Abstract
Pollen dispersal was investigated in five remnant populations of Eucalyptus wandoo, a dominant insect-pollinated tree in the fragmented agricultural region of southern Western Australia. Paternity analysis using six microsatellite loci identified a pollen source for 45% of seedlings, and the remainder were assumed to have arisen from pollen sources outside the stands. Outcrossing was variable, ranging from 52 to 89%, and long distance pollen dispersal was observed in all populations with up to 65% of pollen sourced from outside the populations over distances of at least 1 km. Modelling dispersal functions for pollination events within the two larger populations showed little difference between the four two-parameter models tested and indicated a fat-tailed dispersal curve. Similarity of direct and indirect historical estimates of gene flow indicates maintenance of gene flow at levels experienced prior to fragmentation. The study revealed extensive long distance pollen dispersal in remnant patches of trees within a fragmented agricultural landscape in the southern temperate region and highlighted the role of remnant patches in maintaining genetic connectivity at the landscape scale.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Scattered single trees and small patches of trees (<1 ha) left after land clearing are common features of agricultural landscapes throughout the world, and a growing literature shows that they provide ecological resources and services important for regional conservation and sustainable land-use (Hobbs and Yates 2000; Gibbons and Boak 2002). Single and small patches of trees provide food, such as nectar, pollen and seeds, and nesting sites for many species of small mammals, birds and invertebrates (Lumsden and Bennett 2005; Oliver et al. 2006; Gibbons and Boak 2002). They act as “stepping stones” for many animals, facilitating movement and dispersal between larger fragments thereby contributing to meta-population viability (Gibbons and Boak 2002). Scattered trees and small patches of trees also have a significant influence on soil condition and hydrology (Bird et al. 1991; George et al. 1995; Reid and Landsberg 2000; Jackson and Ash 2001). More recently the role that single scattered trees and small patches might play in facilitating pollen dispersal among larger fragments across the landscape has been recognized (White et al. 2002; Lowe et al. 2005). Extensive pollen dispersal among fragments increases effective population size beyond the boundary of the fragment and will maintain genetic variability by reducing the effects of inbreeding and drift associated with small population size (Lowe et al. 2005).
Pollen dispersal is idiosyncratic and greatly influenced by landscape context and ecological variables (Sork et al. 1999). For animal pollinated species, pollen dispersal following fragmentation will be largely influenced by changes in pollinator behaviour due to the spatial distribution of populations, particularly plant density and population size (Ellstrand 1992; Stacey et al. 1996; Kunin 1997), or by change in the composition of pollinator communities in altered landscapes. The interaction between mating, spatial and ecological factors makes predictions of the level of pollen dispersal in any particular landscape difficult. Population genetic theory predicts that fragmentation of landscapes through anthropogenic disturbance will lead to reduced gene flow, and, in association with decreased population size, reduced genetic diversity and increased genetic structure due to random genetic drift and elevated inbreeding (Young et al. 1996; Hamrick 2004). In contrast to expectations, most tree species in which pollen dispersal has been investigated in fragmented landscapes have shown extensive gene flow that may well have increased due to physical isolation of populations (Nason and Hamrick 1997; Aldrich and Hamrick 1998; White et al. 2002; Dick et al. 2003; Bacles et al 2005). This extensive gene flow has been attributed to a reduction in near-neighbour mating events, either by changes in pollinator behaviour and/or changes in the composition of the pollinator community (Stacy et al. 1996, White et al. 2002, Dick et al. 2003). In addition, reduced size will lead to an increase in the relative level of pollen dispersal into a population since there will be fewer trees within the population to contribute to the pollen pool (Ellstrand 1992).
Thus empirical evidence indicates that pollen dispersal among the scattered trees and remnant populations that are common in many fragmented landscapes provides significant connectivity that increases effective population sizes and thus maintains genetic variability and weakens the diversifying effects of genetic drift. To date these studies have been confined to tropical and northern temperate species, that are generally dioecious, self-incompatible or wind pollinated. The relevance of these studies to the insect pollinated, mixed mating systems of eucalypts that dominate Australian woodland and forest ecosystems is untested. In this study we assessed direct evidence of pollen dispersal in Eucalyptus wandoo, a woodland and forest tree that is characteristic of many eucalypts in fragmented landscapes across southern Australia. It is widely distributed through the agricultural region of Western Australia, and prior to European settlement occurred in relatively large populations in woodlands in a natural heath-woodland mosaic. Widespread land clearing has led to reduced size and increased isolation and fragmentation of populations with many scattered trees in paddocks or on road verges.
Our aim was to measure the level of pollen flow among scattered trees and small patches of E. wandoo by determining (1) the amount of pollen immigration into populations, (2) the minimum distances over which pollen dispersal occurs, and (3) the pattern of localised pollen dispersal within small populations. This was achieved using paternity assignment methods. In addition, the level of current gene flow from direct estimates of pollen dispersal was compared with indirect estimates of historical gene flow based on genetic differentiation between populations. We compare our results with levels of gene flow reported for tree species from other biomes.
Materials and methods
Study species and site details
The study was undertaken in the Dongolocking catchment approximately 250 km south east of Perth in the south-west of Western Australia, an area that has been extensively cleared for intensive cropping and grazing. Approximately 18% of the original vegetation remains as fragments ranging in size from 0.26 to 1,261 ha with a mean area of 24 ha (Wallace 1998). Eucalyptus wandoo is a key component of the woodlands in this region, which, prior to land clearing, were extensive but patchily distributed, forming complex landscape mosaics with heath and shrublands. Extant populations of E. wandoo vary in size and occur across a variety of landscape contexts, ranging from isolated paddock trees to linear strips of trees on road verges to larger woodlands of varying size on private land and in nature reserves. Eucalyptus wandoo is a primarily insect pollinated tree of the Myrtaceae family. It produces masses of small, white to cream-coloured flowers over an extended period of several months from December to March. The flowers are protandrous but geitenogamous pollination is possible because trees are self-compatible and have a large number of flowers occurring simultaneously at various developmental stages.
Pollen dispersal was investigated in five patches of trees, two larger populations of 40 and 46 individuals, and three patches with less than ten individuals. The characteristics of the populations are given in Table 1 and the location of trees in the two larger populations is shown in Fig. 1. The size of each population was determined by direct count and isolation was estimated by measuring the percentage area of remnant vegetation within a 3 km radius of the population using a remnant vegetation overlay in ArcView v3.2. Two of the small populations (J and S) had few neighbouring trees to act as pollen source within a 1 km radius, which allowed estimates of pollen dispersal over this distance. For Population J, there were only 15 trees within a 1 km radius of the population, all located along a track ranging from 290 to 550 m to the north and north-west of the population. The next nearest trees, approximately 50, were along a road verge 1.08 km to the east of the population. All the trees within the 1 km radius were genotyped to assign paternity. For Population S, five scattered trees occurred 300–500 m to the east and the next nearest trees occurred in two patches, 15 trees 640 m to the south-east and five trees 580 m to the south. The five nearest trees were genotyped to allow paternity assignment.
Plant material and genotype analysis
Leaf samples were collected from all trees within each of the five populations (102 trees in total). Extraction of DNA from the leaves was carried out as in Byrne et al. (1998) with 0.1 M sodium sulphite added to the extraction buffer (Byrne et al. 2001). Seed was collected from each tree that produced seed in the small populations and 11–12 trees in the larger populations (33 seed crops in total). Seed was germinated in vermiculite until the cotyledons emerged. DNA was extracted from whole two week old seedlings using a small scale version of the CTAB method of Doyle and Doyle (1990).
DNA samples from the adult trees in the populations and 20 seedlings from each mother tree were assayed for six microsatellite loci using eucalypt primers developed by Brondani et al. (1998) and Steane et al. (2001). Amplification reactions were carried out by polymerase chain reaction with 20 ng of E. wandoo DNA in a 15 μl reaction volume containing 50 mM KCl, 20 mM Tris–HCl pH 8.4, 0.2 mM dNTP’s, 0.3 μM of each primer, 0.5 unit of Taq DNA polymerase and either 1.0 mM (EMBRA 7, EMCRC 12, EMBRA 6), 1.25 mM (EMBRA 10) or 1.5 mM (EMCRC 6, EMBRA 18) MgCl2. Cycling conditions for PCR amplification were 96°C for 2 min; 29 cycles of 30 s at 95°C, 30 s at 56°C, 30 s at 72°C; 5 min at 72°C except for EMCRC 12 which required a touch down reaction of 96°C for 2 min; 20 cycles of 30 s at 94°C, 30 s at 69.5°C with a step down of 0.5°C per cycle, 30 s at 72°C; 10 cycles of 30 s at 94°C, 30 s at 60°C, 30 s at 72°C; 2 min at 72°C. Amplification products from the adults and progeny of the small populations were loaded into 8% non-denaturing polyacrylamide gels and subjected to electrophoresis at 300 V for 4 h. The gels were stained with ethidium bromide and photographed under UV light. Amplification products from the adults and progeny of the larger populations were separated on an Amersham Megabase capillary sequencer using the Genetic Analysis Facility at James Cook University, Queensland. Random sets of samples were rerun to check for repeatability of genotyping and segregation of maternal alleles in the progeny was checked.
Data analysis
The genotype of each sample was scored and alleles were aligned across all families within each population. Diversity parameters for the loci were determined using CERVUS (Marshall et al. 1998). Paternity assignment was undertaken using the maximum likelihood approach in CERVUS using 95% criterion for assignment and allowing mismatch at one locus. Since geitonogamous pollination is possible, assignment of paternity to the mother plant was assumed to be due to selfing. Outcrossing rate was determined as 1−s where s is the proportion of selfed progeny. The level of pollen immigration, t o, was determined as 1−(t i + s) where t i is the proportion of outcross events originating from pollen sources within the population. Assignment based on likelihood may result in cryptic pollinations where paternity is assigned to a most-likely parent that is not the true parent. Cryptic pollination was assessed in FAMOZ (Gerber et al. 2003) using simulations to estimate the number of times paternity was assigned to a genotyped parent when it was not the true parent.
Indirect estimates of historical gene flow were made using differentiation between populations based on the genotypes of adult trees. Due to long generation times and recent land clearing in the last 50 years, these trees were present in the populations prior to fragmentation and therefore differentiation among populations represents pre-fragmentation patterns of genetic structure. Population differentiation was calculated using the RST estimate of Rousset (1996) implemented in FSTAT (Goudet 2001). Calculation of indirect gene flow was made using RST in place of FST in Wright’s (1951) relationship, Nm = (1−FST)/4FST. Differences in selfing and outcrossing between the two large populations were assessed using an unpaired two-tailed t-test assuming unequal variance.
The pattern of pollen dispersal within the two larger populations was evaluated by determining the distance between mother and pollen source for each of the seed sired by trees within the population. The number of actual pollen dispersal events within the two larger populations, R and F, were plotted as a function of distance between parental trees. Four dispersal curves (exponential power, Weibull, geometric and 2Dt functions, see Austerlitz et al. 2004) were fitted using ordinary least squares and the procedure NLIN in the SAS statistical package, version 9.1. As numbers of pollen events were modelled rather than probability values, an additional scaling parameter, z, was added where necessary.
Results
Microsatellite variation
The six microsatellite markers were highly variable within these populations of E. wandoo. We detected a total of 164 alleles in 652 genotyped seeds, with an average of 27.3 alleles per locus. Observed heterozygosity ranged from 0.474 (EMCRC 12) to 0.829 (EMBRA 18) and was generally lower than expected heterozygosity (Table 2). The polymorphic information content of all loci was high with a mean of 0.855. The exclusion probability for paternal assignment for individual loci ranged from 0.584 (EMCRC 6) to 0.865 (EMCRC 12) and the total paternity exclusion probability over the six loci was 0.999.
Paternity assignment
Paternity assignment identified a pollen source within the populations, including self-pollination, for 45% of all seedlings. Paternity of the remaining seedlings was not assigned to any tree within the stands and they were assumed to have originated from pollen sources outside the populations. The level of selfing, pollination from plants within the population, and pollen immigration from outside the population are given in Table 3. Estimates of cryptic pollination for the two larger populations were very low, 0% for population R and 0.13% for population F, indicating that identification of pollination category (i.e. selfing, pollination from plants within the population, and pollen immigration) was highly reliable.
The mean level of outcrossing in the populations varied between 52% (Population S) and 89% (Population R) (Table 3). The mean pollen immigration rates in the populations varied between 33% (Population S) and 65% (Population R). Comparison of the two larger populations, which were of similar size but different density, showed similar levels of cross pollination within the populations, but the level of selfing and the level of pollen immigration were significantly different between the populations (selfing t = 2.11, P = 0.049; external outcrossing t = −2.68, P = 0.015), with less selfing and greater pollen immigration in the denser, patch Population R than in the less dense, linear Population F.
Pollen dispersal
Paternity assignment to neighbouring trees was made for Populations J and S. None of the trees within the 1 km radius of Population J contributed pollen to the seed crops analysed, indicating that pollen dispersal occurred over at least 1 km. For Population S none of five nearest trees contributed pollen to the seed crops analysed, indicating that pollen dispersal occurred over at least 580 m into this population. Pollen dispersal distance into the two larger populations occurred over at least 350 m as these populations were isolated from other trees by this distance, and pollen immigration of 48.7–64.8% was identified for these populations.
The level of population differentiation (RST) between the two larger populations was 0.018, and this gives an indirect estimate of gene flow (Nm) of 13.5. For comparison, direct estimates of gene flow can be obtained from the level of pollen immigration detected in the two larger populations and the current population size, and gave Nm = 13 and 11.5 for Population R and F respectively.
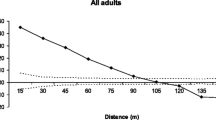
The average pollen dispersal distance within Population R was 30.5 m, which is 63% of the average distance between all trees in the population, and within Population F was 49 m or 57% of the average distance between all trees in the population (Table 4). While the size of the two populations was similar the shape and area they covered differed (Fig. 1). The pattern of pollen dispersal for distance classes in the two populations differed (Fig. 2). Pollen dispersal among trees in the denser, patch shaped Population R showed a steep decay curve with frequent short distance matings and fewer longer distance matings. Pollen dispersal in the linear less dense Population F showed a flatter distribution with fewer short distance matings and a relatively a fat tail. Dispersal curves for the exponential power, geometric, Weibull and 2Dt families were fitted to the observed dispersal events within the populations, and for both populations the 2Dt dispersal curve provided the best fit (Table 5, Fig. 2), although all four dispersal functions were similar in each population.
Discussion
Pollen dispersal
This study has shown maintenance of high gene flow through pollen dispersal in E. wandoo despite occurring in a landscape fragmented by agricultural production. Over 30%, and up to 65%, of pollination occurred from outside the stands, and where the minimum distances could be determined, this pollination occurred from pollen sources 0.6–1.08 km away. This high level of pollen dispersal across the landscape was consistent for all five populations regardless of size or isolation and most of the 33 trees assessed. Thus, pollen dispersal is providing genetic connectivity between trees in these small remnant patches of E. wandoo. The level of pollen dispersal in this southern temperate eucalypt species is similar to that observed in trees from other biomes, particularly insect pollinated tropical species where extensive dispersal occurred over kilometres (Nason and Hamrick 1997; Aldrich and Hamrick 1998; White et al. 2002; Dick et al. 2003). Low density spatial distribution of trees affects pollinator behaviour leading to reduction in near neighbour mating (Chase et al. 1996; Stacy et al. 1996; Dick et al. 2003) and it appears that this is also influencing pollen dispersal patterns in fragmented patches of E. wandoo. It is not unexpected that density would have a strong influence on pollen dispersal since it has been demonstrated that density influences rates of outcrossing in tree species (e.g. Murawski and Hamrick 1991; Aldrich and Hamrich 1998) through affects on pollinator behaviour (Levin and Kerster 1974).
The pattern of pollen dispersal within the two larger populations (R and F) also showed differences that were consistent with differences in the density of trees in the populations. The relatively denser population (R) showed more frequent short distance pollination events than the less dense population (F) where pollen dispersal was more uniform throughout the population. This is consistent with density-dependant foraging of bees where a decrease in plant density was accompanied by an increase in flight distance (Levin and Kerster 1974).
The high level of pollen dispersal across relatively large distances observed in this study is similar to other empirical studies of realised pollen dispersal into sink populations that tend to show a more normal distribution with higher levels of long distance pollen dispersal (Fenster 1991; Dick et al. 2003). This is in contrast to the highly leptokurtic distributions that have been observed in studies of pollinator flight distances (Fenster 1991) and studies of pollen dispersal from a source, where most dispersal occurred at short distances with a long tail of low level pollen dispersal over larger distances (Levin and Kerster 1974; Handel 1983). Estimation of dispersal from the source underestimates pollen dispersal into the sink, since the sink will sample the tails of dispersal curves from many trees (Ellstrand 1992).
Estimation of the dispersal curve at the landscape scale is not possible from this data but the shape of the dispersal curve at the population level may be used as a surrogate. The known dispersal events within the two large populations allowed determination of the best fitting dispersal curves at the population level. For both the larger populations the 2Dt distribution provided the best fit although there was little difference between all of the two-parameter dispersal functions. The 2Dt distribution is less leptokurtic than the Weibull, geometric and exponential distributions and shows a fat tail whatever the value of the shape parameter (Austerlitz et al. 2004). Extrapolation to the landscape level would suggest a fat-tailed dispersal curve with significant levels of pollen dispersal over long distances, which is consistent with the high level of pollen immigration detected over 1 km.
Pollen dispersal in eucalypts
No specific molecular studies of pollen dispersal among natural populations of eucalypts have been undertaken. Most evidence for pollen dispersal in natural populations has come from morphological detection of hybrids in seed crops (see examples in Potts et al. 2003). Recently a more comprehensive study of the pollen dispersal curve from a planted E. nitens population into a natural E. ovata population showed an average of 7% hybrid seed within 100 m of the E. nitens plantation boundary, dropping to 0.7% by 200–300 m, although occasional hybrids were still detected at 1.6 km from the plantation boundary (Barbour et al. 2005). This data has been interpreted as indicating low levels of pollen dispersal in eucalypts and a highly leptokurtic distribution where most dispersal occurs at short distances with a long tail of low level pollen dispersal over larger distances (Levin and Kerster 1974). However, in contrast to the limited data on pollen dispersal in natural populations, estimates of pollen immigration into exotic seed orchards in Brazil and Malagasy have been made. In E. grandis/E. urophylla orchards in Brazil, 14% pollen immigration was detected over 400 m (Campinhos et al. 1998) and 2.8% over 800 m (Jungans et al. 1998), and in a E. grandis orchard in Malagasy 40% pollen immigration was observed over 100 m (Chaix et al. 2003). These studies suggest greater pollen dispersal than the evidence from hybrid detection in natural populations, although dispersal appeared to decay to relatively low levels over larger distances (e.g. 800 m) in comparison to the large amount of pollen immigration still detected over 1 km in this study. Pollen dispersal in seed orchards outside Australia may not be comparable to that in natural eucalypt populations since they occur in quite a different ecological context.
Fragmentation
The extensive pollen dispersal found in this study of patches of E. wandoo is similar to that found in studies of low density tree species in tropical regions, where it was concluded that fragmentation, and associated change in pollinator behaviour, may be increasing pollen dispersal across the landscape (Nason and Hamrick 1997; Aldrich and Hamrick 1998; White et al. 2002; Dick et al. 2003). In this study it is not known what level of pollen dispersal distance is due to the fragmented landscape itself as we do not have estimates of pre-fragmentation dispersal, nor have direct comparison with non-fragmented areas due to the difficulty in determining direct pollen dispersal in larger populations (i.e. having to genotype all plants). However, a measure of indirect gene flow obtained from the level of differentiation between the two larger populations (Nm = 13.6) indicated high historical gene flow that is similar to the direct estimates of current gene flow derived from pollen immigration (Nm = 13 and 11.5 for Population R and F respectively). Pollen dispersal would be the major contributor to historical gene flow because seed dispersal is very limited in eucalypts, and determination of the ratio of gene flow by pollen to seed was high in E. nitens (Byrne et al. 1998). Therefore long distance pollen dispersal may have always been common for woodland eucalypts in this landscape, since the region had a natural level of historical fragmentation as woodlands were interspersed with a mosaic of heath and shrublands. The similarity between direct and indirect estimates of gene flow imply that fragmentation has not led to reduced gene flow in this landscape.
Insects are considered the most common pollinators of E. wandoo although birds (honeyeaters) have been observed visiting E. wandoo flowers (Brown et al. 1997) and would certainly provide a means of long distance pollen dispersal as honeyeaters have been tracked moving up to 12.5 km while foraging (Saunders and Rebeira 1991). Although pollen dispersal may have been historically high, it is also possible that any impact of fragmentation through reduced dispersal has been compensated for by the introduction of honeybees (Apis mellifera) to Western Australia in the nineteenth century. Honeybees are known to forage on many native plants, including eucalypts (Paton 1996; Brown et al. 1997; Horskins and Turner 1999), and are likely to have different foraging behaviour to native bees. Although studies of honeybees show they forage on densely flowering plants and limit interplant movement (Levin and Kerster 1974; Celebrezze and Paton 2004), they are also known to forage over large distances from nests (Goulson 2003). In comparison to social honeybees, most native bees in Australia are solitary and forage over short distances (Mitchener 1970; Schwarz and Hurst 1997). Observations on African honeybees introduced into tropical ecosystems showed long distance pollination in Dinizia excelsa leading to an increase in pollen dispersal in fragmented tropical trees (Dick et al. 2003).
Conservation management
This study has demonstrated extensive pollen dispersal in E. wandoo remnants in a landscape fragmented by agricultural production. While this study is only over one flowering season in one region it suggests that high levels of pollen dispersal are occurring in eucalypts in this fragmented landscape, and that this genetic connectivity between populations may mitigate against inbreeding and loss of genetic diversity in small patches of trees. Similar observations of genetic connectivity were made in two tropical tree species, Swietenia humilis (White et al. 2002) and Dinizia excelsa (Dick et al. 2003), and this genetic connectivity highlights the integral contribution of isolated trees to population dynamics in these fragmented landscapes and challenges the low conservation value previously attributed to such trees (Jansen 1986). Paddock trees and trees on road verges are a significant component of the agricultural landscape in Western Australia, and their contribution to population connectivity through pollen dispersal raises their conservation value. Regeneration through recruitment is very limited in agricultural landscapes, therefore remnant trees are likely to die out due to lack of replacement (Hobbs and Yates 2000; Gibbons and Boak 2002; Dorrough and Moxham 2005) unless active conservation management, such as planting of trees around the edge of paddocks or promotion of natural regeneration through manipulation of grazing regimes or fire treatments, are undertaken to maintain this component of the population system. The high pollen dispersal identified in this study implies that seed quality should not be a limiting factor in the success of natural regeneration in these remnants. Further investigation is required to determine whether this level of pollen dispersal is restricted to tree species or also occurs in other insect-pollinated woody shrubs that are common components of the vegetation.
References
Aldrich PR, Hamrick JL (1998) Reproductive dominance of pasture trees in a fragmented tropical forest mosaic. Science 281:103–105
Austerlitz F, Dick CW, Dutech C, Klein EK, Oddou-Muratorio S, Smouse PE, Sork VL (2004) Using genetic markers to estimate the pollen dispersal curve. Mol Ecol 13:937–954
Barbour RC, Potts BM, Vaillancourt RE (2005) Pollen dispersal from exotic eucalypt plantations. Cons Gen 6:253–257
Bacles CFE, Burczyk J, Lowe AJ, Ennos RA (2005) Historical and contemporary mating patterns in remnant populations of the forest tree Fraxinus excelsior L. Evolution 59:979–990
Bird PR, Bicknell D, Bulman PA, Burke SJA, Leys JF, Parker JN, van der Sommen FJ, Voller P (1991) The role of shelter in Australia for protecting soils, plants and livestock. In: Prinsley RT (ed) The role of trees in sustainable agriculture. Kluwer Academic Publishers, Amsterdam, pp 59–86
Brondani R, Brondani C, Tarchini R, Grattapaglia D (1998) Development, characterisation and mapping of microsatellite markers in Eucalyptus grandis and E. urophylla. Theor Appl Genet 97:816–827
Brown EM, Burbidge AH, Dell J, Edinger D, Hopper SD, Wills RT (1997) Pollination in Western Australia, a database of animals visiting flowers. Handbook No. 15, WA Naturalists’ Club, Perth
Byrne M, Parrish TL, Moran GF (1998) Nuclear RFLP diversity in Eucalyptus nitens. Heredity 81:225–232
Byrne M, Macdonald B, Francki M (2001) Incorporation of sodium sulfite into extraction protocol minimizes degradation of Acacia DNA. Biotech 30:742–748
Campinhos EN, Peters-Robinson I, Bertolucci FL, Alfenas AC (1998) Interspecific hybridisation and inbreeding effect in seed from a Eucalyptus grandis × E. urophylla clonal seed orchard in Brazil. Genet Mol Biol 21:369–374
Celebrezze T, Paton DC (2004) Do introduced honeybees (Apis mellifera, Hymenoptera) provide full pollination service to bird-adapted Australian plants with small flowers? An experimental study of Brachyloma ericoides (Epacridacae). Aust Ecol 29:129–136
Chaix G, Gerber S, Razafimaharo V, Vigneron P, Verhaegen D, Hamon S (2003) Gene flow estimation with microsatellite in a Malagasy seed orcjhard of Eucalyptus grandis. Theor Appl Genet 107:705–712
Chase MR, Moller C, Kesseli R, Bawa KS (1996) Distant gene flow in tropical trees. Nature 383:398–399
Dick CW, Etchelecu G, Austerlitz F (2003) Pollen dispersal of tropical trees (Dinizia excelsa, Fabaceae) by native insects and African honeybees in pristine and fragmented Amazonian rainforest. Mol Ecol 12:753–764
Dorrough J, Moxham C (2005) Eucalypt establishment in agricultural landscapes and implications for landscape-scale restoration. Biol Cons 123:55–66
Doyle JJ, Doyle JL 1990. Isolation of DNA from fresh tissue. Focus 12:13–15
Ellstrand NC (1992) Gene flow by pollen, implications for plant conservation genetics. Oikos 63:77–86
Fenster CB (1991) Gene flow in Chamaecrista fasciculata (Leguminosae) I. Gene dispersal. Evol 45:398–409
George RJ, McFarlane DJ, Speed RJ (1995) The consequences for a changing hydrological environment formative vegetation in southwestern Australia. In: Saunders DA, Craig JL, Mattiske EM (eds) Nature conservation 4: the role of networks. Surrey Beatty and Sons, Chipping Norton, New South Wales, pp 9–22
Gerber S, Chabrier P, Kremer A (2003) FAMOZ, a software for parentage analysis using dominant, codominant and uniparentally inherited markers. Mol Ecol 3:479–481
Gibbons P, Boak M (2002) The value of paddock trees for regional conservation in an agricultural landscape. Ecol Manag Rest 3:205–210
Goudet J (2001) FSTAT, a program to estimate and test gene diversities and fixation indices (2.9.3). Available from http:\\www.unil.ch/softwares/fstat.html
Goulson D (2003) Effects of introduced bees on native ecosystems. Ann Rev Ecol Syst 34:1–26
Hamrick JL (2004) Response of forest trees to global environmental changes. For Ecol Man 197:323–335
Handel SN (1983) Pollination ecology, plant population structure, and gene flow. In: Real L (ed) Pollination Biology. Academic Press, Orlando, pp 163–211
Hobbs RJ, Yates CJ (2000) Temperate eucalypt Woodlands in Australia, biology, conservation, management and restoration. Surrey Beatty and Sons, Chipping Norton, New South Wales
Horskins K, Turner VB (1999) Resource use and foraging patterns of honeybees, Apis mellifera, and native insects on flowers of Eucalyptus costata. Aust J Ecol 24:221–227
Jackson J, Ash AJ (2001) The role of trees in enhancing soil nutrient availability for native perennial grasses in open eucalypt woodlands of north-east Queensland. Aust J Agric Res 52:377–386
Jansen DH (1986) Blurry catastrophes. Oikos 47:1–2
Jungans TG, Peters-Robinson I, Bertolucci FL, Alfenas AC (1998) The use of self-incompatibility in the production of hybrid eucalyptus seed by ‘Aracruz Celulose’ in Brazil. Genet Mol Biol 21:375–379
Kunin WE (1997) Population biology and rarity, on the complexity of density-dependence in insect-plant interactions. In: Kunin WE, Gaston KJ (eds) The Biology of rarity, Causes and consequences of rare-common differences. Chapman & Hall, London, pp 150–173
Levin DA, Kerster HW (1974) Gene flow in seed plants. Evol Biol 7:139–220
Lowe AJ, Boshier D, Ward M, Bacles CFE, Navarro C (2005) Genetic resource impacts of habitat loss and degradation; reconciling empirical evidence and predicted theory for neotropical trees. Heredity 95:255–273
Lumsden LF, Bennett AF (2005) Scattered trees in rural landscapes: foraging habitat for insectivorous bats in south-eastern Australia. Biol Cons 122:205–222
Marshall TC, Slate J, Kruuk LEB, Pemberton JM (1998) Statistical confidence for likelihood-based paternity inference in natural populations. Mol Ecol 7:639–655
Mitchener CD (1970) Superfamily Apoidea. In: Insects of Australia (ed CSIRO) pp 943–951.Melbourne University Press, Parkville
Murawski DA, Hamrick JL (1991) The effect of the density of flowering individuals on the mating systems of nine tropical tree species. J Hered 67:167–174
Nason JD, Hamrick JL (1997) Reproductive and genetic consequences of forest fragmentation, two case studies of neotropical canopy trees. J Hered 88:264–276
Oliver I, Pearce S, Greenslade PJM, Britton DR (2006) Contribution of paddock trees to the conservation of terrestrial invertebrate biodiversity within grazed native pastures. Aust Ecol 31:1–12
Paton DC (1996) Overview of feral and managed honeybees in Australia, Distribution, abundance, extent of interaction with native biota, evidence of impacts and future research. Australian Nature Conservation Agency, Canberra
Potts BM, Barbour RC, Hingston AB, Vaillancourt RE (2003) Genetic pollution of native eucalypt gene pools–identifying the risks. Aust J Bot 51:1–25
Reid N, Landsberg J (2000) Tree decline in agricultural landscapes: what we stand to lose. In: Hobbs RJ, Yates CJ (eds) Temperate eucalypt Woodlands in Australia, biology, conservation, management and restoration. Surrey Beatty and Sons, Chipping Norton, New South Wales, pp 127–166
Rousset F (1996) Equilibrium values of measures of population subdivision for stepwise mutation processes. Genetics 142:1357–1362
Saunders DA, Rebeira CP (1991) Values of corridors to avian populations in a fragmented landscape. In: Saunders DA, Hobbs RJ (eds) Nature Conservation 2, The Role of Corridors. Surrey Beatty & Sons, Chipping Norton, pp 221–240
Schwarz MP, Hurst PS (1997) Effects of introduced honey bees on Australia’s native bee fauna. Vic Nat 114:7–12
Sork VL, Nason J, Campbell DR, Fernandez JF (1999) Landscape approaches to historical and contemporary gene flow in plants. TREE 14:219–224
Stacy EA, Hamrick JL, Nason JD, Hubbell SP, Foster RB, Condit R (1996) Pollen dispersal in low-density populations of three neotropical tree species. Am Nat 148:275–298
Steane DA, Vaillancourt RE, Russell J, Powell W, Marshall D, Potts BM (2001) Development and characterisation of microsatellite loci in Eucalyptus globulus (Myrtaceae). Sil Genet 50:89–91
Wallace KJ (1998) Dongolocking Pilot Planning Project for Remnant Vegetation, Final Report (Phase 1). Department of Conservation and Land Management, Perth
White GM, Boshier DH, Powell W (2002) Increased pollen flow counteracts fragmentation in a tropical dry forest, an example from Swietenia humilis Zuccarini. Proc Nat Acad Sci 99:2038–2042
Wright S (1951) The general structure of populations Ann Eugenics 15:323–354
Young A, Boyle T, Brown AHD (1996) The population genetic consequences of habitat fragmentation for plants. Tree 11:413–418
Acknowledgements
We thank Bronwyn Macdonald for assistance with laboratory work, Richard Fairman for assistance with fieldwork, and Matt Williams for fitting the dispersal curves. This work was supported by Land and Water Australia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Byrne, M., Elliott, C.P., Yates, C.J. et al. Maintenance of high pollen dispersal in Eucalyptus wandoo, a dominant tree of the fragmented agricultural region in Western Australia. Conserv Genet 9, 97–105 (2008). https://doi.org/10.1007/s10592-007-9311-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-007-9311-5