Abstract
The greatest potential for improvement of outcome for patients with Cutaneous Malignant Melanoma lies in the prevention of systemic metastasis. Despite extensive investigation, current prognostic indicators either alone or in combination, although related to melanoma progression, are not sufficient to accurately predict the pattern of progression and outcome for any individual patient. Metastasis related death has been recorded in patients initially diagnosed with early stage tumour as well as in patients many years after initial tumour removal. The trouble finding a predictable pattern in the puzzle of melanoma progression may be linked to the fact that most of the material studied for prognosis is either, cutaneous primaries or metastatic deposits, rather than the melanoma cells in the circulatory system which are responsible for disease progression. In this review article we discuss the potential use of circulating tumour cell (CTC) detection and quantification for identifying patients at risk of metastatic deposits. We also discuss current therapies for the treatment of metastatic melanoma and analyse how CTCs may be used to evaluate the effectiveness of current therapies and to pinpoint patients who require further treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cutaneous Malignant Melanoma (CMM) is the most severe of all skin cancers, accounting for 80% of skin cancer related deaths [1]. CMM is the most common cancer in Australia for persons aged 15–39 and the most common cause of cancer related death in young Australian males. In 2006, in Australia, there were 10,000 new cases and incidence rates continue to rise [2]. In the United States, CMM is an emerging problem as the incidence has increased more than that of any other cancer over the past two decades. More specifically, in 2005 the American Cancer Society recorded approximately 59,580 new cases and over 7,910 new deaths [3, 4].
CMM is often aggressive, unpredictable and difficult to treat. Mortality rates continue to remain high, due mainly to progression of multiple drug resistant metastases for which the cure rate is currently less than 20% [5, 6]. Much effort has been put into identifying prognostic factors that correlate with clinical outcomes to enable The American Joint Committee on Cancer (AJCC) to devise an accurate TNM clinical staging system. Histopathologists determine the tumour thickness (T) component of this system, which most accurately characterises the CMM once excised [7]. Additional information, including ulceration, mitotic rate, clinical or surgical detection of lymph node (N) or distant metastasis (M), is utilised to stage and thus prognosticate and so treat appropriately [8]. Other details including sex, age, LDH levels and anatomical location of the melanoma and metastases, residual disease following initial resection and genomic changes in the primary tumour have been linked with prognostic changes but so far have little material influence on treatment plans [9–11].
Despite extensive investigation the above factors either alone or in combination, although related to melanoma progression, are not sufficient to accurately predict the pattern of progression and outcome for any individual patient.
Metastasis related death has been recorded as long as 35 years following initial removal of the primary tumour (uveal melanoma) [12]. The trouble finding a predictable pattern in the puzzle of melanoma progression may possibly be linked to the fact that most of the material studied, was based either on cutaneous primaries or metastatic deposits, rather than the quantity and phenotype of melanoma cells in the circulatory system. Circulating tumour cell (CTC) detection and quantification may hold the key to identifying patients at risk of metastatic deposits. Analysing CTCs has the potential not only to pinpoint patients who may require further treatment but evaluate the effectiveness of current therapies.
Current methods of treatment of melanoma
CMM, generally referred to as melanoma, arises from melanocytes, cells of neural crest origin which migrate into the skin [13]. CMM can grow laterally in the epidermis or vertically through to the dermis ultimately leading to vascular invasion and metastatic disease. Stage I or II melanoma can be cured by wide surgical excision resulting in a 50–100% 5 year survival rate, decreasing with increasing tumour thickness [14]. Following surgery, patients are examined and depending on the histological features of the primary they may have further investigations including sentinel node biopsy, CT or PET for staging purposes [14]. Patients with tumours under 2 mm do not require any investigations for staging as at this level imaging is not specific enough to identify metastases accurately, even though up to 44% of these people eventually die from metastatic disease despite removal of the primary tumour [14, 15]. Patients diagnosed with lymph node or systemic metastases have a 5 year survival rate that decreases to between 5 and 60% [8, 16]. So far however, there is no specific gene mutation or gene expression profile within the tumour cells in the primary that can accurately predict the aggressiveness of the tumour [17].
There is no doubt that a cancer’s mortality rate is determined by the speed of metastasis [18, 19]. In all tumours, millions of tumour cells (reported to be around 4 × 106 per gram of tumour over a 24 h period [20, 21]) invade the venous and/or lymphatic circulation very early in tumour development and result in disease progression [22–25]. Moreover a meta-analysis has shown that the number of CTCs in a patient’s peripheral blood increases with increasing stages of melanoma [26]. Despite these recent advances there is still much to discover, including the phenotype of the melanoma CTCs that are able to stay dormant in the circulation for many years evading the immune system, and the events leading to their activation, seeding and proliferation in distant body organs.
Once the tumour metastasises, and thus needs systemic treatment, there is a considerable lack of available information and knowledge about how to measure the current disease load and treatment responses. The AJCC currently uses metastatic site and serum LDH to put a number to the 5 year survival rate for prognostication purposes [8]. Treatment is measured in terms of the Objective Response Rate (ORR) defined as the percentage of patients with identifiable tumour shrinkage plus the percentage of patients with complete tumour disappearance following treatment [27].
Analysis of quantity and phenotype of CTCs could have a place in monitoring current treatment responses. Additionally, development of markers that can reliably detect CTCs could potentially identify which tumours may respond to current treatments and highlight possible avenues for future treatment development. Current treatments are purely palliative as they have a response rate under 20% and do not have a significant survival benefit [28, 29]. CTCs as a marker could be used to determine whether the gold standard treatment, DTIC (Dacarbazine), is effective in treating disease as to date there is no placebo controlled trial to confirm its efficacy [28, 29]. Interleukin 2 (IL2) immunotherapy, a treatment developed in the 1990’s, has shown an overall response rate of 16% and these responders had a progression free period of over 5 years [28]. As IL2 is known to have severe side effects it is restricted to those with an excellent performance status and organ function, but ideally it would be desirable to be able to identify responders prior to treatment. There is a possibility that analysis of CTCs could provide a means of identifying responders thus ensuring the most beneficial use of this treatment.
Current therapies also include vaccine treatment with the aim of improving patient survival. Due to the heterogeneity of melanoma cells limited response rates of 12%, are observed, with a mean increased overall survival (OS) of 21.3 months, triple that of DTIC [28, 30]. Analysis of CTCs could provide information on the early response of patients to IL2 or vaccines. Moreover therapies could be developed to target the markers found in patient subsets.
B-RAF inhibitors are an exciting new mode of treatment targeting a mutation in B-RAF, an oncogene that promotes growth and angiogenesis when activated [31]. A selective inhibitor of active B-RAF, PLX4032 has been shown to have a 78% response rate in phase 1 trials, and phase 2 and 3 trials began in September 2009 and January 2010. Despite their initial success, reports indicate that there is rapid acquisition of drug resistance, resulting in a restriction of therapeutic response to an average 8 months [32–34]. There is therefore, an urgent need for early detection of resistance to kinase inhibitors. The method currently used to evaluate the response of this treatment is an overall Response Evaluation Criteria in Solid Tumours (RECIST) which uses current imaging methods to find measurable lesions and monitor size before and after treatment. While current methods can only measure macroscopic metastasis presence/shrinkage, CTCs have the potential to enhance this process by using tumour cell load to monitor microscopic response to treatment and early acquisition of drug resistance.
Knowledge to date on the process of melanoma cell metastasis—not as simple as originally thought
Understanding the processes that lead to metastasis is important for choosing significant molecular markers with which to characterise CTCs for future treatment strategies. The process of metastasis in melanoma requires loss of intracellular adhesion, dermal invasion, migration from the primary site, intravasation, intravascular survival, attachment to the vasculature of target tissues, migration into target organs, proliferation and angiogenesis at the target site [35]. Of particular relevance in this instance, is the process of tumour cell dissemination to seed metastasis.
Melanoma cells arise in the epidermis where initially, like melanocytes, they are tethered tightly to the surrounding melanoma cells, melanocytes and keratinocytes by a complex network of desmosomes, the adhesion molecules and gap junctions which allow transfer of melanin [22, 36, 37]. Once melanoma cells enter the invasive stage they progressively loose these adhesion surface interactions [38–40]. One of the key surface proteins, Epithelial Cadherin (E-Cadherin/CDH1), is bound via its cytoplasmic tail to α- catenin and β-catenin which links it directly to the actin cytoskeleton of the cell [41]. In fact the switching of E-Cadherin/CDH1 with Non-epithelial Cadherin (N-Cadherin/CDH2) indicates the initiation of an epithelial to mesenchymal transition for the cell (EMT) [37]. The EMT process, initially utilized by migrating cells during embryonic development, allows the cell to transform from a polarised epithelial cell to a contractile, motile mesenchymal cell and this process is triggered by secretion of growth factors from fibroblasts and macrophages (e.g. hepatocyte growth factor (HGF), and/or fibroblast growth factor (FGF)). This secretion induces intracellular transduction pathways (Transforming growth factor-β (TGF-β), Platelet Derived Growth Factor (PDGF), Wnt and Notch) which in turn activate transcription factors (Snail and E47) [37, 42]. This cascade leads to the aforementioned switching of E-Cadherin to N-Cadherin, allowing the cell to detach from the epithelium and migrate into the circulation leading to tumour progression [22, 43].
When melanoma cells detach from the primary tumour and enter the bloodstream or lymphatics they can do so actively or passively [18]. Passive entry occurs when cells are simply dislodged from the primary tumour due to increased blood flow and low CDH1 levels [18]. By contrast, active migration occurs in cells when NEDD9, a melanoma metastasis protein, forms a complex with DOCK-3, a guanine nucleotide exchange factor, to activate Rac, a GTPase which acts on actin assembly, actomysin contractility and microtubules [44]. The result of Rac activation is that the cell becomes mobile and elongated so that it can pass between adjacent cells [44]. The movement of these cells is assisted by matrix metalloproteinases that destroy integrins (cell receptors that mediate attachment to surrounding cells) and other extracellular materials [45].
The next step in the active migration process is the attraction of the tumour cells to lymph and/or blood vessels, a process mediated by ligand-receptor interactions between tumour cells and the stroma or endothelial cells. The tumour cells secrete Colony Stimulating Factor 1 (CSF-1) and growth factors such as Epithelial Growth Factor (EGF) which activate the formation and proliferation of tumour associated macrophages in the stroma. These macrophages secrete growth factors, cytokines, chemokines and enzymes that regulate tumour growth, angiogenesis, invasion and/or metastasis [22, 46].
Whether cells actively move toward and into surrounding blood vessels or whether this process is passive and coincidental may be significant. Expression of genes and formation of complexes required for active migration, may enhance survival of the cell once it is in the blood, thus increasing metastatic ability [18]. Markers showing expression of the above factors may therefore provide a better measurement of metastatic potential than sheer quantity of circulating cells.
In order to investigate circulating tumour cells in more detail, models have been developed in which a tumour and its vascular system are isolated and blood vessels are monitored so as to quantify and characterise the cells entering and exiting the tumour [18, 20, 47]. Notably, large volumes (3–4 × 106) of cells are shed every day, with few of these developing into clinically detectable metastases. Characterisation of these circulating cells in patients with advanced breast and prostate cancer indicates that they are predominantly apoptotic and necrotic and are unlikely to survive [48–51]. Furthermore, it is believed that the bulk of circulating cells are destroyed by sheer stress. Moreover circulating immune cells prevent all but the most proficient from producing secondary colonies [38, 52].
Nevertheless, some cells do survive for long periods of time in the vasculature, though not independently. Circulating tumour cells from solid tumours, unlike those from hematopoietic cancers [38, 53], are usually found in clumps, also known as circulating tumour microemboli, which are hidden beneath a “cloak” of platelets and leukocytes for enhanced survival [38, 54, 55]. Additional factors promoting melanoma cell survival in the vasculature are evasion of immune cells including natural killer (NK) cells whose primary role is to pinpoint and destroy cancer cells. The NKG2D ligand is usually expressed on the surface of infected or mutated calls and targets the cell for NK cell mediated destruction, however melanoma cells evade NK cell mediated destruction by intracellular retention of the ligand [56]. Furthermore melanoma cells do not express the oncostatic M receptor, which, when activated by IL-6 prevents cell division [5, 57].
It is not only the immune system in the blood that prevents metastasis but also receptiveness of the distant site and the genetic identity of the cell. The theory of ‘metastatic suppression’ states that despite early dissemination of CTCs to distal sites the gene expression profile of the melanoma cell may decrease metastatic potential without inhibiting primary tumour growth [58–61] (Fig. 1). Metastatic suppression may include inhibition of angiogenesis and increased immune surveillance. Holmgren et al. [52] showed that inhibition of angiogenesis indeed induced tumour dormancy in mice inoculated with Lewis lung carcinoma. Eyles et al. [59] showed that increased immune surveillance suppresses tumour progression in mice bred to develop uveal melanoma. Genes associated with metastatic suppression include Kail [58, 60] which promotes senescence and KISS1 [58, 62] which promotes dormancy of solitary cells. Other genes involved include SMAD7 [58], NM23-H1 [58, 60, 63, 64], GAS1 [65] and BRMS1 [66] which more generally decrease motility, invasiveness and metastasis of CTCs possibly due to identification by immune cells and failed angiogenesis at the distant site.
Models of metastasis. a Multistep model of metastasis. Circulating tumor cells arise from late stage, vessel-infiltrating cancers. In this model, it is hypothesised that circulating tumor cells either fail to enter target organs or undergo lysis or apoptosis. b Single tumor cells enter the circulation and start to spread much earlier than expected and form single-cell metastases. Such single-cell metastases remain dormant for long periods of time,] through metastatic suppression signals that reduce tumor cell proliferation. In the absence of metastatic suppression signals, metastases start to grow rapidly [61]
Given the evidence that melanoma cells disseminate early but may survive as CTCs and micrometastatic deposits not detectable by current staging methods, detection and quantification of CTCs could provide an avenue for detecting clinically occult melanoma progression. It is well established that a melanoma cell may invade the circulation and survive without clinical evidence of a primary tumour, and so, not infrequently a patient may have metastatic melanoma without a known cutaneous primary site [67, 68]. In fact, melanoma can still be lethal even when solid tumours cannot form due to the patient having an inbuilt inability for the tumours to form a vascular supply [4]. In this case the patient developed high levels of cells in the blood and CSF and eventually succumbed to renal failure from tumour lysis syndrome. Taken together with the fact that 0.4–43% of surgical excisions result in recurrences and up to 8% of melanoma in situ diagnoses result in recurrence, it is clear that circulating cells have a significant prognostic value and a more detailed investigation of the phenotype of actively metastasizing CTCs would have major implications for future clinical diagnoses and treatments [69–72].
Current knowledge of the phenotype of melanoma cells
The question remains then, how do we identify the few CTCs that are capable of surviving in the circulation and metastasising? The genetic factors needed to survive in the circulation must be different from those needed to proliferate in situ and also separate from those needed to establish a secondary tumour. Analysis of the cells surviving in the circulation for an extended period of time is required so that this phenotype and mechanisms of survival advantage can be identified. Analysis of the genes expressed in circulating cells compared to primary and secondary tumour cells is one way of highlighting some of the changes in phenotype and adaptability necessary for their survival. This approach has been supported by the observations that different genes are expressed in primary versus circulating cells from the same melanoma patient [23, 24]. It has also been shown that subpopulations of cells within a primary tumour resemble those found in metastatic deposits [73]. Alternately, there is no reason that any cancer cell cannot develop the ability to metastasise, even prior to clinically detectable tumour formation [74]. Therefore, there is a possibility that for heterogeneic tumours like melanoma, an unstable, genetically variant and invasive cell could survive in the circulation but not exist in any great quantity in the primary tumour [22, 75, 76]. Genetic changes that provide an edge in metastasis and proliferation at a secondary site may not be advantageous in the primary tumour and thus too rare to be identified in its population-averaged gene expression profile [22].
In the last few years a number of researchers have shown the existence of a subset of tumour initiating melanoma stem cells within the primary tumour that possess two important features. One of these features is the ability to self renew as well as differentiate into cancer progenitor cells. The other of these features is the innate resistance to chemotherapy and radiation [77–80]. These rare cancer cells are present early on (rather than immediately prior to metastasis as originally thought) and effectively manage the metastatic process when the detectable bulk of the tumour, visible by current techniques has been successfully treated [81] (Figs. 1 and 2). They evade therapy due to their stem cell properties of slow turnover and chemotherapy resistance, and upon reaching their destination would act as a seed for metastasis formation [82]. Melanoma stem cells have been identified in primary tissue and cell lines by expression of recently identified melanoma stem cell markers, namely, Multi-Drug Resistance gene product 1 (MDR-1) [83], CD20 [76, 78, 84, 85], CD44/CDH5/VE-Cadherin [85, 86], CD133 [78, 84, 85, 87], ABCB5 [83–85, 87], ABCC2 [83], ABCG2 [78, 87], human telomerase reverse transcriptase (hTERT) [83], nanog [83] and JARID1B [88]. However unlike normal stem cells, cancer stem cells may be termed stem-cell like since they have mutations in key signalling pathways that may lead to ‘phenotype instability’ enabling cancer cells to switch their phenotype in response to microenvironmental cues [89].
Linear melanoma progression or the melanoma stem-cell model. a Melanoma development is traditionally viewed as a gradual process in which normal mature melanocytes acquire mutations that lead from benign to dysplastic nevi progressing through radial growth phase (RGP) to vertical growth phase (VGP), leading to invasion of the circulation and metastasis. b The melanoma stem-cell model hypothesises that melanoma stem cells lead directly to the observed clinical outcomes without progressing through multiple stages [80]
Evidence of melanoma stem cells in the circulation of melanoma patients remains to be demonstrated but similar cells have been found in the bone marrow in breast cancer patients [89]. If identified in the blood, melanoma stem cells may be effective in pointing to residual melanoma in patients with complete regression according to the current radiological methods of monitoring treatment effectiveness.
Melanoma markers identified
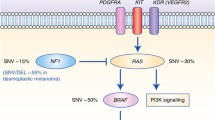
A large volume of studies have been dedicated to identification of markers with sufficient sensitivity and specificity to accurately predict melanoma progression. Even though many of these markers were identified from primary lesion tissues, these markers have been tested in a number of CTC studies [90–93]. Melanocytic and melanoma markers commonly used for CTC analysis include MAGE-A3 [70, 93–95], Mitf [93, 96], MART-1/Melan-A [70, 93–95], Tyr [26, 93, 95] and most recently ABCB5 [79, 84]. As a result of high throughput analyses of melanoma, several key progression pathways have been identified including epithelial-mesenchymal transition [97], but many of these are yet to be tested as informative for CTC analysis using either gene expression or whole cell detection techniques [17, 92, 98, 99]. Key amongst these pathways are: tyrosine kinase receptor (TKR) pathways (e.g. VEGFR, ERBB2 and the TGFβ receptor) [37], the Ras/Raf/MEK/ERK pathway [37], the PI3 K/Akt/PTEN/mTOR pathway [37], cell cycle regulation pathways (Rb/p53/p16INKA/p14ARF/HDM2/CXCR4/CDC42) [100], epigenetic gene expression regulation and DNA repair (DNA methylation, histone mitochondrial pathway and RNA interference) [101, 102], apoptotic pathways (e.g. death receptors: FAS, TRAILR, TNFR; mitochondrial pathway: Bcl2 family) [103], and epithelial to mesenchymal transition (CDH2, SPP1 and SPARC) [104]. Epithelial to mesenchymal transfer (EMT) has been thought to be the key to metastasis by giving malignant melanoma cells stem-like capabilities to migrate within and through tissues. In 2010 Roesch [88] added a further marker to this bank, JARID1B, which identifies slow cycling melanoma stem cells, first identified by Adams and Strasser in 2008 [105]. This marker is expressed on melanoma cells which are slow to divide and so also resistant to current treatments but was not linked with the classic EMT signature or any other stem cell markers suggested to date.
It is possible that these pathways may play a role in CTC survival, proliferation and intra- and extravasation. Furthermore, with continuing discovery of markers specific to melanoma stem cells and metastatic pathways, a marker may be found that reliably detects metastatic melanoma. This would be extremely useful especially in detecting amelanocytic melanoma where there are only small levels of melanocytic gene expression as well as in tracking down CTCs in patients with no primary melanoma.
In summary, conventional staging and treatment of melanoma is not particularly precise and effective. This has the potential to be dramatically improved by investigating molecular and cellular markers/pathways which lead to tumour progression. Such markers could include proteinases (proteinases of the plasmin system, serine proteinases and matrix metalloproteinases) which degrade the extracellular matrix on the path to the circulation or create a basement membrane at the target site. Another possibility is the analysis of the superficial glycoproteins, factors responsible for cell adhesion (integrins) and intercellular communication (cadherins). Furthermore neoangiogenesis markers such as vascular endothelial growth factor (VEGF), endoglin (CD105), a transmembrane glycoprotein which is a component of the receptor for activating TGFβ, as well as neuropilin (NRP1), the co-receptor for VEGF could be included [37]. As previously described, markers of EMT transition (Twist, PI3K/Atk and CDH1) and melanoma stem cell markers (e.g. ABCB5) may also be of value in this analysis.
It has been reported that alteration of the pathways involved in melanocyte development may be the key to acquiring metastatic potential in melanoma [13, 106–109]. In fact, melanoma metastasis mimics the migratory capacity of neural crest cells, which are the embryonic precursors of melanocytes. Some of the genes responsible for melanocyte development have been implicated in the process of melanoma formation, for example, PAX3 [93, 104, 110–112], Mitf [89, 93, 104], dopachrome tautomerase (DCT) [104, 112] and SOX10 [104]. These genes maintain a population of melanoblasts during migration from the neural crest during embryogenesis and from the hair follicle niche in mature skin. It is possible that these cells may also be the key to understanding melanoma stem cell maintenance and migration. The developmental genes and the signalling pathways required for maintenance of stem cell niches may help in the understanding of melanoma heterogeneity and the biological properties of melanoma cell subpopulations [89].
The use of CTCs as a prognostic indicator—valuable or not?
From past studies it has been noted that positivity for CTCs is not a prognostic indicator in itself i.e. not all circulating cells establish successful metastases. Circulating breast cancer cells have been found many years after mastectomy; while only1% of patients develop recurrence, patients with circulating cells shortly after mastectomy are more likely to develop recurrence [113]. In uveal melanoma the quantity and genetic profile of CTCs was more predictive than mere presence of CTCs, which was relatively inaccurate [114]. Moreover there is not yet any evidence that tumour progression is linked to increased genetic marker levels in circulating CTCs. It is possible that an increase in marker level is due to a change in phenotype rather than an increase in the number of CTCs.
A more comprehensive set of analyses of CTCs are required to better understand the diagnostic and prognostic significance of CTCs. We propose that these issues be addressed by isolation, characterisation and quantification of circulating melanoma cells. Additionally, application of newly identified markers to the long term follow up of patients will delineate the metastatic potential of circulating melanoma cells and their usefulness as a prognostic indicator.
Conclusion
This article addresses the transformation of cells from clinically latent to metastatic proliferating cells. We hypothesise that the ability of circulating melanoma cells to become activated, proliferative and migratory from a clinically latent cell depends on several key genes. An alternate hypothesis is that malignant cells disseminate from the primary tumour early in tumour development and remain in a clinically latent state until either the cells themselves or the host environment is receptive to metastasis. Quintana [115] showed that at least 25% of randomly harvested human melanoma cells from 12 different patients will re-establish a melanoma when xenotransplanted as single cells into severely immuno-compromised mice. It is important therefore to identify the pathways responsible for transforming melanoma stem cells, which are normally clinically latent, into cells capable of metastasis. Microarray analysis of tumour cells from different stages of melanoma has demonstrated that the genetic signature of the cell changes as the tumour progresses [24]. This adds to the evidence that analysing the genotype of circulating cells may be beneficial in prognosis.
Current methods use only histological analysis and imaging to determine patient prognosis. While novel therapies are continuing to be trialled for the treatment of melanoma, mortality rates remain high [116]. Measurement of circulating melanoma cells has not yet been refined sufficiently so as to make it an effective diagnostic or prognostic tool. Future studies involving isolation and genetic analysis of circulating cells may provide crucial information about the changes required for metastatic success and the numbers of cells containing these genetic changes needed to establish successful metastasis. As well as the benefit identification of these genes may make to prognostic information, they may also be harnessed as targets for future treatment regimes.
Markers that highlight active, metastatic cells in the circulation would potentially enable understanding of melanoma metastasis like that already identified for breast cancer. Detection of CTCs is already a significant prognostic indicator in breast cancer patients as it relates directly to disease status and treatment efficacy [117, 118]. Indeed it more reliably correlates with overall survival than current imaging methods as they are relatively insensitive, especially in early metastasis [119, 120]. Preferentially, a paradigm embracing the isolation, quantification and characterisation of melanoma stem cells could potentially act as a more reliable and sensitive measure of progression and clearance of malignant melanoma which has already undergone existing or newly developed therapies.
References
Lewis TB et al (2005) Molecular classification of melanoma using real-time quantitative reverse transcriptase-polymerase chain reaction. Cancer 104:1678–1686
Australasian Association of Cancer Registries (2006) Australian Institute of Health and Welfare. http://www.aihw.gov.au/cancer/aacr/. Cited 28/4/10
Jack A et al (2006) The treatment of melanoma with an emphasis on immunotherapeutic strategies. Surg Oncol 15:13–24
Lee RT et al (2008) Melanoma presenting as circulating tumor cells associated with failed angiogenesis. Melanoma Res 18:289–294
Zbytek B et al (2008) Current concepts of metastasis in melanoma. Expert Rev Dermatol 3:569–585
Yang AS, Chapman PB (2009) The history and future of chemotherapy for melanoma. Hematol Oncol Clin North Am 23:583–597
Breslow A (1975) Tumor thickness, level of invasion and node dissection in stage I cutaneous melanoma. Ann Surg 182:572–575
Balch CM et al (2001) Final version of the American Joint Committee on Cancer staging system for cutaneous melanoma. J Clin Oncol 19:3635–3648
Eton O et al (1998) Prognostic factors for survival of patients treated systemically for disseminated melanoma. J Clin Oncol 16:1103–1111
Francken AB et al (2008) Prognosis and determinants of outcome following locoregional or distant recurrence in patients with cutaneous melanoma. Ann Surg Oncol 15:1476–1484
Thompson JF, Scolyer RA, Kefford RF (2009) Cutaneous melanoma in the era of molecular profiling. Lancet 374:362–365
Singh AD et al (2004) The Zimmerman-McLean-Foster hypothesis: 25 years later. Br J Ophthalmol 88:962–967
White RM, Zon LI (2008) Melanocytes in development, regeneration, and cancer. Cell Stem Cell 3:242–252
Roberts DL et al (2002) U.K. guidelines for the management of cutaneous melanoma. Br J Dermatol 146:7–17
Michaelson JS et al (2005) Spread of human cancer cells occurs with probabilities indicative of a nongenetic mechanism. Br J Cancer 93:1244–1249
Wascher RA et al (2003) Molecular tumor markers in the blood: early prediction of disease outcome in melanoma patients treated with a melanoma vaccine. J Clin Oncol 21:2558–2563
Hoek KS et al (2006) Metastatic potential of melanomas defined by specific gene expression profiles with no BRAF signature. Pigment Cell Res 19:290–302
Bockhorn M, Jain RK, Munn LL (2007) Active versus passive mechanisms in metastasis: do cancer cells crawl into vessels, or are they pushed? Lancet Oncol 8:444–448
Chen LL et al (2009) Cancer metastasis networks and the prediction of progression patterns. Br J Cancer 101:749–758
Butler TP, Gullino PM (1975) Quantitation of cell shedding into efferent blood of mammary adenocarcinoma. Cancer Res 35:512–516
Fidler IJ et al (2002) The seed and soil hypothesis: vascularisation and brain metastases. Lancet Oncol 3:53–57
Chiang AC, Massague J (2008) Molecular basis of metastasis. N Engl J Med 359:2814–2823
Ulmer A et al (2008) Visualization of circulating melanoma cells in peripheral blood of patients with primary uveal melanoma. Clin Cancer Res 14:4469–4474
Ulmer A et al (2004) Immunomagnetic enrichment, genomic characterization, and prognostic impact of circulating melanoma cells. Clin Cancer Res 10:531–537
Husemann Y et al (2008) Systemic spread is an early step in breast cancer. Cancer Cell 13:58–68
Mocellin S et al (2006) The prognostic value of circulating tumor cells in patients with melanoma: a systematic review and meta-analysis. Clin Cancer Res 12:4605–4613
Sekine I et al (2002) Relationship between objective responses in phase I trials and potential efficacy of non-specific cytotoxic investigational new drugs. Ann Oncol 13:1300–1306
Bhatia S, Tykodi SS, Thompson JA (2009) Treatment of metastatic melanoma: an overview. Oncol 23:488–496
T FR Crosby, Coles B, Mason M (2000) Systemic treatments for metastatic cutaneous melanoma. Cochrane Database Syst Rev 2:CD001215
Richards JM BA, Gonzalez R, et al. (2005) High-dose Allovectin-7® in patients with advanced metastatic melanoma: final Phase 2 data and design of Phase 3 registration trial. J Clin Oncol 23:16s:7543
Shepherd C, Puzanov I, Sosman JA (2010) B-RAF inhibitors: an evolving role in the therapy of malignant melanoma. Curr Oncol Rep 12:146–152
Fisher DE et al (2010) Melanoma from bench to bedside: meeting report from the 6th international melanoma congress. Pigment Cell Melanoma Res 23:14–26
Modjtahedi H, Essapen S (2009) Epidermal growth factor receptor inhibitors in cancer treatment: advances, challenges and opportunities. Anticancer Drugs 20:851–865
Flaherty KT et al (2010) Inhibition of Mutated, Activated BRAF in Metastatic Melanoma. N Engl J Med 363:809–819
Mazzocca A, Carloni V (2009) The metastatic process: methodological advances and pharmacological challenges. Curr Med Chem 16:1704–1717
Van Den Bossche K, Naeyaert JM, Lambert J (2006) The quest for the mechanism of melanin transfer. Traffic 7:769–778
Moustakas A, Heldin CH (2007) Signaling networks guiding epithelial-mesenchymal transitions during embryogenesis and cancer progression. Cancer Sci 98:1512–1520
Paterlini-Brechot P, Benali NL (2007) Circulating tumor cells (CTC) detection: clinical impact and future directions. Cancer Lett 253:180–204
Pouyssegur J, Dayan F, Mazure NM (2006) Hypoxia signalling in cancer and approaches to enforce tumour regression. Nature 441:437–443
Kageshita T et al (2001) Loss of beta-catenin expression associated with disease progression in malignant melanoma. Br J Dermatol 145:210–216
Vogelmann R et al (2005) TGFbeta-induced downregulation of E-cadherin-based cell-cell adhesion depends on PI3-kinase and PTEN. J Cell Sci 118:4901–4912
Peinado H, Portillo F, Cano A (2004) Transcriptional regulation of cadherins during development and carcinogenesis. Int J Dev Biol 48:365–375
Hsu MY et al (2000) E-cadherin expression in melanoma cells restores keratinocyte-mediated growth control and down-regulates expression of invasion-related adhesion receptors. Am J Pathol 156:1515–1525
Sanz-Moreno V et al (2008) Rac activation and inactivation control plasticity of tumor cell movement. Cell 135:510–523
Egeblad M, Werb Z (2002) New functions for the matrix metalloproteinases in cancer progression. Nat Rev Cancer 2:161–174
Lewis CE, Pollard JW (2006) Distinct role of macrophages in different tumor microenvironments. Cancer Res 66:605–612
Liotta LA, Kleinerman J, Saidel GM (1974) Quantitative relationships of intravascular tumor cells, tumor vessels, and pulmonary metastases following tumor implantation. Cancer Res 34:997–1004
Glinsky GV (1997) Apoptosis in metastatic cancer cells. Crit Rev Oncol Hematol 25:175–186
Swartz MA et al (1999) Cells shed from tumours show reduced clonogenicity, resistance to apoptosis, and in vivo tumorigenicity. Br J Cancer 81:756–759
Larson CJ et al (2004) Apoptosis of circulating tumor cells in prostate cancer patients. Cytometry A 62:46–53
Mehes G et al (2001) Circulating breast cancer cells are frequently apoptotic. Am J Pathol 159:17–20
Holmgren L, O’Reilly MS, Folkman J (1995) Dormancy of micrometastases: balanced proliferation and apoptosis in the presence of angiogenesis suppression. Nat Med 1:149–153
Nguyen DX, Bos PD, Massague J (2009) Metastasis: from dissemination to organ-specific colonization. Nat Rev Cancer 9:274–284
Borsig L et al (2002) Synergistic effects of L- and P-selectin in facilitating tumor metastasis can involve non-mucin ligands and implicate leukocytes as enhancers of metastasis. Proc Natl Acad Sci USA 99:2193–2198
Laubli H et al (2006) L-selectin facilitation of metastasis involves temporal induction of Fut7-dependent ligands at sites of tumor cell arrest. Cancer Res 66:1536–1542
Fuertes MB et al (2008) Intracellular retention of the NKG2D ligand MHC class I chain-related gene A in human melanomas confers immune privilege and prevents NK cell-mediated cytotoxicity. J Immunol 180:4606–4614
Lacreusette A et al (2007) Loss of oncostatin M receptor beta in metastatic melanoma cells. Oncogene 26:881–892
Horak CE et al (2008) The role of metastasis suppressor genes in metastatic dormancy. APMIS 116:586–601
Eyles J et al (2010) Tumor cells disseminate early, but immunosurveillance limits metastatic outgrowth, in a mouse model of melanoma. J Clin Invest 120:2030–2039
Yoshida BA et al (2000) Metastasis-suppressor genes: a review and perspective on an emerging field. J Natl Cancer Inst 92:1717–1730
Röcken M (2010) Early tumor dissemination, but late metastasis: insights into tumor dormancy. J Clin Invest 120:1800–1803
Harms JF, Welch DR, Miele ME (2003) KISS1 metastasis suppression and emergent pathways. Clin Exp Metastasis 20:11–18
Boissan M, Poupon MF, Lacombe ML (2007) NM23 and metastasis suppressor genes: update. Med Sci (Paris) 23:1115–1123
Steeg PS, Horak CE, Miller KD (2008) Clinical-translational approaches to the Nm23–H1 metastasis suppressor. Clin Cancer Res 14:5006–5012
Gobeil S et al (2008) A genome-wide shRNA screen identifies GAS1 as a novel melanoma metastasis suppressor gene. Genes Dev 22:2932–2940
Shevde LA et al (2002) Suppression of human melanoma metastasis by the metastasis suppressor gene, BRMS1. Exp Cell Res 273:229–239
Lee CC et al (2008) Improved survival after lymphadenectomy for nodal metastasis from an unknown primary melanoma. J Clin Oncol 26:535–541
Vijuk G, Coates AS (1998) Survival of patients with visceral metastatic melanoma from an occult primary lesion: a retrospective matched cohort study. Ann Oncol 9:419–422
Zalaudek I et al (2003) Local recurrence in melanoma in situ: influence of sex, age, site of involvement and therapeutic modalities. Br J Dermatol 148:703–708
Koyanagi K et al (2005) Serial monitoring of circulating melanoma cells during neoadjuvant biochemotherapy for stage III melanoma: outcome prediction in a multicenter trial. J Clin Oncol 23:8057–8064
Kujala E, Mäkitie T, Kivelä T (2003) Very long-term prognosis of patients with malignant uveal melanoma. Investig Ophthalmol Vis Sci 44:4651–4659
Wargo JA, Tanabe K (2009) Surgical management of melanoma. Hematol Oncol Clin North Am 23:565–581
Ramaswamy S et al (2003) A molecular signature of metastasis in primary solid tumors. Nat Genet 33:49–54
Bernards R, Weinberg RA (2002) A progression puzzle. Nature 418:823
Waghorne C et al (1988) Genetic evidence for progressive selection and overgrowth of primary tumors by metastatic cell subpopulations. Cancer Res 48:6109–6114
Fang D et al (2005) A tumorigenic subpopulation with stem cell properties in melanomas. Cancer Res 65:9328–9337
Grichnik JM et al (2006) Melanoma, a tumor based on a mutant stem cell? J Invest Dermatol 126:142–153
Monzani E et al (2007) Melanoma contains CD133 and ABCG2 positive cells with enhanced tumourigenic potential. Eur J Cancer 43:935–946
Schatton T et al (2008) Identification of cells initiating human melanomas. Nature 451:345–349
Zabierowski SE, Herlyn M (2008) Melanoma stem cells: the dark seed of melanoma. J Clin Oncol 26:2890–2894
Wicha MS (2006) Cancer stem cells and metastasis: lethal seeds. Clin Cancer Res 12:5606–5607
Pantel K, Alix-Panabieres C, Riethdorf S (2009) Cancer micrometastases. Nat Rev Clin Oncol 6:339–351
Keshet GI et al (2008) MDR1 expression identifies human melanoma stem cells. Biochem Biophys Res Commun 368:930–936
Schatton T, Frank MH (2008) Cancer stem cells and human malignant melanoma. Pigment Cell Melanoma Res 21:39–55
Smalley KS, Herlyn M (2009) Integrating tumor-initiating cells into the paradigm for melanoma targeted therapy. Int J Cancer 124:1245–1250
Hess AR, Seftor EA, Gruman LM, Kinch MS, Seftor RE, Hendrix MJ (2006) VE-cadherin regulates EphA2 in aggressive melanoma cells through a novel signaling pathway: implications for vasculogenic mimicry. Cancer Biol Ther 5:228–233
La Porta C (2009) Cancer stem cells: lessons from melanoma. Stem Cell Rev 5:61–65
Roesch A et al (2010) A temporarily distinct subpopulation of slow-cycling melanoma cells is required for continuous tumor growth. Cell 141:583–594
Hoek KS, Goding CR (2010) Cancer stem cells versus phenotype-switching in melanoma. Pigment Cell Melanoma Res 23:746–759
Bennett DC (2008) How to make a melanoma: what do we know of the primary clonal events? Pigment Cell Melanoma Res 21:27–38
Bosserhoff AK (2006) Novel biomarkers in malignant melanoma. Clin Chim Acta 367:28–35
Gogas H et al (2009) Biomarkers in melanoma. Ann Oncol 20(Suppl 6):vi8–vi13
Medic S et al (2007) Molecular markers of circulating melanoma cells. Pigment Cell Res 20:80–91
Koyanagi K et al (2006) Association of circulating tumor cells with serum tumor-related methylated DNA in peripheral blood of melanoma patients. Cancer Res 66:6111–6117
Xi L et al (2007) Optimal markers for real-time quantitative reverse transcription PCR detection of circulating tumor cells from melanoma, breast, colon, esophageal, head and neck, and lung cancers. Clin Chem 53:1206–1215
Koyanagi K et al (2006) Microphthalmia transcription factor as a molecular marker for circulating tumor cell detection in blood of melanoma patients. Clin Cancer Res 12:1137–1143
Alonso SR et al (2007) A high-throughput study in melanoma identifies epithelial-mesenchymal transition as a major determinant of metastasis. Cancer Res 67:3450–3460
Mandruzzato S et al (2006) A gene expression signature associated with survival in metastatic melanoma. J Transl Med 4:50
Talantov D et al (2005) Novel genes associated with malignant melanoma but not benign melanocytic lesions. Clin Cancer Res 11:7234–7242
Tucci MG et al (2007) Involvement of E-cadherin, beta-catenin, Cdc42 and CXCR4 in the progression and prognosis of cutaneous melanoma. Br J Dermatol 157:1212–1216
Kauffmann A et al (2008) High expression of DNA repair pathways is associated with metastasis in melanoma patients. Oncogene 27:565–573
Winnepenninckx V et al (2006) Gene expression profiling of primary cutaneous melanoma and clinical outcome. J Natl Cancer Inst 98:472–482
Eberle J et al (2008) Apoptosis pathways and oncolytic adenoviral vectors: promising targets and tools to overcome therapy resistance of malignant melanoma. Exp Dermatol 17:1–11
Medic S, Ziman M (2009) PAX3 across the spectrum: from melanoblast to melanoma. Crit Rev Biochem Mol Biol 44:85–97
Adams JM, Strasser A (2008) Is tumor growth sustained by rare cancer stem cells or dominant clones? Cancer Res 68:4018–4021
Carreira S et al (2006) Mitf regulation of Dia1 controls melanoma proliferation and invasiveness. Genes Dev 20:3426–3439
Gupta PB et al (2005) The evolving portrait of cancer metastasis. Cold Spring Harb Symp Quant Biol 70:291–297
McArdle L et al (2005) Microarray analysis of phosphatase gene expression in human melanoma. Br J Dermatol 152:925–930
Topczewska JM et al (2006) Embryonic and tumorigenic pathways converge via Nodal signaling: role in melanoma aggressiveness. Nat Med 12:925–932
Kubic JD et al (2008) Pigmentation PAX-ways: the role of Pax3 in melanogenesis, melanocyte stem cell maintenance, and disease. Pigment Cell Melanoma Res 21:627–645
Lang D et al (2005) Pax3 functions at a nodal point in melanocyte stem cell differentiation. Nature 433:884–887
Plummer RS et al (2008) PAX3 expression in primary melanomas and nevi. Mod Pathol 21:525–530
Meng S et al (2004) Circulating tumor cells in patients with breast cancer dormancy. Clin Cancer Res 10:8152–8162
Cools-Lartigue JJ et al (2008) Immunomagnetic isolation and in vitro expansion of human uveal melanoma cell lines. Mol Vis 14:50–55
Quintana E et al (2008) Efficient tumour formation by single human melanoma cells. Nature 456:593–598
Lewis KD et al (2008) A phase II study of the heparanase inhibitor PI-88 in patients with advanced melanoma. Invest New Drugs 26:89–94
Tewes M et al (2009) Molecular profiling and predictive value of circulating tumor cells in patients with metastatic breast cancer: an option for monitoring response to breast cancer related therapies. Breast Cancer Res Treat 115:581–590
Fehm T et al (2008) Micrometastatic spread in breast cancer: detection, molecular characterization and clinical relevance. Breast Cancer Res 10(Suppl 1):S1
Cristofanilli M et al (2004) Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med 351:781–791
Dawood S, Cristofanilli M (2007) Integrating circulating tumor cell assays into the management of breast cancer. Curr Treatment Options Oncol 8:89–95
Acknowledgments
We would like to thank Peter Mathews for helpful discussions and acknowledge funding from The Cancer Council of Western Australia, the National Health and Medical Research Council of Australia and the Cancer and Palliative Care Research and Evaluation Unit.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ireland, A., Millward, M., Pearce, R. et al. Genetic factors in metastatic progression of cutaneous melanoma: the future role of circulating melanoma cells in prognosis and management. Clin Exp Metastasis 28, 327–336 (2011). https://doi.org/10.1007/s10585-010-9368-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-010-9368-2