Abstract
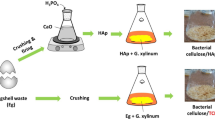
In the present work, bionanocomposites based on bacterial cellulose (BC) obtained from alternative sources (cashew juice and sisal liquid waste) and hydroxyapatite (HA) were developed. BC–HA composites were prepared through alternate immersion in CaCl2 and Na2HPO4 solutions. Cellulose was successfully produced from the alternative sources of media without the need for additional supplementation and HA crystals that homogeneously precipitated onto the BC surface. The Ca/P ratio ranged from 1.53 to 1.58, indicating the presence of calcium-deficient HA in the composites; this is a phase similar to biological apatite. After immersion into synthetic body fluid, the HA layer formed on the surface of pure BC and the composites, attesting the material’s bioactivity. Moreover, apatite deposition on the composites was up to three times higher than observed on pure cellulose with no significant desorption of apatite from the composites. These results support that the BC derived from agroindustrial wastes have potential to produce nanocomposites of cellulose/HA for use in bone tissue regeneration.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Annually, millions of people suffer from bone defects caused by trauma, tumors, or bone related diseases. Auto- and allografts bones have traditionally been the main sources for bone substitutes, in which autografts have the most osteogenic potential followed by allografts. However, the former is limited by donor-site morbidity and supply, whereas the latter may carry the risk of disease transmission and immune response (Moore et al. 2001). These concerns have led to the search for alternatives through the development of synthetic grafts.
The ideal bone graft must meet a number of requirements. They must provide enough mechanical support without being overly stiff, be capable of integrating with the host bony tissue, be biocompatible, enhance bone regeneration, and improve the biomechanical stability of the treated bony defects (Giannoudis et al. 2005; Janicki and Schmidmaier 2011). Hydroxyapatite (HA) is widely used as a biomaterial in various biomedical applications due to their biocompatibility and osteoinductive properties; several studies revealed that nanocomposites of cellulose/HA exhibit many of these required properties, and are thus promising for applications in bone regeneration (Fang et al. 2009; Fricain et al. 2002; Grande et al. 2009; He et al. 2012; Nge et al. 2010; Saska et al. 2011; Zadegan et al. 2011; Zimmermann et al. 2011).
Bacterial cellulose (BC) produced by Acetobacter organisms is a biomaterial that has garnered interest in the field of tissue engineering due to its unique properties, including high water holding capacity, crystallinity, purity, and tensile strength. The in vivo biocompatibility of BC was also evaluated (Andrade et al. 2012; Helenius et al. 2006; Pértile et al. 2012; Schumann et al. 2009; Wippermann et al. 2009). Although chemically identical to plant cellulose, BC has a unique ultrafine fibrillar nanostructure, which determines its superior physical and mechanical properties. Moreover, BC is also distinguished amongst plant-derived celluloses in that it is environmentally safer. Plant-derived cellulose usually requires the removal of hemicellulose and lignin, which cause environmental damage from the use of environmentally harsh purifying techniques like Kraft pulping (Andrade et al. 2010). BC is usually produced using synthetic culture media that contains glucose as a main carbon source and other nutrients; these results in high production costs, limiting the use of this material (Gomes et al. 2013; Wu and Liu 2013). Culture medium may represent about 30 % of the total cost of microbial fermentation processes (Rivas et al. 2004).
Therefore, the use of cheaper nutrient sources, such as agroindustrial residues, is a strategy to overcome this limitation (Gomes et al. 2013; Cavka et al. 2013), favoring large-scale, low cost production and reducing the environmental impact (Huang et al. 2014). Therefore, the aim of this work was the development of a bionanocomposite based on HA and BC from alternative sources, including cashew juice and sisal juice (sisal liquid waste).
Experimental section
Microorganism and bacterial cellulose production from agroindustrial wastes
Gluconacetobacter hansenii (CCT 1431) purchased from Coleção de Culturas Tropical da Fundação André Tosello (Campinas-SP, Brazil) was used as the BC producing bacteria. Sisal and cashew juices were provided by Embrapa Algodão (Campina Grande-PB, Brazil) and Embrapa Agroindústria Tropical (Pacajus-CE, Brazil), respectively. The agroindustrial materials were filtered through a commercial fabric filter to remove debris and autoclaved (121 °C, 15 min). The pH and °Brix values from the juices were determined before bacterial cultivation. The synthetic medium (Hestrin and Schramm 1954) was used as reference medium.
The inoculum was cultivated in HS medium at 30 °C for 48 h. The reference and alternative media were cultured with 3 % (v/v) of the inoculum and statically incubated at 30 °C for 7 days. After fermentation, BC pellicles were purified through immersion in 2 % (w/v) SDS (200 mL, 25 °C, 1 min), then washed with water, followed by 4 % (w/v) NaOH treatment (600 mL, at 80 °C, 30 min). After purification, the BC pellicles were washed several times with distilled water until neutral pH was achieved.
Preparation of bacterial cellulose/hydroxyapatite nanocomposites
The BC–HA composites were prepared according to Hutchens et al. (2006). After purification, the BC pellicles (21 × 13 cm), with thickness of about 15 µm, were immersed in 800 mL of CaCl2 solution (11 g L−1) and mildly stirred at 30 °C for 24 h. Thereafter, for the removal of excess CaCl2, the pellicles were washed with deionized water, followed by immersion in 800 mL of Na2HPO4 solution (8.52 g L−1) using the same conditions previously described. This alternate soaking process (immersion in calcium and phosphate solutions) was repeated six times with the samples rinsed thoroughly with deionized water between each soaking cycle (~500 mL of deionized water for 10 min at 25 °C). Finally, these nanocomposites were removed, dried in an air oven at 70 °C to a constant weight and stored in a desiccator until use.
The nanocomposites obtained from the BC pellicles produced in HS, sisal liquid, and cashew juice were designated as HS-HA, Sisal-HA and Cashew-HA, respectively.
Thermal analyses (TGA/dTG)
Thermal analyses (TGA and dTG) were performed on a thermogravimetric analyzer (Shimadzu, TGA STA 6000), with 5–7 mg of dried samples in the range of 0–700 °C under a nitrogen atmosphere (40 mL/min) at a heating rate of a 10 °C min−1 to evaluate their thermal stability.
X-ray diffraction
X-ray diffraction analyses were performed on an X-ray diffractometer (Rigaku, DMAXB), using CuKα radiation. The diffraction data were collected in the 2θ range from 10° to 40° at a scanning speed of 0.5° min−1. The crystallinity index (CI) is a measure of the percentage of crystalline material in a given sample and it is also correlated to the degree of order within the crystals. Cellulose crystallinity index was determined as described by Segal et al. (1959), and the HA crystalline index was calculated using the Landi et al. (2000) method (Segal et al. 1959; Landi et al. 2000). The crystalline species (cellulose and HA) were identified using the X-pert Highscore 1.0 and comparison of the XRD pattern of each sample on the database (JCPDS).
Fourier transform infrared spectroscopy (FTIR) studies
Samples were examined using a Fourier transform infrared spectrometer (FTIR, Nicolet 800 combined with a MTech PAS cell). Samples were ground and pelletized using KBr (1:10, w/wt), followed by uniaxillary pressure under vacuum. Spectra were obtained between 4000 and 400 cm−1 at a 4 cm−1 of resolution.
SEM and EDS
Cellulose pellicles and CB-HA composites were analyzed by scanning electron microscopy (SEM) on a Zeiss DSM-940A microscope at 30 kV after gold-sputtering treatment. The average diameters of the nanofibers and globular crystals of at least 50 structures were determined using the ImageJ program (National Institute of Health-NIH). Energy dispersive X-ray spectroscopy (EDS) was used to determine the C/O molar ratio from purified BC pellicles and the Ca/P molar ratio from the composites.
Desorption study
Desorption was assessed by measuring the variation of weight loss, attributed to apatite release, from the composite. Samples that were 1 cm2 in size, were weighed and immersed in 0.1 mol L−1 phosphate buffered solution (PBS) at pH 7.25 and 37 °C for 30 days. After, the samples were washed with deionized water, dried in an air oven at 70 °C to a constant weight.
Bioactivity assessment
A bioactivity assay was developed using the synthetic body fluid (SBF) technique described by Bohner and Lemaitre (2009). Nanocomposite samples (4 cm2) were completely immersed in 40 mL of SBF at 37 °C for 14 days. The samples were then dried in an air oven at 70 °C to a constant weight. The amount of deposited apatite on the composite surface was determined by the mass difference between the initial and the final mass.
Protein (BSA) adsorption
BSA Adsorption onto the composite materials was performed as described by Bundela and Bharadwaj (2012). Fresh BSA solutions (0.5 mg mL−1) were prepared in 0.5 M PBS (Phosphate Buffer Solution) at pH 7.4. The composite samples (200 mg) were equilibrated with PBS for 24 h and then gently shaken for 120 min. The composites were removed, and the concentration of non-adsorbed protein in the liquid medium was measured by recording the absorbance of protein solution at 272 nm with a UV spectrophotometer (Cirrus 60 ST, Femto) and quantified using a BSA standard curve.
Statistical analysis
The statistical analysis was carried out using the software OriginPro 7.0 (OriginLab Corporation). Each assay was conducted in triplicate. The statistical significance of the evaluated data was analyzed by one-way analysis of variance (ANOVA) and an unpaired F-test. The values were considered significant when α < 0.05.
Results and discussion
Bacterial cellulose production from agroindustrial wastes
The agroindustrial wastes had a combination of °Brix and pH close to the HS medium (Table 1).
In the °Brix analysis for sugar content estimation, the nearly equivalent values between the alternative media and the standard medium suggest the existence of carbohydrates in the cashew and sisal juices that can be used by G. hansenii for the metabolic process. Both juices maintained a pH close to 5, which was within the optimum pH range (4–6) for Gluconacetobacter growth (Chawla et al. 2009). After the production and purification, all the pellicles were gelatinous, resistant to handling, and translucence. The yield of BC production from HS and sisal juice was about the same (0.54 g L−1) as well as their microfibrillar network structure. BC obtained from cashew juice presented the minor yield (~0.28 g L−1) with loosely network structure.
Thermal analyses (TGA/dTG)
The first instance of mass loss for the BC pellicles (4–6 %) and for the BC–HA composites (6–11 %) occurred around 100 °C due to the evaporation of water adsorbed on the cellulose. Between 280 and 310 °C (Table 2), there was another mass loss event caused by the cellulose degradation processes (depolymerization, dehydration, and decomposition of the glycoside units) followed by carbonic residue formation (Saska et al. 2011; Barud et al. 2007, 2008a; Wan et al. 2007) Although this thermal event is the main characteristic of cellulose degradation, these mass losses are not fixed values, and they depend on the cellulosic source (Uesu et al. 2000). In general, the HA has thermal stability in this temperature range. The decomposition of HA is usually accompanied by a phase change, and tends to be evidenced at temperatures above 700 °C, which leads to a high amount of residues. After the analysis, a carbonaceous residue around 24.8 % was observed for BC samples, whereas the BC–HA composites exhibited a residue around 68.1 %. This evidence confirms HA deposition on the BC membrane, and the HA content was estimated to be around 43.3 %. Table 2 presents the percentages of the 2nd mass loss (2nd event) and their respective temperatures for pure BC and BC–HA composites (Fig. 1).
X-ray diffraction
The BC (Fig. 2) displayed three diffraction peaks at 2θ = 15°, 17°, and 22.5°, which are usually attributed to the interplanar distance characteristic of Iα and Iβ phases of elementary crystalline structure of cellulose. Planes (100)Iα, (110)Iβ, and (010)Iβ are related to the angle of 15°, whereas the planes (110)Iα and (200)Iβ are associated to the angle 22.5° (Saska et al. 2011; Barud et al. 2008b). The absence of a high-intensity peak at 34.5° indicates that the sample is mainly composed of Iα cellulose. The CI of the BC pellicles were 77.4 % for HS, 69.3 % for cashew, and 72.7 % for sisal. These values are within the expected crystallinity range (Andrade et al. 2010).
In the BC–HA composites, there were peaks at 2θ = 15° and 22.5°, as similar to the neat pellicles (Barud et al. 2008b), while the main characteristic diffraction peaks of HA were observed at 2θ = 29°, 32°, 40°, and 51° (Fig. 2). The crystallinity of HA in the composites was 38.3 % (HS-HA), 23.4 % (Cashew-HA), and 27.8 % (Sisal-HA). The poor crystalline apatite formation by biomimetic route is in agreement with literature (Roveri et al. 2010).
Fourier transform infrared spectroscopy (FTIR) studies
Expected cellulose vibrational bands were found in the FTIR spectra (Fig. 3; Table 3) of the BC pellicles and BC–HA composites. The pellicle purification was effective, since no additional bands, which could be related to contaminant substances, were detected.
The band in the region 3500–3200 cm−1, assigned to cellulose hydroxyl groups, was observed to have decreasing relative intensity and band broadening for the composite in comparison with the pure BC membrane. This evidence suggests that the presence of HA crystals affects the cellulose hydroxyl groups. Moreover, deviation of the band assigned to intramolecular hydrogen bonding (~3500 cm−1) confirms the strong interaction between the OH groups and apatite. It is important to emphasize that the presence of remanescent water molecules may also have some influence on broadening of OH region.
The characteristic bands of phosphate group (PO4 3−) at 569–573, 601–603, and 1030 cm−1 (Table 3) indicate the formation of HA (Zhai et al. 2005; Barrère et al. 2006). The presence of the PO4 3− doublet band at 602 and 561 cm−1 suggests that the precursor phase of the HA was octacalcium phosphate (OCP), Ca8H2(PO4)6·5H2O (Saska et al. 2011). OCP has a chemical structure close to HA, Ca10(PO4)6(OH)2 and is considered a biological precursor of HA. Several studies have been performed to understand the role played by OCP in the formation of the mineral phase of calcified tissues and in the processes of bone remodeling (Suzuki 2010; Elliot 1994). Moreover, the band, which was ~877 cm−1, corresponds to the stretching mode of CO3 2− ions, suggesting absorption of CO2 from the air (Saska et al. 2011). Thus, the FTIR peaks at around 872–874 cm−1 indicate carbonated apatite deposition on BC substrates.
Generally, there were no differences among the BC spectra and among the BC–HA composite spectra. This observation confirms the similarities between the BC from different media and the similarities between the HA deposition on all BC nanofibers.
SEM and EDS
A typical dry BC membrane is depicted in Fig. 4a with a continuous network containing ultra-thin fibers (nanocellulose). The mean fiber diameter in BC pellicles was measured as about 92.2 ± 4.2 nm (HS), 92.3 ± 4.7 nm (Sisal) and 84.5 ± 3.6 nm (Cashew) as measured using ImageJ. The SEM images confirmed the characteristic three-dimensional BC structure, as well as the absence of contaminants. Moreover, there were no significant differences between the membranes from different media.
This nanostructure provides a large surface area for stabilization of adsorbed particles on it (Saska et al. 2011). The SEM images of the dry membranes of BC–HA nanocomposites (Fig. 4b–d) depict typical clustered crystallites, as described by others authors (Morejón-Alonso et al. 2007, 2008), precipitated on the BC. Although the HA crystal exhibited a larger particle size distribution (Fig. 4c), most of them were in the microscale (~2 µm).
From all the EDS analysis of the BC pellicles (Table 4), the major components were carbon (~53 % w/wt) and oxygen (~44 %) with some minor content of adsorbed ions from the culture media and washing solutions. The BC–HA nanocomposites presented a Ca/P mass ratio ranging from 1.53 to 1.58. The ratio of Ca/P is one of the methods used to characterize the composition of the different calcium phosphates, especially apatite, which may vary in composition. The formula of stoichiometric HA is Ca10(PO4)6(OH)2, with a Ca/P mass ratio around 1.67. The Hutchens technique (Hutchens et al. 2006) involves the production of non-stoichiometric HA, reflecting the mineral formation of a calcium-deficient specimen.
The EDS, XRD, and FTIR results indicate that the deposited material is calcium-deficient carbonate-containing hydroxyapatite (CDHA), which is a similar phase to the biological apatite. This phase is desirable to facilitate the proliferation of osteoblast cells, which will synthesize the organic component for bone matrix regeneration (Le Guéhennec et al. 2004).
Desorption study
After 30 days of saline solution immersion, all the BC–HA nanocomposites presented no statistical difference (p value > 0.05) between the initial and the final mass (Table 5). Thus, there is no sign of HA desorption from the composite. Loose particles can lead to particle migration which may result in damage to surrounding tissues (Mohamed 2012).
Kim et al. (2004) observed a weight loss of up to 25 % after 160 h of in vitro degradation of a HA/poly(ε-caprolactone) composite (Kim et al. 2004), while Alge et al. (2012) noted that the dicalcium phosphate dihydrate cement had a mass loss between 5 and 20 % in 2 weeks. These findings provide evidence that BC could hold the HA crystals in a way that prevented leaching (Alge et al. 2012).
Bioactivity assessment
The formation of a HA layer on the surface of composite materials after immersion in SBF (Fig. 5; Table 5) attests the composite bioactivity. BC films are smooth (Fig. 5a, d, and g), in contrast to BC–HA composites (Fig. 5b, e, and h), which have a rough and granular surface. After immersion in the SBF (Fig. 5c, f, and i), the composites appear to be less granular and more smooth, because of the formation of the HA layer, increasing the crystal size and cementing the voids between the granules. Materials that exhibit this property have potential for use in bone tissue regeneration (Coleman et al. 2007).
Hydroxyapatite crystals can act as nucleation points, helping ion precipitation and increasing the mass of the composite. The mass gain in the BC–HA (8.4–9.2 mg) was superior to the BC pellicles (2.9–3.8 mg). Moreover, the percent increment was superior in the BC pellicles made from sisal juice (21.5 %) and cashew juice (23.1 %) than in the BC from HS medium (17.8 %). This corroborates the notion that G. hansenii produced similar materials in synthetic and natural media.
Protein (BSA) adsorption
BSA adsorption in the BC–HA nanocomposites (Table 5) indicates the biomaterial from sisal juice had a higher index of protein adsorption, followed by HS and cashew juice. When a foreign material comes into contact with the body, proteins will immediate adsorb on its surface, and the adsorbed protein layer will determine future events, such as platelet adhesion, aggregation, or inflammatory response (Wang et al. 2012). Basu et al. (2013) observed an adsorption of about 51.3 mg of BSA per scaffold gram (Basu et al. 2013), similar to the data from this article, and Swain and Sarkar (2013) verified an adsorption between 23 and 28 % mg/g in different kinds of HA crystals (Swain and Sarkar 2013).
The tissue response to implanted materials arises mainly from differences in the adsorption of proteins that, in turn, depend on parameters of the surface topography, such as roughness, porosity, and pore size, as well as chemical composition among others factors. The BSA deposition in the BC–HA is due to the electrostatic interaction between the Ca2+ and the PO4 3− of HA with the COO− and the NH3 + of the BSA protein, respectively. All the nanocomposites presented good protein deposition properties, indicating possible osteoinductivity capacity.
Conclusion
Bacterial celluloses from agro-waste juices were similar to each other and to the standard culture medium. The HA nanocomposites with celluloses also did not exhibit substantial variation. HA was deposited with some uniformity in the spaces between the fibers of biopolymers and exhibited similar chemical characteristics to biological apatite. Preliminary in vitro studies showed that materials were biocompatible. In general, the BC membranes proved to be suitable for the preparation of nanocomposites for applications in the biomedical field.
References
Alge DL, Goebel WS, Chu TMG (2012) In vitro degradation and cytocompatibility of dicalcium phosphate dihydrate cements prepared using the monocalcium phosphate monohydrate/hydroxyapatite system reveals rapid conversion to HA as a key mechanism. J Biomed Mater Res B Appl Biomater 100B:595–602
Andrade FK, Pértile RAN, Dourado F, Gama FMP (2010) Bacterial cellulose: properties, production and applications. In: Lejeune A, Deprez T (eds) Cellulose: structure and properties, derivatives and industrial uses. Nova Science Publishers, Hauppage, pp 427–458
Andrade FK, Alexandre N, Amorim I, Gartner F, Maurício AC, Luís AL et al (2012) Studies on the biocompatibility of bacterial cellulose. J Bioact Compat Polym 28:97–112
Barrère F, Blitterswijk CA, Groot K (2006) Bone regeneration: molecular and cellular interactions with calcium phosphate ceramics. Int J Nanomed 1:317–332
Barud HS, Ribeiro C, Crespi M, Martines M, Dexpert-Ghys J, Marques R et al (2007) Thermal characterization of bacterial cellulose-phosphate composites membranes. J Therm Anal Calorim 87:815–818
Barud HS, Assunção RMN, Martines MAU, Dexpert-Ghys J, Marques RFC, Messaddeq Y et al (2008a) Bacterial cellulose-silica organic-inorganic hybrids. J Sol Gel Sci Technol 46:363–367
Barud HS, de Araújo Júnior AM, Santos DB, de Assunção RMN, Meireles CS, Cerqueira DA et al (2008b) Thermal behavior of cellulose acetate produced from homogeneous acetylation of bacterial cellulose. Thermochim Acta 471:61–69
Basu B, Swain SK, Sarkar D (2013) Cryogenically cured hydroxyapatite-gelatin nanobiocomposite for bovine serum albumin protein adsorption and release. RSC Adv 3:14622–14633
Bohner M, Lemaitre J (2009) Can bioactivity be tested in vitro with SBF solution? Biomaterials 30:2175–2179
Bundela H, Bharadwaj V (2012) Synthesis and characterization of hydroxyapatite poly (vinyl alcohol) based nanocomposites for their perspective use as bone substitutes. Polym Sci Ser A 54:299–309
Cavka A, Guo X, Shui-Jia T, Winestrand S, Jönsson LJ, Hong F (2013) Production of bacterial cellulose and enzyme from waste fiber sludge. Biotechnol Biofuels 6:1–10
Chawla PR, Bajaj IB, Survase SA, Singhal RS (2009) Microbial cellulose: fermentative production and applications. Food Technol Biotechnol 47:107–124
Coleman NJ, Nicholson JW, Awosanya K (2007) A preliminary investigation of the in vitro bioactivity of white Portland cement. Cement Concrete Res 37:1518–1523
Elliot JC (1994) Structure and chemistry of the apatite and other calcium orthophosphates, 1st edn. Elsevier, Amsterdam
Fang B, Wan YZ, Tang TT, Gao C, Dai KR (2009) Proliferation and osteoblastic differentiation of human bone marrow stromal cells on hydroxyapatite/bacterial cellulose nanocomposite scaffolds. Tissue Eng Part A 15:1091–1098
Fricain JC, Granja PL, Barbosa MA, de Jeso B, Barthe N, Baquey C (2002) Cellulose phosphates as biomaterials. In vivo biocompatibility studies. Biomaterials 23:971–980
Giannoudis PV, Dinopoulos H, Tsiridis E (2005) Bone substitutes: an update. Injury 36:S20–S27
Gomes FP, Silva NHCS, Trovatti E, Serafim LS, Duarte MF, Silvestre AJD et al (2013) Production of bacterial cellulose by Gluconacetobacter sacchari using dry olive mill residue. Biomass Bioenerg 55:205–211
Grande CJ, Torres FG, Gomez CM, Carmen Baño MC (2009) Nanocomposites of bacterial cellulose/hydroxyapatite for biomedical applications. Acta Biomater 5:1605–1615
He M, Chang C, Peng N, Zhang L (2012) Structure and properties of hydroxyapatite/cellulose nanocomposite films. Carbohydr Polym 87:2512–2518
Helenius G, Backdahl H, Bodin A, Nannmark U, Gatenholm P, Risberg B (2006) In vivo biocompatibility of bacterial cellulose. J Biomed Mater Res A 76A:431–438
Hestrin S, Schramm M (1954) Synthesis of cellulose by acetobacter-xylinum.2. Preparation of freeze-dried cells capable of polymerizing glucose to cellulose. Biochem J 58:345–352
Huang Y, Zhu C, Yang J, Nie Y, Chen C, Sun D (2014) Recent advances in bacterial cellulose. Cellulose 21:1–30
Hutchens SA, Benson RS, Evans BR, O’Neill HM, Rawn CJ (2006) Biomimetic synthesis of calcium-deficient hydroxyapatite in a natural hydrogel. Biomaterials 27:4661–4670
Janicki P, Schmidmaier G (2011) What should be the characteristics of the ideal bone graft substitute? Combining scaffolds with growth factors and/or stem cells. Injury 42:S77–S81
Kim HW, Knowles JC, Kim HE (2004) Hydroxyapatite/poly(e-caprolactone) composite coatings on hydroxyapatite porous bone scaffold for drug delivery. Biomaterials 25:1279–1287
Landi E, Tampieri A, Celotti G, Sprio S (2000) Densification behaviour and mechanisms of synthetic hydroxyapatites. J Eur Ceram Soc 20:2377–2387
Le Guéhennec L, Layrolle P, Daculsi G (2004) A review of bioceramics and fibrin sealant. Eur Cell Mater 8:1–10
Mohamed KR (2012). Biocomposite materials, composites and their applications. In: Prof. Ning H (ed.) InTech, pp 113–146
Moore WR, Graves SE, Bain GI (2001) Synthetic bone graft substitutes. ANZ J Surg 71:354–361
Morejón-Alonso L, Carrodeguas RG, García-Menocal JAD, Pérez JAA, Manent SM (2007) Effect of sterilization on the properties of CDHA-OCP-β-TCP biomaterial. Mater Res 10:15–20
Morejón-Alonso L, Carrodeguas RG, García-Menocal JAD (2008) Transformations in CDHA/OCP/β-TCP scaffold during ageing in simulated body fluid at 36.5 °C. J Biomed Mater Res B Appl Biomater 84B:386–393
Nge TT, Sugiyama J, Bulone V (2010) Bacterial cellulose-based biomimetic composites. In: Elnashar M (ed) Biopolymers. InTech, Rijeka, pp 345–368
Pértile RA, Moreira S, Costa RM, Correia A, Guardao L, Gartner F et al (2012) Bacterial cellulose: long-term biocompatibility studies. J Biomater Sci Polym 23:1339–1354
Rivas B, Moldes AB, Dominguez JM, Parajo JC (2004) Development of culture media containing spent yeast cells of Debaryomyces hansenii and corn steep liquor for lactic acid production with Lactobacillus rhamnosus. Int J Food Microbiol 97:93–98
Roveri N, Foresti E, Lelli M, Lesci IG, Marchetti M (2010) Microscopic investigations of synthetic biomimetic hydroxyapatite. Microsc Sci Technol Appl Educ 3:1868–1879
Saska S, Barud HS, Gaspar AMM, Marchetto R, Ribeiro SJL, Messaddeq Y (2011) Bacterial cellulose-hydroxyapatite nanocomposites for bone regeneration. Int J Biomater 2011:1–8
Schumann DA, Wippermann J, Klemm DO, Kramer F, Koth D, Kosmehl H et al (2009) Artificial vascular implants from bacterial cellulose: preliminary results of small arterial substitutes. Cellulose 16:877–885
Segal L, Creely JJ, Martin AE, Conrad CM (1959) An empirical method for estimating the degree of crystallinity of native cellulose using the X-ray diffractometer. Text Res J 29:786–794
Suzuki O (2010) Octacalcium phosphate: osteoconductivity and crystal chemistry. Acta Biomater 6:3379–3387
Swain SK, Sarkar D (2013) Study of BSA protein adsorption/release on hydroxyapatite nanoparticles. Appl Surf Sci 286:99–103
Uesu NY, Pineda EA, Hechenleitner AA (2000) Microcrystalline cellulose from soybean husk: effects of solvent treatments on its properties as acetylsalicylic acid carrier. Int J Pharm 206:85–96
Wan YZ, Huang Y, Yuan CD, Raman S, Zhu Y, Jiang HJ et al (2007) Biomimetic synthesis of hydroxyapatite/bacterial cellulose nanocomposites for biomedical applications. Mat Sci Eng C Bio S 27:855–864
Wang K, Zhou C, Hong Y, Zhang X (2012) A review of protein adsorption on bioceramics. Interface Focus 2:259–277
Wippermann J, Schumann D, Klemm D, Kosmehl H, Salehi-Gelani S, Wahlers T (2009) Preliminary results of small arterial substitute performed with a new cylindrical biomaterial composed of bacterial cellulose. Eur J Vasc Endovasc Surg 37:592–598
Wu JM, Liu RH (2013) Cost-effective production of bacterial cellulose in static cultures using distillery wastewater. J Biosci Bioeng 115:284–290
Zadegan S, Hosainalipour M, Rezaie HR, Ghassai H, Shokrgozar MA (2011) Synthesis and biocompatibility evaluation of cellulose/hydroxyapatite nanocomposite scaffold in 1- n-allyl-3-methylimidazolium chloride. Mater Sci Eng C 31:954–961
Zhai Y, Cui FZ, Wang Y (2005) Formation of nano-hydroxyapatite on recombinant human-like collagen fibrils. Curr Appl Phys 5:429–432
Zimmermann KA, LeBlanc JM, Sheets KT, Fox RW, Gatenholm P (2011) Biomimetic design of a bacterial cellulose/hydroxyapatite nanocomposite for bone healing applications. Mater Sci Eng C 31:43–49
Acknowledgments
The authors are thankful to the Brazilian Research Agency EMBRAPA and the following laboratories from Federal University of Ceará (UFC): Laboratory of Biomaterials, Laboratory of Characterization of Materials (LACAM), X-rays Laboratory, and Laboratory of Bioinorganic. This research was financially supported by the following fellowships: Fundação Cearense de Apoio ao Desenvolvimento Científico e Tecnológico (FUNCAP, Brazil), and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES, Brazil).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Duarte, E.B., das Chagas, B.S., Andrade, F.K. et al. Production of hydroxyapatite–bacterial cellulose nanocomposites from agroindustrial wastes. Cellulose 22, 3177–3187 (2015). https://doi.org/10.1007/s10570-015-0734-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-015-0734-8