Abstract
The Puerto Rican crested toad (Peltophryne lemur) is currently composed of a single wild population on the south coast of Puerto Rico and two captive populations founded by animals from the northern and southern coasts. The main factors contributing to its decline are habitat loss, inundation of breeding ponds during storms, and impacts of invasive species. Recovery efforts have been extensive, involving captive breeding and reintroductions, habitat restoration, construction of breeding ponds, and public education. To guide future conservation efforts, genetic variation and differentiation were assessed for the two captive colonies and the remaining wild population using the mitochondrial control region and six novel microsatellite loci. Only two moderately divergent mitochondrial haplotypes were found, with one fixed in each of the southern and northern lineages. Moderate genetic variation exists for microsatellite loci in all three groups. The captive southern population has not diverged substantially from the wild population at microsatellite loci (F ST = 0.03), whereas there is little allelic overlap between the northern and southern lineages at five of six loci (F ST > 0.3). Despite this differentiation, they are no more divergent than many populations of other amphibian species. As the northern breeding colony may not remain viable due to its small size and inbred nature, it is recommended that a third breeding colony be established in which northern and southern individuals are combined. This will preserve any northern adaptive traits that may exist, and provide animals for release in the event that the pure northern lineage becomes extirpated.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The evaluation of current taxonomy and identification of genetic lineages within threatened species are vital to developing efficient and optimal recovery programs. For example, the use of morphological traits to designate taxonomic entities often does not reflect patterns of genetic variation (Burbrink et al. 2000; Culver et al. 2000; Nittinger et al. 2007), which can waste limited resources on groups that are not distinct and may have little value to the conservation of the species as a whole (Avise and Nelson 1989; Zink et al. 2000). A lack of distinct lineages can also facilitate recovery, as it may increase the number of source populations for efforts such as captive breeding or translocation between isolated populations (Eizirik et al. 2001; Godoy et al. 2004). In contrast, early detection of multiple lineages may enable intervention before an evolutionarily significant lineage becomes irreversibly imperiled (Nielson et al. 2001; Shaffer et al. 2004a; Vredenburg et al. 2007), as occurred for the tuatara (Daugherty et al. 1990). Knowledge of the genetic structure of a species can identify the most closely related population or subspecies for use in reintroductions following extirpation (Drew et al. 2003; Kraaijeveld-Smit et al. 2005), while individuals from divergent populations may be incorporated to maximise genetic variation (Madsen et al. 1999; Land et al. 2004). Finally, assessment of the genetic composition of existing captive programs can determine whether animals are suitable for further breeding (Brock and White 1992; Ruokonen et al. 2000) or reintroduction (Garcia-Moreno et al. 1996; Wyner et al. 1999).
The Puerto Rican crested toad (Peltophryne lemur) is a highly endangered bufonid for which two separate lineages are currently maintained in captivity. Captive breeding was initiated prior to the frequent use of molecular markers to identify genetic structure within species. In this study, mitochondrial DNA sequences and six novel microsatellite loci are used to investigate the genetic variation and differentiation of the two P. lemur captive breeding colonies, as well as the sole remaining wild population. This information will be used to determine the optimal future management actions to ensure the persistence of P. lemur in the wild.
Biology and conservation history of Peltophryne lemur
The only toad endemic to Puerto Rico, P. lemur has been collected from at least eight sites across Puerto Rico and one on Virgin Gorda of the British Virgin Islands (Fig. 1). It generally occupies low-elevation (<200 m), arid or semi-arid coastal regions and is often associated with limestone karst formations that provide fissures into which it retreats during the day (Rivero et al. 1980; Miller 1985). Breeding is sporadic, and occurs in temporary pools formed by heavy rains (USFWS 1992). Peltophryne lemur has not been seen on Virgin Gorda for over 40 years, and was thought to be extinct on Puerto Rico until 1965 (USFWS 1992). Populations have since been found on both the northern (Quebradillas and Isabela) and southern (Guánica State Forest Reserve) coasts of Puerto Rico (Rivero et al. 1980; USFWS 1992), although no toads have been seen in the north since 1988 (Barber 2007). It is therefore probable that Guánica, which may have numbered as few as 80 individuals in 2003 (Hedges et al. 2004), is the only remaining wild population. P. lemur is listed as critically endangered by the International Union for Conservation of Nature. The primary cause of decline is habitat loss and alteration, including drainage of breeding sites and spraying of surrounding areas with pesticides (Johnson 1990; USFWS 1992). Predation and/or competition by introduced species (e.g., cane toad, Bufo marinus; mongoose, Herpestes javanicus; feral dogs and cats) may have contributed to declines (Rivero et al. 1980; USFWS 1992). The small size and isolation of the remaining population in Guánica indicates that it is also vulnerable to stochastic environmental (e.g., hurricanes Johnson 1990) and demographic events.
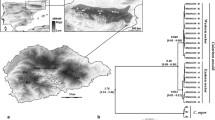
Map of Puerto Rico and the British Virgin Islands showing historical locations of Peltophryne lemur (●) and sites represented by captive zoo programs (*). San Juan (⊙) is the capital of Puerto Rico and is shown for reference only. The three main physiographic regions of Puerto Rico are indicated: the karst belt in the northeast, the mountainous interior, and the discontinuous coastal plain (after Monroe 1976)
Recovery efforts for P. lemur have been extensive. The Guánica breeding pond is closed for three weeks during the breeding season and is no longer drained for parking (Johnson 1990). Toads have been radiotracked to monitor movements, and habitat characteristics have been evaluated (Johnson 1990; Matos-Torres 2006). Wild individuals were collected during the 1980s to establish two breeding colonies representing the northern (four founders) and southern (32 founders) populations. To date, more than 110,000 southern tadpoles and toads have been released into constructed ponds in Guánica Forest, and releases of northern animals began at Arecibo in 2006 (Barber 2007). In 2003 and 2005, captive-born adult toads returned to the southern release site to breed (PRCT SSP 2006). Additional property is being secured and several ponds constructed in both regions, while public outreach and education are a continuing component of the recovery program (Barber 2007).
In order to maintain the optimal recovery strategy for P. lemur, several important issues must be resolved using genetic data. On average, a founding population of 20 individuals will capture >95% of heterozygosity and ~87% of allelic diversity (Frankham et al. 2002). While the southern captive colony may have sampled sufficient genetic variation from the wild to maintain a healthy population for many generations, the northern colony likely had low variation from its initiation. The potential for increased homozygosity due to inbreeding and loss of genetic variation through drift are factors that must also be considered in the breeding populations. This is especially applicable to the northern colony, as the 123 individuals currently in captivity and used for breeding are the product of four siblings that themselves are descended from two first cousins. In addition, the isolation of the captive colonies from each other and the wild population may have led to genetic or morphological divergence between them (Rowe et al. 1998; Shuster et al. 2005; Hakansson et al. 2007). The degree of genetic differentiation between the northern and southern populations also requires assessment. Fossil evidence suggests that P. lemur was widespread throughout Puerto Rico during its history (Pregill 1981); however, it is currently restricted to low-elevation areas on opposite coasts, separated by the Cordillera Central mountain range. Previous allozyme assays suggested that the two populations may be divergent (Lacy and Foster 1987). It thus remains unclear whether the northern and southern populations have been separated throughout their history, or if population declines and habitat fragmentation have isolated them more recently.
The genetic data obtained here will be used to address three main questions: (1) do the captive populations suffer reduced genetic diversity relative to the wild population; (2) has the captive southern population diverged genetically from the wild southern population; and (3) what is the level of differentiation between the populations from the northern and southern coasts? Given the precarious state of the northern breeding colony and its probable extirpation in the near future, the long-term reintroduction of purely northern individuals is not realistic. The information from this study will thus be used to discriminate between the few remaining options for this lineage: (1) continue breeding the northern and southern populations separately. If the northern colony becomes extirpated, terminate reintroductions in this region; (2) continue breeding the northern and southern populations separately. If the northern colony becomes extirpated, reintroduce individuals from the southern lineage to the northern region; or (3) create an additional breeding colony in which northern and southern individuals are mixed. If adaptive differences exist between the northern and southern populations, this approach will facilitate the preservation of these traits. In the event that the pure northern colony becomes extirpated, these mixed individuals can be reintroduced to the north and may experience greater survival over purely southern animals due to the presence of some northern adaptations.
Methods
Sample collection and DNA extraction
Muscle or organs (heart, kidney, liver) were collected post-mortem from individuals of the northern and southern breeding colonies at the Toronto Zoo and Audubon Nature Institute in New Orleans. In total, four individuals from the northern colony and 58 from the southern colony were sampled. During a breeding of wild toads in 2003 at Tamarindo Pond in Guánica State Forest, Puerto Rico, an additional 43 tadpoles that had died were collected opportunistically. The southern breeding colony was founded by individuals collected from several breeding ponds, including Tamarindo, during the 1980s. Differential mortality of tadpoles may reflect the genotype at the major histocompatibility complex (Barribeau et al. 2008), suggesting that the targeted collection of dead individuals may not constitute a random genetic sample. However, the perilous state of P. lemur in the wild mandates that sampling impacts be minimised. Furthermore, the high mortality rate of amphibian larvae, particularly in temporary ponds such as those used by P. lemur (Duellman and Trueb 1986), indicates that assumptions of genotype interactions may be premature without supporting information. Hereafter, the southern breeding colony will be called Guánica, the northern breeding colony will be called Quebradillas, and the wild southern population will be called Tamarindo.
All 105 samples were genotyped at the six microsatellite loci to ensure that no rare alleles were missed. However, maternal inheritance of the mitochondrial genome means that all offspring of a female possess the same haplotype; thus, mitochondrial DNA was sequenced for only a subset of samples from the breeding colonies. According to the P. lemur studbook maintained by the Toronto Zoo, the 58 Guánica samples employed in this study trace their lineage to six wild females and 14 wild males. Thirty captive-bred individuals representing these six matrilineal lineages were sequenced, as were five male founders that could potentially harbour different mitochondrial lineages. Due to the small sample size for Quebradillas, all samples were sequenced. Because the genealogy of the Tamarindo individuals is unknown, all 44 samples were sequenced.
Although the number of samples used for the northern colony is small, it must be emphasised that a very limited gene pool is available for profiling. Only four wild individuals (two male and two female) were used to establish the colony, and all current breeding individuals are descendants of two cousins. Two of the samples profiled here are direct offspring of these cousins, and the other two are the product of a mating between two of these offspring (i.e. between full siblings). Tissue samples of earlier generations are not available. Thus, a maximum of only one mitochondrial lineage and four alleles for each microsatellite locus are possible in any combination of samples from this population. Additional sampling would therefore not be informative, and the four samples analysed here are representative of the variation present in the northern colony.
Samples were prepared for extraction by dissolving 10 mg of tissue in 500 μl of 1× lysis buffer (2 M urea, 0.1 M NaCl, 0.25% n-lauroyl sarcosine, 5 mM CDTA, 0.05 M Tris–HCl pH 8), and digesting with 50 μl of proteinase K. Total genomic DNA was extracted with the DNeasy Tissue Kit (Qiagen Inc.) and quantified with the PicoGreen Quantitation Kit (Molecular Probes) on a FLUOStar Galaxy (BMG Labtech).
Mitochondrial control region sequencing
A fragment of the control region was amplified with the primers CytbA-L and ControlP-H (Goebel et al. 1999) in a 25 μl reaction containing 1–5 ng DNA, 1× PCR buffer, 2 mM MgCl2, 0.2 mM dNTPs, 0.1 mg/ml BSA, 0.2 μM each primer, and 2 U Taq polymerase (Invitrogen). Thermal cycling used an MJ Research PTC-225 thermal cycler with the following conditions: denaturation at 94°C for 5 min, 30 cycles of denaturation at 94°C for 30 s, annealing at 55°C for 30 s, and extension at 72°C for 45 s, with a final extension of 72°C for 10 min. PCR product was visualised with ethidium bromide on 1% agarose gels, and 5 μl was purified with ExoSAP-IT (USB). Both strands were sequenced using the PCR primers with the DYEnamic ET-Terminator cycle sequencing kit on a MegaBACE 1000 DNA Analysis System (GE Healthcare).
Microsatellite development and profiling
Tetranucleotide microsatellites were isolated according to the protocol of Beauclerc et al. (2007). In total, 115 clones were sequenced, of which 83 were positive for a tetranucleotide repeat, with five duplicate clones found. Primer pairs were designed manually for 20 positive clones that possessed sufficient flanking sequence. Individual loci were amplified in 10 μl volumes containing 5 ng genomic DNA, 1× PCR buffer, 1.5 mM MgCl2, 0.2 mM each dNTP, 0.3 μM each primer, and 0.5 U Taq polymerase using the following thermal cycling conditions: denaturation at 94°C for 5 min, 30 cycles of denaturation at 94°C for 30 s, annealing at the optimal temperature (see Table 1) for 1 min, and extension at 72°C for 1 min, with a final extension of 60°C for 45 min. Nine and three individuals from Guánica and Quebradillas, respectively, were screened for polymorphism by visualising PCR products on 4% agarose gels stained with SYBR Green I (Molecular Probes). Six loci amplified reliably and were polymorphic. To profile all samples, each locus was amplified individually and pooled into two multiplex reactions prior to desalting (Table 1). All PCR product was desalted using Sephadex G50, profiled on a MegaBACE 1000 DNA Analysis System, and scored using genetic profiler v.2.0 (GE Healthcare).
Data analysis
Sequences were aligned in clustalx v.1.81 (Thompson et al. 1997) and edited by eye using bioedit v.7.0.533 (Hall 1999). Pairwise sequence divergence was calculated using mega v.3.1 (Kumar et al. 2004). Microsatellite diversity indices (number and frequencies of alleles, observed and expected heterozygosity) were calculated using genepop v.3.4 (Raymond and Rousset 1995), and conformation to Hardy–Weinberg equilibrium was tested by estimation of exact P values with a Markov chain method (Guo and Thompson 1992). Allelic richness (number of alleles per population) and private allelic richness were calculated by standardisation to the smallest sample size (N = 4) using rarefaction in the program hp-rare v.1.0 (Kalinowski 2005).
Population differentiation was estimated between all pairs of populations for all loci with Nei’s genetic distance D S (1972) and the genotype likelihood ratio distance D LR (Paetkau et al. 1997) using software from http://www2.biology.ualberta.ca/jbrzusto/ (J. Brzustowski, unpublished). These distance measures are calculated in very different manners, and Paetkau et al. (1997) recommended their use to provide independent estimates of genetic distance between populations. Population structure was evaluated with θ W (Weir and Cockerham 1984), the unbiased estimator of Wright’s (1951) F ST, using arlequin v.3.1 (Excoffier et al. 2005). Significance was determined by a permutation test of 1,000 iterations. Genetic differentiation was further evaluated using the clustering program structure v.2.0 (Pritchard et al. 2000). The likelihood that the samples represented between one and five genetic clusters (K = 1–5) was determined, using the following model: admixture, correlated allele frequencies, burn-in of 500,000 steps, and 1,000,000 iterations of the Markov Chain Monte Carlo algorithm. The model was run four times for each value of K to ensure consistency between runs. The largest value of ∆K (Evanno et al. 2005) was used to determine the most likely number of populations.
Results
Mitochondrial sequencing
Quality sequence was obtained for 963 bp of the mitochondrial control region, yielding two haplotypes (GenBank accession numbers EU149946 and EU149947): PleCR-1 occurred only in Guánica and Tamarindo, and PleCR-2 was exclusive to Quebradillas. Sixteen variable sites were found: 13 transitions, one transversion, and two indels of one base pair. Pairwise sequence divergence between the haplotypes was 0.015.
Microsatellite profiling
The total number of alleles for the six microsatellite loci was 46, with a range from 4 to 10 and an average of 7.7 alleles per locus (Table 2). Guánica possessed the largest number of alleles; however, the disparate sample size between groups clearly contributed to this outcome. When standardised by rarefaction, allelic richness was nearly equal for all three populations. Each population also had several private alleles (Fig. 2; Table 2): Guánica possessed six, Tamarindo three, and Quebradillas twelve. The sampling of amphibian tadpoles from a single location is typically discouraged as it may consist of a group of siblings and yield biased data. However, the observation of seven alleles at locus PleMs-3 in the Tamarindo samples indicates that this group is descended from at least four parents, and individuals are therefore not all full siblings.
Observed and expected heterozygosities ranged from 0.41 to 1.00 and 0.43 to 0.84, respectively (Table 3). Following sequential Bonferroni correction, all loci were in Hardy–Weinberg equilibrium and linkage equilibrium in Quebradillas and Tamarindo. In Guánica, however, loci PleMs-8 and PleMs-14 continued to deviate significantly from Hardy–Weinberg equilibrium due to heterozygote excess, and significant linkage disequilibrium remained between loci PleMs-14/PleMs-10, PleMs-14/PleMs-3, PleMs-10/PleMs-11 and PleMs-10/PleMs-3. It is probable that the disequilibrium in Guánica is due to the managed breeding of this closed population. The fact that all loci are in equilibrium in the wild population, which presumably experiences random mating, suggests that the loci are not linked or under selection and are therefore suitable for use in further analyses.
Guánica and Tamarindo shared the greatest number of microsatellite alleles, 25 out of 46 total alleles (54%; Fig. 2). When excluding alleles found only in Quebradillas, Guánica and Tamarindo shared 74% of their alleles (25 out of 34). Very few alleles were common between Quebradillas and the southern populations: none were shared for loci PleMs-4 and PleMs-8, and only one was shared at loci PleMs-10, PleMs-11, and PleMs-14. In total, Quebradillas shared only five alleles (11%) with both Guánica and Tamarindo; its remaining 12 alleles were unique. Both Guánica and Tamarindo are much more distant from Quebradillas than they are from each other based on the pairwise genetic distance measures D S and D LR (Table 4). F ST values were significant at P = 0.01 between all pairs of populations, but values were much larger between either of the southern populations and Quebradillas (Table 4).
The average log probability of K [ln Pr(X|K)] increased with the number of clusters (data not shown). However, the rate of change of the log probability of K (∆K) was largest for K = 3 (∆K 2 = 2.6; ∆K 3 = 551.5; ∆K 4 = 117.1). This was therefore chosen as the number of groups most likely represented by the microsatellite data. Guánica and Tamarindo individuals were both assigned in similar proportions and nearly equally to clusters 1 and 2, with no individuals assigned to cluster 3 at >2% of their genome (data not shown). All Quebradillas individuals were strongly assigned to cluster 3 (>98% of each genome).
Discussion
Genetic diversity within populations
The control region is the most variable section of the mitochondrial genome (Moritz et al. 1987), making the very low haplotypic diversity of P. lemur somewhat surprising. This bufonid has one of the lowest mitochondrial diversities among a number of amphibian species: multiple haplotypes typically exist for this and less variable regions (Bos and Sites 2001; Austin et al. 2002; Smith and Green 2004), and often over relatively small scales (Shaffer et al. 2004b; Dever 2007). However, Rowe et al. (2006) found that all British and Irish individuals of Bufo calamita possessed a single control region haplotype.
Peltophryne lemur is moderately variable at the six microsatellite loci. Within-population genetic diversity (HE) is similar to or greater than that found for microsatellite loci in several other amphibians (Rowe et al. 1998; Palo et al. 2003; Brede and Beebee 2004; Martinez-Solano et al. 2005; Arens et al. 2006), while only a few have been found to have substantially greater HE (Austin et al. 2003; Burns et al. 2004; Hoffman et al. 2004). Allelic diversity for P. lemur is comparable to several species examined over similar and larger geographical scales, despite much larger sample sizes for some (Rowe et al. 1998; Palo et al. 2003; Brede and Beebee 2004; Kraaijeveld-Smit et al. 2005; Arens et al. 2006). This diversity is surprising, considering the small sample sizes, relatedness of individuals, and limited variation in mtDNA. It thus appears that neither the captive nor wild populations of P. lemur have suffered a severe reduction in heterozygosity due to small population sizes and prolonged isolation. In fact, the Guánica captive colony had greater heterozygosity than expected at two loci. This likely reflects the minimisation of kinship management system used to designate pairs for breeding (Johnson 1990; Ballou and Lacy 1995), which reduces the tendency to homozygosity via inbreeding and the loss of alleles through genetic drift (Montgomery et al. 1997). By comparison, random mating in the wild Tamarindo population, combined with a possible bottleneck of 80 individuals (Hedges et al. 2004), has allowed these processes to reduce heterozygosity below that of Guánica for several loci.
Comparison of populations
There appears to be very little difference in the amount of genetic variation found in Guánica and Tamarindo. Both populations possess similar numbers of alleles and H E estimates for most loci. There is also little divergence between them: a single mitochondrial haplotype exists in all individuals, there are very few unshared alleles, and the measures of genetic distance and differentiation are very low. Comparable estimates of these measures have been found over similar distances in several other amphibian species (Newman and Squire 2001; Brede and Beebee 2004; Burns et al. 2004; Funk et al. 2005; Arens et al. 2006). In addition, the structure algorithm could not distinguish individuals from these two groups. The genetic similarity of these populations is not surprising, as the Guánica captive colony is recently descended from the wild Tamarindo population. Although gene flow between captive-bred individuals released into Guánica State Forest and wild individuals located at Tamarindo pond cannot be completely excluded, it is highly unlikely as P. lemur has been shown to move only up to 2 km (Johnson 1999) and these sites are separated by 20 km of migratory distance.
Despite the fact that only a small number of related, inbred individuals were sampled, moderate genetic variability exists at nuclear loci in Quebradillas. It is probable that most alleles present in the northern population were detected during this analysis, as 50–100% of the maximum number of possible alleles (four) was observed for all loci. Thus, more extensive sampling would likely have revealed little additional variation. Although it possesses fewer alleles than Guánica or Tamarindo based on direct counts, it is nearly equal when all groups are standardised to N = 4. As well, H E is very similar to both southern populations. Quebradillas is moderately divergent from the southern populations at mtDNA, with 1.5% sequence divergence between the two haplotypes. A comparison with other amphibian mitochondrial studies shows that many species possess deeper divergences in the control region (Shaffer et al. 2004b) and more conserved regions (Lougheed et al. 1999; Austin et al. 2002; Vredenburg et al. 2007). In contrast to the mtDNA data, Quebradillas is quite differentiated from the southern populations at nuclear microsatellite loci. It possesses far more private alleles than either Guánica or Tamarindo, and shares more than a single allele with them at only one of the six loci. This abundance of private alleles in Quebradillas is likely not an artifact of sample size, as one would expect the populations with larger sample sizes (i.e. Guánica or Tamarindo) to detect a greater number of rare or unique alleles. Furthermore, structure clearly distinguished the northern individuals, and the measures of genetic distance and differentiation are much greater between Quebradillas and both southern populations than between the two southern populations. These F ST values of >0.3 are quite large compared to some other amphibian microsatellite studies (Newman and Squire 2001; Brede and Beebee 2004; Johansson et al. 2005), particularly given the small distance separating the two populations (~55 km). However, some studies have found similar or greater values over very short geographic distances (Kraaijeveld-Smit et al. 2005; Arens et al. 2006), and several report values >0.4 across the distribution of a single species (Rowe et al. 1998; Burns et al. 2004; Funk et al. 2005).
For small animals with low vagility such as amphibians, it is often not the geographic distance that determines levels of gene flow between populations, but rather the ecological and/or geographical barriers that separate them such as mountain ridges (Funk et al. 2005; Kraaijeveld-Smit et al. 2005; Martinez-Solano et al. 2005), unsuitable habitat (Johansson et al. 2005), or even ancient geological features that are no longer apparent (Lougheed et al. 1999). The Cordillera Central mountain range, with a maximum elevation >1,300 m (Pico 1974), extends across the interior of Puerto Rico in an east-west direction (Fig. 1). The strong differentiation between northern and southern populations of P. lemur is thus not unexpected, as this species is restricted to low-elevation coastal areas (Rivero et al. 1980). The genetic data, combined with P. lemur’s historical distribution and probable inability to traverse the mountainous interior, suggests that northern and southern populations have likely not experienced gene flow for a substantial time.
Conservation and management recommendations
An important finding of this study is that the breeding of P. lemur in AZA (Association of Zoos and Aquariums) institutions has been very successful in terms of genetic management: the captive Guánica population has not suffered reduced genetic diversity relative to the wild Tamarindo population, nor has it diverged substantially during its isolation. Although only neutral molecular markers were evaluated, this suggests that adaptation to captivity has been minimal (e.g., Ford 2002) and that the captive southern population is suitable for continued reintroduction to the south coast of Puerto Rico. Additional supplementation of the captive colony by wild individuals does not appear to be immediately necessary for genetic purposes.
The future of P. lemur in northern Puerto Rico, however, is in jeopardy. Although the Quebradillas breeding colony possesses adequate variation at this time, it likely cannot be sustained much longer as the 123 individuals currently in captivity are descended from four inbred siblings. It was previously believed that the colony had reached reproductive senility as no clutches had been obtained for some time (Bloxam and Tonge 1995). While this is likely in part due to the age of the individuals, it is also possible that inbreeding avoidance (Waldman et al. 1992) and/or depression have contributed to lower fitness. Successful breeding of northern animals has occurred recently, with releases of tadpoles on the north coast of Puerto Rico. However, it is unlikely that individuals have become established since it required nearly 20 years and over 100,000 released individuals before breeding animals were observed in the south. This indicates that there will be limited opportunities to restock the captive northern colony should it become depleted. The prospects for the northern lineage of P. lemur are therefore dire and it is no longer sufficient to maintain separate populations in captivity; in order to ensure the persistence of P. lemur in the north, less conventional management options must be explored.
The presence of an exclusive mitochondrial haplotype and many unique microsatellite alleles in Quebradillas suggests that the northern and southern populations of P. lemur are quite distinct and have not experienced gene flow for some time. This isolation may have enabled unique, adaptive characteristics to evolve in the north, facilitated by differences in habitat and climatic conditions: Guánica is located in an arid coastal plain, receiving ~30–35 inches of rainfall annually and possessing dry limestone vegetation, while Quebradillas receives 40–60 inches of annual rainfall and consists of limestone hills with humid limestone vegetation (Pico 1974). Any unique adaptations that northern individuals may possess have the potential to greatly assist with the establishment of reintroduced individuals to this region and should be preserved. It is therefore recommended that whilst the Quebradillas breeding colony is successful in producing offspring for release, the Guánica and Quebradillas colonies should continue to be maintained separately and animals released into their respective regions. In order to ensure the continuation of the northern lineage, however, a third breeding colony consisting of both northern and southern individuals should be created. This will increase the genetic diversity and number of breeding individuals for the Quebradillas colony, thereby increasing its health and longevity. Initially, this would serve as an experimental colony to determine whether mixed individuals possess greater or diminished fitness prior to their release into the wild. In the event that the pure Quebradillas lineage becomes extirpated, these animals could then be used for release to the northern coast of Puerto Rico. This strategy would preserve the most overall genetic diversity, including any unique genetic adaptations that may be present in the northern lineage. The alternative option to release pure Guánica individuals into the north should the Quebradillas colony become extirpated is less desirable, as they would lack any potentially adaptive traits that may increase their survival in the conditions found in northern Puerto Rico.
The mixing of distinct lineages is generally avoided because of the possibility of reduced fitness due to outbreeding depression. However, it “should not be dismissed lightly as a management option, especially where there is evidence that remnant populations have reduced viability because of inbreeding depression” (Moritz 1999). Inbreeding depression is generally considered a more serious problem than outbreeding depression, and the negative impacts of outbreeding depression may be overemphasised while the benefits of mixing lineages are overlooked (Frankham et al. 2002). “Genetic rescue” (e.g., Tallmon et al. 2004) by mixing distinct lineages has successfully reduced inbreeding depression and increased fitness for several threatened species (Westemeier et al. 1998; Madsen et al. 1999; Land et al. 2004) and continues to be recommended (Soltis and Gitzendanner 1999; Wyner et al. 1999; Godoy et al. 2004). Experimentally inbred populations of plants (Richards 2000; Newman and Tallmon 2001) and animals (Bryant et al. 1999; Ball et al. 2000) also experienced increased fitness when immigrants were introduced. Very low levels of migration (e.g., one individual per generation) may be sufficient for successful genetic rescue, which minimises genetic swamping and allows local adaptation to proceed (Hedrick 1995; Newman and Tallmon 2001; Palo et al. 2003; Vila et al. 2003). Despite the considerable divergence of northern and southern P. lemur populations, they do not appear to warrant classification as separate subspecies. Many other amphibian species are much more differentiated at mtDNA, and the F ST estimates fall within those for several other amphibians. Furthermore, husbandry protocols are the same for the northern and southern captive colonies, suggesting that critical reproductive and rearing aspects do not differ (Lentini 2000). Although the potential for reduced fitness due to outbreeding depression cannot be completely excluded, these factors indicate that it should be minimal. In contrast, it is known that the northern colony of P. lemur is highly inbred. While the extent of inbreeding depression has not been thoroughly evaluated and can vary within a single lineage (Pray and Goodnight 1995), it was suggested over a decade ago that the northern colony may be suffering from reduced fitness (Bloxam and Tonge 1995). Given the available evidence, inbreeding depression is a much greater concern for the future of the northern lineage than should be outbreeding depression in a mixed population.
The establishment of a third breeding colony of mixed northern and southern P. lemur lineages can be viewed as insurance for securing some northern traits, as it is probable that the pure northern colony will become extirpated due to demographic stochasticity or inbreeding depression. Because outbreeding depression is due primarily to the breakup of coadapted gene complexes between divergent lineages, it is probable that negative effects would not manifest until the F 3 generation when such complexes have undergone recombination (Tallmon et al. 2004). The mixed colony should therefore be created immediately so that the fitness of mixed individuals can be monitored for several generations prior to release into the wild (e.g., Moritz 1999). Although fitness in captivity cannot necessarily be extrapolated to the wild, monitoring should provide an indication of the overall health and reproductive capacity of the interbred colony relative to the pure northern colony. Sufficient individuals are generally produced in captivity that these efforts should not affect the existing breeding colonies or release efforts. Its initiation would also not require a large investment of resources, as several facilities currently house P. lemur and much is known about its husbandry (Miller 1985; Lentini 2000). Future work should develop models to determine the minimum number of southern individuals necessary to effect genetic rescue of the northern population while minimising genetic swamping, as was done for the Florida panther (Hedrick 1995).
Not only will a mixed breeding colony facilitate the establishment of P. lemur populations in northern Puerto Rico by preserving adaptive traits, its experimental nature enables these recovery efforts to contribute to the development of reintroduction biology as a science (sensu Seddon et al. 2007). It can also provide insight into the actual repercussions of outbreeding depression, allowing future conservation biologists to assess the potential for adverse effects on a given species. Finally, this study demonstrates that it is not always possible to follow generalised conservation guidelines for all species; rather, each species must be evaluated individually and decisions made to maximise the retention of genetic diversity, thereby maximising the species’ prospects for recovery.
References
Arens P, Bugter R, van’t Westende W et al (2006) Microsatellite variation and population structure of a recovering tree frog (Hyla arborea L.) metapopulation. Conserv Genet 7:825–835. doi:10.1007/s10592-005-9112-7
Austin JD, Lougheed SC, Neidrauer L, Chek AA, Boag PT (2002) Cryptic lineages in a small frog: the post-glacial history of the spring peeper, Pseudacris crucifer (Anura: Hylidae). Mol Phylogenet Evol 25:316–329. doi:10.1016/S1055-7903(02)00260-9
Austin JD, Davila JA, Lougheed SC, Boag PT (2003) Genetic evidence for female-biased dispersal in the bullfrog, Rana catesbeiana (Ranidae). Mol Ecol 12:3165–3172. doi:10.1046/j.1365-294X.2003.01948.x
Avise JC, Nelson WS (1989) Molecular genetic relationships of the extinct dusky seaside sparrow. Science 243:646–648. doi:10.1126/science.243.4891.646
Ball SJ, Adams M, Possingham HP, Keller MA (2000) The genetic contribution of single male immigrants to small, inbred populations: a laboratory study using Drosophila melanogaster. Heredity 84:677–684. doi:10.1046/j.1365-2540.2000.00721.x
Ballou JD, Lacy RC (1995) Identifying genetically important individuals for management of genetic diversity in pedigreed populations. In: Ballou JD, Gilpin M, Foose TJ (eds) Population management for survival and recovery: analytical methods and strategies in small population conservation. Columbia University Press, New York
Barber D (2007) Cooperative Amphibian Programs in AZA: Puerto Rican Crested Toad SSP. Connect February: 13
Barribeau SM, Villinger J, Waldman B (2008) Major histocompatibility complex based resistance to a common bacterial pathogen of amphibians. PLoS ONE 3:1–9. doi:10.1371/journal.pone.0002692
Beauclerc KB, Johnson B, White BN (2007) Characterization, multiplex conditions, and cross-species utility of tetranucleotide microsatellite loci for Blanchard’s cricket frog (Acris crepitans blanchardi). Mol Ecol Notes 7:1338–1341. doi:10.1111/j.1471-8286.2007.01874.x
Bloxam QMC, Tonge SJ (1995) Amphibians: suitable candidates for breeding-release programmes. Biodivers Conserv 4:634–644. doi:10.1007/BF00222519
Bos DH, Sites JW (2001) Phylogeography and conservation genetics of the Columbia spotted frog (Rana luteiventris; Amphibia, Ranidae). Mol Ecol 10:1499–1513. doi:10.1046/j.1365-294X.2001.01295.x
Brede EG, Beebee TJC (2004) Contrasting population structures in two sympatric anurans: implications for species conservation. Heredity 92:110–117. doi:10.1038/sj.hdy.6800391
Brock MK, White BN (1992) Application of DNA fingerprinting to the recovery program of the endangered Puerto Rican parrot. Proc Natl Acad Sci USA 89:11121–11125. doi:10.1073/pnas.89.23.11121
Bryant EH, Backus VL, Clark ME, Reed DH (1999) Experimental tests of captive breeding for endangered species. Conserv Biol 13:1487–1496. doi:10.1046/j.1523-1739.1999.98440.x
Burbrink FT, Lawson R, Slowinski JB (2000) Mitochondrial DNA phylogeography of the polytypic North American rat snake (Elaphe obsoleta): a critique of the subspecies concept. Evolution 54:2107–2118
Burns EL, Eldridge MDB, Houlden BA (2004) Microsatellite variation and population structure in a declining Australian Hylid Litoria aurea. Mol Ecol 13:1745–1757. doi:10.1111/j.1365-294X.2004.02190.x
Culver M, Johnson WE, Pecon-Slattery J, O’Brien SJ (2000) Genomic ancestry of the American puma (Puma concolor). J Hered 91:186–197. doi:10.1093/jhered/91.3.186
Daugherty CH, Cree A, Hay JM, Thompson MB (1990) Neglected taxonomy and continuing extinctions of tuatara (Sphenodon). Nature 347:177–179. doi:10.1038/347177a0
Dever JA (2007) Fine-scale genetic structure in the threatened foothill yellow-legged frog (Rana boylii). J Herpetol 41:168–179. doi:10.1670/0022-1511(2007)41[168:FGSITT]2.0.CO;2
Drew RE, Hallett JG, Aubry KB et al (2003) Conservation genetics of the fisher (Martes pennanti) based on mitochondrial DNA sequencing. Mol Ecol 12:51–62. doi:10.1046/j.1365-294X.2003.01715.x
Duellman WE, Trueb L (1986) Biology of amphibians. Johns Hopkins University Press, Baltimore
Eizirik E, Kim J, Menotti-Raymond M et al (2001) Phylogeography, population history and conservation genetics of jaguars (Panthera onca, Mammalia, Felidae). Mol Ecol 10:65–79. doi:10.1046/j.1365-294X.2001.01144.x
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software structure: a simulation study. Mol Ecol 14:2611–2620. doi:10.1111/j.1365-294X.2005.02553.x
Excoffier L, Laval G, Schneider S (2005) arlequin ver. 3.0: An integrated software package for population genetics data analysis. Evol Bioinform 1:47–50
Ford MJ (2002) Selection in captivity during supportive breeding may reduce fitness in the wild. Conserv Biol 16:815–825. doi:10.1046/j.1523-1739.2002.00257.x
Frankham R, Ballou JD, Briscoe DA (2002) Introduction to Conservation Genetics. Cambridge University Press, Cambridge
Funk WC, Blouin MS, Corn PS et al (2005) Population structure of Columbia spotted frogs (Rana luteiventris) is strongly affected by the landscape. Mol Ecol 14:483–496. doi:10.1111/j.1365-294X.2005.02426.x
Garcia-Moreno J, Matocq MD, Roy MS, Geffen E, Wayne RK (1996) Relationships and genetic purity of the endangered Mexican wolf based on analysis of microsatellite loci. Conserv Biol 10:376–389. doi:10.1046/j.1523-1739.1996.10020376.x
Godoy JA, Negro JJ, Hiraldo F, Donazar JA (2004) Phylogeography, genetic structure and diversity in the endangered bearded vulture (Gypaetus barbatus, L.) as revealed by mitochondrial DNA. Mol Ecol 13:371–390. doi:10.1046/j.1365-294X.2003.02075.x
Goebel AM, Donnelly JM, Atz ME (1999) PCR primers and amplification methods for 12S ribosomal DNA, the control region, cytochrome oxidase I, and cytochrome b in bufonids and other frogs, and an overview of PCR primers which have amplified DNA in amphibians successfully. Mol Phylogenet Evol 11:163–199. doi:10.1006/mpev.1998.0538
Guo SW, Thompson EA (1992) Performing the exact test of Hardy–Weinberg proportion for multiple alleles. Biometrics 48:361–372. doi:10.2307/2532296
Hakansson J, Bratt C, Jensen P (2007) Behavioural differences between two captive populations of red jungle fowl (Gallus gallus) with different genetic background, raised under identical conditions. Appl Anim Behav Sci 102:24–38. doi:10.1016/j.applanim.2006.03.013
Hall TA (1999) bioedit: a user-friendly biological sequence alignment editor and analysis program for Windows 95–98-NT. Nucleic Acids Symp Ser 41:95–98
Hedges B, Joglar R, Thomas R (2004) Bufo lemur. In: 2007 IUCN red list of threatened species (www.iucnredlist.org). Accessed June 11 2008
Hedrick PW (1995) Gene flow and genetic restoration: the Florida panther as a case study. Conserv Biol 9:996–1007. doi:10.1046/j.1523-1739.1995.9050996.x
Hoffman EA, Schueler FW, Blouin MS (2004) Effective population sizes and temporal stability of genetic structure in Rana pipiens, the northern leopard frog. Evolution 58:2536–2545
Johansson M, Primmer CR, Sahlsten J, Merila J (2005) The influence of landscape structure on occurrence, abundance and genetic diversity of the common frog, Rana temporaria. Glob Change Biol 11:1664–1679. doi:10.1111/j.1365-2486.2005.1005.x
Johnson RR (1990) Release and translocation strategies for the Puerto Rican crested toad, Peltophryne lemur. Endanger Species Update 8:54–57
Johnson B (1999) Recovery of the Puerto Rican Crested Toad. Endanger Species Bull 24:8–9
Kalinowski ST (2005) hp-rare 1.0: a computer program for performing rarefaction on measures of allelic richness. Mol Ecol Notes 5:187–189. doi:10.1111/j.1471-8286.2004.00845.x
Kraaijeveld-Smit FJL, Beebee TJC, Griffiths RA, Moore RD, Schley L (2005) Low gene flow but high genetic diversity in the threatened Mallorcan midwife toad Alytes muletensis. Mol Ecol 14:3307–3315. doi:10.1111/j.1365-294X.2005.02614.x
Kumar S, Tamura K, Nei M (2004) mega3: Integrated software for Molecular Evolutionary Genetics Analysis and sequence alignment. Brief Bioinform 5:150–163. doi:10.1093/bib/5.2.150
Lacy R, Foster M (1987) Summary of electrophoretic analyses for Puerto Rican crested toad. Report, Chicago Zoological Society, 2 pp
Land D, Shindle D, Cunningham M, Lotz M, Ferree B (2004) Florida panther genetic restoration and management annual report 2003–2004. Florida Fish and Wildlife Conservation Commission, Tallahassee
Lentini A (2000) Puerto Rican Crested Toad (Peltophryne lemur) SSP Husbandry Manual. Keeper and Curator Edition. Toronto Zoo, Scarborough, Ontario. 48 pp
Lougheed SC, Gascon C, Jones DA, Bogart JP, Boag PT (1999) Ridges and rivers: a test of competing hypotheses of Amazonian diversification using a dart-poison frog (Epipedobates femoralis). Proc R Soc Lond B Biol Sci 266:1829–1835. doi:10.1098/rspb.1999.0853
Madsen T, Shine R, Olsson M, Wittzell H (1999) Restoration of an inbred adder population. Nature 402:34–35. doi:10.1038/46941
Martinez-Solano I, Rey I, Garcia-Paris M (2005) The impact of historical and recent factors on genetic variability in a mountain frog: the case of Rana iberica (Anura: Ranidae). Anim Conserv 8:431–441. doi:10.1017/S136794300500243X
Matos-Torres JJ (2006) Habitat characterization for the Puerto Rican crested toad (Peltophryne lemur) at Guanica State Forest, Puerto Rico. Unpublished M.Sc. dissertation, University of Puerto Rico, Mayaguez
Miller TJ (1985) Husbandry and breeding of the Puerto Rican toad (Peltophryne lemur) with comments on its natural history. Zoo Biol 4:281–286. doi:10.1002/zoo.1430040308
Monroe WH (1976) The karst landforms of Puerto Rico. USGS Professional Paper 899. Washington, DC
Montgomery ME, Ballou JD, Nurthen RK et al (1997) Minimizing kinship in captive breeding programs. Zoo Biol 16:377–389. doi:10.1002/(SICI)1098-2361(1997)16:5<377::AID-ZOO1>3.0.CO;2-7
Moritz C (1999) Conservation units and translocations: strategies for conserving evolutionary processes. Hereditas 130:217–228. doi:10.1111/j.1601-5223.1999.00217.x
Moritz C, Dowling TE, Brown WM (1987) Evolution of animal mitochondrial DNA: relevance for population biology and systematics. Annu Rev Ecol Syst 18:269–292. doi:10.1146/annurev.es.18.110187.001413
Nei M (1972) Genetic distance between populations. Am Nat 106:283–292. doi:10.1086/282771
Newman RA, Squire T (2001) Microsatellite variation and fine-scale population structure in the wood frog (Rana sylvatica). Mol Ecol 10:1087–1100. doi:10.1046/j.1365-294X.2001.01255.x
Newman D, Tallmon DA (2001) Experimental evidence for beneficial fitness effects of gene flow in recently isolated populations. Conserv Biol 15:1054–1063. doi:10.1046/j.1523-1739.2001.0150041054.x
Nielson M, Lohman K, Sullivan J (2001) Phylogeography of the tailed frog (Ascaphus truei): implications for the biogeography of the Pacific Northwest. Evolution 55:147–160
Nittinger F, Gamauf A, Pinsker W, Wink M, Haring E (2007) Phylogeography and population structure of the saker falcon (Falco cherrug) and the influence of hybridization: mitochondrial and microsatellite data. Mol Ecol 16:1497–1517. doi:10.1111/j.1365-294X.2007.03245.x
Paetkau D, Waits LP, Clarkson PL, Craighead L, Strobeck C (1997) An empirical evaluation of genetic distance statistics using microsatellite data from bear (Ursidae) populations. Genetics 147:1943–1957
Palo JU, O’Hara RB, Laugen AT et al (2003) Latitudinal divergence of common frog (Rana temporaria) life history traits by natural selection: evidence from a comparison of molecular and quantitative genetic data. Mol Ecol 12:1963–1978. doi:10.1046/j.1365-294X.2003.01865.x
Pico R (1974) The geography of Puerto Rico. Aldine Publishing Co., Chicago
Pray LA, Goodnight CJ (1995) Genetic variation in inbreeding depression in the red flour beetle Tribolium castaneum. Evolution 49:176–188
PRCT SSP (2006) The Puerto Rican crested toad species survival plan website. (www.crestedtoadssp.org). Accessed June 4 2008
Pregill G (1981) Late Pleistocene herpetofaunas from Puerto Rico. University of Kansas Miscellaneous Publication #71, Lawrence
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Raymond M, Rousset F (1995) genepop (Version 1.2): Population genetics software for exact tests and ecumenicism. J Hered 86:248–249
Richards CM (2000) Inbreeding depression and genetic rescue in a plant metapopulation. Am Nat 155:383–394. doi:10.1086/303324
Rivero JA, Mayorga H, Estremera E, Izquierdo I (1980) Sobre el Bufo lemur (Cope) (Amphibia, Bufonidae). Caribb J Sci 15:33–40
Rowe G, Beebee TJC, Burke T (1998) Phylogeography of the natterjack toad Bufo calamita in Britain: genetic differentiation of native and translocated populations. Mol Ecol 7:751–760. doi:10.1046/j.1365-294x.1998.00387.x
Rowe G, Harris DJ, Beebee TJC (2006) Lusitania revisited: a phylogeographic analysis of the natterjack toad Bufo calamita across its entire biogeographical range. Mol Phylogenet Evol 39:335–346. doi:10.1016/j.ympev.2005.08.021
Ruokonen M, Kvist L, Tegelstroem H, Lumme J (2000) Goose hybrids, captive breeding and restocking of the Fennoscandian populations of the lesser white-fronted goose (Anser erythropus). Conserv Genet 1:277–283. doi:10.1023/A:1011509922762
Seddon PJ, Armstrong DP, Maloney RF (2007) Developing the science of reintroduction biology. Conserv Biol 21:303–312. doi:10.1111/j.1523-1739.2006.00627.x
Shaffer HB, Fellers GM, Voss SR, Oliver JC, Pauly GB (2004a) Species boundaries, phylogeography and conservation genetics of the red-legged frog (Rana aurora/draytonii) complex. Mol Ecol 13:2667–2677. doi:10.1111/j.1365-294X.2004.02285.x
Shaffer HB, Pauly GB, Oliver JC, Trenham PC (2004b) The molecular phylogenetics of endangerment: cryptic variation and historical phylogeography of the California tiger salamander, Ambystoma californiense. Mol Ecol 13:3033–3049. doi:10.1111/j.1365-294X.2004.02317.x
Shuster SM, Miller MP, Lang BK et al (2005) The effects of controlled propagation on an endangered species: genetic differentiation and divergence in body size among native and captive populations of the Socorro Isopod (Crustacea: Flabellifera). Conserv Genet 6:355–368. doi:10.1007/s10592-005-5003-1
Smith MA, Green DM (2004) Phylogeography of Bufo fowleri at its northern range limit. Mol Ecol 13:3723–3733. doi:10.1111/j.1365-294X.2004.02301.x
Soltis PS, Gitzendanner MA (1999) Molecular systematics and the conservation of rare species. Conserv Biol 13:417–483. doi:10.1046/j.1523-1739.1999.97286.x
Tallmon DA, Luikart G, Waples RS (2004) The alluring simplicity and complex reality of genetic rescue. Trends Ecol Evol 19:489–496. doi:10.1016/j.tree.2004.07.003
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The clustal_x windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882. doi:10.1093/nar/25.24.4876
USFWS (US Fish and Wildlife Service) (1992) Recovery plan for the Puerto Rican crested toad (Peltophryne lemur). USA Department of Interior, Atlanta
Vila C, Sundqvist A-K, Flagstad O et al (2003) Rescue of a severely bottlenecked wolf (Canis lupus) population by a single immigrant. Proc R Soc Lond B Biol Sci 270:91–97. doi:10.1098/rspb.2002.2184
Vredenburg VT, Bingham R, Knapp R et al (2007) Concordant molecular and phenotypic data delineate new taxonomy and conservation priorities for the endangered mountain yellow-legged frog. J Zool (Lond) 271:361–374. doi:10.1111/j.1469-7998.2006.00258.x
Waldman B, Rice JE, Honeycutt RL (1992) Kin recognition and incest avoidance in toads. Am Zool 32:18–30
Weir BS, Cockerham CC (1984) Estimating F-statistics for the analysis of population structure. Evolution 38:1358–1370. doi:10.2307/2408641
Westemeier RL, Brawn JD, Simpson SA et al (1998) Tracking the long-term decline and recovery of an isolated population. Science 282:1695–1698. doi:10.1126/science.282.5394.1695
Wright S (1951) The genetical structure of populations. Ann Eugen 15:323–354
Wyner YM, Amato G, Desalle R (1999) Captive breeding, reintroduction, and the conservation genetics of black and white ruffed lemurs, Varecia variegata variegata. Mol Ecol 8:S107–S115. doi:10.1046/j.1365-294X.1999.00815.x
Zink RM, Barrowclough GF, Atwood JL, Blackwell-Rago RC (2000) Genetics, taxonomy, and conservation of the threatened California gnatcatcher. Conserv Biol 14:1394–1405. doi:10.1046/j.1523-1739.2000.99082.x
Acknowledgments
Many thanks go to M. Canals of the Guánica State Forest and R. Dietz of the Audubon Nature Institute for providing tissue samples. A. Lentini coordinated samples from the Toronto Zoo and answered many questions during the work. Funding was provided by the Toronto Zoo, a Natural Sciences and Engineering Research Council (NSERC) operating grant to BNW, and NSERC PGS-A and PGS-B to KBB. T. Frasier and two anonymous referees provided valuable comments on earlier versions of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beauclerc, K.B., Johnson, B. & White, B.N. Genetic rescue of an inbred captive population of the critically endangered Puerto Rican crested toad (Peltophryne lemur) by mixing lineages. Conserv Genet 11, 21–32 (2010). https://doi.org/10.1007/s10592-008-9782-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-008-9782-z