Abstract
Survival following diagnosis of liver metastasis remains poor and improved treatment strategies to combat liver metastases are needed. Synthetic triterpenoids, including 1-[2-cyano-3-,12-dioxooleana-1,9(11)-dien-28-oyl]imidazole (CDDO-Imidazolide or CDDO-Im), have been shown to inhibit primary tumor growth and lung metastasis in experimental models. Oral administration of CDDO-Im results in relatively high liver concentrations, suggesting that CDDO-Im may provide an approach to treatment of liver metastases. Here we assessed the effect of CDDO-Im on liver metastasis, using B16F1 (mouse melanoma) and HT-29 (human colon carcinoma) cells. In vitro, nanomolar concentrations of CDDO-Im arrested proliferation or induced cell death in both cell lines. In vivo, cells were injected via a surgically exposed mesenteric vein to target cells to the liver of mice. Mice were then treated with CDDO-Im (800 mg/kg diet) or vehicle control. Livers were removed at endpoint and metastatic burden was quantified by standard histology. In addition, a novel whole liver magnetic resonance imaging (MRI) technique was used to assess the effect of CDDO-Im on growing metastases as well as on non-dividing, solitary cancer cells present in the same livers. CDDO-Im treatment significantly decreased liver metastasis burden in both HT-29 (n = 8 treated, 10 control) and B16F1 (n = 15 treated, 16 control) injected mice (>60%, P < 0.05), but did not reduce the numbers of solitary B16F1 cancer cells (hypo-intensity) in the same livers (P = 0.9). This study demonstrates that CDDO-Im may be useful for the treatment metastatic liver disease as it successfully inhibits growth of actively proliferating liver metastases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Liver is a common site of metastatic disease from a number of cancers, including colon and melanoma, and is associated with poor survival [1–3]. Improvements in surgical techniques and chemotherapy treatment have increased mean survival times, however 5 year survival rates remain low even after all clinically detectable metastases are surgically removed and adjuvant therapy administered [4–6]. Thus, new approaches to treat liver metastases are needed.
The synthetic triterpenoid 1-[2-cyano-3-,12-dioxooleana-1,9(11)-dien-28-oyl]imidazole (CDDO-Imidazolide or CDDO-Im) is one of a number of synthetic oleanane triterpenoids that have been shown to elicit multifunctional effects dependent on both cell type and drug concentration [7]. These effects include the potential to induce cell differentiation, cell cycle arrest [8] and apoptosis (independent of multidrug resistance status) [9, 10] in a number of different cancer cell lines [7, 11]. Pharmacodynamic characterization of CDDO-Im following oral dosing has revealed that CDDO-Im is stable in the digestive system, readily absorbed and pharmacologically active in a number of organs including the liver [12], suggesting a potential role for CDDO-Im in treatment of liver metastases. Yet while synthetic triterpenoids have been shown to successfully inhibit orthotopic and ectopic tumors, lung metastasis and carcinogen induced primary tumor growth in multiple experimental models [8, 10, 13–16], their effect on metastatic liver disease is unknown.
Although liver metastasis is often fatal, experimental models have revealed that metastasis is an inefficient process whereby the majority of metastatic cells that arrive in the liver do not form large vascularized metastases [17–21]. Instead, many cells arriving in a secondary site will undergo apoptosis, and some may remain, often for extended periods of time, as solitary dormant cells alone or amidst proliferating metastases [17–19, 22–28]. The heterogeneous nature of the metastatic cell population makes it unlikely that all cells will respond uniformly to treatment. This situation has been demonstrated in experimental metastasis model in which doxorubicin significantly inhibited the growth of large actively growing metastases, but had no effect on the number and viability of solitary cells or their ability to form late occurring metastases following treatment [29, 30]. Thus, new therapies should be tested for their effects on both sub-populations of metastatic cells, those that are actively proliferating and those that are dormant, in order to better understand and use new treatments.
Here we assessed the effect of the synthetic triterpenoid CDDO-Im on the metastatic cell lines B16F1 (murine melanoma) and HT-29 (human colon carcinoma) in 2D culture and in experimental liver metastasis models in vivo. We found that CDDO-Im inhibited proliferation and induces cell death of HT-29 and B16F1 cells in culture, in a concentration dependent manner, similar to findings reported for other cell lines [7, 11, 14, 31–36]. We used standard histology to assess the effect on metastatic burden, and found that CDDO-Im inhibited growth of metastasis from both cell lines. We also used a novel MRI technique to simultaneously quantify the effect of CDDO-Im on both actively proliferating B16F1 metastases as well as solitary, dormant cells in whole livers. This approach confirmed the results from standard histology, showing that CDDO-Im treatment significantly decreased metastatic tumor burden, size and volume, and further showed that treatment had no effect on the numbers of dormant cells present in the livers. These results suggest that while additional treatment strategies are likely required to kill solitary dormant metastatic cells, CDDO-Im may have a role to play in the treatment of actively proliferating liver metastases from multiple tumor types.
Materials and methods
Cell culture and MPIO labeling
Cell culture and MPIO labeling were performed as described previously [19, 24, 37]. Briefly, B16F1 (ATCC CRL-6323) murine melanoma and HT-29 (ATCC HTB-38) human colon carcinoma cells (both from the American Type Culture Collection, Manassas, VA) were maintained in alpha-minimal essential media (α-MEM) (Invitrogen, Burlington, Canada) containing 10% fetal bovine serum (FBS) (Sigma, Mississauga, Canada) at 37°C and 5% CO2. As recommended by ATCC, cell morphology, rate of growth and mycoplasma testing were routinely performed. B16F1 cells were labeled with micrometer-sized iron oxide nanoparticles (MPIO) in a T75 tissue culture flask using media with FBS when 70–90% confluent. MPIO beads (312.5 μl, 0.9 μm diameter, 63% magnetite, labeled with Dragon Green, catalog #MC05F; Bangs Laboratories Inc, Fishers, IN, USA) were then added to 10 ml of media with FBS/flask and incubated for 24 h, as described [26, 30]. Cells were washed thoroughly with serum free α-MEM then centrifuged and resuspended in serum free α-MEM at the appropriate concentration for injection.
CDDO-Im treatment, imaging of 2D culture and flow cytometry
Details regarding the synthesis of a number of synthetic triterpenoids, including CDDO-Im (1-[2-cyano-3-,12-dioxooleana-1,9(11)-dien-28-oyl]imidazole), has been published previously [13, 38, 39]. Stock vials of CDDO-Im were kept frozen in DMSO and diluted to the appropriate concentration in cell culture media as required. To determine the effect of CDDO-Im on HT-29 and B16F1 cells in 2D cell culture, cells were seeded in a 6 or 24 well tissue culture plates. 48 h following seeding of cells, CDDO-Im in DMSO (or DMSO alone) was diluted with alpha-MEM plus 10% FBS to the appropriate concentration and added to each well. As triterpenoids exhibit time and concentration dependent effects on cell cycle progression, cell survival and morphology, cell populations were monitored longitudinally. Images were acquired from each well, using a Zeiss Axiovert 200 incubated (CO2, humidity and temperature control) inverted microsope system, every 24–48 h following treatment until day 9. Three images per well (3–4 wells per concentration) were analyzed in order to determine the number of cells per field of view (10×).
Analysis of cellular cleaved Caspase-3 was performed 48 h after exposure to CDDO-Im for both cell lines. Cells were trypsinized, washed and harvested into single-cell suspensions in ice-cold flow buffer (phosphate-buffered saline [PBS] + 2% FBS). An unconjugated anticaspase-3 antibody used was to bind cleaved Caspase-3 (Asp175—Rabbit mAb) (Cell Signaling Technologies, Boston, MA), following the manufacturer’s protocol. The secondary antibody used was a phycoerythrin (PE) conjugated anti-rabbit antibody (R&D Systems, Minneapolis, MN). Non-specific binding was accounted for using the secondary antibody alone. Samples were incubated sequentially with primary and secondary antibodies and re-suspended in cold flow buffer prior to analysis on an EPICS XL-MCL flow cytometer (Beckman Coulter, Mississauga, ON).
Experimental metastasis assay and in vivo CDDO-Im treatment
Female 6–10 weeks old C57BL/6 (for the B16F1 cells) (Harlan, Indianapolis, IN) or immunocompromised NIH III mice (for the HT-29 cells) (Charles River, Wilmington, MA) were cared for in accordance with the Canadian Council on Animal Care, under a protocol approved by the University of Western Ontario Council on Animal Care. For experimental liver metastasis assays, mice were anesthetized with an i.p. injection of xylazine/ketamine (2.6 mg ketamine and 0.13 mg xylazine per 20 g body mass). Anesthetized mice received mesenteric injections of 100 μl of B16F1 (3 × 105 cells) or 200 μl of HT-29 (2 × 106 cells) cells suspended in alpha-MEM to target cells directly to liver as described previously [19]. Mice were then treated with CDDO-Im (800 mg/kg diet) mixed in a powdered diet (NIH-31 modified mouse diet, Harlan). NIH III mice were treated with CDDO-Im 5 days a week while C57BL/6 mice were treated continuously for the duration of the experiment. Treatment schedules differed between mouse strains due to the 5–8 fold difference in length of the experiments. Controlled quantities of diet with CDDO-Im or vehicle control (ethanol and neobee oil) were replaced every 48 h for NIH III mice and every 72 h for C57Bl/6 mice, and consumption was approximately 4.5 and 3.5 g per day per mouse, respectively. Treatment via diet commenced immediately following surgery, however mice did not begin to consume significant amounts of food until at least 24 h following surgery. Weight of all mice was recorded prior to cell injection, throughout experiments and at the time of sacrifice. At endpoint (56 days for HT-29 and 7–11 days for B16F1) mice were sacrificed, livers were removed and fixed in formalin for at least 48 h prior to sectioning for histology or imaging by MRI.
Tumor burden, volume and solitary cell quantification and statistics
Metastatic tumor burden from histological sections was determined by quantifying the area of tumor and normal tissue from at least 5 random hematoxylin and eosin (H&E) stained sections of the liver as described previously [29]. Liver tumor volume (apparent as signal hyperintensity; i.e., dark regions) and MPIO labeled cells/signal void area (signal hypointensity; i.e., white regions) were also quantified in some experiments using a novel MRI technique we recently developed [30]. Briefly, at endpoint livers were removed and fixed in 10% neutral buffered formalin for at least 48 h. Intact livers were then scanned using a clinical 3T GE EXCITE whole body MR system with customized animal gradient insert coil and radiofrequency (RF) coil. Tumor volume and signal void area were quantified using a semi-automated procedure in which a fixed pixel intensity was manually defined and subsequent volume and area measurements were automated using VgStudio (Dresden, Germany) or Image J (NIH, Bethesda, MD) software, respectively.
All statistics were performed with GraphPad Prism software (La Jolla, CA). T-tests were performed to determine if significant differences existed in observed means of tumor burden, tumor volume and solitary cell signal void area. All statistics are reported as mean value ± standard error.
Results
CDDO-Im inhibited growth, altered morphology and induced cell death in B16F1 and HT-29 cells in vitro
To determine the effects of CDDO-Im on B16F1 and HT-29 cells in 2D cell culture, cells were treated with CDDO-Im or vehicle control as described above. Dose dependent and multifunctional effects of CDDO-Im were observed in the response of both B16F1 (Fig. 1a–c) and HT-29 cells (Fig. 1d–f) to treatment. Longitudinal observation of cells following exposure to CDDO-Im revealed readily apparent changes in morphology as well as growth arrest at concentrations between 100 and 200 nM. As seen in Fig. 1a and d, cell morphology and growth arrest were evident by 48 h following CDDO-Im treatment. At higher concentrations (greater than 200–400 nM), CDDO-Im induced cell death (cell shrinkage, rounding, detachment and apoptotic bodies observed) in both B16F1 and HT-29 cells (Fig. 1a, d). Cell death was verified by trypan blue staining (data not shown). The ability of CDDO-Im to cause cell death by apoptosis was confirmed by flow cytometry analysis of cellular cleaved Caspase-3. 48 h post treatment an increased percentage of cleaved Caspase-3 positive cells was observed in the 200 nM (B16F1) and 400 nM (B16F1 and HT29) treatment groups when compared to the no treatment groups (Fig. 1c, f). These results are consistent with results depicted in Fig. 1b and e, indicating growth inhibition at lower doses (200 nM—HT-29) and apoptosis at higher doses.
CDDO-Im inhibited growth and induced apoptosis in B16F1 and HT-29 cells in vitro. Nanomolar concentrations of CDDO-Im arrested growth or induced apoptosis in both B16F1 (a–c) and HT-29 (d–f) cells. 200 nM CDDO-Im was sufficient to inhibit proliferation and alter cell morphology (arrows) while at 300–400 nM apoptosis was induced (b, c, e, f). Flow cytometry to detect cleaved caspase-3 positive cells was used to confirm apoptosis in both B16F1 (c) and HT-29 (f) cells
CDDO-Im inhibited in vivo growth of HT-29 liver metastases
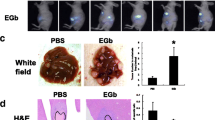
To determine the effect of CDDO-Im treatment on liver metastases from HT-29 human colon carcinoma cells, we used an experimental liver metastasis model. NIH III mice were first injected with 2 × 106 HT-29 cells via the mesenteric vein to target liver and then treated with CDDO-Im (800 mg/kg diet) or vehicle control commencing immediately following cell injections and continued for 56 days. At endpoint livers were removed, weighed and fixed for histological analysis. CDDO-Im treatment resulted in a decrease in the size of metastases (white areas on liver surface) that were apparent in surface images of whole intact livers (Fig. 2a, b—arrows). Subsequent quantification of tumor burden from random histological samples (Fig. 2a, b—right panels) showed that CDDO-Im treatment significantly decreased metastatic burden by more than 70% (t-test, P < 0.05, Control n = 10, 31.9 ± 9.4%, Treated n = 8, 8.5 ± 3.5%) (Fig. 2c). No significant difference in total mouse weight was observed at endpoint in this or any subsequent in vivo experiments (P > 0.05).
CDDO-Im inhibited HT-29 liver metastasis growth in vivo. 2 × 106 HT-29 cells were delivered to the liver via mesenteric vein injection. CDDO-Im treatment, or vehicle control, via diet (800 mg/kg diet) immediately following cell injection and continued 5 days a week until endpoint. Metastasis burden was determined histologically. Livers were removed at day 56, fixed, sectioned and stained by H&E. At least 5 liver sections per mouse were used to determine liver metastasis burden. CDDO-Im treatment significantly decreased (t-test, P < 0.05, Control n = 10, 31.9 ± 9.4%, Treated n = 8, 8.5 ± 3.5%)
CDDO-Im inhibited growth of B16F1 liver metastases but did not decrease the number of solitary cells
To determine the effect of CDDO-Im treatment on liver metastases from B16F1 mouse melanoma cells, C57BL/6 mice were injected with 3 × 105 B16F1 cells via the mesenteric vein to target liver and treated with CDDO-Im (800 mg/kg diet) or vehicle control. Treatment commenced immediately following surgery and lasted for the duration of the experiment. At endpoint livers were removed, fixed and scanned using a 3T clinical MR scanner as described above. As apparent in images of whole livers (Fig. 3a, b), CDDO-Im treatment resulted in an observable decrease in the size of metastases apparent on the surface of the liver. Inhibition of metastasis growth was quantified by analysis of MR images in order to determine tumor burden (tumor volume/total volume). Liver tumor burden in CDDO-Im treated mice was found to be decreased by 50% compared to vehicle control treated mice (t-test, P < 0.05, Control n = 10, 30.5 ± 3.9%, Treated, n = 10, 14.7 ± 3.0%) (Fig. 3c).
CDDO-Im inhibited B16F1 liver metastasis growth in vivo. B16F1 metastases are apparent on the surface of whole livers as black areas (a, b). Metastases on the surface (white arrows) of whole livers appear to be smaller in CDDO-Im treated (b) than vehicle control (a) groups. Metastasis burden was determined by MRI. Analysis of MR images of whole livers (100 × 100 × 200 μm) revealed that CDDO-Im treatment significantly reduced B16F1 liver metastases burden (c) (t-test, P < 0.05, Control n = 10, 30.5 ± 3.9%, Treated n = 10, 14.7 ± 3.0%)
In order to assess the effect of CDDO-Im treatment on the majority of the metastatic cell population, including both macroscopic metastases as well as solitary cells, we used a recently developed MRI technique capable of quantifying both tumor volume and the number of MPIO labeled solitary cells (signal void area) in the same liver. 3 × 105 B16F1 cells labeled with MPIO and injected via the mesenteric vein. Livers were recovered at day 11 and MR images were subsequently acquired by a single 6 min scan of whole livers described above. Images were then analyzed to determine tumor volume (image hyperintensity volume) and signal void area from MPIO labeled cells (image hypointensity) as recently described (30). A reduction in size of B16F1 metastases was once again apparent in examination of the surface of whole livers (Fig. 4a). This decrease in metastasis size was also apparent in 2D MR images as the areas of hyperintensity, due to B16F1 metastases, were decreased in images of CDDO-Im treated mice (Fig. 4b—arrows). 3D images of whole intact livers, in which metastases are pseudocolored green, were rendered from 2D images (Fig. 4c). MR images of the entire liver were used in order to quantify total metastatic tumor volume and signal void area (i.e., number of MPIO labeled cells). CDDO-Im treatment resulted in a more than 60% reduction in tumor volume quantified from the MR images of the entire liver (t-test, P < 0.05, Control n = 6, 4577 ± 1001 mm3, Treated, n = 5, 1640 ± 498 mm3) (Fig. 5a). Despite the significant reduction in metastatic tumor volume, no significant change in the number of solitary dormant cells (signal void area) was observed (t-test, P = 0.9, Control = 747 ± 122 mm2, Treated = 770 ± 105 mm2) (Fig. 5b).
CDDO-Im inhibited B16F1 liver metastasis growth. The reduction in metastasis size is readily apparent from surface image of whole livers (a). The reduction is also apparent as smaller areas of hyperintensity (arrows) in 2D MR images (b) and in green pseudocolored tumor on the surface of rendered 3D images of whole livers (c)
CDDO-Im inhibited B16F1 liver metastasis growth but did not affect the number of solitary dormant cells (signal void area) in vivo. Tumor volume, quantified from signal hyperintensity in MR images, was significantly reduced in CDDO-Im treated mice (t-test, P < 0.05, Control n = 6, Treated n = 5). No significant difference was observed in signal void area (MPIO labeled cells) in liver from CDDO-Im treated versus vehicle control mice (t-test, P = 0.9)
Discussion
While members of the synthetic triterpenoid family have been shown to inhibit primary tumor growth and subsequent spontaneous lung metastasis [8, 10, 13, 16], the effect of CDDO-Im on metastasis growth of liver metastasis was unknown. Because CDDO-Im can accumulate in liver tissue following oral administration, here we assessed the ability of this compound to inhibit liver metastasis. We showed that CDDO-Im arrested proliferation of and/or induced apoptosis in a concentration dependent manner in metastatic cell lines of two tumor types, B16F1 melanoma and HT-29 colon carcinoma, consistent with results described previously in other cancer cell lines [7, 11, 14, 31–36]. Oral treatment of mice injected with cancer cells via the mesenteric vein (experimental metastasis assays) with CDDO-Im (800 mg/kg diet) was found to significantly inhibit liver metastasis growth from both cell lines in vivo by 50–70%, as detected both as surface metastases and by histology. We then used a novel MR procedure, able to simultaneously quantify total whole-liver metastatic volume from growing B16F1 metastases, as well as the population of non-dividing cancer cells that persist in the liver. This approach confirmed the results from histology, that CDDO-Im significantly reduced metastatic burden by ~60%, and further showed that the population of solitary B16F1 cells present in the same livers was not reduced.
Liver is the second most common organ in which metastatic disease is found to occur and is significant cause of cancer related deaths [1]. Even after initial treatment that leaves no clinically apparent metastases, recurrence and limited 5 year survival remain significant problems [4–6]. As such, new treatment strategies are needed in order to more successfully treat metastatic liver disease. The ability to be dosed orally, to induce apoptosis or arrest growth in a variety of cancer cell lines, and the relatively high concentration that can accumulate in liver make CDDO-Im a logical candidate for study as a treatment of liver metastasis. Additionally, as CDDO-Im is capable of both inhibiting proliferation and inducing apoptosis in cancer cells (depending on concentration) it has a useful therapeutic property in that even at doses that are not lethal to cancer cells, CDDO-Im may inhibit tumor growth by causing growth arrest or differentiation.
The mechanisms by which synthetic triterpenoids exert their diverse effects remain incompletely understood [7]. This complexity is amplified by the cellular responses that differ based on concentration (not simply increasing the same effect) and on cell type. Synthetic triterpenoids may have an inhibitory affect on the growth of tumors via a number of mechanisms that include, inhibition of angiogenesis [40], pro-apoptotic signaling via both intrinsic and extrinsic (increase DR4 and DR5 expression/decrease activity of the anti-apoptotic protein cFLIP) pathways [9, 15] and inhibition of HER2 tyrosine kinase activity [16]. In addition to the ability to inhibit tumor growth, synthetic triterpenoids have been shown to have a cytoprotective effect on non-cancer cells in experiments in which orally administered treatment reduced aflatoxin induced liver metastases [41] and vinyl carbamate induced lung tumors [42]. Thus, synthetic triterpenoids have multifunctional treatment potential as they reduce carcinogenesis [41], inhibit growth [8, 16] and induce apoptosis in cancer cells [7, 11]. Such multi-functional treatment strategies will likely be necessary to target the entire metastatic cells population.
It has been established previously that treatment with the cytotoxic chemotherapy doxorubicin is capable of reducing liver tumor burden and volume, but does not affect the number of dormant metastatic cells or their ability to proliferate and form late developing metastases [29, 30]. Here we show that CDDO-Im significantly inhibits metastatic tumor burden (HT-29—by histology) and volume (B16F1—by MRI) by 50–80%, but does not decrease the number of solitary cells. However, as evident in the 2D culture treatment of HT-29 and B16F1 cells, at concentrations insufficient to induce apoptosis, cellular proliferation is inhibited. Thus, while the number of solitary dormant metastatic cells was not affected by CDDO-Im treatment, it is possible that continued treatment would maintain some of these cells in a dormant state. Indeed, the synthetic triterpenoid CDDO-Me has recently been shown to inhibit growth and induce apoptosis via Akt and mTOR [10], the later having recently been shown to promote survival of dormant cells in vivo [43]. It is worth noting that while the size of metastases appear smaller in surface images of livers from CDDO-Im treated mice, consistent with the reduction in histologically determined metastatic burden in the liver in both models, a significant number of metastases are still visible (Figs. 2 and 3), suggesting the slowing of growth, not the elimination of metastatic cells. Further studies with models of metastatic dormancy are required to more precisely address any effect of CDDO-Im treatment on individual solitary cells.
Here we have shown that the synthetic triterpenoid CDDO-Im significantly inhibits growth of metastases, with no effect on the number of solitary cells, in experimental metastasis models in which cells are injected directly to target them to the liver. It may be that in order to further reduce the number of macroscopic metastases and/or solitary dormant metastatic cells, other synthetic triterpenoids or combination treatment may be required. Indeed, prevention and treatment effects of synthetic triterpenoids have both been found to be synergistic with the rexinoid LG100268 [44, 45]. Limitations and further characterization required for the MRI technique used here have been discussed previously [30]. In particular, while the ultimate fate and rate of iron clearance remain to be fully understood, greater than two thirds of the initial signal voids (iron loaded B16F1 single cell population) are no longer apparent at endpoint, consistent with rate of loss of cells reported in the liver by previous optical and MRI quantification experiments [19, 30]. Our work presented here with CDDO-Im suggests that synthetic triterpenoids are able to inhibit liver metastasis growth and that they deserve further evaluation and development as a treatment for metastatic disease.
References
Pickren J, Tsukada Y, Lane W (1982) Liver metastasis. In: Weiss L, Gilbert L (eds) Liver metastasis. G.K. Hall Medical Publishers, Boston, MA
Lee SM, Betticher DC, Thatcher N (1995) Melanoma:chemotherapy. Br Med Bull 51:609–630
Fong Y, Kemeny N, Paty P et al (1996) Treatment of colorectal cancer: hepatic metastasis. Semin Surg Oncol 12:219–252
Lochan R, White SA, Manas DM (2007) Liver resection for colorectal liver metastasis. Surg Oncol 16:33–45
Rivoire M, Kodjikian L, Baldo S et al (2005) Treatment of liver metastases from uveal melanoma. Ann Surg Oncol 12:422–428
Sharma S, Camci C, Jabbour N (2008) Management of hepatic metastasis from colorectal cancers: an update. J Hepatobiliary Pancreat Surg 15:570–580
Liby KT, Yore MM, Sporn MB (2007) Triterpenoids and rexinoids as multifunctional agents for the prevention and treatment of cancer. Nat Rev Cancer 7:357–369
Ling X, Konopleva M, Zeng Z et al (2007) The novel triterpenoid c-28 methyl ester of 2-cyano-3,12-dioxoolen-1,9-dien-28-oic acid inhibits metastatic murine breast tumor growth through inactivation of stat3 signaling. Cancer Res 67:4210–4218
Hyer ML, Shi R, Krajewska M et al (2008) Apoptotic activity and mechanism of 2-cyano-3,12-dioxoolean-1,9-dien-28-oic-acid and related synthetic triterpenoids in prostate cancer. Cancer Res 68:2927–2933
Deeb D, Gao X, Jiang H et al (2009) Oleanane triterpenoid cddo-me inhibits growth and induces apoptosis in prostate cancer cells by independently targeting pro-survival akt and mtor. Prostate 69:851–860
Suh N, Wang Y, Honda T et al (1999) A novel synthetic oleanane triterpenoid, 2-cyano-3,12-dioxoolean-1,9-dien-28-oic acid, with potent differentiating, antiproliferative, and anti-inflammatory activity. Cancer Res 59:336–341
Yates MS, Tauchi M, Katsuoka F et al (2007) Pharmacodynamic characterization of chemopreventive triterpenoids as exceptionally potent inducers of nrf2-regulated genes. Mol Cancer Ther 6:154–162
Place AE, Suh N, Williams CR et al (2003) The novel synthetic triterpenoid, cddo-imidazolide, inhibits inflammatory response and tumor growth in vivo. Clin Cancer Res 9:2798–2806
Lapillonne H, Konopleva M, Tsao T et al (2003) Activation of peroxisome proliferator-activated receptor gamma by a novel synthetic triterpenoid 2-cyano-3,12-dioxooleana-1,9-dien-28-oic acid induces growth arrest and apoptosis in breast cancer cells. Cancer Res 63:5926–5939
Hyer ML, Croxton R, Krajewska M et al (2005) Synthetic triterpenoids cooperate with tumor necrosis factor-related apoptosis-inducing ligand to induce apoptosis of breast cancer cells. Cancer Res 65:4799–4808
Konopleva M, Zhang W, Shi Y et al (2006) Synthetic triterpenoid 2-cyano-3,12-dioxooleana-1,9-dien-28-oic acid induces growth arrest in her2-overexpressing breast cancer cells. Mol Cancer Ther 5:317–328
Logan PT, Fernandes BF, Di Cesare S et al (2008) Single-cell tumor dormancy model of uveal melanoma. Clin Exp Metastasis 25:509–516
Naumov GN, MacDonald IC, Weinmeister PM et al (2002) Persistence of solitary mammary carcinoma cells in a secondary site: a possible contributor to dormancy. Cancer Res 62:2162–2168
Luzzi KJ, MacDonald IC, Schmidt EE et al (1998) Multistep nature of metastatic inefficiency: dormancy of solitary cells after successful extravasation and limited survival of early micrometastases. Am J Pathol 153:865–873
Chambers AF, MacDonald IC, Schmidt EE et al (2000) Clinical targets for anti-metastasis therapy. Adv Cancer Res 79:91–121
Chambers AF, Groom AC, MacDonald IC (2002) Dissemination and growth of cancer cells in metastatic sites. Nat Rev Cancer 2:563–572
Aguirre-Ghiso JA (2007) Models, mechanisms and clinical evidence for cancer dormancy. Nat Rev Cancer 7:834–846
Suzuki M, Mose ES, Montel V, Tarin D (2006) Dormant cancer cells retrieved from metastasis-free organs regain tumorigenic and metastatic potency. Am J Pathol 169:673–681
Heyn C, Ronald JA, Mackenzie LT et al (2006) In vivo magnetic resonance imaging of single cells in mouse brain with optical validation. Magn Reson Med 55:23–29
Shapiro EM, Sharer K, Skrtic S et al (2006) In vivo detection of single cells by mri. Magn Reson Med 55:242–249
Heyn C, Ronald JA, Ramadan SS et al (2006) In vivo mri of cancer cell fate at the single-cell level in a mouse model of breast cancer metastasis to the brain. Magn Reson Med 56:1001–1010
Goodison S, Kawai K, Hihara J (2003) Prolonged dormancy and site-specific growth potential of cancer cells spontaneously disseminated from nonmetastatic breast tumors as revealed by labeling with green fluorescent protein. Clin Cancer Res 9:3808–3814
Cameron MD, Schmidt EE, Kerkvliet N et al (2000) Temporal progression of metastasis in lung: cell survival, dormancy, and location dependence of metastatic inefficiency. Cancer Res 60:2541–2546
Naumov GN, Townson JL, MacDonald IC et al (2003) Ineffectiveness of doxorubicin treatment on solitary dormant mammary carcinoma cells or late-developing metastases. Breast Cancer Res Treat 82:199–206
Townson J, Ramadan S, Simedrea C et al (2009) Three-dimensional imaging and quantification of both solitary cells and metastases in whole mouse liver by magnetic resonance imaging. Cancer Res 69:8326–8331
Chintharlapalli S, Papineni S, Konopleva M et al (2005) 2-cyano-3,12-dioxoolean-1,9-dien-28-oic acid and related compounds inhibit growth of colon cancer cells through peroxisome proliferator-activated receptor gamma-dependent and -independent pathways. Mol Pharmacol 68:119–128
Ikeda T, Sporn M, Honda T et al (2003) The novel triterpenoid cddo and its derivatives induce apoptosis by disruption of intracellular redox balance. Cancer Res 63:5551–5558
Kim KB, Lotan R, Yue P et al (2002) Identification of a novel synthetic triterpenoid, methyl-2-cyano-3,12-dioxooleana-1,9-dien-28-oate, that potently induces caspase-mediated apoptosis in human lung cancer cells. Mol Cancer Ther 1:177–184
Stadheim TA, Suh N, Ganju N (2002) The novel triterpenoid 2-cyano-3,12-dioxooleana-1,9-dien-28-oic acid (cddo) potently enhances apoptosis induced by tumor necrosis factor in human leukemia cells. J Biol Chem 277:16448–16455
Konopleva M, Tsao T, Ruvolo P et al (2002) Novel triterpenoid cddo-me is a potent inducer of apoptosis and differentiation in acute myelogenous leukemia. Blood 99:326–335
Gao X, Deeb D, Hao J et al (2010) Synthetic triterpenoids inhibit growth, induce apoptosis and suppress pro-survival Akt, mTOR and NF-{kappa}B signaling proteins in colorectal cancer cells. Anticancer Res 30:785–792
Graham KC, Wirtzfeld LA, MacKenzie LT et al (2005) Three-dimensional high-frequency ultrasound imaging for longitudinal evaluation of liver metastases in preclinical models. Cancer Res 65:5231–5237
Liby K, Voong N, Williams CR et al (2006) The synthetic triterpenoid cddo-imidazolide suppresses stat phosphorylation and induces apoptosis in myeloma and lung cancer cells. Clin Cancer Res 12:4288–4293
Honda T, Honda Y, Favaloro FGJ et al (2002) A novel dicyanotriterpenoid, 2-cyano-3,12-dioxooleana-1,9(11)-dien-28-onitrile, active at picomolar concentrations for inhibition of nitric oxide production. Bioorg Med Chem Lett 12:1027–1030
Vannini N, Lorusso G, Cammarota R et al (2007) The synthetic oleanane triterpenoid, cddo-methyl ester, is a potent antiangiogenic agent. Mol Cancer Ther 6:3139–3146
Yates MS, Kwak M, Egner PA et al (2006) Potent protection against aflatoxin-induced tumorigenesis through induction of nrf2-regulated pathways by the triterpenoid 1-[2-cyano-3-,12-dioxooleana-1,9(11)-dien-28-oyl]imidazole. Cancer Res 66:2488–2494
Liby K, Royce DB, Williams CR et al (2007) The synthetic triterpenoids cddo-methyl ester and cddo-ethyl amide prevent lung cancer induced by vinyl carbamate in a/j mice. Cancer Res 67:2414–2419
Schewe DM, Aguirre-Ghiso JA (2008) Atf6alpha-rheb-mtor signaling promotes survival of dormant tumor cells in vivo. Proc Natl Acad Sci USA 105:10519–10524
Liby K, Risingsong R, Royce DB et al (2008) Prevention and treatment of experimental estrogen receptor-negative mammary carcinogenesis by the synthetic triterpenoid cddo-methyl ester and the rexinoid lg100268. Clin Cancer Res 14:4556–4563
Liby K, Black CC, Royce DB et al (2008) The rexinoid lg100268 and the synthetic triterpenoid cddo-methyl amide are more potent than erlotinib for prevention of mouse lung carcinogenesis. Mol Cancer Ther 7:1251–1257
Acknowledgments
The authors thank Carl Postenka and Carmen Simedrea for their technical assistance. Supported by grant #42511 from the Canadian Institutes of Health Research (to AFC and ICM). JLT was supported by a doctoral research award from the Canadian Institutes of Health Research. AFC is Canada Research Chair in Oncology, and receives salary support from the Canada Research Chairs Program
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Townson, J.L., MacDonald, I.C., Liby, K.T. et al. The synthetic triterpenoid CDDO-Imidazolide suppresses experimental liver metastasis. Clin Exp Metastasis 28, 309–317 (2011). https://doi.org/10.1007/s10585-011-9374-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-011-9374-z