Abstract
The purpose of this study was to investigate and compare the specificity, sensitivity, and area under curve (AUC) of the lactulose/mannitol ratio, lactulose/creatinine ratio, and lactulose recovery and their diagnostic value for intestinal permeability assessment within the absorption lactulose/mannitol (L/M) test. Results The value of the lactulose/mannitol ratio, lactulose/creatinine ratio, and the percentage of lactulose recovery in Crohn’s disease (0.0763 ± 0.0369; 99.62 ± 67.87; 1.0478 ± 0.6148) and in liver cirrhosis (0.0517 ± 0.0365; 54.65 ± 53.26; 0.838 ± 0.929) were significantly different from the values measured in the control group (0.0123 ± 0.0081; 10.95 ± 7.07; 0.2438 ± 0.1568), P < 0.0001–0.002). In Crohn’s disease, specificity, sensitivity, and AUC were 100%, 89.5%, and 0.987, respectively, of the lactulose/mannitol ratio at a cut-off level of 0.022. In liver cirrhosis, the test characteristics were 88.5%, 84.2%, and 0.910 at a cut-off level of 0.018. Conclusion The lactulose/mannitol ratio was evaluated to have the highest diagnostic value to assess intestinal permeability.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Intestinal permeability may vary with changing pathological circumstances transiently as well as persistently. The variations can occur due to liver cirrhosis, nonspecific intestinal inflammations, acute pancreatitis, chemotherapy, etc. [1–5].
Many agents were tested to assess their characteristic for measuring intestinal permeability. Currently, two agents are used as optimal markers: mannitol/l-rhamnose to evaluate transcellular absorption and lactulose/cellobiose to evaluate paracellular absorption.
Lactulose and mannitol are convenient and the most frequently used agents in present clinical practice, enabling a test of the intestinal permeability (test L/M). They present insignificant affinity for the monosaccharide transporter system, therefore being passively absorbed and having the advantage of not being metabolized and excreted in urine unchanged [4, 6, 7].
Determination of lactulose and mannitol are performed using the gas chromatography (GC) and high-performance liquid chromatography (HPLC) method, and enzymatic methods [8–11].
Medical literature and clinical practice report different methods for expression and evaluation of measured (obtained) values of lactulose and mannitol in urine: the lactulose/mannitol ratio, the lactulose/creatinine ratio, and lactulose excretion in urine collected during the test duration, which is usually 5 h, measured as a percentage of the orally administered dose. The purpose of our study was to evaluate the diagnostic value (sensitivity, specificity, and AUC), and its separate aspects, provided by the L/M test in a cohort of patients with Crohn’s disease and liver cirrhosis.
Study Cohort and Methods
The study included 21 men with a mean age of 57 (47–67) years and five women with a mean age of 52 (40–61) years, diagnosed with alcohol-related liver cirrhosis, CHILD score B–C. Clinically significant ascites was present in six patients.
The cohort of patients with Crohn’s disease included seven men with a mean age of 31 (20–45) years and 13 women with a mean age of 33 (22–47) years with the active form of Crohn’s disease (A—CDAI of 220–280) affecting terminal ileum and colon. The control group consisted of nine men with a mean age of 35 (24–60) years and 11 women with a mean age of 27 (22–50) years; these subjects were either employees of University Hospital Brno or students of the medical faculty presenting with no symptoms of disease of the digestive tract.
L/M Test Description
In the morning, the examined subject was asked to empty the urinary bladder and consequently drink on an empty stomach a test solution consisting of 5 g lactulose, 10 g mannitol, and 40 g sucrose diluted in 100 mL water.
The patient’s urine was then collected for the next 5 h. The urine of each subject was stored in a vessel containing chlorhexidine as a conservative. The urine was stirred, the volume precisely measured, and an aliquot specimen sent to the laboratory. Until the laboratory analysis, the samples were stored at −20°C.
Evaluation Methods
To detect lactulose and mannitol in urine, we used the set sugar absorption test; lactulose, mannitol in urine-enzymatic, Instruchemie, NL.
Lactulose determination is a fully enzymatic method. In the first stage, lactulose is hydrolysed into fructose and galactose using β-galactosidase. The fructose formed is further quantitatively converted through three consecutive enzymatic reactions, in the presence of hexokinase, glucose phosphate isomerase, and glucose 6-phosphate dehydrogenase, to gluconate-6-phosphate and NADPH. The activity of mannitol dehydrogenase catalyzes the conversion of mannitol to fructose and NADH.
The NADPH and NADH formed in the reactions were estimated by measuring the change in absorbance at 340 nm.
The methods were performed by using a Cobas Mira analyzer. Creatinine in urine was measured on a Modular (Roche) analyzer, with a kinetic blanked compensated Jaffé method. The values obtained allowed the calculation of the lactulose/mannitol ratio, the lactulose/creatinine ratio, and the percentage of urinary lactulose excretion of the ingested dose.
Statistical Methods
The normal (Gaussian) distribution of all the parameters under study was verified with the Kolmogorov–Smirnov test.
For each parameter, in each patient cohort, the data collected were processed with Student’s t-test (significant at P < 0.05), calculating the mean, the standard deviation (SD), and the statistical significance of the differences versus the control group.
The values of the cut-off levels, the sensitivity, the specificity, and the area under curve (AUC) were calculated to determine the tested parameters in both cohorts.
The diagnostic value of the lactulose/mannitol ratio, the lactulose/creatinine ratio, and the percentage of urinary lactulose excretion of the ingested dose were expressed as the comparison of their receiver operating characteristic (ROC) curves.
MedCalc for Windows version 9.3.1 was used for the statistical calculations, receiver operating characteristic (ROC), cut-off levels for maximal sensitivity and specificity, and the graphical representations.
Results
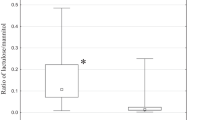
The basic statistical indicators of the tested parameters in the L/M test in the cohort of patients with Crohn’s disease and liver cirrhosis versus the control cohort are presented in Table 1. In both cohorts, the mean value of the three tested parameters was highly significantly different from the control group. The sensitivity, specificity, and AUC of the determined cut-off levels in Crohn’s disease and in liver cirrhosis are presented in Table 2A, B. In the cohort of patients with Crohn’s disease, the calculated lactulose/mannitol ratios demonstrated nearly ideal values. The next two indicators, the lactulose/creatinine ratio and the percentage of lactulose recovery showed nonideal but still very reliable and comparable values. In the cohort of patients with liver cirrhosis, the calculated values of the sensitivity, the specificity, and the AUC were lower then in the foregoing cohort, nonetheless, the priority given to the lactulose/mannitol ratio was preserved.
The area under the curve (ROC curves area) is a suitable parameter to summarize the overall discriminative or diagnostic value of a model featuring Crohn’s disease (Fig. 1a) and liver cirrhosis (Fig. 1b). For both diseases, the diagnostic value of the tested parameters was assessed as follows (in order of decreasing importance): lactulose/mannitol, lactulose/creatinine, and lactulose recovery.
Discussion
Morphological and functional integrity of the small intestine underlies two opposite processes: the highly effective mechanism of nutrient absorption that includes specific transport movements and, in the opposite direction, a highly effective and complex defence mechanism that serves as a protective barrier to prevent harmful bacteria, toxins, antigens, and other pathogens from entering the body. When the epithelium of the small intestine become impaired, it can lose the tight cell junctions and allow bacteria and toxins to enter the blood stream, which may cause sepsis and multiple organ failure.
Macromolecules with antigen characteristics may produce local or system response of the immune system, leading to serious allergies. The pathological symptoms and reactions that are a consequence of increased intestinal permeability is a condition called leaky gut syndrome [12, 13].
Schematically, the complex processes of the intestinal mucous membrane can be divided into transcellular and paracellular fluxes. It follows that mucosal intestinal permeability can be assessed on the basis of the quality and functions of paracellular structures of the intestinal mucous membrane.
The assessment of the mucosal intestinal integrity (permeability) is measured by urinary excretion of orally administered test unmetabolizable sugars. The usual scheme of the test lies in oral administration of two sugars of a different molecular size and with a different mechanism of absorption. Monosaccharides, such as l-rhamnose or mannitol, are absorbed through the transcellular pathway and reflect the degree of absorption of small molecules (<0.65 nm). Disaccharides, such as lactulose or cellobiose, are absorbed through the paracellular junction complex, which corresponds to the permeability of larger molecules (>0.93 nm).
The results of our investigation justify the priority given to the L/M ratio used to determine and appraise the degree of intestinal permeability within the L/M test. A further advantage of this ratio is the elimination of errors due to nonmucosal factors, because variables such as rate of gastric emptying, intestinal transit, impairment of renal function, and diuresis should affect both sugars similarly [14–18].
References
Ammori BJ, Leeder PC, King RF et al (1999) Early increase in intestinal permeability in patients with severe acute pancreatitis: correlation with endotoxemia, organ failure, and mortality. J Gastrointest Surg 3:252–262
Daniele B, Secondulfo M, De Vivo R et al (2001) Effect of chemotherapy with 5-fluorouracil on intestinal permeability and absorption in patients with advanced colorectal cancer. J Clin Gastroenterol 3:228–230
Penalva JC, Martínez A, Esteban A et al (2002) Intestinal permeability alteration in acute pancreatitis. Pancreatology 2:167–187
Welcker K, Martin A, Kolle P, Siebeck M, Gross M (2004) Increased intestinal permeability in patients with inflammatory bowel disease. Eur J Res 9:456–460
Soderholm JD, Olaison G, Lindberg E et al (1999) Different intestinal permeability patterns in relatives and spouses of patients with Crohn's disease: an inherited defect in mucosal defence? GUT 44:96–100
Cox MA, Lewis KO, Cooper BT (1999) Measurement of small intestinal permeability markers, lactulose, and mannitol in serum: results in coeliac disease. Dig Dis Sci 2:402–406
Feld JJ, Meddings J, Heathcote EJ (2006) Abnormal intestinal permeability in primary biliary cirrhosis. Dig Dis Sci 9:1607–1613
Marsilio R, D'Antiga L, Zancan L, Dussini N, Zacchello F (1998) Simultaneous HPLC determination with light-scattering detection of lactulose and mannitol in studies of intestinal permeability in pediatrics. Clin Chem 44:1685–1691
Miki K, Butler R, Moore D, Favidson G (1996) Rapid and simultaneous quantification of rhamnose, mannitol, and lactulose in urine by HPLC for testing intestinal permeability in pediatric practice. Clin Chem 42:71–75
Hessels J, Snoeyink EJ, Platenkamp AJ, Voortman G, Steggink J, Eidhof HH (2003) Assessment of intestinal permeability: enzymatic determination of urinary mannitol, raffinose, sucrose and lactulose on Hitachi analyzer. Clin Chem Lab Med 1:33–38
Liverani E, Silveri NG, Gasbarrini G, Mingrone G (2000) Intestinal permeability increases with the severity of abdominal trauma: a comparison between gas liquid chromatographic and enzymatic method. Hepatogastroenterol 34:1037–1041
Zhengwu S, Xiandong W, Andersson R (1998) Role of intestinal permeability in monitoring mucosal barrier function. History, methodology, and significance of pathophysiology. Dig Surg 5:386–397
Keshavarizian A, Holems EW, Patel M, Iber F, Fields JZ, Pethkar S (1999) Leaky gut in alcoholic cirrhosis: a possible mechanism for alcohol-induced liver damage. Am J Gastroenterol 1:200–207
Di Leo V, D´Inca R, Diaz-Granado N et al (2003) Lactulose/mannitol test has high efficacy for excluding organic causes of chronic diarrhea. Am J Gastroenterol 10:2245–2252
Indrio F, Raimondi F, Laforgia N, Riezzo G, Polimeno L, Francavilla R (2007) Effect of hyperbilirubinemia on intestinal permeability in healthy term newborn. Acta Pediatr 1:73–75
Pascula S, Such J, Esteban A et al (2003) Intestinal permeability is increased in patients with advanced cirrhosis. Hepatogastroenterol 53:1482–1486
Spahr L, Bresson-Handi S, Amann P et al (2007) Allopurino, oxidative stress and intestinal permeability in patients with cirrhosis: an open-label pilot study. Liver Int 1:54–60
Colome G, Sierra C, Blasco J, Garcia MV, Valverde E, Sanchez E (2007) Intestinal permeability in different feedings in infancy. Acta Pediatr 1:69–72
Acknowledgment
This work was supported by the Grant Agency of the Health Ministerium, Czech Republic NR9084-3.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dastych, M., Dastych, M., Novotná, H. et al. Lactulose/Mannitol Test and Specificity, Sensitivity, and Area under Curve of Intestinal Permeability Parameters in Patients with Liver Cirrhosis and Crohn's Disease. Dig Dis Sci 53, 2789–2792 (2008). https://doi.org/10.1007/s10620-007-0184-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-007-0184-8