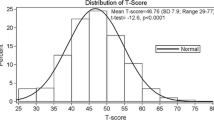
Abstract
In addition to chronic hepatitis, many individuals infected with hepatitis C virus (HCV) suffer from fatigue, which may compromise their health-related quality of life (HRQL). To assess systematically health-related quality of life (HRQL) in patients with chronic hepatitis C and to determine if any clinical, biochemical, virologic, demographic, and histologic features are associated with HRQL status. In this cross-sectional observational study, one hundred thirty patients with chronic HCV infection (HCV RNA positive by PCR) and 61 healthy controls were enrolled from a tertiary care teaching medical center. All patients and controls completed one generic HRQL questionnaire (MOS SF-36) and one liver-disease specific instrument (Chronic Liver Disease Questionnaire, CLDQ). Ninety-five HCV patients and all the controls also completed a fatigue questionnaire (Chronic Fatigue Screener, CFS) and had immunologic markers determined (Cryoglobulin, Soluble IL-2 receptors, Rheumatoid Factor). We compared the HRQL of HCV-infected patients to the controls and, using data from other studies, to the general population, patients with diabetes, and patients with chronic low back pain. Patients with chronic HCV had greater HRQL impairment than healthy controls and those with type II diabetes. Fatigue was the most important symptom with negative impact on HRQL. Sixty-one percent of HCV-infected patients reported fatigue-related loss of activity. Additionally, other factors associated with HRQL were gender and histologic cirrhosis. Chronic HCV infection has a profound negative impact on patients’ HRQL. Disabling fatigue is the most important factor that contributes to loss of well-being in this relatively young group of patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hepatitis C virus (HCV) is one of the most important causes of chronic liver disease affecting nearly 170 million individuals worldwide [1]. In the United States, 3.9 million individuals have evidence of HCV infection, which is most prevalent in the most productive years of patient’s lives. HCV infection leads to chronic hepatitis in up to 75–85% of patients, with 20–25% developing cirrhosis [2–4]. Additionally, HCV infection is also associated with a variety of nonhepatic manifestations including vasculitis, arthritis, thyroid disease, and fatigue [5–10]. Both hepatic and the nonhepatic manifestations of HCV have a profound negative impact on patient’s health-related quality of life (HRQL) [11–14]. Psychosocial factors, the severity of the chronic liver disease (cirrhosis), and the extrahepatic manifestations of HCV, may all influence the degree of HRQL compromise.
Although autoimmune markers such as antinuclear antibody (ANA), rheumatoid factor (RF), cryoglobulins, sedimentation rate (ESR), antibodies directed against cardiolipin, thyroid, smooth muscle, and liver/kidney/microsomal (LKM) proteins are commonly found in this infection, their clinical implication remain unknown. From all these factors, RF is detected in 70–76% and cryoglobulins 40–45% of infected individuals, respectively [5–9]. In addition to these autoimmune markers, soluble interleukin-2 receptors (SIL-2R), another marker of immune activation, may also correlate with disease activity [10]. Although the exact role of these autoimmune markers is not clear, they may be associated with rheumatologic symptoms negatively affecting patients’ health-related quality of life (HRQL).
We therefore undertook a systematic study of HRQL and fatigue in HCV patients to evaluate the potential impact of a variety of clinicodemographic variables on patients’ well-being.
Methods
Patient selection
Consecutive patients with the diagnosis of chronic hepatitis C infection were enrolled from the outpatient practices of the Cleveland Clinic Foundation. Diagnosis of chronic hepatitis C was based on the presence of hepatitis C antibody (ELISA II), elevated ALT and detectable HCV RNA. We excluded patients if they had received interferon therapy within three months or had another major chronic illness requiring treatment.
A sample of healthy people (controls) was also recruited from primary care practices. These individuals were selected if they agreed to participate in the study, if they were being seen for their yearly physical examinations, and if they had no specific complaints or chronic illness requiring treatment.
Protocol
All patients with chronic hepatitis C and controls completed two HRQL questionnaires and underwent a complete physical examination. The initial 95 HCV patients also completed a chronic fatigue questionnaire and had immunologic markers determined (cryoglobulins, rheumatoid factor, and soluble interleukin-2 receptor). Similarly, 53 controls completed chronic fatigue screener.
Details of the questionnaires used for assessment of HRQL and fatigue [15–21] are as follows:
-
1.
Medical Outcomes Study Short Form 36 (SF-36), a widely used and validated generic HRQL questionnaire [17] includes 36 items divided into eight scales, which can be aggregated into two summary scores, a mental component summary (MCS) and a physical component summary (PCS). Scale scores for SF-36 range between 0 and 100, while PCS scores range between 8 and 73 and MCS score between 10 and 74. From the results of the medical outcomes study (MOS) and the National Survey of Health Status, scores for SF-36 are available for a variety of chronic conditions and the U.S. general population. In our study, SF-36 scores for patients with chronic hepatitis C were compared to another slowly progressive chronic condition, type II diabetes. Scores for patients with diabetes and the U.S. general population were obtained from SF-36 User’s Manual [18].
-
2.
The Chronic Liver Disease Questionnaire (CLDQ), a liver-disease specific HRQL questionnaire that includes 29 items divided into six domains (Abdominal Symptoms, Activity, Emotional Function, Fatigue, Systemic Symptoms, and Worry). Summary scores for each domain range from 1 (most impairment) to 7 (least impairment), and one can calculate an overall CLDQ score, also from 1 to 7. CLDQ is reliable and valid both for measuring differences between individuals at a point in time, and for longitudinal change over time [19–21]. CLDQ scores of HCV infected patients were compared to those of 61 healthy controls (Age = 44 ± 14, 35% male).
-
3.
Chronic Fatigue Screener (CFS) is a questionnaire [15] designed to focus on the features of chronic fatigue as defined by the Centers for Disease Control and Prevention and described by the International Chronic Fatigue Syndrome Study Group [16]. Extensively used in the studies of chronic fatigue, this questionnaire consists of 23 items based on yes/no answers. The first five items of this questionnaire deal with duration and severity of fatigue:
-
a.
Fatigue for at least 6 months.
-
b.
Lifelong fatigue.
-
c.
Fatigue-related reduction in occupational, educational, social, and personal activities.
-
d.
Fatigue related to exertion or work.
-
e.
Fatigue relieved by rest.
The next nine items are related to symptoms associated with chronic fatigue syndrome (chronic fatigue syndrome items):
-
a.
Impairment of short-term memory.
-
b.
Sore throat.
-
c.
Tender lymph nodes.
-
d.
Muscle pain.
-
e.
Unrefreshing sleep.
-
f.
Joint pain.
-
g.
Headaches
-
h.
Malaise lasting greater than 24 h after exertion.
-
i.
Presence of these symptoms simultaneously for the past 6 months.
The last nine items in the CFS questionnaire relates to patients’ psychological health.
-
a.
Using this questionnaire and established guidelines [16], chronic fatigue alone [CF(+)] was defined as fatigue that persists ≥6 months (that is, a “YES” answer to the first question). By contrast, chronic fatigue syndrome [CFS(+)] was diagnosed if in addition to fatigue at 6 months, patients answer “YES” to four of the chronic fatigue syndrome items being present concurrently in the past 6 months.
Using the chronic fatigue screener, HCV patients and controls were divided into those with chronic fatigue only [CF(+)], chronic fatigue syndrome [CFS(+)], or no fatigue.
Clinical and laboratory data
The following clinical and laboratory data were obtained:
Cryoglobulins (Cryo): Ten mL of serum in three 7 mL red top tubes were placed in 37 degree Celsius water and delivered to the Immunopathology Lab. Cryoglobulins were quantitatively determined by spectrophotometry as previously described (reference range 0–50 micrograms/mL) [22].
Rheumatoid Factor (RF): One mL of serum in 6 mL serum separator tube was sent to immunopathology lab and used for the test. RF is quantitatively measured by the nephelometry method with a reference range 0–19 SIU/mL (established on adult sera during June 1996 at the Cleveland Clinic Foundation) [23].
Soluble Interleukin-2 Receptor(SIL-2R): Three mL of serum in 6 mL serum separator tube at room temperature were delivered to Immunopathology Lab within 2 hours and stored. Soluble Interleuken-2 Receptor (SIL-2R) is quantitatively measured by an in vitro enzyme-linked immunosorbent assay (ELISA), with results being reported as U/mL (0–6400 U/mL).
Other Clinical Data: All patients had routine chemistries including aminotransferases, total bilirubin, serum albumin, and prothrombin time determined. Level of viremia was determined by HCV RNA by PCR with threshold for detection at 1×103 copies /mL (Amplicore HCV Monitor7, Roche Molecular Systems, Brachburg, NJ).
Diagnosis of cirrhosis was established histologically and disease severity was measured using modified Child-Pugh classification [25, 26].
Histologic severity of liver disease was determined by histologic activity index (HAI). Widely used in most clinical studies of hepatitis C, HAI provides a numerical score for both inflammatory and fibrosis components of liver biopsy specimens obtained in patients with hepatitis C. A higher inflammatory and fibrosis score suggests more severe histologic disease [27].
The Institutional Review Board of the Cleveland Clinic approved this protocol.
Statistical analysis
We used ANOVA for multiple group comparisons unless there was less than 10 subjects in any particular group, then we used Kruskal-Wallis tests for multiple comparisons (P-values significant at 0.05) and Dunn tests for pair-wise comparisons (P-values significant at 0.0167 level with Bonferroni correction). The R-square value resulting from ANOVA tests were used to quantify the predictive ability of SF-36 summary scores and CLDQ overall scores. Chi-square tests were used in comparing the ratios for two groups when each combination had at least five subjects; otherwise, we calculated Fisher’s Exact tests. We used the Mantel-Haenszel procedure to compare ratios while controlling for confounding factors. We used Pearson moment-product correlations to establish the magnitude of the relationship between variables. All tests were two-sided with a significance level of 0.05 unless otherwise noted.
Results
Clinical and demographic data
One hundred thirty patients with chronic hepatitis C and 61 controls fulfilled the inclusion/exclusion criteria and agreed to participate in the study. All patients and controls completed HRQL questionnaires. Ninety-five HCV patients had additional testing for immunologic markers and completed the chronic fatigue screener. Table 1 summarizes the clinical characteristics of all HCV infected patients.
Mean SF-36 scores in patients with chronic hepatitis C are compared to those of general population and patients with type II diabetes. All scale scores and summary scores of HCV patients were significantly lower than general population (* p-value < 0.05). Patients with HCV had significantly lower scores in MH, VT, and SF scales (** p-value < 0.05). All other scores were not significantly different (p-value > 0.05). BP = Bodily Pain, GH = General Health, MH = Mental Health, PF = Physical Functioning, RE = Role Emotion, RP = Role Physical, SF = Social Functioning, VT = Vitality, MCS = Mental Component Summary Scores, PCS = Physical Component Summary.
Chronic fatigue scores
Chronic fatigue screener determined that 71% (67/95) of HCV infected patients suffered from chronic fatigue for at least 6 months as compared to only 25% (13/52) of controls (p-value 0.001). Only 9% of HCV patients compared to 6% of controls had lifelong fatigue (p-value 0.75). Symptoms suggestive of chronic fatigue syndrome were seen in 27% (26/95) of HCV patients and 11% (6/53) of controls (p-value 0.001).
Of the HCV patients who reported fatigue for 6 months, 57% (38/67) reported that it was relieved by rest, as did 54% (7/13) of controls (p-value 0.86). Similarly, 61% (40/66) of HCV patients reported loss of their usual activity (occupational, educational, social, and personal) related to fatigue as compared to 69% (9/13) of the controls (p-value = 0.76). Chronic fatigue markers were not associated with route of transmission (prior intravenous drug use), age (>44 to <44), gender, or presence of cirrhosis.
Health-related quality of life in HCV and other patient groups
Patients infected with HCV had significantly lower scores than general population and healthy controls in all aspect of their HRQL (Fig. 1). HRQL scores from patients with hepatitis C (measured by SF-36) were similar to diabetics, except for MH, VT, and SF being lower in HCV patients (Fig. 1).
Factors associated with HRQL
-
1.
Cirrhosis: Patients with HCV-related cirrhosis had lower scores in all the CLDQ domains than those without cirrhosis (overall CLDQ score: 3.94 ± 1.30 vs. 4.83 ± 1.27, p-value = 0.0004). Although SF-36 PCS, RP, BP, and GH scale scores were lower for patients with cirrhosis (PCS: 34.46 ± 12.07 vs. 46.30 ± 14.68, p-value = 0.0001), MH, RE, SF, VT, and MCS scores were either not different or higher in cirrhotics (MCS: 45.78 ± 6.87 vs. 42.09 ± 6.35, p-value = 0.006). As disease severity worsened by Child-Pugh classification, all the CLDQ scores and some of the SF-36 scores (mainly those dealing with physical heath) became lower (Figs. 2 and 3).
Fig. 2 Mean CLDQ scores in patients with chronic hepatitis C are compared according to disease severity (NC = Non Cirrhotics, A = Child’s A, B/C = Child’s B and C). All scale scores and summary scores fell with disease severity (* p-value < 0.05, ** p-value < 0.001). AB = Abdominal Symptoms, FA = Fatigue, SY = Systemic Symptoms, AC = Activity, EM = Emotional Function, WO = Worry, and overall CLDQ = overall CLDQ scores
Fig. 3 Mean SF-36 scores in patients with chronic hepatitis C are compared according to disease severity (NC=Non Cirrhotics, A=Child’s A, B/C=Child’s B and C). PF, RP GH, and PCs scores were significantly lower in Child’s B/C (p-value < 0.001). Additionally, BP and RE were also significantly lower in Child’s B/C (p-value < 0.05). All other scores were not significantly different (p-value>0.05). BP=Bodily Pain, GH=General Health, MH=Mental Health, PF=Physical Functioning, RE=Role Emotion, RP=Role Physical, SF=Social Functioning, VT=Vitality, MCS=Mental Component Summary Scores, PCS=Physical Component Summary
-
2.
Gender: Both fatigue domain and overall CLDQ scores were lower in female patients (Fig. 4). Although SF-36 RP, PF and PCS scores were also lower for female patients, other scores were not different (Figs. 4 and 5).
Fig. 4 Fig. 5 Fig. 6 Mean CLDQ scores in patients with hepatitis C and controls divided by fatigue categories. Scores for nonfatigued controls (Control/No Fatigue) were compared to nonfatigued HCV (HCV/No Fatigue). HCV/No Fatigue had lower scores for WO and CLDQ (p-value<0.001). Additionally, scores of HCV patients with chronic fatigue (HCV/CF) were lower than those of controls with chronic fatigue (Control/CF) only in WO (p-value = 0.02)
Fig. 7 Mean SF-36 scores in patients with hepatitis C and controls divided by fatigue categories. Scores for nonfatigued controls (Control/No Fatigue) were compared to nonfatigued HCV (HCV/No Fatigue). HCV/No Fatigue had lower scores for GH (p-value = 0.04), MH (p-value = 0.007), SF (p-value = 0.05), and MCS (p-value = 0.002)
-
3.
Chronic Fatigue: HCV patients and controls were divided into those with chronic fatigue alone [CF (+)], chronic fatigue syndrome [CFS (+)], and no fatigue. HRQL scores (both measured by SF-36 and CLDQ) were lowest for individuals designated as chronic fatigue syndrome [CFS (+)]. There was no difference between HCV patients with CFS and controls with CFS.
The next lowest HRQL scores were in individuals with chronic fatigue only [CF (+)]. HCV patients and the controls who suffered from chronic fatigue had similar impairment of HRQL, except for Worry domain of CLDQ which was lower in HCV infected patients.
However, the nonfatigued HCV patients’ CLDQ scores remained lower than the nonfatigued controls [Worry (WO) and Overall CLDQ scores and General Health (GH), Mental Health (MH), Social Functioning (SF), and Mental Component summary scale (MCS) scores of SF-36].
With respect to comparison between the CF (+), CFS (+), and no fatigue groups, R-square values were computed for the SF-36 MCS and PCS scores as well as for the overall CLDQ scores (for both HCV and control individuals). The R-square for MCS was 0.25, for the PCS it was 0.41, and for the overall CLDQ score it was 0.51.
-
4.
Other Factors: Factors not associated with HRQL (both by CLDQ and SF-36) included route of transmission, age, race, and total bilirubin. Additionally, SIL-2R, levels of cryoglobulins, rheumatoid factor, and viremia did not significantly correlate with HRQL.
Discussion
Following the discovery of Hepatitis C virus, investigators have advanced our understanding of the molecular biology of the virus, the mechanism of chronicity, the epidemiology of chronic HCV, and the developed increasingly effective therapy. In addition, there is a growing appreciation of HCV-related impairment of patients’ well-being. Our data suggest that chronic HCV infection has a profound deleterious effect on HRQL. Patients with HCV infection have similar HRQL scores to patients with diabetes. Chronic fatigue, which is commonly seen in patients infected with HCV, can explain some of this loss in well-being. HCV-related fatigue is not associated with viral markers, patient demographics (gender, age, etc.), severity of liver disease (cirrhosis), or the route of transmission of HCV. Although chronic fatigue was more common in HCV infected individuals, measures of severity of fatigue (fatigue relieved by rest and loss of activity due to fatigue) were similar to non-HCV fatigue. Some aspects of HRQL in nonfatigued HCV patients’ were significantly impaired than nonfatigued controls (worry, overall CLDQ, general health, mental health, and social functioning). This finding suggests that the emotional impact of carrying this infection to the patients’ overall health may be substantial.
The relationship between SF-36, CLDQ, and fatigue was measured by R-square (coefficient of determination) statistics, which quantifies the proportion of total variance associated with the use of an independent variable (CLDQ score, PCS, or MCS). We found that the CLDQ captures more variance than both summary scores of SF-36.
In addition to fatigue, other physical factors also play a role in reducing well-being. Even though cirrhosis did not affect the fatigue component (as measured by chronic fatigue screener), it affects the overall HRQL (measured by CLDQ) of patients infected with HCV. Patients with cirrhosis had lower CLDQ scores than those without cirrhosis. Disease-specific HRQL scores measured by the CLDQ fell progressively with disease severity. Although some of the physical aspects of SF-36 also fell, mental aspects of SF-36 did not change or improved as the disease severity worsens. Few other disease features are associated with the magnitude of HRQL impairment. Specifically, we found no relation with mode of transmission, and only very weak associations with immunologic markers.
Our study had some limitations. First, all subjects in our study were recruited from one tertiary health care center, potentially introducing problems with external validity of our results to patients seen in the primary care centers. Second, the cross-sectional design of our study does not allow us to describe how progression of hepatitis C as it relates to changes in HRQL. However, the fact that substantial impairment of HRQL was found in those with early liver disease suggests that a recruitment bias does not account for these findings. Additionally, progressive worsening of CLDQ scores with disease severity suggests that the relationship between disease severity and HRQL will remain the same in the future prospective studies of hepatitis C.
Our study suggests that although investigators strive to develop new targeted antiviral agents to treat chronic hepatitis C, we must consider the major impact of HCV on patients’ HRQL. HRQL impairment is important for its own sake but also because of its association with lower productivity and increased disability claims. The success of future therapy for HCV rests not only in developing strategies to change the natural history of this disease but also in improving patient’s HRQL.
References
World Health Organization (2000) Hepatitis C – Factsheet No 164. Revised October 2000. WHO, Geneva
Davis G (guest ed) (1997) Hepatitis C. Clin Liver Dis 1(3):494–704
Alter M (1997) Epidemiology of hepatitis C. Hepatology 26(3, Suppl. 1):62S–65S
Hoofnagle J, Tralka TS (1997) The national institute of health consensus development conference: management of hepatitis C. Hepatology 26(3, Suppl. 1):1S–10S
McMurray RW, Elbourne K (1997) Hepatitis C virus infection and autoimmunity. Semin Arthritis Rheum 26(4): 689–701
Pawlotsky JM, Yahia MB, Andre C et al (1994) Immunological disorders in C virus chronic active hepatitis: a prospective case-control study. Hepatology 19(4):841–848
Sawada T, Hirohata S, Inoue T, Ito K (1991) Development of rheumatoid arthritis after hepatitis C virus infection. Arthritis Rheum 34:1620–1621
Siegel L, Cohn L, Nashel D (1993) Rheumatic manifestations of hepatitis C infection. Semin Arthritis Rheum 23(3):149–154
Adinolfi LE, Utili R, Attanasio V, Zampino R, Ragone E, Tripodi M-F, Ruggiero G (1996) Epidemiology, clinical spectrum and prognostic value of mixed cyroglobulinemia in hepatitis C virus patients: a prospective study. Ital J Gastroenterol 28:1–9
Rubin LA, Nelson DL (1990) The soluble interleukin—2 receptor: biology, function, and clinical application. Ann Int Med 113:619–627
Davis G, Balart L, Schiff E, Lindsay K, Bodenheimer, Perrillo R, Carey W, Jacobson, Payne J, Dienstag J, van Thiel D, Tamburro C, Martino F, Sangvhi B, Albrecht J (1994) Assessing health-related quality of life in chronic hepatitis C using the sickness impact profile. Clin Ther 16:334–345
Carithers R, Sugano D, Bayliss M (1996) Health assessment for chronic HCV: results of quality of life. Dig Dis Sci 41(12, Suppl.):75S
Foster G, Goldin R, Thomas H (1998) Chronic hepatitis C infection causes a significant reduction in quality of life in the absence of cirrhosis. Hepatology 27(1):209–212
Bonkovsky H, Woolley J, Consensus Interferon Study Group (1999) Reduction of health-related quality of life in chronic hepatitis C and improvement with interferon therapy. Hepatology 29(1):264–269
Bombardier CH, Buchwald D (1995) Outcome and prognosis of patients with chronic fatigue vs. chronic fatigue syndrome. Arch Intern Med 155(19):2105–2110
Fukuda K, Straus S, Hickie I, Sharpe M, Dobbins J, Komaroff A, International Chronic Fatigue Syndrome Study Group (1994) The chronic fatigue syndrome: a comprehensive approach to its definition and study. Ann Intern Med 121(12):953–959
Ware J, Sherbourne C (1992) The MOS 36-item short form health survey (SF-36). I. Conceptual framework and item selection. Med Care 30:473–482
Ware J, Kosinski M, Keller S (1994) SF-36 physical and mental health summary scales: a user’s manual. The Health Institute, Boston, MA
Younossi Z, Guyatt, G, Kiwi M, King D, Bohprai N (1999) Development of a disease-specific health-related quality of life index for chronic liver disease. GUT 44:1–6
Guyatt G, Walter S, Norman G (1987) Measuring change over time: assessing the usefulness of evaluative instrument. J Chron Dis 40:171–178
Jaeschke R, Guyatt G, Keller J, Singer J (1989) Measurement of health status: ascertaining the meaning of a change in quality-of-life questionnaire score. Controlled Clin Trials 10:407–415
Henry JB (1979) Clinical diagnosis and management by laboratory methods (16th ed.). Philadelphia, PA: WB Saunders Company
Norbert W, Tietz M (1978) Fundamentals of clinical chemistry (3rd ed.). Philadelphia, PA: WB Saunders Company
Rubin L, Nelson D (1990) The soluble interleukin-2 receptor: biology, function, and clinical application. Ann Intern Med 113:619–627
Turcotte JG, Lambert MJ III (1973) Variceal hemorrhage, hepatic cirrhosis, and portacaval shunts. Surgery 73:810–817
Pugh RN, Murray-Lyon IM, Dawson JL, Pietroni MC, Williams R (1973) Transection of the oesophagus varices. Br J Surg 60:646–649
Acknowledgment
This research was supported in part by grants from the I.H. Page Centre for Clinical Effectiveness of the Cleveland Clinic Foundation and an unrestricted educational grant from WT Grant to LC.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kallman, J., O’Neil, M.M., Larive, B. et al. Fatigue and Health-Related Quality of Life (HRQL) in Chronic Hepatitis C Virus Infection. Dig Dis Sci 52, 2531–2539 (2007). https://doi.org/10.1007/s10620-006-9708-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-006-9708-x