Abstract
Cellulose nanofibers have a bright future ahead as components of nano-engineered materials, as they are an abundant, renewable and sustainable resource with outstanding mechanical properties. However, before considering real-world applications, an efficient and energetically friendly production process needs to be developed that overcomes the extensive energy consumption of shear-based existing processes. This paper analyses how the charge content influences the mechanical energy that is needed to disintegrate a cellulose fiber. The introduction of charge groups (carboxylate) is achieved through periodate oxidation followed by chlorite oxidation reactions, carried out to different extents. Modified samples are then subjected to different levels of controlled mechanical energy and the yields of three different fractions, separated by size, are obtained. The process produces highly functionalized cellulose nanofibers based almost exclusively on chemical reactions, thus avoiding the use of intensive mechanical energy in the process and consequently reducing drastically the energy consumption.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
From the most basic to the most advanced use, cellulose seems always to be one step ahead of any other material, be it natural or synthetic. Besides being the most abundant biopolymer on earth, as well as being renewable, biodegradable and carbon-neutral, cellulose has unique properties that have been crucial for the existence of life on earth. It has served mankind as the primary source of heat, clothes and building material, to cite the most relevant ones. Because of its proven record of applications, it is not surprising that the use of cellulose nanostructures, especially cellulose nanofibers (CNF) or nanofibrils, promises to play an essential role in the development of the next generation high-tech nanostructured materials.
The cellulose fiber wall, with a typical diameter (d) ranging 15–35 μm, is a compounded material mainly composed of cellulose microfibers (d ~ 40–100 nm), arranged in different orientations, embedded in a polymeric network of hemicelluloses, pectins and lignins (Somerville et al. 2004), with the percentage of each constituent varying in the radial direction through well defined layers. The microfibers themselves are composed of several nanofibrils (d ~ 2–10 nm) made of crystalline and amorphous domains. Whether these domains are arranged in an alternating configuration or a core–shell distribution (Ding and Himmel 2006) is still an open question, although traditionally the first possibility has been the most widely accepted (Habibi et al. 2010). Finally, the number of cellulose polymeric chains that builds up one nanofibril is also a matter of discussion, but lately a molecular model consisting of a 36-glucan-chain elementary fibril forming both crystalline and subcrystalline structures is being preferentially considered (Ding and Himmel 2006; Gross and Chu 2010). However, cellulose nanofibrils from different sources are known to have different diameters and thus a different number of elementary chains associated with them.
CNF are then the primary complete building entities in the hierarchy of plants. From the point of view of materials science, their fibrillar shape of small diameter and very high aspect ratio makes them ideal to be used as reinforcing elements, but by themselves they are also ideal to form strong and transparent films (Henriksson et al. 2008; Siro and Plackett 2010; Saito et al. 2009) that can compete with polymeric ones. However, there are still two major problems that require solution before considering real-world applications for the CNF (Hubbe et al. 2008; Siro and Plackett 2010): first, finding an efficient and energetically favourable way to isolate them. Because neighbouring nanofibrils are either chemically cross-linked (Somerville et al. 2004) or physically entangled by single-chain polysaccharides (Keckes et al. 2003), it seems that their isolation always requires a considerable amount of shear, i.e. mechanical action, regardless of the type of pretreatment. So far, existing methods (Henriksson et al. 2007; Herrick et al. 1983; Hubbe et al. 2008; Isogai et al. 2011; Siro and Plackett 2010; Turbak et al. 1983) make use of a considerable amount of mechanical energy to disrupt the fiber wall, a process that, in addition to other environmental implications, requires a high energy input and high cost. The second step to be mastered has to do with the problem of dispersing hydrophilic CNF into hydrophobic media, e.g. polymeric matrices. Despite several strategies that have been developed to minimize this effect, such as grafting hydrophobes onto them (Siro and Plackett 2010) or coating them with surfactans (Heux et al. 2000), the high crystallinity is often an issue since it limits reactivity. It is very likely that without fully addressing these two features, CNF will have a hard time to find their way out of the laboratories and into the factories.
In recent years, the use of enzymatic or chemical pretreatments on cellulose fibers has become popular with the aim of reducing the amount of mechanical energy required to liberate the nanostructures. The enzymatic route typically involves mixtures of various cellulases which are able to partially digest both the crystalline and amorphous regions (Paakko et al. 2007; Henriksson et al. 2007) facilitating the subsequent mechanical disintegration of the fibers. Alternatively, the introduction of carboxylate groups (COO) onto the surface of the nanofibrils leads under mild alkaline conditions to the appearance of repulsive forces that also weaken the structure. In this direction the preferred pathway is the 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO) radical-mediated oxidation with hypochlorite and chlorite salts as the most common oxidizing agents (Iwamoto et al. 2010; Saito et al. 2009, 2010; Fukuzumi et al. 2009; Siro and Plackett 2010; Isogai et al. 2011), by which one of the three hydroxyl groups in the accessible glucose units of cellulose is converted to a carboxylic group. The use of such nitroxyl radicals and nitrosonium salts as an oxidative route to transform hydroxyl functions into carboxyl and/or aldehyde groups is disclosed elsewhere (Bobbitt and Flores 1988; Chang and Robyt 1996).
Both enzymatic and chemical modifications allow reducing the disintegration energy of cellulose fibers from somewhere in the order of 100 kWh/kg for unmodified cellulose preparations to as little as 1–2 kWh/kg (Isogai 2009; Siro and Plackett 2010), depending on the extent of the treatment. These new limits are comparable with those required to produce so called mechanical pulps out of wood, which means that are industrially viable. The main drawback of these two approaches, however, is that either they require the input of a substantial amount of energy or they fail to provide reasonable production yields. Another issue, concerning the chemical pretreatments, is that the maximum carboxylic content that can be introduced by means of TEMPO oxidation is limited (in case of TEMPO/NaBr/NaClO around 1.7 mmol/g, i.e. millimoles of COO per gram of dried fibers, and, if TEMPO/NaClO2/NaClO is used, below 1 mmol/g (Okita et al. 2010; Isogai et al. 2011). In principle, if one considers that the interior of the crystalline domains is not accessible, the combination of the amorphous regions and the exterior surfaces of the crystals would still account for more than 3 mmol/g, taking an average degree of crystallinity of 60 % (corresponding to a ECF-bleached pine kraft pulp (Liitia et al. 2003)), assuming that the nanofibrils are composed of 36 cellulose chains in a hexagonal conformation (Ding and Himmel 2006) and considering that only every second glucose unit has the primary hydroxyl pointing out of the crystal. Apparently, diffusion problems prevent taking the modification further toward the theoretical maximum. Other estimations, however, claim that 1.7 mmol/g correspond to the entire surface oxidation of cellulose nanofibrils of wood origin (Okita et al. 2010).
In order to surpass TEMPO moderate oxidation limits, this work uses a different and well studied oxidation route, namely periodate oxidation (Potthast et al. 2009; Potthast et al. 2007; Kim et al. 2000), to produce dialdehyde cellulose, followed by chlorite oxidation to convert aldehydes into carboxylic groups. It has been recently established that periodate oxidation attacks the crystalline domains of CNF already in the early stages of the treatment (Potthast et al. 2009). Such chemical treatment allows reaching carboxylic contents in the order of 6.5 mmol/g (Yang 2011), although in this work only up to 3.5 mmol/g is reported. The paper analyses the relation between the carboxylic content of cellulose fibers and the disintegration energy required to convert them into nanofibrils. It shows that the oxidative treatment ultimately results in the spontaneous liberation of the CNF from the cell wall without the necessity of applying any mechanical energy other than that required to stir fiber suspensions during the chemical treatments. However, the length and especially the crystallinity of the nanostructures are severely affected, which in turn could become beneficial for certain applications such as biofuel production. The study brings some new light in understanding the mechanisms that hold the nanofibrils together inside the fiber cell wall and anticipates the production of CNF exclusively by chemical means by defining a charge threshold beyond which cellulose fibers need no mechanical energy to be disintegrated. Finally, since the process involves the introduction of a large amount of functional groups onto the CNF surfaces, the final product obtained is expected to show a higher reactivity and thus to be more prone to further derivatization than any previous preparation.
Materials and methods
Materials
Unbeaten bleached softwood kraft pulp (SKP), supplied by Domtar Inc. Canada as never-dried pulp, was used as raw material for the chemical treatments. Sodium meta-periodate (NaIO4; Sigma-Aldrich), sodium chloride (NaCl; ACP Chemicals Inc.), hydroxylamine hydrochloride (NH2OH·HCl; Sigma-Aldrich), hydrochloric acid (HCl; ACP Chemicals Inc.), sodium hydroxide (NaOH; ACP Chemicals Inc.), sodium chlorite (NaClO2), hydrogen peroxide (H2O2; Sigma-Aldrich), 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO; Sigma-Aldrich), sodium phosphate buffer, sodium hypochlorite (NaClO), ethanol (ACP Chemicals Inc.) and a mix-bed ion exchange resin (Sigma-Aldrich) were used as received. Double deionized water was used throughout the experimentation, except for the dialysis purification where Milli-Q ultrapure water (Millipore Corp.) was used.
Chemical treatments
Cellulose fibers in the form of pulp suspensions were subjected to two successive chemical treatments, carried out to various extents in order to achieve various degrees of oxidation. Initially, periodate oxidation was carried out in aqueous media using a glass beaker with overhead stirrer, with the following reaction conditions: bleached softwood kraft pulp (3 g), NaIO4 (1.98 g; 10.75 mmol; 50 mol % based on moles of anhydroglucose in pulp) and NaCl (11.7 g; 1 M based on overall solution) were added to 200 mL water. The beaker was totally covered with aluminium foil before starting the reaction, in order to prevent light from activating side reactions, and the mixture was gently stirred at room temperature. After the desired reaction time, the modified pulp (dialdehyde cellulose) was filtered out and thoroughly washed with deionized water repeatedly. In order to convert aldehyde moieties into carboxylic groups, periodate-oxidized pulp (3.5 g), NaClO2 (80 % pure; 2.76 g; 24.5 mmol) and H2O2 (30 wt.% solution; 2.76 g; 24.5 mmol) were added to 150 mL water. This mixture was stirred at room temperature for 20 h, during which the pH was kept at 5 by drop wise addition of NaOH solution (especially necessary during the first 3 h).
Determination of aldehyde and carboxylate content
The aldehyde content of the dialdehyde cellulose produced by the periodate oxidation reaction was determined using the hydroxylamine-hydrochloride (NH2OH·HCl) titration method, by which the HCl released from its reaction with aldehydes is back-titrated with a NaOH solution of known concentration. More specifically, a water suspension of periodate-oxidized cellulose fibers (20 mL; 0.65 g dry basis) was mixed with 40 mL of isopropanol, making a final proportion of isopropanol/water of 2/1 v/v, and the mixture was sufficiently stirred to prepare a well-dispersed slurry. The pH of the mixture was then adjusted to 2–3 by adding a few drops of concentrated HCl and then carefully adjusted to 3.5 with NaOH 0.1 N. 10 mL of 10 wt.% NH2OH·HCl solution was added to this mixture, allowing it to react for 10 min. Finally, the HCl released from the reaction was titrated with 0.5 N NaOH solution until pH 3.5 was reached again. The aldehyde content was then calculated using the following equation:
Here Aald is the aldehyde content (mmol/g cellulose), VNaOH the volume of NaOH (mL) consumed in the titration, N is the normality of the NaOH (eq/L) and wcell the weight of dry cellulose (g) initially suspended.
The carboxylate content of the samples was determined by conductometric titration. To a 120 mL of 0.02 wt.% water suspension of the cellulosic product (20.4 mg dry basis) 2.5 mL of a 0.02 M NaCl solution was added and the mixture was gently stirred. Then 0.1 M HCl was slowly added to the mixture to set the pH value in the range of 2.5–3.0. Using an 836 Titrando titrator (Metrohm, Switzerland) a 0.005 M NaOH solution was added at a rate of 0.05 mL/min until the mixture had reached pH 11. The carboxylate content of the sample was determined from the conductivity curves using the following equation:
Here [COO] is the carboxylate content in mmol per gram cellulose, VNaOH is the volume of NaOH (mL) required for the deprotonation of carboxylic groups, MNaOH is the normality of NaOH (eq/L) and wcell is the amount of dry cellulose product initially used (g).
Disintegration process
Two different devices were used in the disintegration process: a domestic blender (Braun hand held blender, type 4191) and a double-cylinder type homogenizer (Brinkmann Polytron homogenizer PT-35/4), which were always used in combination with a stirring plate to keep the pulp suspensions homogeneously dispersed.
These systems were initially characterized in order to have control over their mechanical action and energy consumption. First, to obtain a reliable measurement of the amount of electrical energy consumed by each set up, an ammeter was connected in series with the stirring plate and the mixer, and a data logging program was run on a computer connected to them. Assuming that the voltage remained constant, the electrical energy was calculated using Ohm’s Law:
where Ee is electrical energy (J), V is voltage (V), I is current (A) and t is time (s). The electrical input was recorded at different settings over time periods that would be used later for the experimentation and the most stable one, showing a linear consumption with a squared regression factor higher than 0.99, was chosen for each device.
The second part of the characterization attempted to approximate the amount of mechanical action that is actually exerted by each device and which obviously differs from the electrical input; in our approach we correlate this parameter to the thermal energy that such mechanical action releases. With that aim we carried out a Joule experiment, following the idea that the energy transferred to the water in the form of kinetic energy must be dissipated by viscous forces and transformed into thermal energy. We used a thermocouple to measure the increase in temperature of 250 mL of water caused by the blender and the homogenizer at the predefined settings; the water was contained in a glass beaker insulated with a Styrofoam home-made shell. The mass and specific heats of the water and the glass allows the calculation of the thermal energy released:
where Et is the thermal energy (J), m is the mass (kg), c is the specific heat (J/kg °C), ΔT is the temperature difference (°C), and the sub-indices w and g stand for “water” and “glass”, respectively. From the characterization of both disintegration equipments we defined an efficiency parameter for each device, as follows:
This characterization was meant to provide a fair approximation rather than an accurate determination.
Having characterized the equipments, the disintegration experiments were performed on 40 mL of 0.25 wt.% modified pulp suspensions at the preselected settings for different amounts of time. Each processing time corresponded to a given electricity consumption and thermal energy released, which we used as a measure of the amount of mechanical energy applied (Thermal Energy ≈ Mechanical Energy).
Isolation
The following process was used to isolate the different cellulosic fractions after the reactions and the mechanical treatments were completed: a filtration at atmospheric pressure through a 20 μm-pore size nylon cloth was carried out to separate out the macroscopic fraction composed of non-disintegrated fibers still maintaining their original size; the resulting suspension was vacuum-filtered through a surface-hydrophilized polytetrafluoroethylene (PTFE) membrane with 0.1 μm pore size (Advantec Tokyo, Japan), which caused the retention of the CNF; finally, the resulting filtrate containing mainly dissolved cellulose derivatives was oven dried. Each fraction was weighted and a mass balance was calculated.
Film characterization
Cellulose nanofibrils obtained in the form of films and oven dried at 60 °C for 8 h were characterized by X-ray diffraction (XRD) analysis and atomic force microscopy (AFM). The XRD measurements were performed on a Bruker D8 Discover two dimensional diffractometer with VANTEC 2D detector and CuKα radiation (λ = 1.54 Å). Diffractograms were acquired with a 2θ (Bragg angle) range of 12.5–32.5° at a scan rate of 0.005°/s. A crystallinity index (CI) was calculated empirically (Segal et al. 1959) as 100 × (Imax° − I18°)/Imax, where Imax is the maximum intensity of the 002 lattice diffraction and I18° the intensity recorded at that particular angle, both in arbitrary units.
The surface morphology of the films was examined by AFM. The images were acquired with a MFP-3D atomic force microscope (Asylum Research, Santa Barbara, CA) on the films glued to a glass slide with double-sided tape. The experiments were conducted in tapping mode using silicon cantilevers (Nanoworld) with force constant 42 N/m, tip length 125 μm, tip radius 8 nm or less and resonance frequencies 320 kHz. Similar AFM conditions were employed to measure CNF that had been liberated from those films by magnetic stirring in water for 5 days.
Results and discussion
Figure 1 illustrates the effect of carboxylate concentration on the energy required to disintegrate cellulose fibers by plotting some of the limits reported in the literature and recalculating others from the experimental details provided there. The references to the disintegration energy in most of the works are often very vague and rarely contain a production yield associated to it, but similar limits appear repeatedly in one form or the other in patents and scientific papers (Engelhardt et al. 2009; Isogai 2009; Saito et al. 2009, 2008; Siro and Plackett 2010; Turbak et al. 1983). What these publications do not tackle, however, is a complete analysis of how the energy required to disintegrate a fiber is affected by the charge content, and more importantly, how the combination of these two parameters affects the production yield and the associated mechanisms.
Estimates of the energy required to disintegrate cellulose fibers into cellulose nanofibrils according to data available in the literature (1 Siro and Plackett 2010, 2 Isogai 2009, 3 Turbak et al. 1983, 4 Saito et al. 2009, 5 Engelhardt et al. 2009, 6 Saito et al. 2008). Cloud shapes represent unmodified (lowest COO content) and TEMPO-oxidized wood cellulose fibers. The green dashed line shows how, apparently, the disintegration energy would approach zero energy at [COO] ~ 3 mmol/g. The arrows in the insets illustrate the carboxylation of primary (blue dots) or secondary (green dots) hydroxyl groups achieved respectively through TEMPO or Periodate + Chlorite oxidations, and the different charge densities that result in each case. (Color figure online)
Mechanically fibrillated cellulose, with or without pretreatment, has been extensively studied for the last two decades. Figure 2 shows atomic force microscopy (AFM) images of two different nanofibrillar cellulose preparations. Figure 2a corresponds to CNF extracted from coir (Cocos nucifera) fibers after being disintegrated exclusively by mechanical action (20 passes in Niro Soavi high pressure homogenizer with 500 bar pressure drop (Tejado et al. 2006)). The image shows the intrinsic limitations of this technique: damaging of the nanofibrils and difficulty to totally disintegrate the cellulose fibers (Siro and Plackett 2010; Henriksson et al. 2007). Figure 2b shows CNF isolated from softwood kraft pulp after TEMPO/NaClO/NaClO2 treatment ([COO] = 0.8 mmol/g) following a previous work (Saito et al. 2009); oxidized fibers were disintegrated in a domestic blender (Waring Commercial Blender, Model 51BL31) for 15 min and then sonicated (Vibra-Cell, Sonics & Materials Inc.) for 5 min before imaging. Nanofibrils look much longer and better dispersed than in the previous case.
AFM images of cellulose nanofibrils from a coir, after intensive mechanical homogenization (Tejado et al. 2006), and b TEMPO-oxidized softwood kraft pulp after disintegration with a domestic blender followed by sonication
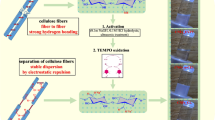
The chemistry used in the present work for the introduction of charges on cellulose fibers is schematically shown in Fig. 3. Compared to TEMPO-mediated oxidation it allows the introduction of a larger amount of carboxylic groups and consequently the study of the effect of the charge content on the disintegration energy through a wider range, including the limit where, according to Fig. 1, the energy is expected to approach zero. Four samples with different carboxylate content (1.0, 2.0, 2.5 and 3.5 mmol/g, obtained from conductometric titrations) were used for this study.
The four modified cellulose samples were subjected to mechanical treatments of different intensities, by varying the amount of time, with either a domestic blender or a homogenizer and the electrical energy consumed during each treatment was recorded. The characterization of both disintegration equipments showed that the efficiency (see section “Disintegration process”) was around 27 % for the domestic blender but only about 9 % for the homogenizer at the selected settings. According to our measurements, that means that the homogenizer wastes most of the electricity consumed in internal processes and heat released from the engine, and only around 10 % is actually taken into the suspension. On the other hand, the domestic blender transfers more than one fourth of the electricity into the pulp preparations being thus much more efficient for this application. As expected, after applying the efficiency correction factor, the thermal energy input for the blender and the homogenizer fell into a comparable range (1.9 and 2.4 kJ/min, resp.) despite having very disparate electric consumptions (7.2 and 26.0 kJ/min, resp.). This agreement actually proofs the reliability of the energy measurements since, as will be seen later, the mass yields for all three cellulosic fractions fit perfectly with the thermal energy recorded regardless the type of device used.
The effect of the mechanical action on the yield of non-disintegrated cellulose fibers, dissolved carboxylated cellulosic and cellulose nanofibrils, respectively, is shown in Fig. 4a–c for the four different samples. Data obtained with the blender and the homogenizer are mixed without specifically mentioning their origin. Figure 4a shows that the amount of non-disintegrated fibers decreases with increasing carboxylate content at a given energy level, and also with increasing energy at a given concentration. This is exactly the kind of behaviour that is expected, as the charge groups create repulsion between the nanofibrils, thus weakening the macrostructure, and the mechanical action separates them physically. It is remarkable to see that already at 1 mmol/g only about 55 wt.% gets caught on the 20 μm pores while the rest passes through. Maybe more interesting is to note that at 3.5 mmol/g such amount gets close to zero, even at the minimum energy level tested. Such observation strongly supports the prediction highlighted in Fig. 1, which shows that, above certain charge content, cellulose fibers need almost no energy to get disintegrated. Indeed, according to present results, the threshold value may well lay around 3 mmol/g as suggested from that graph.
Figure 4b shows the cellulose fraction that passed through the 0.1 μm pore size filter and which we generally correlate with dissolved carboxylated cellulose. The trend is the opposite than that for the non-disintegrated fibers, with the yield being favoured by both carboxylate content and energy applied. In this case both the 3.5 and 2.5 mmol/g samples show an almost constant yield, ranging from 35 to 40 wt.%, while the less charged samples seem to approach this value as the energy is increased. These results suggest that the liberation of the CNF is driven by the dissolution of large cellulose fractions. The chemical treatment would preferentially oxidize the amorphous domains, as they are more accessible and thus more reactive, which would get released by a solvating force upon reaching a certain charge concentration. That would cause the spontaneous liberation of the CNF, a mechanism illustrated in Fig. 5.
Suggested mechanism for the liberation of CNF in the absence of mechanical energy. The preferential oxidation of the amorphous domains of cellulose leads, upon surpassing a threshold value, to their solubilisation with the subsequent liberation of cellulose nanostructures of varying length. Green glossy dots represent (charged) carboxylate groups. This process starts at the readily available surface of the fibers and continues inward (Varma et al. 1997). (Color figure online)
Finally Fig. 4c shows the evolution for the CNF fraction. As a general rule, the yield follows the same trend observed in Fig. 4b and increases with increasing carboxylate content for a given energy level and with increasing energy input for a given charge content. However, for a carboxylate concentration of 3.5 mmol/g a plateau value is observed regardless the amount of energy applied, suggesting that the maximum possible yield has been reached and, again, that the energy applied has played little or no role on it. The same plateau, in the range 50–60 wt.%, is reached for the 2.5 mmol/g sample after applying 1.25 kWh/kg. A similar plateau value has been recently observed by other authors upon the study of different catalysts as potential TEMPO substitutes (Iwamoto et al. 2010). The authors also relate the yield to the charge content and obtain similar maxima. Although not calculated specifically, the disintegration specifications (domestic blender, 5 min) suggest that the amount of mechanical energy applied in those cases is several times greater than the highest level used in our study. Nevertheless, other studies using the TEMPO route (Isogai et al. 2011) claim that up to 95 % of the original cellulose can be converted into CNF if enough energy is applied.
The CNF fractions, obtained originally as films, were characterized through AFM and XRD. Figure 6 shows images from (a) one of these films, corresponding to [COO] = 2.5 mmol/g sample, and (b) CNF with [COO] = 1 mmol/g after being liberated from the corresponding film. Cellulose nanofibers with diameters in the order of 10–20 nm are clearly visible in both samples. In addition to CNF, Fig. 6a shows large regions within the film where nanofibers are absent. Those areas could be made of amorphous cellulose, in agreement with previous observations in which cellulose becomes increasingly amorphous with progressive periodate oxidation leading to new cellulose derived products (Varma et al. 1997). Another possibility is the presence of dissolved carboxylated cellulose that had deposited on top of a CNF network during the filtration process, favoured by the blocking of the 0.1 μm pores. Figure 6b shows well defined CNF of varying width, including both individualized nanofibrils and small bundles of them which were never completely disintegrated. Despite these bundles, the degree of isolation of individual CNF achieved is shown to be very high considering the limited mechanical treatment applied, and must be ascribed almost completely to the chemical modification carried out.
In the XRD curves of these films (Fig. 7) a typical X-ray diffraction pattern of oxidized native softwoods is observed in all cases, similar to that shown elsewhere (Okita et al. 2010). The crystallinity indices obtained from these curves (66.2, 68.2, 69.8 and 73.5 %, respectively for 1.0, 2.0, 2.5 and 3.5 mmol/g carboxylate content) also correspond to samples with both crystalline and amorphous parts, instead of a highly crystalline CNF sample as would be expected. It has been reported that periodate oxidation of cellulose leads to a severe loss of crystallinity (Kim et al. 2000; Hou et al. 2007; Varma et al. 1997), what would explain the low CI values. The increase in CI with increasing charge content could be related to a higher degradation of the more oxidized samples leading to a lower deposition of dissolved carboxylated cellulose onto CNF networks.
The film characterization, however, confirms that cellulose nanofibrils have been liberated almost in the absence of mechanical treatment; at the same time it suggests that the separation method should be improved in order to have well defined fractions, which will be attempted in future work. The fact that periodate oxidation leads to cellulose degradation could be also related to the decreased CNF yields described before. According to our mass balances, this yield loss is due to the solubilisation of amorphous carboxylated cellulose prior to the liberation of the nanofibrils. Such disintegrating mechanism leads to a reduced CNF production yield but at the same time allows an energetically friendly production. Moreover, the total disintegration of cellulose fibers could find an interesting field of application in biofuel production, where the decreases in crystallinity and polymerization degree of carbohydrates are highly desired, as they favour their digestibility (Mora-Pale et al. 2011).
The practical implications of this work are very interesting. It not only suggests that cellulose nanofibers can be produced almost in the absence of mechanical energy, but also that samples can be prepared in such a way that they need a predefined amount of shear to be torn apart. This would facilitate some problematic aspects related to nanofibrillar cellulose preparations, such as their transportation and elevated viscosity. For example, one big problem that prevents cellulose nanofilms to be produced in a continuous industrial process is the high viscosity that CNF suspensions already show at very low concentrations (Siro and Plackett 2010). This could be avoided by preparing suspensions of modified cellulose fibers (having a viscosity close to that of water), which had been previously oxidized to the extent where they become disintegrated by a precise mechanical energy (Tejado et al. 2011). Using a papermaking machine as an example, that mechanical energy would be the one exerted in the headbox at the time the pulp suspension is delivered onto the forming wire. Before getting to the headbox, no difference with regular pulp suspensions should be encountered, thus no viscosity problems are expected; only upon passing through the slice opening, the fibers would be disintegrated and deposited on a forming wire as nanofibrils. The same could apply to mixtures of regular and oxidized fibers, where the latter would act as a reinforcing element of paper, or to mixtures of polymer matrices and CNF, which could take advantage of the shear imparted by an extruder or a mixer.
Conclusions
This paper shows that the isolation of cellulose nanofibers can be achieved almost in the absence of mechanical energy if the oxidation is sufficiently high. The mechanism responsible for this “spontaneous” disintegration is shown to be the dissolution of overcharged amorphous domains, which become solubilised upon surpassing a certain charge threshold value set around 3 mmol/g, according to experimental results. A maximum of 50–60 wt.% of the original cellulose is converted into cellulose nanofibers what seems to be a direct consequence of the chemistry applied (periodate-chlorite oxidations). This study opens up the possibility for the energy-friendly production of cellulose nanofibers.
References
Bobbitt JM, Flores MCL (1988) Organic nitrosonium salts as oxidants in organic chemistry. Heterocycles 27(2):509–533
Chang PS, Robyt JF (1996) Oxidation of primary alcohol groups of naturally occurring polysaccharides with 2,2,6,6-tetramethyl-1-piperidine oxoammonium ion. J Carbohydr Chem 15(7):819–830
Ding SY, Himmel ME (2006) The maize primary cell wall microfibril: a new model derived from direct visualization. J Agric Food Chem 54(3):597–606. doi:10.1021/jf051851z
Engelhardt J, Fischer S, Hettrich K, Krueger C, Nachtkamp K, Pinnow M (2009) Preparing dispersion containing a particle made of amorphous cellulose. Germany Patent, WO2009021687-A1
Fukuzumi H, Saito T, Wata T, Kumamoto Y, Isogai A (2009) Transparent and high gas barrier films of cellulose nanofibers prepared by TEMPO-mediated oxidation. Biomacromolecules 10(1):162–165. doi:10.1021/bm801065u
Gross AS, Chu JW (2010) On the molecular origins of biomass recalcitrance: the interaction network and solvation structures of cellulose microfibrils. J Phys Chem B 114(42):13333–13341. doi:10.1021/jp106452m
Habibi Y, Lucia LA, Rojas OJ (2010) Cellulose nanocrystals: chemistry, self-assembly, and applications. Chem Rev 110(6):3479–3500. doi:10.1021/cr900339w
Henriksson M, Henriksson G, Berglund LA, Lindstrom T (2007) An environmentally friendly method for enzyme-assisted preparation of microfibrillated cellulose (MFC) nanofibers. Eur Polym J 43:3434–3441. doi:10.1016/j.eurpolymj.2007.05.038
Henriksson M, Berglund LA, Isaksson P, Lindstrom T, Nishino T (2008) Cellulose nanopaper structures of high toughness. Biomacromolecules 9(6):1579–1585. doi:10.1021/bm800038n
Herrick FW, Casebier RL, Hamilton JK, Sandberg KR (1983) Microfibrillated cellulose: morphology and accessibility. J Appl Polym Sci: Appl Polym Symp 37:797–813
Heux L, Chauve G, Bonini C (2000) Nonflocculating and chiral-nematic self-ordering of cellulose microcrystals suspensions in nonpolar solvents. Langmuir 16:8210–8212
Hou QX, Liu W, Liu ZH, Bai LL (2007) Characteristics of wood cellulose fibers treated with periodate and bisulfite. Ind Eng Chem Res 46:7830–7837. doi:10.1021/ie0704750
Hubbe MA, Rojas OJ, Lucia LA, Sain M (2008) Cellulosic nanocomposites, review. Bioresources 3(3):929–980
Isogai A (2009) Individualization of nano-sized plant cellulose fibrils achieved by direct surface carboxylation using TEMPO catalyst. Paper presented at the 2009 International Conference on Nanotechnology for the Forest Products Industry, Edmonton, AB, Canada, June 23–26
Isogai A, Saito T, Fukuzumi H (2011) TEMPO-oxidized cellulose nanofibers. Nanoscale 3(1):71–85. doi:10.1039/c0nr00583e
Iwamoto S, Kai WH, Isogai T, Saito T, Isogai A, Iwata T (2010) Comparison study of TEMPO-analogous compounds on oxidation efficiency of wood cellulose for preparation of cellulose nanofibrils. Polym Degrad Stab 95(8):1394–1398. doi:10.1016/j.polymdegradstab.2010.01.017
Keckes J, Burgert I, Fruhmann K, Muller M, Kolln K, Hamilton M, Burghammer M, Roth SV, Stanzl-Tschegg S, Fratzl P (2003) Cell-wall recovery after irreversible deformation of wood. Nat Mat 2(12):810–814. doi:10.1038/nmat1019
Kim UJ, Kuga S, Wada M, Okano T, Kondo T (2000) Periodate oxidation of crystalline cellulose. Biomacromolecules 1(3):488–492
Liitia T, Maunu SL, Hortling B, Tamminen T, Pekkala O, Varhimo A (2003) Cellulose crystallinity and ordering of hemicelluloses in pine and birch pulps as revealed by solid-state NMR spectroscopic methods. Cellulose 10(4):307–316
Mora-Pale M, Meli L, Doherty TV, Linhardt RJ, Dordick JS (2011) Room temperature ionic liquids as emerging solvents for the pretreatment of lignocellulosic biomass. Biotechnol Bioeng 108(6):1229–1245. doi:10.1002/bit.23108
Okita Y, Saito T, Isogai A (2010) Entire surface oxidation of various cellulose microfibrils by TEMPO-mediated oxidation. Biomacromolecules 11(6):1696–1700. doi:10.1021/bm100214b
Paakko M, Ankerfors M, Kosonen H, Nykanen A, Ahola S, Osterberg M, Ruokolainen J, Laine J, Larsson PT, Ikkala O, Lindstrom T (2007) Enzymatic hydrolysis combined with mechanical shearing and high-pressure homogenization for nanoscale cellulose fibrils and strong gels. Biomacromolecules 8(6):1934–1941. doi:10.1021/bm061215p
Potthast A, Kostic M, Schiehser S, Kosma P, Rosenau T (2007) Studies on oxidative modifications of cellulose in the periodate system: molecular weight distribution and carbonyl group profiles. Holzforschung 61(6):662–667. doi:10.1515/hf.2007.099
Potthast A, Schiehser S, Rosenau T, Kostic M (2009) Oxidative modifications of cellulose in the periodate system—reduction and beta-elimination reactions. Holzforschung 63(1):12–17. doi:10.1515/hf.2009.108
Saito T, Nishiyama Y, Isogai A, Pouteau J, Vinion M (2008) Fine cellulose fiber for dispersion used as functional additives such as gelatinizer and emulsifier. Japan Patent, JP2008001728-A
Saito T, Hirota M, Tamura N, Kimura S, Fukuzumi H, Heux L, Isogai A (2009) Individualization of nano-sized plant cellulose fibrils by direct surface carboxylation using TEMPO catalyst under neutral conditions. Biomacromolecules 10(7):1992–1996. doi:10.1021/bm900414t
Saito T, Hirota M, Tamura N, Isogai A (2010) Oxidation of bleached wood pulp by TEMPO/NaClO/NaClO2 system: effect of the oxidation conditions on carboxylate content and degree of polymerization. J Wood Sci 56(3):227–232. doi:10.1007/s10086-009-1092-7
Segal L, Creely JJ, Martin AE, Conrad CM (1959) An empirical method for estimating the degree of crystallinity of native cellulose using the X-ray diffractometer. Textile Res J 29:786–794
Siro I, Plackett D (2010) Microfibrillated cellulose and new nanocomposite materials: a review. Cellulose 17(3):459–494. doi:10.1007/s10570-010-9405-y
Somerville C, Bauer S, Brininstool G, Facette M, Hamann T, Milne J, Osborne E, Paredez A, Persson S, Raab T, Vorwerk S, Youngs H (2004) Toward a systems approach to understanding plant-cell walls. Science 306(5705):2206–2211. doi:10.1126/science.1102765
Tejado A, Labidi J, Peña C, Álvarez de Arcaya P, Mondragon I (2006) Isolation and characterization of cellulose microfibrils from exotic natural fibers. Paper presented at the 9th European Workshop on Lignocellulosics and Pulp (EWLP), Vienna
Tejado A, van de Ven TGM, Alam MN, Antal M (2011) Highly charge-group modified cellulose fibers which can be made into cellulose nanostructures or super-absorbing cellulosic materials and method of making them. Canada Patent, US Provisional Patent Application 61450222
Turbak AE, Snyder FW, Sandberg KR (1983) Microfibrillated cellulose production using high pressure homogeniser gives reduced costs and eliminates cellulose degradation. Germany Patent, US4374702-A
Varma AJ, Chavan VB, Rajmohanan PR, Ganapathy S (1997) Some observations on the high-resolution solid-state CP-MAS C13-NMR spectra of periodate-oxidised cellulose. Polym Degrad Stab 58(3):257–260
Yang H (2011) Investigation and characterization of oxidized cellulose and cellulose nanofiber films. M.Sc. Thesis, McGill University, Montreal
Acknowledgments
This work was funded by an Industrial Research Chair, funded by the National Sciences and Engineering Research Council of Canada (NSERC) and FPInnovations. The contribution of undergraduate students who participated in an industrial design project course in Chemical Engineering Department, McGill University, in which some of the data were collected, is also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tejado, A., Alam, M.N., Antal, M. et al. Energy requirements for the disintegration of cellulose fibers into cellulose nanofibers. Cellulose 19, 831–842 (2012). https://doi.org/10.1007/s10570-012-9694-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-012-9694-4