Abstract
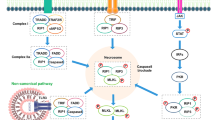
Epilepsy is one of the most common serious brain diseases worldwide. Programmed cell death (PCD), a cellular self-destruction phenomenon, has been greatly documented in neurodegenerative diseases. Pyroptosis is a well-known pro-inflammatory PCD, and its involvement in epilepsy has been reported in animal models of epilepsy and also epileptic patients. Canonical (caspase-1-dependent) and non-canonical (caspase-1-independent) pathways are two main mechanisms implicated in pyroptotic cell death. Mouse caspase-11 or human analogues caspase-4/5 induce the non-canonical pathway. In both pathways, membrane gasdermin (GSDMD) pores contribute to pro-inflammatory cytokine release and lead to membrane destabilization and cell lysis. IL-1β and IL-18 are pro-inflammatory cytokines that are released following pyroptotic PCD. Brain inflammation increases excitability in the nervous system, promotes seizure activity, and is probably associated with the molecular and synaptic changes involved in epileptogenesis. Pro-inflammatory cytokines affect the glutamate and GABA neurotransmitter release as well as their receptors, thereby resulting in seizure activity. This review is intended to provide an overview of the current published works on pyroptotic cell death in epilepsy. The mechanisms by which pro-inflammatory cytokines, including IL-1β and IL-18 can promote epileptic discharges were also collected. According to this survey, since the involvement of pyroptosis in the development of epilepsy has been established, pyroptosis-targeted therapies may represent a novel anti-epileptogenic strategy.

Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Andoh T, Kishi H, Motoki K, Nakanishi K, Kuraishi Y, Muraguchi A (2008) Protective effect of IL-18 on kainate-and IL-1β-induced cerebellar ataxia in mice. J Immunol 180(4):2322–2328. https://doi.org/10.4049/jimmunol.180.4.2322
Argaw AT, Zhang Y, Snyder BJ, Zhao M-L, Kopp N, Lee SC, Raine CS, Brosnan CF, John GR (2006) IL-1β regulates blood-brain barrier permeability via reactivation of the hypoxia-angiogenesis program. J Immunol 177(8):5574–5584. https://doi.org/10.4049/jimmunol.177.8.5574
Assuncao Guimaraes C, Linden R (2004) Programmed cell deaths: apoptosis and alternative deathstyles. Eur J Biochem 271(9):1638–1650. https://doi.org/10.1111/j.1432-1033.2004.04084.x
Beghi E (2020) The epidemiology of epilepsy. Neuroepidemiology 54(2):185–191. https://doi.org/10.1159/000503831
Bergsbaken T, Fink SL, Cookson BT (2009) Pyroptosis: host cell death and inflammation. Nat Rev Microbiol 7(2):99–109. https://doi.org/10.1038/nrmicro2070
Bertheloot D, Latz E, Franklin BS (2021) Necroptosis, pyroptosis and apoptosis: an intricate game of cell death. Cell Mol Immunol 18(5):1106–1121. https://doi.org/10.1038/s41423-020-00630-3
Blamire A, Anthony D, Rajagopalan B, Sibson N, Perry V, Styles P (2000) Interleukin-1β-induced changes in blood–brain barrier permeability, apparent diffusion coefficient, and cerebral blood volume in the rat brain: a magnetic resonance study. J Neurosci 20(21):8153–8159. https://doi.org/10.1523/JNEUROSCI.20-21-08153.2000
Boucher D, Monteleone M, Coll RC, Chen KW, Ross CM, Teo JL, Gomez GA, Holley CL, Bierschenk D, Stacey KJ (2018) Caspase-1 self-cleavage is an intrinsic mechanism to terminate inflammasome activity. J Exp Med 215(3):827–840. https://doi.org/10.1084/jem.20172222
Broz P (2015) Caspase target drives pyroptosis. Nature 526(7575):642–643. https://doi.org/10.1038/nature15632
Castañeda-Cabral JL, Ureña-Guerrero ME, Beas-Zárate C, Colunga-Durán A, MdlA N-L, Orozco-Suárez S, Alonso-Vanegas M, Guevara-Guzmán R, Deli MA, Rocha L (2020) Increased expression of proinflammatory cytokines and iNOS in the neocortical microvasculature of patients with temporal lobe epilepsy. Immunol Res 68(3):169–176. https://doi.org/10.1007/s12026-020-09139-3
de Brito Toscano EC, Vieira ÉLM, Dias BBR, Caliari MV, Gonçalves AP, Giannetti AV, Siqueira JM, Suemoto CK, Leite REP, Nitrini R (2021) NLRP3 and NLRP1 inflammasomes are up-regulated in patients with mesial temporal lobe epilepsy and may contribute to overexpression of caspase-1 and IL-β in sclerotic hippocampi. Brain Res 1752:147230. https://doi.org/10.1016/j.brainres.2020.147230
DeSena AD, Do T, Schulert GS (2018) Systemic autoinflammation with intractable epilepsy managed with interleukin-1 blockade. J Neuroinflamm 15(1):38. https://doi.org/10.1186/s12974-018-1063-2
Dilena R, Mauri E, Aronica E, Bernasconi P, Bana C, Cappelletti C, Carrabba G, Ferrero S, Giorda R, Guez S (2019) Therapeutic effect of anakinra in the relapsing chronic phase of febrile infection–related epilepsy syndrome. Epilepsia Open 4(2):344–350. https://doi.org/10.1002/epi4.12317
Downs KP, Nguyen H, Dorfleutner A, Stehlik C (2020) An overview of the non-canonical inflammasome. Mol Asp Med 76:100924. https://doi.org/10.1016/j.mam.2020.100924
Fang Y, Tian S, Pan Y, Li W, Wang Q, Tang Y, Yu T, Wu X, Shi Y, Ma P (2020) Pyroptosis: a new frontier in cancer. Biomed Pharmacother 121:109595. https://doi.org/10.1016/j.biopha.2019.109595
Frank D, Vince JE (2019) Pyroptosis versus necroptosis: similarities, differences, and crosstalk. Cell Death Differ 26(1):99–114. https://doi.org/10.1038/s41418-018-0212-6
Fricker M, Tolkovsky AM, Borutaite V, Coleman M, Brown GC (2018) Neuronal cell death. Physiol Rev 98(2):813–880. https://doi.org/10.1152/physrev.00011.2017
Fu C-Y, He X-Y, Li X-F, Zhang X, Huang Z-W, Li J, Chen M, Duan C-Z (2015) Nefiracetam attenuates pro-inflammatory cytokines and GABA transporter in specific brain regions of rats with post-ischemic seizures. Cell Physiol Biochem 37(5):2023–2031. https://doi.org/10.1159/000438562
Fujikawa DG, Shinmei SS, Cai B (2000) Seizure-induced neuronal necrosis: implications for programmed cell death mechanisms. Epilepsia 41:S9–S13. https://doi.org/10.1111/j.1528-1157.2000.tb01549.x
He Y, Hara H, Núñez G (2016) Mechanism and regulation of NLRP3 inflammasome activation. Trends Biochem Sci 41(12):1012–1021. https://doi.org/10.1016/j.tibs.2016.09.002
Henshall DC, Simon RP (2005) Epilepsy and apoptosis pathways. J Cereb Blood Flow Metab 25(12):1557–1572. https://doi.org/10.1038/sj.jcbfm.9600149
Hong Y, Deng N, Jin H-N, Xuan Z-Z, Qian Y-X, Wu Z-y, Xie W (2018) Saikosaponin A modulates remodeling of Kv4. 2-mediated A-type voltage-gated potassium currents in rat chronic temporal lobe epilepsy. Drug Des Dev Ther 12:2945–2958. https://doi.org/10.2147/DDDT.S166408
Jiang Q, Tang G, Zhong XM, Ding DR, Wang H, Li JN (2021) Role of Stat3 in NLRP3/caspase-1-mediated hippocampal neuronal pyroptosis in epileptic mice. Synapse 75(12):e22221. https://doi.org/10.1002/syn.22221
Jorgensen I, Miao EA (2015) Pyroptotic cell death defends against intracellular pathogens. Immunol Rev 265(1):130–142. https://doi.org/10.1111/imr.12287
Jung HK, Ryu HJ, Kim M-J, Kim WI, Choi HK, Choi H-C, Song H-K, Jo S-M, Kang T-C (2012) Interleukin-18 attenuates disruption of brain–blood barrier induced by status epilepticus within the rat piriform cortex in interferon-γ independent pathway. Brain Res 1447:126–134. https://doi.org/10.1016/j.brainres.2012.01.057
Jyonouchi H, Geng L (2016) Intractable epilepsy (IE) and responses to anakinra, a human recombinant IL-1 receptor agonist. J Clin Cell Immunol 7(5):1000456. https://doi.org/10.4172/2155-9899.1000456
Kamali AN, Zian Z, Bautista JM, Hamedifar H, Hossein-Khannazer N, Hosseinzadeh R, Yazdani R, Azizi G (2021) The potential role of pro-inflammatory and anti-inflammatory cytokines in epilepsy pathogenesis. Endocr Metab Immune Disord-Drug Targets 21(10):1760–1774. https://doi.org/10.2174/1871530320999201116200940
Kang R, Lotze MT, Zeh HJ, Billiar TR, Tang D (2014) Cell death and DAMPs in acute pancreatitis. Mol Med 20(1):466–477. https://doi.org/10.2119/molmed.2014.00117
Kayagaki N, Stowe IB, Lee BL, O’Rourke K, Anderson K, Warming S, Cuellar T, Haley B, Roose-Girma M, Phung QT (2015) Caspase-11 cleaves gasdermin D for non-canonical inflammasome signalling. Nature 526(7575):666–671. https://doi.org/10.1038/nature15541
Kelley N, Jeltema D, Duan Y, He Y (2019) The NLRP3 inflammasome: an overview of mechanisms of activation and regulation. Int J Mol Sci 20(13):3328. https://doi.org/10.3390/ijms20133328
Kenney-Jung DL, Vezzani A, Kahoud RJ, LaFrance-Corey RG, Ho ML, Muskardin TW, Wirrell EC, Howe CL, Payne ET (2016) Febrile infection-related epilepsy syndrome treated with anakinra. Ann Neurol 80(6):939–945. https://doi.org/10.1002/ana.24806
Klapal L, Igelhorst BA, Dietzel-Meyer ID (2016) Changes in neuronal excitability by activated microglia: differential Na+ current upregulation in pyramid-shaped and bipolar neurons by TNF-α and IL-18. Front Neurol 7:44. https://doi.org/10.3389/fneur.2016.00044
Klement W, Garbelli R, Zub E, Rossini L, Tassi L, Girard B, Blaquiere M, Bertaso F, Perroy J, de Bock F (2018) Seizure progression and inflammatory mediators promote pericytosis and pericyte-microglia clustering at the cerebrovasculature. Neurobiol Dis 113:70–81. https://doi.org/10.1016/j.nbd.2018.02.002
Knodler LA, Crowley SM, Sham HP, Yang H, Wrande M, Ma C, Ernst RK, Steele-Mortimer O, Celli J, Vallance BA (2014) Noncanonical inflammasome activation of caspase-4/caspase-11 mediates epithelial defenses against enteric bacterial pathogens. Cell Host Microbe 16(2):249–256
Kolbrink B, Riebeling T, Kunzendorf U, Krautwald S (2020) Plasma membrane pores drive inflammatory cell death. Front Cell Dev Biol 8:817
Laflamme N, Lacroix S, Rivest S (1999) An essential role of interleukin-1β in mediating NF-κB activity and COX-2 transcription in cells of the blood–brain barrier in response to a systemic and localized inflammation but not during endotoxemia. J Neurosci 19(24):10923–10930
Lee VLL, Shaikh MF (2019) Inflammation: cause or consequence of epilepsy? Epilepsy-advances in diagnosis and therapy. IntechOpen, London
Lei Q, Yi T, Chen C (2018) NF-κB-Gasdermin D (GSDMD) axis couples oxidative stress and NACHT, LRR and PYD domains-containing protein 3 (NLRP3) inflammasome-mediated cardiomyocyte pyroptosis following myocardial infarction. Med Sci Monit 24:6044–6052. https://doi.org/10.12659/MSM.908529
Li Z, Zhao F, Cao Y, Zhang J, Shi P, Sun X, Zhang F, Tong L (2018) DHA attenuates hepatic ischemia reperfusion injury by inhibiting pyroptosis and activating PI3K/Akt pathway. Eur J Pharmacol 835:1–10. https://doi.org/10.1016/j.ejphar.2018.07.054
Li X, Lin J, Hua Y, Gong J, Ding S, Du Y, Wang X, Zheng R, Xu H (2021) Agmatine alleviates epileptic seizures and hippocampal neuronal damage by inhibiting gasdermin D-mediated pyroptosis. Front Pharmacol 12:627557. https://doi.org/10.3389/fphar.2021.627557
Liang Z, Damianou A, Di Daniel E, Kessler BM (2021) Inflammasome activation controlled by the interplay between post-translational modifications: emerging drug target opportunities. Cell Commun Signal 19(1):23. https://doi.org/10.1186/s12964-020-00688-6
Liston A, Masters SL (2017) Homeostasis-altering molecular processes as mechanisms of inflammasome activation. Nat Rev Immunol 17(3):208–214. https://doi.org/10.1038/nri.2016.151
Liu S, Du J, Li D, Yang P, Kou Y, Li C, Zhou Q, Lu Y, Hasegawa T, Li M (2020) Oxidative stress induced pyroptosis leads to osteogenic dysfunction of MG63 cells. J Mol Histol 51(3):221–232. https://doi.org/10.1007/s10735-020-09874-9
Mangan MS, Olhava EJ, Roush WR, Seidel HM, Glick GD, Latz E (2018) Targeting the NLRP3 inflammasome in inflammatory diseases. Nat Rev Drug Discov 17(8):588–606. https://doi.org/10.1038/nrd.2018.97
Marchi N, Fan Q, Ghosh C, Fazio V, Bertolini F, Betto G, Batra A, Carlton E, Najm I, Granata T (2009) Antagonism of peripheral inflammation reduces the severity of status epilepticus. Neurobiol Dis 33(2):171–181. https://doi.org/10.1016/j.nbd.2008.10.002
Maroso M, Balosso S, Ravizza T, Iori V, Wright CI, French J, Vezzani A (2011) Interleukin-1β biosynthesis inhibition reduces acute seizures and drug resistant chronic epileptic activity in mice. Neurotherapeutics 8(2):304–315. https://doi.org/10.1007/s13311-011-0039-z
Menu P, Vince J (2011) The NLRP3 inflammasome in health and disease: the good, the bad and the ugly. Clin Exp Immunol 166(1):1–15
Mishra AP, Salehi B, Sharifi-Rad M, Pezzani R, Kobarfard F, Sharifi-Rad J, Nigam M (2018) Programmed cell death, from a cancer perspective: an overview. Mol Diagn Ther 22(3):281–295. https://doi.org/10.1007/s40291-018-0329-9
Mohseni-Moghaddam P, Sadr SS, Roghani M, Arabzadeh S, Khamse S, Zamani E, Hosseini M, Moradi F (2019) Huperzine A ameliorates cognitive dysfunction and neuroinflammation in kainic acid-induced epileptic rats by antioxidant activity and NLRP 3/caspase-1 pathway inhibition. Clin Exp Pharmacol Physiol 46(4):360–372. https://doi.org/10.1111/1440-1681.13064
Mohseni-Moghaddam P, Roghani M, Khaleghzadeh-Ahangar H, Sadr SS, Sala C (2021) A literature overview on epilepsy and inflammasome activation. Brain Res Bull 172:229–235. https://doi.org/10.1016/j.brainresbull.2021.05.001
Motamedi G, Meador K (2003) Epilepsy and cognition. Epilepsy Behav 4(Suppl 2):S25-38. https://doi.org/10.1016/j.yebeh.2003.07.004
Pilla DM, Hagar JA, Haldar AK, Mason AK, Degrandi D, Pfeffer K, Ernst RK, Yamamoto M, Miao EA, Coers J (2014) Guanylate binding proteins promote caspase-11–dependent pyroptosis in response to cytoplasmic LPS. Proc Natl Acad Sci USA 111(16):6046–6051. https://doi.org/10.1073/pnas.1321700111
Ramazi S, Fahanik-Babaei J, Mohamadi-Zarch S-M, Tashakori-Miyanroudi M, Nourabadi D, Nazari-Serenjeh M, Roghani M, Baluchnejadmojarad T (2020) Neuroprotective and anticonvulsant effects of sinomenine in kainate rat model of temporal lobe epilepsy: involvement of oxidative stress, inflammation and pyroptosis. J Chem Neuroanat 108:101800. https://doi.org/10.1016/j.jchemneu.2020.101800
Rana A, Musto AE (2018) The role of inflammation in the development of epilepsy. J Neuroinflamm 15(1):144. https://doi.org/10.1186/s12974-018-1192-7
Rogers C, Erkes DA, Nardone A, Aplin AE, Fernandes-Alnemri T, Alnemri ES (2019) Gasdermin pores permeabilize mitochondria to augment caspase-3 activation during apoptosis and inflammasome activation. Nat Commun 10(1):1689. https://doi.org/10.1038/s41467-019-09397-2
Ruan J, Xia S, Liu X, Lieberman J, Wu H (2018) Cryo-EM structure of the gasdermin A3 membrane pore. Nature 557(7703):62–67. https://doi.org/10.1038/s41586-018-0058-6
Rühl S, Broz P (2015) Caspase-11 activates a canonical NLRP3 inflammasome by promoting K+ efflux. Eur J Immunol 45(10):2927–2936. https://doi.org/10.1002/eji.201545772
Schmid-Burgk JL, Gaidt MM, Schmidt T, Ebert TS, Bartok E, Hornung V (2015) Caspase-4 mediates non-canonical activation of the NLRP3 inflammasome in human myeloid cells. Eur J Immunol 45(10):2911–2917. https://doi.org/10.1002/eji.201545523
Sharma D, Kanneganti T-D (2016) The cell biology of inflammasomes: mechanisms of inflammasome activation and regulation. J Cell Biol 213(6):617–629. https://doi.org/10.1083/jcb.201602089
ShiZhaoWangShiWangHuangZhuangCaiWangShao JYKXYHYTFF (2015) Cleavage of GSDMD by inflammatory caspases determines pyroptotic cell death. Nature 526(7575):660–665. https://doi.org/10.1038/nature15514
Shi J, Gao W, Shao F (2017) Pyroptosis: gasdermin-mediated programmed necrotic cell death. Trends Biochem Sci 42(4):245–254. https://doi.org/10.1016/j.tibs.2016.10.004
Shorvon SD (2011) The etiologic classification of epilepsy. Epilepsia 52(6):1052–1057. https://doi.org/10.1111/j.1528-1167.2011.03041.x
Smith DB, Craft BR, Collins J, Mattson RH, Cramer JA (1986) Behavioral characteristics of epilepsy patients compared with normal controls. Epilepsia 27(6):760–768. https://doi.org/10.1111/j.1528-1157.1986.tb03607.x
Soltani Khaboushan A, Yazdanpanah N, Rezaei N (2022) Neuroinflammation and proinflammatory cytokines in epileptogenesis. Mol Neurobiol 59(3):1724–1743. https://doi.org/10.1007/s12035-022-02725-6
Strowig T, Henao-Mejia J, Elinav E, Flavell R (2012) Inflammasomes in health and disease. Nature 481(7381):278–286. https://doi.org/10.1038/nature10759
Tan C-C, Zhang J-G, Tan M-S, Chen H, Meng D-W, Jiang T, Meng X-F, Li Y, Sun Z, Li M-M (2015) NLRP1 inflammasome is activated in patients with medial temporal lobe epilepsy and contributes to neuronal pyroptosis in amygdala kindling-induced rat model. J Neuroinflamm 12(1):18. https://doi.org/10.1186/s12974-014-0233-0
Tang T, Lang X, Xu C, Wang X, Gong T, Yang Y, Cui J, Bai L, Wang J, Jiang W (2017) CLICs-dependent chloride efflux is an essential and proximal upstream event for NLRP3 inflammasome activation. Nat Commun 8(1):202. https://doi.org/10.1038/s41467-017-00227-x
Vezzani A, Moneta D, Conti M, Richichi C, Ravizza T, De Luigi A, De Simoni M, Sperk G, Andell-Jonsson S, Lundkvist J (2000) Powerful anticonvulsant action of IL-1 receptor antagonist on intracerebral injection and astrocytic overexpression in mice. Proc Natl Acad Sci USA 97(21):11534–11539. https://doi.org/10.1073/pnas.190206797
Vezzani A, Balosso S, Maroso M, Zardoni D, Noé F, Ravizza T (2010) ICE/caspase 1 inhibitors and IL-1beta receptor antagonists as potential therapeutics in epilepsy. Curr Opin Investig Drugs 11(1):43–50
Vezzani A, French J, Bartfai T, Baram TZ (2011) The role of inflammation in epilepsy. Nat Rev Neurol 7(1):31–40. https://doi.org/10.1038/nrneurol.2010.178
Vezzani A, Aronica E, Mazarati A, Pittman QJ (2013a) Epilepsy and brain inflammation. Exp Neurol 244:11–21. https://doi.org/10.1016/j.expneurol.2011.09.033
Vezzani A, Friedman A, Dingledine RJ (2013b) The role of inflammation in epileptogenesis. Neuropharmacology 69:16–24. https://doi.org/10.1016/j.neuropharm.2012.04.004
Volchuk A, Ye A, Chi L, Steinberg BE, Goldenberg NM (2020) Indirect regulation of HMGB1 release by gasdermin D. Nat Commun 11(1):4561. https://doi.org/10.1038/s41467-020-18443-3
Wang Y, Jin S, Sonobe Y, Cheng Y, Horiuchi H, Parajuli B, Kawanokuchi J, Mizuno T, Takeuchi H, Suzumura A (2014) Interleukin-1β induces blood–brain barrier disruption by downregulating sonic hedgehog in astrocytes. PLoS ONE 9(10):e110024. https://doi.org/10.1371/journal.pone.0110024
Wree A, Eguchi A, McGeough MD, Pena CA, Johnson CD, Canbay A, Hoffman HM, Feldstein AE (2014) NLRP3 inflammasome activation results in hepatocyte pyroptosis, liver inflammation, and fibrosis in mice. Hepatology 59(3):898–910. https://doi.org/10.1002/hep.26592
Wu J, Chen L, Wang S, Li Y, Liu L, Chen G, Wang S, Zhou S (2019) Negative regulation of autophagy in activating nucleotide-binding oligomerization domain-like receptor family pyrin domain-containing 3 inflammasomes in the hippocampus of an epilepsy rat model. Nanosci Nanotechnol Lett 11(7):947–959. https://doi.org/10.1166/nnl.2019.2958
Xia S, Yang P, Li F, Yu Q, Kuang W, Zhu Y, Lu J, Wu H, Li L, Huang H (2021) Chaihu-longgu-muli decoction exerts an antiepileptic effect in rats by improving pyroptosis in hippocampal neurons. J Ethnopharmacol 270:113794. https://doi.org/10.1016/j.jep.2021.113794
Xia L, Liu L, Cai Y, Zhang Y, Tong F, Wang Q, Ding J, Wang X (2022a) Inhibition of gasdermin D-mediated pyroptosis attenuates the severity of seizures and astroglial damage in kainic acid-induced epileptic mice. Front Pharmacol 12:751644. https://doi.org/10.3389/fphar.2021.751644
Xia L, Liu L, Wang Q, Ding J, Wang X (2022) Relationship between the pyroptosis pathway and epilepsy: a bioinformatic analysis. Front Neurol 12:782739. https://doi.org/10.3389/fneur.2021.782739
Xiang-Tao Z, Wei-Wei C, Fang-Chen C, Ying C, Er-Zhen C (2021) The role and mechanism of pyroptosis and potential therapeutic targets in sepsis: a review. Front Immunol 12:711939. https://doi.org/10.3389/fimmu.2021.711939
Xiao Z, Peng J, Wu L, Arafat A, Yin F (2017) The effect of IL-1β on synaptophysin expression and electrophysiology of hippocampal neurons through the PI3K/Akt/mTOR signaling pathway in a rat model of mesial temporal lobe epilepsy. Neurol Res 39(7):640–648. https://doi.org/10.1080/01616412.2017.1312070
Xiaoqin Z, Zhengli L, Changgeng Z, Xiaojing W, Li L (2005) Changes in behavior and amino acid neurotransmitters in the brain of rats with seizure induced by IL-1β or IL-6. J Huazhong Univ Sci Tech 25(3):236–239. https://doi.org/10.1007/BF02828129
Xu S, Li X, Liu Y, Xia Y, Chang R, Zhang C (2019) Inflammasome inhibitors: promising therapeutic approaches against cancer. J Hematol Oncol 12(1):64. https://doi.org/10.1186/s13045-019-0755-0
Xu Z, Chen Z-m, Wu X, Zhang L, Cao Y, Zhou P (2020) Distinct molecular mechanisms underlying potassium efflux for NLRP3 inflammasome activation. Front Immunol 11:609441. https://doi.org/10.3389/fimmu.2020.609441
Yabal M, Calleja DJ, Simpson DS, Lawlor KE (2019) Stressing out the mitochondria: mechanistic insights into NLRP3 inflammasome activation. J Leukoc Biol 105(2):377–399. https://doi.org/10.1002/JLB.MR0318-124R
Yang J, Zhao Y, Shao F (2015) Non-canonical activation of inflammatory caspases by cytosolic LPS in innate immunity. Curr Opin Immunol 32:78–83. https://doi.org/10.1016/j.coi.2015.01.007
Yang Y, Wang H, Kouadir M, Song H, Shi F (2019) Recent advances in the mechanisms of NLRP3 inflammasome activation and its inhibitors. Cell Death Dis 10(2):128. https://doi.org/10.1038/s41419-019-1413-8
Ye M, Bi Y-F, Ding L, Zhu W-W, Gao W (2016) Saikosaponin a functions as anti-epileptic effect in pentylenetetrazol induced rats through inhibiting mTOR signaling pathway. Biomed Pharmacother 81:281–287. https://doi.org/10.1016/j.biopha.2016.04.012
Ye S, Zheng Q, Zhou Y, Bai B, Yang D, Zhao Z (2019) Chlojaponilactone B attenuates lipopolysaccharide-induced inflammatory responses by suppressing TLR4-mediated ROS generation and NF-κB signaling pathway. Molecules 24(20):3731. https://doi.org/10.3390/molecules24203731
Yu P, Zhang X, Liu N, Tang L, Peng C, Chen X (2021) Pyroptosis: mechanisms and diseases. Signal Transduct Targ Ther 6(1):128. https://doi.org/10.1038/s41392-021-00507-5
Zhang R, Sun L, Hayashi Y, Liu X, Koyama S, Wu Z, Nakanishi H (2010a) Acute p38-mediated inhibition of NMDA-induced outward currents in hippocampal CA1 neurons by interleukin-1β. Neurobiol Dis 38(1):68–77. https://doi.org/10.1016/j.nbd.2009.12.028
Zhang X-M, Jin T, Quezada HC, Mix E, Winblad B, Zhu J (2010b) Kainic acid-induced microglial activation is attenuated in aged interleukin-18 deficient mice. J Neuroinflamm 7(1):26. https://doi.org/10.1186/1742-2094-7-26
Zhao LR, Xing RL, Wang PM, Zhang NS, Yin SJ, Li XC, Zhang L (2018) NLRP1 and NLRP3 inflammasomes mediate LPS/ATP-induced pyroptosis in knee osteoarthritis. Mol Med Rep 17(4):5463–5469. https://doi.org/10.3892/mmr.2018.8520
Zhu H, Jian Z, Zhong Y, Ye Y, Zhang Y, Hu X, Pu B, Gu L, Xiong X (2021) Janus kinase inhibition ameliorates ischemic stroke injury and neuroinflammation through reducing NLRP3 inflammasome activation via JAK2/STAT3 pathway inhibition. Front Immunol 12:714943. https://doi.org/10.3389/fimmu.2021.714943
Acknowledgements
This study did not receive any financial support.
Funding
This study did not receive any financial support.
Author information
Authors and Affiliations
Contributions
PMM contributed to the study conception and design. The first draft of the manuscript was written by HKA (the introduction part), and PMM (all of the other parts). RA commented on previous versions of the manuscript. He provided valuable scientific and linguistic editing of the manuscript. NE contributed to the designing of the figure. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that they have no conflict of interest.
Ethical Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Atabaki, R., Khaleghzadeh-Ahangar, H., Esmaeili, N. et al. Role of Pyroptosis, a Pro-inflammatory Programmed Cell Death, in Epilepsy. Cell Mol Neurobiol 43, 1049–1059 (2023). https://doi.org/10.1007/s10571-022-01250-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-022-01250-3