelial cells, with greater expression in poorly differentiated tumor cells (Fig. 2D) (P = 0.008). In the Gross type (Borrmann) classification, p-Akt1 staining was significantly different in different types of tumor (P = 0.001). With regard to TNM stage, p-Akt1 staining was much greater for patients in stages III + IV than for those in stages I + II (P = 0.031). There was a statistically significant difference between p-Akt1 staining in tumors with nodal metastasis and in those without (P = 0.022) (Table 1).
Levels of p-Akt1 Are Increased in Gastric Cancer Cells Under Serum Deprivation
Because we found Akt1 to be highly expressed in human gastric cancer, we next analyzed the expression of Akt1 in four gastric cancer cell lines and in an immortal gastric epithelial cell line grown under normal conditions (10% FBS) or deprived of serum overnight (0.1% FBS). Figure 3A shows that under normal growth conditions phosphorylation of S473 is observed for all five cell lines, and that levels of native p-Akt1 varied slightly among cell lines. When immortal GES-1 cells were deprived of serum, little S473 phosphorylation was observed. Levels of phosphorylated S473 were greatest in the MKN45 cells and moderate in AGS, SGC7901, and MGC803 cells. That S473 phosphorylation was maintained in most gastric cancer cell lines under serum deprivation, a condition in which Akt1 phosphorylation is commonly reduced or absent, suggested that Akt1 was activated in gastric cancer cells and may affect gastric cancer survival.
Akt protein and mRNA levels in human gastric cancer cells. A: Immunoblot analysis of Akt activity in five gastric cancer cell lines. Cells were exposed to a high (HS) or low (LS) concentration of serum for 12 h. Phosphorylation of Akt1 at S473 is shown in the top panel. Total levels of Akt1 protein are shown in the bottom panel. This shows that levels of p-Akt1 are increased in gastric cancer cells under serum deprivation. B: LY294002 (LY) inhibited phosphorylation of S473. Cells were placed in LS for 12 h. LY294002 (50 mmol L−1) was added 4 h before preparation of cell lysates. Phosphorylation of Akt at S473 is shown in the top panel. Total levels of Akt protein levels are shown in the bottom panel. LY294002 inhibited phosphorylation of S473. C: RT-PCR analysis of Akt1, Akt2, and Akt3 mRNA expression in different human gastric cancer cell lines. The Akt1 mRNA was ubiquitously expressed in all the cell lines examined, irrespective of their origin (top panel). Interestingly, expression of Akt2 and Akt3 mRNA was variable among the gastric cancer cell lines (2nd and 3rd panel). Positive signals for Akt2 and Akt3 were obtained for GES, MKN45, MGC803 and SGC7901cells. The β-actin mRNA (bottom panel) was amplified as an internal control. M, marker; NGM, normal gastric mucosa. A representative experiment from three independent experiments is shown
To demonstrate that Akt1 phosphorylation depended on PI3K activity, we tested whether Akt1 phosphorylation was inhibited by LY294002, a PI3K inhibitor [22]. Figure 3B shows that LY294002 inhibited phosphorylation of S473 in the gastric cancer cell lines that maintain S473 phosphorylation under serum deprivation. Native Akt1 levels did not change.
To determine the expression profile of Akt isoforms expressed in these five cell lines, we performed RT-PCR with isoform specific primers. Figure 3C shows that the mRNA of both Akt isoforms was expressed in the five cell lines tested. Akt1 expression was ubiquitous whereas expression of Akt2 and Akt3 was variable, and even low in AGS cells. The expression pattern did not reveal any obvious correlation with cell type.
LY294002 Inhibits Proliferation of Gastric Cancer Cells and Regulates G1 Cell Cycle Progression
P-Akt was mainly observed in the proliferative region of gastric glands and in the area of intestinal metaplasia, suggesting that Akt activation increases cell proliferation by promoting cell-cycle progression [23]. To demonstrate that active Akt promotes cellular proliferation of gastric cancer cell lines, we initially used a pharmacological approach to alter Akt activity and assess cell proliferation. Five gastric cell lines were seeded and cultured for 24 h, then incubated in the presence or absence of the PI3K inhibitor, LY294002. Total cells were harvested 24, 48, and 72 h after incubation. As shown in Fig. 4A, proliferation of GES-1 cells was slightly reduced by high concentrations of LY294002 only (25.5 and 50 μmol L−1) 24 h after the treatment. After treatment for 48 and 72 h, however, proliferation of the cells was significantly inhibited by LY294002 in a dose and time-dependent manner. When gastric cancer cells containing active Akt1 were treated with LY294002, typical growth inhibition was observed. Cell proliferation was significantly inhibited by LY294002 in a dose-dependent manner over a period of 72 h (Fig. 4B–E). These results indicate that PI3K/Akt may affect the proliferation of gastric cancer cells.
PI3K/Akt was required for the proliferation and G1 progression of gastric cancer cells. A–E: Cells were then cultured in the absence or presence of LY294002 (12.5, 25 and 50 μmol L−1) for 24, 48, and 72 h. Cell number was indicated by MTT assay. The data represents means and standard error from three independent experiments. The proliferation assays were performed in triplicate. Normalized cell number was significantly decreased compared to the control at the same time point (P < 0.05; Student’s T test). (A: GES-1, B: MGC803, C: AGS, D: SGC7901, E: MKN45) F: Inhibition of PI3K/Akt leads to G1 arrest in gastric cancer cells. Cells were cultured in a 90 mm plate at a density of 1 × 106 cells/plate in RPMI1640 supplemented with 10% FBS for 24 h. The cells were then cultured in the absence or presence of LY294002 (10 μmol L−1) for 24 h. Cell cycle was analyzed
To determine whether inhibition of Akt activity by LY294002 affects cell cycle progression, cells were treated with LY294002 (0 or 10 μmol L−1) and the cell cycle distribution was analyzed by flow cytometry. A histogram for G1 DNA content is shown in Fig. 4F. Treatment of highly Akt1-active cell lines with 10 μmol L−1 LY294002 for 24 h increased the percentage of the cell population at the G1 phase whereas for low Akt1-activity GES-1 cells inhibition of Akt activity had only a slight effect on cell cycle distribution. Because GES-1 is an immortal cell line, however, its regulation mechanisms might be quite different from those of other tumor cells. These data indicate that PI3K/Akt signaling is required for the G1 cell cycle. Inhibition of PI3K/Akt activity may block cellular entry into the S phase by arresting cells at G1 phase.
Because LY294002 inhibits gastric cancer cell proliferation, we then used a genetic approach to alter Akt activity and assessed cell proliferation, to confirm and extend the data generated by use of LY294002. Because levels of Akt phosphorylation are moderate in AGS cells, and are somewhat sensitive to serum deprivation, we chose AGS cells for the transfection experiments. AGS cells were transfected with HA-tagged K179M-Akt1 or myr-Akt1. Compared with controls, growth and colony formation were significantly reduced when the cells were transfected with HA-K179M-Akt1 whereas the opposite results were obtained for cells transfected with HA-myr-Akt1. Review of the cell cycle revealed retardation of HA-K179M-Akt1 transfectants in the G1 phase and a higher proportion in the S phase for HA-myr-Akt1 (Fig. 5).
Proliferation of Akt mutant transfectants of AGS cells. A: Monolayer cell growth. 103 cells were seeded in 96-well plates and grown under normal conditions. Viable cells were analyzed using MTT methods at the indicated times. B: Anchorage-independent growth in soft agar of AGS cells. AGS transfectants were grown in 0.3% agarose. Colonies greater than or equal to 100 μm in diameter were counted after 3 weeks and the cell cycle was analyzed. The data are means and standard error from three independent experiments. *Indicates that the number of cells was significantly different from the control at the same time (P < 0.05; One-way ANOVA)
Discussion
Several studies have discovered Akt gene amplification or have investigated kinase and protein levels in human cancers. Possible genetic alterations related to Akt were first observed in gastric tumors. Although initial studies by Staal [4] may have been premature, they still form an excellent rationale for further investigation of the involvement of Akt in gastric cancer. Nam et al. [23] also have published a good report on the prognostic implications of p-Akt in human gastric cancer. Our study emphasizes the role of Akt1 in the development and malignancy of gastric carcinoma in vivo and in vitro.
Gastric carcinogenesis is a multi-step process with morphological progression involving multiple genetic and epigenetic events [24]. Our immunohistochemistry analysis shows that p-Akt1 was up-regulated at an early stage during this process, for example in intestinal metaplasia. p-Akt1 may, therefore, affect the onset of gastric carcinogenesis.
Western blot analysis showed levels of p-Akt1, but not t-Akt1, were significantly different in tumor and non-tumor tissue from gastric cancer patients. This is consistent with previous reports on gastric cancer [23]. Further evaluation here revealed p-Akt levels were even higher in poorly differentiated tumors and in tumors with nodal metastasis or in the late TNM clinical stage. These findings indicate that p-Akt1 might also participate in the progression of gastric cancer. These findings for gastric cancer are not consistent with those from Nam’s study, in which expression of p-Akt was high in early-stage pTNM [23]. This discrepancy may be because the samples were not identical—in our study samples were mainly in pTNM stages II and III whereas in their study approximately 43% (148/347) of the samples were in pTNM I stage.
Subcellular location is also important for Akt activation. In resting nonstimulated cells, most phospho-Akt resides in the cytoplasm. Activation of Akt occurs at the plasma membrane and has been shown, in several studies, to be followed by its translocation to both the cytoplasm and nuclei [25–27]. Here we have shown that in normal gastric mucosa p-Akt1 is expressed mainly in the cytoplasm of a very limited number of cells, and less in the plasma membrane; this might contribute to gland proliferation or acid secretion. In gastric cancer tissues, however, p-Akt1 is expressed in both the cytoplasm and the membrane, even in nuclei in some specimens. It is worth noting that many of the substrates of Akt are proteins that function in the membranes and nuclei.
Possible mechanisms of Akt phosphorylation and activation include activation of upstream kinases, for example PI3K, and/or inhibition of lipid or protein phosphatases, for example PTEN, or mutation of Akt itself. Because t-Akt protein levels did not change in our experiments, although not examined in cohorts, the causes of increased Akt phosphorylation in gastric cancers may include mutation of PI3K or deletion/inactivation of PTEN [1]. The ability of PI3K inhibitors to reduce Akt1 phosphorylation suggests that activation of Akt1 is PI3K-dependent, although we have not directly assayed PI3K activity in the tumor lysates and these cells. Kinases or GFRs upstream from PI3K could also be activated and responsible for Akt activation. We have detected high expression of EerbB2, a member of the growth factor glycoprotein family, in some gastric cancer cells, for example SGC7901 [28]. The stress of serum deprivation in the study might have a hint effect in activating PI3K in these cells. Aberrant phosphatase function may also contribute to Akt activation in gastric cancer cells. The lipid phosphatase PTEN has been shown to regulate Akt activity and has been reported to reduce expression in gastric cancers. But, as Sato reported [29], only one of the 58 primary tumors (1.7%) carried a somatic 5-bp deletion in intron 7 of PTEN, which did not alter the mRNA sequence, and no mutations were detected in any of the ten gastric cancer cell lines. Similar levels of PTEN mRNA expression were observed in all cell lines and primary tumors studied by RT-PCR, and PTEN promoter CpG islands remained unmethylated. It therefore seemed that PTEN does not participate in gastric carcinogenesis as a tumor-suppressor gene and inhibitor of Akt. We generally regard PI3K as one of the reasons for the activity of Akt1.
By semiquantitative RT-PCR, we observed that Akt1 was up-regulated in most gastric cancer cell lines at the mRNA level, suggesting that the up-regulation of Akt1 occurred at the transcriptional level in gastric cancer cells. There was, however, no significant difference between Akt1 mRNA expression in well-differentiated and poorly differentiated cells, which did not correlate well with Akt1 protein expression. Hence non-transcriptional mechanisms may also exist that result in up-regulation of Akt1 protein expression in gastric cancer cell lines. In addition, semiquantitative RT-PCR analysis also provided evidence for upregulation of Akt2 andAkt3 mRNAs in most gastric cancer cell lines.
It has been reported that Akt1 can affect proliferation as a result of signals to the cell-cycle machinery. In this study, we found that LY294002 and kinase-dead Akt1 (K179M-Akt1) inhibited cell growth and colony formation and induced G1 cell-cycle arrest whereas constitutively active Akt1(myr-Akt1) enhanced cell growth and G1 cell cycle progression to some distance. The cell cycle is regulated by the coordinated action of Cyclin/Cdk complexes and CKIs. Levels of Cyclin D are controlled at the levels of transcription, translation, and protein stability, which are important in the G1/S phase-transcriptional and post-transcriptional levels, by distinct mechanisms. PI3K/Akt signaling has recently been shown to stimulate induction of Cyclin D1 expression [30] and to be important in preventing Cyclin D1 degradation by regulating the activity of the Cyclin D1 kinase glycogen synthase-3β(GSK3β) [31]. P21cip1/waf1 and p27kip1 are two major cyclin/CDK inhibitors. Akt phosphorylates p21cip1/waf1 on residue Thr-145 and p27kip1 on residue Thr-157. The phosphorylation on these residues inhibits p21 or p27 nuclear localization and affinity to Cdk, leading to activation of cyclin/CDK and DNA replication [9–12]. We believe p-Akt1 might regulate the cell cycle by targeting these cell-cycle-related proteins.
In conclusion we have identified Akt as an activited kinase that contributes to gastric cancer progression and promotes proliferation of gastric cancer cells.
References
Vivanco CL (2002) The phosphatidylinositol 3-kinase-AKT pathway in human cancer. Nature Rev 2:489–501
Cheng JQ, Lindsley CW, Cheng GZ, Yang H, Nicosia SV (2005) The Akt/PKB pathway: molecular target for cancer drug discovery. Oncogene 24:7482–7492
Laine J, Kunstle G, Obata T, Noguchi M (2002) Differential regulation of Akt kinase isoforms by the members of the TCL1 oncogene family. J Biol Chem 227:3743–3751
Staal SP (1987) Molecular cloning of akt oncogene and its human homologues AKT1 and AKT2: amplification of AKT1 in a primary adenocarcinoma. Proc Natl Acad Sci USA 84:5034–5037
Bellacosa A, de Feo D, Godwin AK, Bell DW, Cheng JQ, Altomare M, Wan M, Dubeau L, Scambia G (1995) Molecular alterations of the AKT2 oncogene in ovarian and breast carcinomas. Int J Cancer 64:280–285
Cheng JQ, Ruggeri B, Klein WM, Sonoda G, Altomare DA, Watson DK, Testa JR (1996) Amplification of AKT2 in human pancreatic cells and inhibition of AKT2 expression and tumorigenicity by antisense RNA. Proc Natl Acad Sci USA 93:3636–3641
Mende I, Malstrom S, Tsichlis PN, Vogt PK, Aoki M (2001) Oncogenic transformation induced by membrane-targeted Akt2 and Akt3. Oncogene 20:4419–4423
Malik SN, Brattain M, Ghosh PM, Troyer DA, Prihoda T, Bedolla R, Kreisberg JI (2002) Immunohistochemical demonstration of phospho-Akt in high Gleason grade prostate cancer. Clin Cancer Res 8:1168–1171
Li Y, Dowbenko D, Lasky LA (2002) AKT/PKB phosphorylation of p21Cip/WAF1 enhances protein stability of p21Cip/WAF1 and promotes cell survival. J Biol Chem 277:11352–11361
Shin I, Yakes FM, Rojo F, Shin NY, Bakin AV, Baselga J, Arteaga CL (2002) PKB/Akt mediates cell-cycle progression by phosphorylation of p27(Kip1) at threonine 157 and modulation of its cellular localization. Nat Med 8:1145–1152
Dan HC, Sun M, Yang L, Feldman RI, Sui XM, Ou CC, Nellist M, Yeung RS, Halley DJ, Nicosia SV, Cheng JQ (2002) Phosphatidylinositol 3-kinase/Akt pathway regulates tuberous sclerosis tumor suppressor complex by phosphorylation of tuberin. J Biol Chem 277:35364–35370
Vousden KH, Lu X (2002) Live or let die: the cell’s response to p53. Nat Rev 2:594–604
Wu K, Crusius JBA, Shivanarda S, Fan DM, Pena AS (2002) The immunogenetic and pathogenesis of gastric cancer. Drugs Today 38:391–417
Alberts SR, Cervantes A, van de Velde CJ (2003) Gastric cancer: epidemiology, pathology and treatment, Ann. Oncol 14:ii31–ii36
Ren J, Chen Z, Zhou SJ, Zhang XY, Pan BR, Fan DM (2000) Detection of circulating gastric carcinoma associated antigen MG7-Ag in human sera using an established single determinant immuno-polymerase chain reaction technique. Cancer 88:280–285
Roder DM (2002) The epidemiology of gastric cancer. Gastric Cancer 5:5–11
Yang K, Tao N (1994) Establishment and biological characterization of human gastric epithelial cell lines GES-1. Chin J Oncol 16:7–10
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chariyalertsak S, Sirikulchayanonta V, Mayer D, Chariyalertsak S, Sirikulchayanonta V, Mayer D, Kopp-Schneider A, Furstenberger G, Marks F, Muller-Decker K (2001) Aberrant cyclooxygenase isozyme expression in human intrahepatic cholangiocarcinoma. Gut 48:80–86
Maaser K, Daubler P, Barthel B, Heine B, von Lampe B, Stein H, Hoffmeister B, Scherer H, Scherubl H (2003) Oesophageal squamous cell neoplasia in head and neck cancer patients: upregulation of COX-2 during carcinogenesis. Br J Cancer 88:1217–1222
Okano J, Gaslightwala I, Birnbaum MJ, Rustgi AK, Nakagawa H (2000) Akt/protein kinase B isoforms are differentially regulated by epidermal growth factor stimulation in esophageal cancer cells. J Biol Chem 275:30934–30942
Cross MJ, Stewart A, Hodgkin MN, Kerr DJ, Wakelam MJ (1995) Wortmannin and its structural analogue demethoxyviridin inhibit stimulated phospholipase A2 activity in Swiss 3T3 cells. Wortmannin is not a specific inhibitor of phosphatidylinositol 3-kinase. J Biol Chem 270:25352–25355
Nam SY, Lee HS, Jung GA, Choi J, Cho SJ, Kim MK, Kim WH, Lee BL (2003) Akt/PKB activation in gastric carcinomas correlates with linicopathologic variables and prognosis. APMIS 11:1105–1113
Correa P (1988) A human model of gastric carcinogenesis. Cancer Res 48:3554–3560
Andjelkovic M, Alessi DR, Meier R, Fernandez A, Lamb NJ, Frech M, Cron P, Cohen P, Lucocq JM, Hemmings BA (1997) Role of translocation in the activation and function of protein kinase B. J Biol Chem 272:31515–31524
Filippa N, Sable CL, Hemmings BA, Van Obberghen E (2000) Effect of phosphoinositide-dependent kinase 1 on protein kinase B translocation and its subsequent activation. Mol Cell Biol 20:5712–5721
Dufner A, Andjelkovic M, Burgering BM, Hemmings BA, Thomas G (1999) Protein kinase B localization and activation differentially affect S6 kinase 1 activity and eukaryotic translation initiation factor 4E-binding protein 1 phosphorylation. Mol Cell Biol 19:4525–4534
Bi F, Fan DM, Hui HX, Wang CJ, Zhang XY (2001) Reversion of the malignant phenotype of gastric cancer cell SGC7901 by c-e-rbB-z-specific hammerhead ribozyme. Cancer Gene Ther 8:835–842
Sato K, Tamura G, Tsuchiya T, Endoh Y, Sakata K, Motoyama T, Usuba O, Kimura W, Terashima M, Nishizuka S, Zou T, Meltzer SJ (2002) Analysis of genetic and epigenetic alterations of the PTEN gene in gastric cancer. Virchows Arch 440:160–165
Borgatti P, Martelli AM, Bellacosa A, Casto R, Massari L, Capitani S, Neri LM (2000) Translocation of Akt/PKB to the nucleus of osteoblast-like MC3T3-E1 cells exposed to proliferative growth factors. FEBS Lett 477:27–32
Paradis S, Ruvkun G (1998) Caenorhabditis elegans Akt/PKB transduces insulin receptor-like signals from AGE-1 PI3 kinase to the DAF-16 transcription factor. Genes Dev 12:2488–2498
Acknowledgment
This work was supported by National Natural Sciences Foundation of China. (No. 30400530 and 30200121).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Han, Z., Wu, K., Shen, H. et al. Akt1/protein Kinase Bα is Involved in Gastric Cancer Progression and Cell Proliferation. Dig Dis Sci 53, 1801–1810 (2008). https://doi.org/10.1007/s10620-007-9824-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-007-9824-2