Abstract
Identifying molecular targets for treatment of pancreatic cancer metastasis is critical due to the high frequency of dissemination prior to diagnosis of this lethal disease. Because the KISS1 metastasis suppressor is expressed at reduced levels in advanced pancreatic cancer, we hypothesized that re-expression of KISS1 would reduce metastases. Highly metastatic S2VP10 cells expressing luciferase (S2VP10L) were transfected with a FLAG-tagged version of KISS1 (KFM), KFMΔSS (with deleted secretion signal sequence), or pcDNA3 control plasmid (CP) and expression was confirmed by RTQ-PCR. SCID mice were implanted orthotopically with S2VP10L cells or transfectants and tumor growth and metastases were monitored using bioluminescence imaging. Mice with S2VP10L-KISS1 tumors developed fewer liver (98%) and lung (99%) metastases than S2VP10L. Unexpectedly, mice with S2VP10L-KFMΔSS tumors also had reduced liver and lung metastases, but had more metastases than mice with S2VP10L-KISS. KISS1 protein was found in the cytoplasm of both KFMΔSS and KISS1-expressing orthotopic tumors by immunohistochemistry. Metastases were not found in lungs of mice with S2VP10L-KISS1 tumors; whereas, KFMΔSS lung sections had regions of concentrated KISS1 staining, suggesting that secretion of KISS1 is needed to reduce metastasis significantly. These data suggest induction of KISS1 expression has potential as an adjuvant treatment for pancreatic cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Human pancreatic cancer has a very poor prognosis and is currently the fourth leading cause of cancer death in the United States [1]. Pancreatic adenocarcinoma patients have a median survival of <6 months and a dismal 5-year survival rate of 5% [2, 3]. The lethal nature of pancreatic adenocarcinoma is strongly associated with its early metastases to distant organs. The aggressive, but largely asymptomatic, nature of this disease leads to late clinical presentation which, unfortunately, remains mostly incurable [4]. Conventional therapies such as chemotherapy, radiation, and surgery, are largely ineffective in treating pancreatic adenocarcinoma; therefore, novel treatment strategies are desperately needed [5]. Since dissemination has usually occurred before clinical detection, preventing the proliferation of already disseminated cells (i.e., colonization) offers promise for patients with pancreatic adenocarcinoma.
Metastasis suppressors represent a potential new strategy for treatment of locally advanced and metastatic pancreatic cancer. Metastasis suppressors, by definition, inhibit metastasis without blocking orthotopic tumor growth [6]. Metastasis suppressors are found in every cellular compartment, including some that are secreted. Each regulates one or more steps in the metastatic cascade, including regulation of signaling pathways that inhibit proliferation at secondary sites [7–9]. Many also exhibit an inverse correlation with tumor grade and the likelihood of recurrence with metastatic disease [10].
Nascent KISS1 is a secreted suppressor of metastases and is processed into a number of peptides, called kisspeptins [11]. A 54-amino acid polypeptide (metastin or kisspeptin-54 (kp-54)) is the ligand for a G-protein coupled receptor KISS1R, alternatively known as GPR54, hOT7T175 or AX0R12 [6, 9, 12]. Re-expression of KISS1 inhibits metastasis of human melanoma, breast, and ovarian cancer in xenograft models while still allowing growth of orthotopic tumors in vivo [11, 12]. KISS1 expression has been reported to be significantly lower in human pancreatic cancer tissue than in normal pancreas and to be associated with advanced disease [13, 14]. Although KISS1 expression is inversely associated with disease progression by immunohistochemistry or in situ hybridization analyses, no statistically significant correlation has been yet found between plasma levels of secreted KISS1 (plasma metastin-L1) and tumor grade [15].
The aim of this study was to examine whether re-expression of KISS1 in pancreatic cancer cells suppresses metastasis of the human pancreatic cancer cell line S2VP10 in an orthotopic mouse xenograft model.
Materials and methods
Cells and cell culture
A metastatic subclone of the SUIT-2 pancreatic adenocarcinoma cell line, S2VP10, was provided by Dr. Michael A. Hollingsworth at the University of Nebraska [16–18]. Cells were grown in Dulbecco’s-modified Eagle’s medium containing 4.5 g/L glucose (DMEM) supplemented with 10% fetal bovine serum (FBS) and 2 mM L-glutamine and were maintained in a humidified 5% CO2 atmosphere at 37°C. S2VP10 cells were transduced with a non-replicating retrovirus containing firefly luciferase gene (Stratagene, La Jolla, CA). A single cell clone, S2VP10L, was isolated and bioluminescence signal was confirmed using the IVIS 100 bioluminescence system (Caliper, Hopkinton, MA). The growth rates of parental S2VP10 cells and S2VP10L were identical. We have been unable to identify another pancreatic xenograft model, including L3.6pl, whose rate of metastasis is adequate for our study.
Parental cell lines were analyzed for expression of KISS1R and KISS1 by real-time RT-PCR, flow cytometry and ELISA using a monoclonal antibody recognizing KISS1 that was developed by the UAB Tissue Characterization and Immunoreagent Resource, as described below.
KISS1 transfection
Plasmids containing an internal FLAG epitope and either KISS1 (KFM) or a deleted secretion sequence (KFMΔSS) were previously generated using pcDNA3 (Invitrogen, Carlsbad, CA) according to manufacturer’s instructions [19]. The presence of the internal FLAG epitope did not modify KP-54 function [19]. Transfected cells were selected with G418 for 3 passages. S2VP10L cells transfected with control plasmid pCMV served as a negative control (S2VP10L-CP). For conceptual simplicity, we will use KISS1 (for KFM) and KFMΔSS, respectively.
Detection of KISS1 and KISS1R
For RTQ-PCR, mRNA was isolated from cells or tissue using Trizol (Invitrogen, Carlsbad, CA), according to manufacturer’s instructions and RNA was stored at −80°C up to 6 months prior to analysis. RNA quantity was assessed with a Nanodrop (Thermo Scientific, Wilmington, DE). Primers for KISS1 (Hs00158486m1), KISS1R (Hs00261399m1), S9 (Hs00396989m1) and 18S (Hs99999901s1) were purchased from ABI, Foster City, CA. The Taqman One Step RT-PCR (ABI) was used for the reaction according to protocols. Samples were measured in an ABI7500 Real Time PCR system. Samples were measured in triplicate [20]. Normalization was performed using S9 for cultured cells and 18S for tissue samples.
To examine KISS1R expression on SVP10L cells and their respective variants flow cytometry was performed using a FACSCalibur flow cytometer (BD Biosciences, San Jose, CA). The following antibodies were used: anti-KISS1R (LifeSpan Biosciences; #LS-A1929), anti-rabbit IgG-FITC (Sigma, #F-9887) and rabbit IgG (Abcam) was used as an isotype control. Cell preparations were stained in FACS buffer (HBSS with 3% FBS). Living cells were distinguished from dead cells using propidium iodide (PI) uptake.
Human pancreatic cancer orthotopic xenograft mouse models
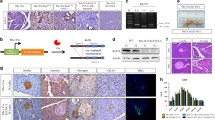
Female severe combined immunodeficient (SCID) mice 2 months of age (Frederick Animal Production Program, Frederick, MD) were allowed to acclimate for 4 weeks before use. Strict adherence to the UAB Institutional Animal Care and Use Committee (IACUC) approved protocol was maintained throughout the study. Orthotopic cell implantation followed the procedure in DeRosier et al. [21]. Briefly, mice were anesthetized with i.p. xylazine and ketamine. A 1-cm incision was made in the left upper abdominal quadrant in a sterile hood. The spleen was located and used to indirectly position the tail of the pancreas, carefully avoiding direct pancreatic manipulation. Mice (8/group) received S2VP10L, S2VP10L-CP, S2VP10L-KFMΔSS1, or S2VP10L-KISS1 cells. The cellular suspension was stored on ice in a sterile tube and then drawn up using a 28-gauge needle to aliquot 1 × 105 cells/40 μl which was injected into the tail of the pancreas. A sterile cotton tipped applicator was used to cover the injection site for 30 s to prevent peritoneal leakage. The organs were returned to the abdomen and the skin and peritoneum were closed in a single layer closure with 5-0 Prolene sutures. Animals recovered on a warming blanket and received liquid acetaminophen for 24 h with food and water ad libitum.
Metastasis monitoring by bioluminescence imaging
Bioluminescence imaging was used to assess orthotopic implantation in all mice immediately following surgery with the IVIS-100 bioluminescence imaging system (Caliper). Mice with detectable leakage from the pancreas were removed from the study. Mice were injected i.p. with 2.5 mg luciferin 10 min prior to imaging. Each mouse was imaged weekly to monitor orthotopic tumor and metastatic growth. Region of interest (ROI) analysis was used to measure light emitted for both orthotopic and metastatic sites using the IVIS Living Image software.
Experimental metastasis
Mice were implanted orthotopically with S2VP10L, S2VP10L-CP, S2VP10L-KFMΔSS 1, or S2VP10L-KISS1 cells as indicated above, and study group was reduced to 6/group due to detectable cell leakage from the pancreas in some mice. Lung, liver, diaphragm, and pancreas were removed and examined with bioluminescence imaging 15 and 30 days post-injection (3 mice/treatment). Bioluminescent signal was quantified using the Region of Interest (ROI) method for each organ and means were compared using ANOVA test. To determine background luminescence signal, organs were removed from 3 naïve mice and imaged using the ROI method. The signal obtained in the organs of naïve mice were: lung (1.92 × 102 counts/s), liver (2.50 × 102 counts/s), and diaphragm (2.32 × 102 counts/s). Immediately following bioluminescence imaging, organs were divided into portions for histology or RTQ-PCR analyses.
For histologic analyses, tumor xenografts as well as other tissues were harvested 15 and 30 days after cell injection, formalin fixed, and processed into paraffin blocks using standard methods. Metastases were detected using a monoclonal mouse anti-KISS1 antibody (6a4.27) generated at the UAB Tissue Characterization and Immunoreagent Resource. Antibody specificity was determined by co-localization of 6a4.27 staining with anti-FLAG staining on immunoblots as well as recognition of mass spectrometry-verified kisspeptins (B.H. Beck, K.T. Nash, and D.R. Welch, unpublished observations).
Formalin-fixed, paraffin-embedded tumors were cut into 4 μm sections and attached to slides. After deparaffinization and rehydration, antigen retrieval was preformed in a pressure cooker 121°C for 20 min in pH 8 EDTA buffer (luciferase or CD31 IHC) or pH 9 EDTA buffer (KISS1 IHC). After H2O2 quench and blocking with 3% goat serum, slides were incubated with the primary antibody at 1:1000 dilution (luciferase (Chemicon, Billerica, MA) or CD31 (Abcam Cambridge, MA) or 10 μg/ml (KISS1) for 20 min followed by Signet HRP label for 20 min and ScyTeck DAB (Logan, UT) for 7 min. Slides were counterstained with hematoxylin. Sections were viewed using a Nikon microscope and photographs were acquired using a Spot Camera (Diagnostic Instruments). Luciferase and KISS1 staining were based on 3 images per group.
Statistics
Organ metastasis levels were compared among groups for each organ using ANOVA with SAS software. When the ANOVA was statistically significant (P < 0.05), Tukey’s test was used to determine the source of the difference.
Results
Confirmation of KISS1 gene transfection and KISS1R expression
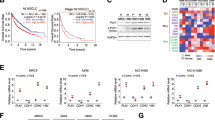
KISS1 and KISS1R expression were examined in stably transfected S2VP10L cells by RTQ-PCR (KISS1 and KISS1R), ELISA (KISS1) and flow cytometry (KISS1R) (Fig. 1). KISS1 and KFMΔSS mRNA levels were higher in transfected S2VP10L cells than S2VP10L-CP or non-transfected control cells. S2VP10L-KFMΔSS cells had 10-fold higher KISS1 mRNA than S2VP10L-KISS1 cells. Consistent with other cells that have been suppressed for metastasis following re-expression of KISS1, S2VP10L cells and derivative cells did not express KISS1R (Fig. 1).
KISS1 gene transfection and KISS1R expression. a Cells were transfected with plasmids containing KISS1, KFMΔSS, or control (CP). KISS1 mRNA levels were determined by RTQ-PCR. KISS1 levels relative to S2VP10L are shown. b SVP10L cells express very low or no KISS1R mRNA (Bar Graph showing real-time quantitative RT-PCR) or protein (Flow cytometry). T47D human breast cancer cells, which express low levels of KISS1R, are included as a positive control
KISS1 expression suppresses metastatic ability in vivo
To test the effect of KISS1 on metastasis, an orthotopic pancreatic cancer mouse model and bioluminescence imaging were used. At day 15 post-tumor cell injection, S2VP10L and S2VP10L-CP tumor-bearing mice had similar tumor growth and metastatic efficiency (Fig. 2). Mice injected with S2VP10L-KFMΔSS 1 cells developed fewer pulmonary (50% of both controls), hepatic (95% of both controls), and diaphragmatic (97% of both controls) metastasis compared to mice implanted with parental (S2VP10L) or vector-only (S2VP10L-CP) control cells (P = 0.04, P = 0.009, and P = 0.006 respectively). S2VP10-KISS1 tumor bearing mice had reduced metastases in lung (90%), liver (99%), and diaphragm (97%) (P = 0.007, P = 0.002, and P = 0.006 respectively). As predicted, mice implanted with S2VP10L-KISS1 cells developed fewer metastases in lung (77%) and liver (23%) compared to mice implanted with S2VP10L-KFMΔSS 1 cells (P = 0.04 and P = 0.034, respectively). Interestingly, mice implanted with S2VP10L-KISS1 cells did not have reduced diaphragmatic metastases compared to S2VP10L-KFMΔSS 1 cells.
At day 15 post implant, S2VP10L and S2VP10L-CP mice had similar primary and metastasis tumor levels in all organs sampled as indicated by bioluminescence imaging. S2VP10L-KFMΔSS had reduced pulmonary metastasis (50%) and hepatic metastasis (95%) as compared to controls (P = 0.04 and P = 0.009). S2VP10-KISS1 mice had reduced metastasis by 90% in lung and 99% in liver as compared to controls (P = 0.007 and P = 0.002, respectively). Group means and standard deviations are presented
At day 30 post cell implant, the effects of KISS1 were even more pronounced. KISS1-expressing S2VP10L cells had 99% fewer pulmonary (P < 0.013) and 98% fewer hepatic (P < 0.001) metastases than control S2VP10L implanted mice detectable by bioluminescence. At day 30, orthotopic tumor burdens were indistinguishable amongst all groups (P = 0.53) (Fig. 3). Mice with S2VP10L-KFMΔSS tumors had significantly reduced pulmonary metastasis (77%) and hepatic metastasis (81%) compared to S2VP10L-CP and S2VP10L injected mice (P = 0.04 and P = 0.009, respectively). Mice implanted with S2VP10L cells expressing KISS1 had significantly fewer lung and liver metastases than mice implanted with S2VP10L- KFMΔSS cells(P = 0.03 and P = 0.04, respectively).
Metastasis measured by bioluminescence imaging in mice and mouse organs 30 days post implant. Tissues (Lu = Lung; Li = liver, D = diaphragm; and P = pancreas) were removed 30 days post implant. S2VP10L-KISS1 had reduced lung metastasis (P < 0.013), liver metastasis (P < 0.001), and disease in the diaphragm (P < 0.008) as compared to either S2VP10L-CP or S2VP10L. Pancreas levels were similar among all groups (P = 0.553). Both S2VP10L and S2VP10L-CP had extensive metastasis in each organ as identified in whole body images of mouse and specific organs. S2VP10L-KISS1 mice had minimal metastasis in whole body of mice or in specific organs. Left and Center panels contain a single representative mouse and organs from this representative mouse. In contrast, group mean and standard deviation are presented in Right panel
Mice injected with S2VP10L, S2VP10L-CP, or S2VP10L-KFMΔSS cells developed macroscopic liver, lung, and diaphragm metastases that were detectable using bioluminescence. Metastases were confirmed by immunohistochemical staining of luciferase (Fig. 4). The bioluminescence signals corresponded proportionately to metastatic burden extrapolated from organ sections (Figs. 3, 4). Strikingly, pulmonary or hepatic metastases could not be detected by immunohistochemistry in S2VP10L-KISS1 (Fig. 4).
Immunohistochemistry confirmed luciferase positive metastasis levels in lung sections 30 days post tumor cell injection (bar = 100 μm). Representative S2VP10L (a) and S2VP10L-CP (b) lung sections had multiple metastases (indicated by arrows). S2VP10L-KFMΔ SS (c) had moderate lung metastases (indicated by arrows). S2VP10L-KISS1 (d) had no detectable metastases
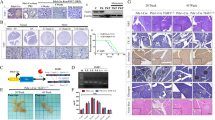
KISS1 expression confirmed by histology and RTQ-PCR
KISS1 protein was visualized by immunohistochemistry in the cytoplasm of cells in the S2VP10L-KISS1 and -KFMΔSS pancreatic tumors at days 15 (data not shown) and 30 post-tumor cell injection (Fig. 5). KISS1 protein was elevated in S2VP10L-KISS1 tumor sections as compared to S2VP10L controls. KISS1 protein appeared to be more concentrated in the cytoplasm of S2VP10L-KFMΔSS tumors than tumors expressing full-length KISS1, which is consistent with loss of the secretion signal sequence. In mice implanted with S2VP10L and S2VP10L-CP cells, pulmonary metastatic lesions showed similar low cytoplasmic KISS1 protein levels as observed in the corresponding orthotopic tumors. Likewise, mice with S2VP10L-KFMΔSS tumors expressed readily detectable KISS1 protein in pulmonary metastases. Mice injected with S2VP10L-KISS1 did not develop detectable pulmonary metastases (Figs. 3, 4). The antibody also recognizes murine KISS1, so it is challenging to detect tumor cells amongst the background staining in normal lung.
Immunohistochemistry confirmed KISS1 protein levels in pancreatic tumor sections (bar = 50 μm) and lung sections (bar = 100 μm) at day 30 post tumor cell injection. a Representative S2VP10L pancreatic tumor sections had low levels of KISS1 protein (indicated by brown staining). b S2VP10L-KFMΔ SS had high KISS1 levels (indicated by dark brown staining) and concentrated KISS1 levels in some cells which suggests that KISS1 is not secreted in some cells containing the KFMΔ SS plasmid. c S2VP10L-KISS1 had weak cytoplasmic staining and cell membrane staining (indicated by brown staining), but did not contain a high concentration of KISS1 as found in S2VP10L-KFMΔ SS 1 which suggests that KISS1 is secreted. d S2VP10L metastasis in the lung contained low KISS1 protein levels. e S2VP10L-KFMΔ SS 1 lung sections had concentrated KISS1 protein staining in metastatic nodules. f S2VP10L-KISS1 lung section had no detectable metastasis and the lung had similar KISS1 staining to normal mouse lung
To assess KISS1 expression in orthotopic and ectopic cells, mRNA was isolated from intra-pancreatic and lung tumor tissues (Fig. 6). S2VP10L and S2VP10L-CP pancreatic tumors had the lowest KISS1 levels; whereas KISS1 levels of S2VP10L-KFMΔSS tumors were 3,500X higher and S2VP10L-KISS1 was 3,000X higher than controls. KISS1 mRNA levels in the pancreatic tumors were roughly proportionate to in vitro levels (Fig. 1). KISS1 mRNA and KISS1 protein expression (by IHC) corresponded well in pancreatic tumors (Figs. 5, 6). KISS1 mRNA levels in lung samples were similar among treatment groups (Figs. 5, 6) as well as normal mouse lung (data not shown).
KISS1 mRNA levels were determined in tissue from mice at day 30 post tumor cell injection. RNA levels are expressed on a per cell basis. S2VP10L and S2VP10L-CP had low levels of KISS1 mRNA in pancreatic samples. S2VP10L-KFMΔSS 1 and S2VP10L-KISS1 had higher levels of KISS1 mRNA in pancreatic samples. Lung samples among the 4 groups had similar KISS1 mRNA levels (P > 0.05). Group means and standard deviations are presented
Orthotopic tumor growth assessed by bioluminescence imaging
Orthotopic tumor growth was assessed longitudinally by bioluminescence imaging. All mice had similar pancreatic luciferase signals after tumor cell injection (Fig. 7). The pancreatic tumors of S2VP10L-KISS1-injected mice had significantly reduced signal compared to controls or S2VP10L-KFMΔSS tumors at day 7 (P = 0.04), but, by day 14, the signals were again similar (Fig. 7). Tumor burden, as indicated by the luciferase signal, was not significantly different for S2VP10L-KISS1 pancreatic tumors compared to the other tumors at either day 14 or 30 post-tumor cell injection (Figs. 2, 3, 7).
Tumor growth monitored by bioluminescence imaging. Immediately after cell injection, tumors were measured using bioluminescence imaging. Tumor growth was monitored weekly and luciferase signal was normalized. All tumors had similar luciferase signal after implantation. Left panel contains a representative mouse for each group. In contrast, group mean and standard deviation are presented in Right panel. S2VP10L-KISS1 pancreatic tumors had decreased signal at day 7 in comparison to controls (P = 0.04). S2VP10L-KFMΔSS pancreatic tumors grew at similar rates (day 7 and 14) as the control (S2VP10L), but by day 14 had reduced metastasis levels. At day 14, S2VP10L-KISS1 pancreatic tumors were similar to control pancreatic tumors, but had no detectable levels of metastasis
Discussion
The majority of patients with pancreatic cancer already have metastases at the time of diagnosis and very few survive 5 years [3, 23]. In an initial report by Masui on 30 patients, KISS1 levels were significantly lower in pancreatic carcinomas than normal pancreatic tissue, a finding later confirmed in 53 patients [13, 14]. Generally, cancer patients with higher expression of KP-54 and KISS1R had longer survival [22–25]. Taken together, these facts coupled with KISS1 being a secreted protein, makes KISS1 an interesting candidate for suppression of pancreatic metastasis. Our study is the first to examine the effects of over-expressing KISS1 in pancreatic cancer in vivo. As hypothesized, KISS1 over-expression significantly suppressed pancreatic carcinoma metastasis to lung and liver (99% and 98%, respectively (Fig. 3)). These findings provide a further basis to study the actions and mechanisms of KISS1 and its potential as a new target for prevention and/or progression of metastasis.
KISS1 re-expression in ovarian and melanoma cancer cells reduced metastasis (50% ovarian and 90% melanoma) [11, 19]. Although secretion of KISS1 was required for metastasis suppressor activity in the melanoma model [19], KISS1 suppressed pancreatic carcinoma metastasis in our orthotopic peritoneal model. Whereas the findings in melanoma precluded an intracrine loop, the findings in pancreatic adenocarcinoma leave open this possibility, albeit all of the cells suppressed for metastasis failed to express detectable KISS1R. We acknowledge the possibility that an undetectable fraction of KISS1 might be secreted. Likewise, as we have speculated previously, other biologically active fragments of KISS1 (i.e., besides KP-54) may be released into the surrounding milieu. Experiments to ascertain this possibility are underway.
Consistent with the functional definition of metastasis suppressors, KISS1 did not completely block orthotopic tumor growth. In the S2VP10L model, orthotopic tumors did develop, but not without a transient delay during the first week after injection (Fig. 7). Because orthotopic pancreatic tumors were of similarly sized by day 30, any initial delay in the primary tumor growth is unlikely explain differences in metastatic outgrowth observed at the time of experiment termination. An explanation for this initial growth delay is not readily apparent, as differences in angiogenesis (i.e., blood vessel density as measured by anti-CD31 staining) were not observed (Fig. 8). The effect of KISS1 on metastatic tumor outgrowth beyond 30 days was not possible because all animals had to be euthanized because of ethical considerations.
Although the lung samples of S2VP10L-KISS1 mice had a detectable, but weak, bioluminescence signal (Fig. 3), metastatic lesions were not observed by IHC in step sections. These findings are consistent with prior publications in breast and melanoma models that KISS1-expressing cells are able to disseminate, but not colonize ectopic sites [26, 27]. This property suggests that KISS1 could be useful for controlling pancreatic tumor cells that have all-too-often previously seeded other tissues before initial diagnosis.
In summary, identification of KISS1 as a metastasis suppressor in pancreatic cancer has expanded the horizon of our understanding of the function of this gene. KISS1 and its signaling pathways may represent new targets for the treatment of metastatic pancreatic cancer. Strategies could be developed to target metastatic pancreatic tumors by the delivery of KISS1 and/or kisspeptins which may result in static disease. Re-expression of KISS1 may prove to be a valuable treatment to suppress metastasis of pancreatic cancer.
References
Hezel AF, Kimmelman AC, Stanger B, Bardeesy N, DePinho RA (2006) Genetics and biology of pancreatic ductal adenocarcinoma. Genes Develop 20:1218–1249
Wray CJ, Ahmad SA, Matthews JB, Lowy AM (2005) Surgery for pancreatic cancer: recent controversies and current practice. Gastroenterology 128:1626–1641
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ (2009) Cancer statistics, 2009. CA Cancer J Clin 59:225–249
Breslin TM et al (2001) Neoadjuvant chemoradiotherapy for adenocarcinoma of the pancreas: treatment variables and survival duration. Ann Surg Oncol 8:123–132
Stathopoulos GP, Androulakis N, Souglakos J, Stathopoulos J, Georgoulias V (2008) Present treatment and future expectations in advanced pancreatic cancer. Anticancer Res 28:1303–1308
Rinker-Schaeffer CW, O’Keefe JP, Welch DR, Theodorescu D (2006) Metastasis suppressor proteins: discovery, molecular mechanisms, and clinical application. Clin Cancer Res 12:3882–3889
Chirco R, Liu XW, Jung KK, Kim HR (2006) Novel functions of TIMPs in cell signaling. Cancer Metastasis Rev 25:99–113
Freije JM, MacDonald NJ, Steeg PS (1998) Nm23 and tumour metastasis: basic and translational advances. Biochem Soc Symposia 63:261–271
Stafford LJ, Vaidya KS, Welch DR (2008) Metastasis suppressors genes in cancer. Int J Biochem Cell Biol 40:874–891
Steeg PS (2003) Metastasis suppressors alter the signal transduction of cancer cells. Nat Rev Cancer 3:55–63
Jiang Y, Berk M, Singh LS, Tan HY, Yin LH, Powell CT et al (2005) KISS1 suppresses metastasis in human ovarian cancer via inhibition of protein kinase C alpha. Clin Exp Metastasis 22:369–376
Stafford LJ, Xia CZ, Ma WB, Cai Y, Liu MY (2002) Identification and characterization of mouse metastasis-suppressor KISS1 and its G-protein-coupled receptor. Cancer Res 62:5399–5404
Masui T, Doi R, Mori T et al (2004) Metastin and its variant forms suppress migration of pancreatic cancer cells. Biochem Biophys Res Commun 315:85–92
Nagai K, Doi R, Katagiri F, Ito T, Kida A, Koizumi M, Masui T, Kawaguchi Y, Tomita K, Oishi S, Fujii N, Uemoto S (2009) Prognostic value of metastin expression in human pancreatic cancer. J Exp Clin Cancer Res 21:28–29
Katagiri F, Nagai K, Kida A, Tomita K, Oishi S, Takeyama M, Doi R, Fujii N (2009) Clinical significance of plasma metastin level in pancreatic cancer patients. Oncol Rep 21:815–819
Akisawa N, Nishimori I, Iwamura T, Onishi S, Hollingsworth MA (1999) High levels of ezrin expressed by human pancreatic adenocarcinoma cell lines with high metastatic potential. Biochem Biophys Res Commun 258:395–400
Kitamura N, Iwamura T, Taniguchi S, Yamanari H, Kawano K, Holli SR, Bramhall J, Schulz J, Nemunaitis PD, Brown BailletM, Buckels JA (2002) A double-blind placebo-controlled, randomised study comparing gemcitabine and marimastat with gemcitabine and placebo as first line therapy in patients with advanced pancreatic cancer. Br J Cancer 87:161–167
Hollingsworth MA, Setoguchi T (2001) High collagenolytic activity in spontaneously highly metastatic variants derived from a human pancreatic cancer cell line (SUIT-2) in nude mice. Clin Exp Met 18:561–571
Nash KT, Phadke PA, Navenot JM, Hurst DR, Accavitti-Loper MA, Sztul E, Vaidya KS, Frost AR, Kappes JC, Peiper SC, Welch DR (2007) Requirement of KISS1 secretion for multiple organ metastasis suppression and maintenance of tumor dormancy. J Natl Cancer Inst 99:309–321
Johnson MR, Wang K, Smith JB, Heslin MJ, Diasio RB (2000) Quantitation of dihydropyrimidine dehydrogenase expression by real-time reverse transcription polymerase chain reaction. Anal Biochem 278:175–184
DeRosier LC, Buchsbaum DJ, Oliver PG, Huang ZQ, Sellers JC, Grizzle WE, Wang W, Zhou T, Zinn KR, Long JW, Vickers SM (2007) Combination treatment with TRA-8 anti death receptor 5 antibody and CPT-11 induces tumor regression in an orthotopic model of pancreatic cancer. Clin Cancer Res 13:5535s–5543s
Longo R, Cacciamani F, Naso G, Gasparini G (2008) Pancreatic cancer: from molecular signature to target therapy. Crit Rev Oncol Hematol 68:197–211
Dhar DK, Naora H, Kubota H, Maruyama R, Yoshimura H, Tonomoto Y et al (2004) Downregulation of KiSS-1 expression is responsible for tumor invasion and worse prognosis in gastric carcinoma. Int J Cancer 111:868–872
Kostadima L, Pentheroudakis G, Pavlidis N (2007) The missing kiss of life: transcriptional activity of the metastasis suppressor gene KISS1 in early breast cancer. Anticancer Res 27:2499–2504
Prentice, LM, Klausen, C, Kalloger, S, Köbel M, McKinney S, Santos JL, et al (2007) Kisspeptin and GPR54 immunoreactivity in a cohort of 518 patients defines favourable prognosis and clear cell subtype in ovarian carcinoma. BMC Med 5:33
Lee JH, Miele ME, Hicks DJ, Phillips KK, Trent JM, Weissman BE, Welch DR (1996) KiSS-1, a novel human malignant melanoma metastasis-suppressor gene. J Natl Cancer Inst 88:1731–1737
Lee JH, Welch DR (1997) Suppression of metastasis in human breast carcinoma MDA-MB-435 cells after transfection with the metastasis suppressor gene, KiSS-1. Cancer Res 57:2384–2387
Acknowledgments
This study funded by the NIH Spore in Pancreatic Cancer P20CA101955 (DJB), NIH-1K99CA139050 (LRM), NIH-CA134981 (DRW) and CA87728 (DRW), and National Foundation for Cancer Research Center for Metastasis Research (DRW). This study was supported by UAB’s Small Animal Imaging Shared Facility (P30CA013148), Mass Spectrometry and Proteomics Shared Facilities, and Tissue Characterization and Immunoreagent Resource. We would also like to thank S. Lagan for assistance in manuscript preparation.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
McNally, L.R., Welch, D.R., Beck, B.H. et al. KISS1 over-expression suppresses metastasis of pancreatic adenocarcinoma in a xenograft mouse model. Clin Exp Metastasis 27, 591–600 (2010). https://doi.org/10.1007/s10585-010-9349-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-010-9349-5