Abstract
Apoptosis is a highly regulated and programmed cell breakdown process characterized by numerous changes. It was reported as the major mechanism of anticancer drug-induced cells death. Unfortunately, many of these drugs are non-specific and cause severe side effects. The effects of 5-flourouracil (5-FU) on the apoptotic events in normal murine thymus were evaluated using an in vivo model. A single dose of 5-FU (150 mg/kg ip) was injected to CF-1 mice. A multiparametric analysis of thymic weight, cellularity, viability, architectural organization, apoptosis, DNA fragmentation, and the expression of several apoptotic proteins was evaluated in 10 days time-course study post-5-FU dosing. Total organ weights, thymocyte counts, and cell viabilities diminished drastically from the second day. The thymus architecture assessed through electron scanning microscopy revealed deep alterations and the lost of cell–cell contact between the first and the third days. DNA fragmentation and apoptotic indexes (May Grünwald Giemsa staining, double flourescent dyes, and TdT-mediated dUTP nick-end labeling assay) revealed that cell death was maximal on the second day (three times over control). Furthermore, the pro-apoptotic proteins FAS and Bax were strongly up-regulated during the first 2 days. The aforementioned morphological and biochemical changes were also accompanied within the same period by caspase 3 activation. This study revealed that in vivo apoptosis in normal thymus after 5-FU administration is related to FAS, Bax, and Caspase 3 co-expressions under the current experimental conditions, these findings, therefore, contribute to a new insight into the molecular mechanisms involved during 5-FU administration upon the thymus and the possible events committed in the lymphophenia associated with chemotherapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Apoptosis is a well-controlled, tightly regulated physiological mechanism that is used extensively during the development, tissue homeostasis, and maintenance of the immune system in adult organisms (Jacobson 1997). This process involves the activation of several molecular events, leading to cell death that is characterized by morphological changes, chromatin condensation, and apoptotic bodies that are associated with DNA fragmentation and caspases activation (Kerr 2002). Caspases, also known as cysteine aspartate-specific proteases, are a family of intracellular proteins that exist as dormant proenzymes in healthy cells and are activated through proteolysis. Once activated, they cleave cellular substrates, leading to morphological hallmarks of apoptosis (Zhang et al. 2000a, b; Savill and Fadok 2000; Ashkenazi and Dixit 1998; Thornberry and Lazebnik 1998).
It has been reported that apoptosis is the major mechanism of anticancer drug-induced cells death (Ricci and Zong 2006). 5-Fluorouracil (5-FU) is an antimetabolic chemotherapeutic agent with multiple mechanisms of action, including inhibition of the synthesis of thymidine nucleotides and incorporation into RNA (Parker and Cheng 1990; Pritchard et al. 1997). Although 5-FU is a widely used antineoplastic agent, the cytotoxicity of 5-FU is not limited to tumor tissue. Hematopoietic cells and normal epithelial cells of gastrointestinal tract are susceptible to 5-FU-induced cytotoxicity, which produces severe leucopenia and intestinal toxicity, leading to lethal translocation of intestinal microflora (Yeager et al. 1983; Ijiri and Potten 1987; Rocha et al. 1994). In addition, in vitro and in vivo studies showed that 5-FU-induced apoptosis upon the thymus is partially mediated via death receptor CD95 (Eichhorst et al. 2001). Moreover, there is widespread evidence that another potential apoptotic-promoting system may be involved in the thymus post-5-FU, such as those involving the homodimerization of death promoting molecule Bax and/or Bak that regulate mitochondria-mediated apoptosis by direct modulation of mitochondrial permeabilization (Miyashita et al. 1995; Gottlieb and Oren 1998). Experimental evidence has suggested that Bax is one of the crucial proteins in apoptosis regulation upon 5-FU treatment. In vitro studies using oral squamous carcinoma cells (Takemura et al. 2005) and colon carcinoma cells (Kobayashi et al. 2000) showed an increased expression of Bax after 5-FU treatment. Furthermore, mutant colorectal cancer cells that lack functional Bax gene exhibit partial resistance to 5-FU (Zhang et al. 2000a, b). In addition, 5-FU induced-apoptosis was accompanied by increased expression of Bax in colon cancer cells with wild-type p53 (Nita et al. 1998). This in vitro experimental data strongly suggests that Bax expression is linked to 5-FU-induced apoptosis.
It is also noteworthy that degradation of antiapoptotic proteins such as Bcl-2 and Bcl-xL induces dimerization of Bax, involving the release of cytochrome c from mitochondria, caspases cascade activation, and cell death (Fincucane et al. 1999; Alnemri 1997; Henkart 1996; Martin and Green 1995; Chinnaiyan and Dixit 1996).
Although the apoptotic mechanisms are well known, cellular response triggered by 5-FU varies depending on experimental conditions. For instance, in vitro studies using HT 29 colon carcinoma cells could not show significant DNA fragmentation during the first 24 h after 5-FU (Horowitz et al. 1997), whereas LoVo colorectal cancer cells showed minimal DNA fragmentation at 48 h (Werner et al. 2007). Nevertheless, in vivo studies showed DNA fragmentation in breast cancer tissue removed after 5-FU treatment (Asaga et al. 2001). Unfortunately, little is known about the possible in vivo side effects of 5-FU, specifically upon normal thymic tissue and its apoptotic mechanism.
Therefore, in the current study, the effect of 5-FU on murine thymus using an in vivo normal model through several experimental procedures was studied. Thus, thymic weight and structure, apoptotic indexes (TdT-mediated dUTP nick-end labeling, TUNEL; DNA fragmentation; light; and fluorescence microscopy), as well as FAS, Bax, and caspase 3 expressions (immunoblottings) were evaluated after a single dose of 5-FU over a period of 10 days.
Experimental data strongly suggests that 5-FU induces thymocytes apoptosis 6 h post-drug dosing, reaching the maximum injury on the second day. In spite of the reduction of glands weight and the deep alterations of the thymic structure, the apoptotic related proteins (FAS, Bax, and Caspase 3) were expressed in a coordinated way along the acute apoptotic phase and the further recovery.
Data from this study may contribute to the understanding of 5-FU immunotoxicity and the underlying mechanisms involved in this process.
Material and methods
Animals
Adult female CF-1 Swiss mice (age, 5–6 weeks; average weight, 23–25 g) were used all through the experimental protocol. They were housed in cages in an air-conditioned room with controlled temperature (24°C) for 5 days before the experiment and maintained in a 12-h light–dark cycles. They were allowed to access freely to pelleted food and water. Experiments were conducted according to the principles in the “Guide for the Care and Use of Laboratory Animals” (NIH, Bethesda, MD, 1996).
Experimental design
Mice were divided randomly into three groups. The first group (n = 96) was subdivided into two subsets. Mice of the first subset (n = 48) were treated with saline for controls, whereas mice of the second subset (n = 48) were injected with 5-FU (F 6627–5G; Sigma, St. Louis, MO). 5-FU was dissolved in sterile saline solution (NaCl 0.9%), and it was injected i.p. at a single dose of 150 mg/kg. Animals from the first group were used to assess cellularity, viability, thymic weight, and apoptotic assays. Six mice were used for control, and six ones were used for 5-FU-treated mice at each time point of the study for the above-mentioned assays.
The second group of animals (n = 18) was used for DNA laddering assay and scanning electronic microscopy. Two animals were used at each time point for these assays.
Finally, the third group (n = 27) was used to obtain thymic extracts for Western blotting. Three animals were used for obtaining lysates at each time point of the experimental schedule. Experimental data from 5-FU-injected mice of the second and third groups of rodents were compared to the control animals (day 0).
Animals were euthanized by cervical dislocation under pentobarbital (100 mg/kg i.p.) anesthesia at 0, 6, and 12 h, and 1, 2, 3, 5, 7, and 10 days, respectively, then glands were immediately removed and used for the different assays.
Determination of thymus cellularity and viability
Thymus tissue was placed in ice-cold phosphate buffer saline (PBS, pH 7.4) and passed through a sieve. Cell suspensions were washed several times with minimum essential medium (MEM, Alpha Modification Sigma Co., USA) and were centrifuged at 500 rpm for 10 min to avoid remaining debris.
Thymic cell suspensions were flushed with 500 μl of MEM, and cellularity, as well as viability, was determined using a hemocytometer. Viability was assessed by the Trypan Blue (0.2%) exclusion assay. Cellularities are expressed as total nucleated cells × 106/thymus, and thymus viabilities are expressed as a percentage of viable cells.
Both parameters were determined in thymic cell suspensions from three independent experiments at the indicated time points after injection of 5-FU.
Scanning electronic microscopy
This technique aims to preserve the tissue, particularly for a direct observation of the thymic architecture during post-5-FU recovery. Thymi were removed and placed in fresh cold fixative solution (5% formaldehyde, 6% acetic acid, 65% ethanol, and 24% water) to be observed under the scanning electronic microscope. The samples were dehydrated and dried to critical point (Dento Drier). Then, they were exposed to gold–palladium coating for 3 min. The sections were examined through the scanning electronic microscope (Jepl-JSM-5800 L.V.) and images (1,500–2,000×) were obtained in triplicate at the indicated time points of the protocol.
Methodological aspects of apoptosis detection
-
(a)
May Gründwald–Giemsa (MGG) staining
May Gründwald–Giemsa staining (Merck, Germany) was use on methanol-fixed cytospin preparations following the morphological criteria to identify apoptosis. Typical apoptotic findings consist of chromatin condensation, cell shrinkage, and formation of apoptotic bodies (Kerr et al. 1994).
Apoptotic indexes were evaluated from three independent assays by dividing the number of recognizable apoptotic cells to 1,000 cells counted randomly (400×). Results are expressed as apoptotic percentage at the indicated time points of the protocol.
-
(b)
Fluorescence microscopic analysis of thymocytes death
A slight modified method of double fluorescent staining was used to evaluate thymocyte cell death (Jones and Senft 1985). Briefly, cells were incubated in PBS containing a combination of 75 μg/ml fluorescein diacetate and 100 μg/ml propidium iodide (IP). Fluorescein diacetate is cell permeant and metabolized in the cytosol to fluorescein, which is retained by cells with normally impermeant plasma membranes. Viable and early apoptotic cells exclude PI, a nuclear stain; hence, this method is not suitable to distinguish between them. Moreover, late-apoptotic as necrotic cells take up IP because of cell membrane disruption, although it is also difficult to determine whether they are apoptotic or necrotic.
The identification of late-apoptotic cells was based on nuclear morphology and fluorescent brightness.
Apoptotic cells were counted in randomly selected fields (×400). A total of 500 cells were counted to determine the percentage of apoptosis at the indicated time points of the experimental protocol.
-
(c)
Apoptosis: TUNEL assay
Apoptosis in thymocytes after 5-FU treatment was confirmed by TUNEL assay as described previously (Romero Benitez et al. 2004). Briefly, samples fixed in 4% paraformaldehyde were assayed using the ApoptoTag fluorescein direct in situ apoptosis kit (Intergen Co., New York, USA) according to manufacturer’s instructions. The assay was also performed in control slides. Apoptotic nuclei were identified under a fluorescence microscope. Nuclei of apoptotic cells were stained positive for green fluorescence, whereas counterstaining showed red fluorescence with PI staining.
The percentages of apoptotic cells were calculated from random fields on each slide. One hundred cells were counted up in each field. A total of 500 cells were counted up for every sample taken. Results obtained from thymic images, taken in triplicate (×400) are expressed as a percentage of apoptotic cells.
-
(d)
Isolation of apoptotic DNA fragments
Thymocytes (4 × 107) were treated in 0.2 ml of lysis buffer (50 mM Tris, pH 7.5, 20 mM ethylene-diamine-tetra-acetic acid, and 1% Nonidet P-40) for 40 s. Supernatants with apoptotic DNA were collected after centrifugation at 2,000 rpm for 5 min at room temperature. They were treated with 6 μl of 100 mg/ml RNAase A (Sigma Co.) and 13 μl of 10% sodium dodecyl sulfate (SDS) plus 6 μl of water and were incubated at 56°C for 2 h. Then, 50 μl of proteinase K (100 μg/ml, Sigma Co.) were added to each sample. They were digested for 4 h at 37°C. DNA was precipitated by adding 450 μl of absolute ethanol. After precipitation, DNA was resuspended in 50 μl of water, and 10 μl of the samples were electrophoresed in 1.5% agarose gels containing 0.5 μg/ml ethidium bromide. DNA bands were visualized using 312 nm UV radiation.
Western blotting
To study FAS, Bax, and Caspase 3 expressions by Western blotting, different protein extractions were used.
FAS and Bax were performed essentially as described previously (Juaristi et al. 2007). Briefly, at each time points after 5-FU injection, thymi were treated with radioimmunoprecipitation assay buffer (50 mM Tris, 150 mM NaCl, 2.5 mg/ml deoxycholic acid, 1 mM EDTA, 10 μg/ml Nonidet-40, pH 7.4, supplemented with protease inhibitors: 2.5 mM phenylmethylsulfonyl fluoride).
Several authors have reported that cytosol is a suitable source of pro-caspase 3 and the cleaved forms of caspase 3 (Samali et al. 1999; Yang et al. 2005; Lee et al. 2006), consequently; caspase 3 was evaluated in cytosolic thymic extracts. Thymic cell suspensions were rinsed twice with PBS and then lysed with ice-cold buffer (10 mM 4-2-hydroxyethyl-1-piperazineethanesulfonic acid, pH 7.4, 10 mM KCl, 1.5 mM MgCl2, 0.5 mM dithiotreitol, 0.1% Igepal (Sigma Co. 630), 2.5 mM phenylmethane-sulfonylfluoride, and supplemented with protease inhibitors: 2.5 μg/ml leupeptin, 0.95 μg/ml aprotinin) for 30 min. Cells were briefly sonicated, centrifuged at 14.000×g for 20 min at 4°C, and the supernatant was used as cytosolic fraction.
Proteins from whole cell lysates and cytoplasmatic extracts were loaded onto 12% SDS–polyacrylamide gel electrophoresis (40 μg/well). Proteins were transferred to nitrocellulose membranes to perform Western blottings. As blocking solution, 5% of low-fat milk in Tris-buffered saline–Tween (TBS-T) buffer (50 mM Tris(hydroxymethyl)aminomethane, 175 mM NaCl, adjusted to pH 7.5, with HCl and supplemented with 0.1% Tween-20) was used.
FAS rabbit polyclonal (SC-7886), Bax rabbit polyclonal (SC-6236), and Caspase 3 goat polyclonal (SC-1225; Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA.) were used as primary antibodies. They were diluted in blocking solution at 1/200, 1/400, and 1/100 for FAS, Bax, and Caspase 3, respectively.
Secondary antibodies: IgG goat anti-rabbit (CN 111-035-045) and IgG donkey anti-goat (CN 705-035-147), horseradish peroxidase (HRP) labeled (Jackson Immunoresearch Inc.), were diluted 1/1,000 and 1/500, respectively, in blocking solution. Incubations were performed for 1 h at room temperature. Bound HRP was detected by Opti4CN kit (BioRad). Band intensities were quantified using Scion Image 3.0 Software.
Values that are expressed in arbitrary optical density units (UA) were obtained as previously described (Juaristi et al. 2007).
Image and statistical analysis
All results are expressed as mean ± standard error of mean (SEM). Thymocytes cellularities and viabilities from the 5-FU-treated and the control groups were statistically analyzed using a one-way analysis of variance (ANOVA) test corrected by Bonferroni. Data from all other experiences were compared by ANOVA followed by Dunnett’s test. Analyses were performed using the software package Instat and Prism 4.0 (GraphPad Software Inc., San Diego, USA). A P value < 0.05 was considered statistically significant. Correlation analyses between apoptosis-related proteins and apoptosis indexes were performed using Spearman rank correlations test.
Results
To assess whether 5-FU affects murine normal thymus, a time course study was designed (0–10 days) after a single dose of 5-FU (150 mg/kg) i.p., administration through several experimental procedures.
Thymic weight, cellularity, and viability post-5-FU dosing
Thymus of 5-FU-injected mice showed an acute weight reduction within the first 7 days (p < 0.01). Thymic mass loss started from the sixth hour after 5-FU dosing (80 ± 0.2 mg), reaching low values between days 2 (35 ± 0.10 mg) and 7 (34 ± 0.4 mg) compared to control values (120 ± 0.7 mg). Thymus weight failed to recover to normality through the experience (Fig. 1).
5-FU induces time-dependent weight loss in murine thymus. The thymus was obtained at the indicated time points after 5-FU administration as described in “Materials and methods .” Control weight was set as 100%. Organ weights diminished from 6 h after injection, reaching their lowest values between 48 and 120 h post-5-FU treatment (approximately 75% lower than control). Results were obtained from six mice/group (controls) and six mice/group (5-FU-treated animals) at each time points of the experimental study and expressed as mean ± SEM. **P < 0.01 indicates significant differences between the control and the 5-FU-treated animals
Reduction of this parameter was also reflected in thymocyte counts. Thymic cellularities decreased progressively during the experimental schedule, showing the minimal cell count from day 2 (18.03 ± 1.82 x 106 cells p < 0.01) until the end of the study compared to control values (80.33 ± 12.01 x 106 cells). As regards thymus viabilities, they showed significant reduction between day 1 (79.6 ± 2.5%) and 7 (74.8 ± 0.6%), compared to control values (96.7 ± 1.9%, p < 0.01). Data is shown in Table 1.
Thymic morphologic changes
Interactions between stromal cells and thymocytes play a crucial role in T cell development (Anderson et al. 1996). To assess changes in the thymic architecture post-5-FU injury, scanning electronic microscopy (SEM) images were obtained from 5-FU-injected mice at each time points of the study (Fig. 2).
Effects of 5-FU on thymic architecture. The figure depicts representative scanning electron microscopic images (×2,000) of thymic samples untreated (a) and 5-FU-treated mice at 12 h (b), 2 days (c), 7 days (d), and 10 days (e) post-5-FU. The main feature of 5-FU-induced damage is the acute alteration in the inner thymic architecture. Arrowheads indicate loss of cell–cell contact. Hypocellularity was noticed from the first 12 h (b) to the second day post-treatment (c). An apparent morphological recovery of the thymic architecture was seen from the seventh day onward (d, e). Two mice were used for each time point of the experimental schedule (n = 18)
Architectural changes were observed from the 12th hour to the fifth day post-5-FU injection.
Important cell reductions, as well as the loss of cell–cell contact, were noticed between the first and the third day post-5-FU dosing. Thymus seemed to exhibit a moderate reorganization on day 7, and it was almost normal by day 10.
These results provide substantial evidence of 5-FU toxicity upon thymic tissue, suggesting that thymic hypocellularity is the additive result from the toxic effects of the drug and serious damage of the thymic microenvironment.
To determine whether apoptosis was involved in this process, additional experimental approaches were combined.
Thymic apoptosis
Taking into account that apoptosis has been considered a major mechanism of chemotherapy-induced cell death (Ricci and Zong 2006), apoptosis values of the thymus were determined to elucidate whether this process is involved during 5-FU injury.
Apoptosis was assessed by May Grünwald–Giemsa staining (Fig. 3a), double fluorescent dyes (fluorescein diacetate and PI; Fig. 3b), and TUNEL assay (Fig. 3c), as described in “Materials and Methods.” Percentages of apoptotic cells were obtained by TUNEL, a widely used confirmatory assay for programmed cell death (Fig. 3d).
5-FU-induced thymocyte apoptosis in vivo. Thymic cells were isolated from control and 5-FU-treated animals and fixed as described in “Materials and methods.” Apoptotic biochemical and morphological changes were examined along the experience under light and fluorescence microscopes and TUNEL assay. Representative images were obtained from the control group and 5-FU-treated group 48 h post-injection. a May Grünwald–Giemsa (MG-G) staining of apoptotic cells exhibit shrinkages bodies, with nuclear condensation and fragmented nuclei. b Fluorescence assays with fluorescein diacetate (DAF) and propidium iodide (IP) show viable cells with homogeneous green fluorescence of their cytoplasm. In contrast, apoptotic cells show nuclei with irregular bright red fluorescence as a result of chromatin condensation and nuclear fragmentation. c TUNEL assay was used as a confirmatory technique to assess thymic apoptosis. Nuclei of apoptotic cells were stained positive for green fluorescence. d Percentages of apoptotic cells related to total cells (mean ± SEM) determined by TUNEL assays were represented. Five hundred cells were counted for each sample taken on the scheduled days. Results were obtained from six mice/group (controls) and six mice/group (5-FU-treated animals) at each time points of the experimental study and expressed as mean ± SEM. *P < 0.05 and **P < 0.01 indicate significant differences between apoptotic percentages of the untreated control group and the 5-FU-treated groups
5-FU-injected mice showed an acute increase of apoptotic percentages from the sixth hour (8.5 ± 1.1, p < 0.05) to the fifth day (8.7 ± 0.04, p < 0.05), compared to the control group (3.3 ± 1.07). Maximal apoptotic values were observed on the second day (13.5 ± 1.5 p < 0.01). Apoptotic percentages returned almost to basal values on day 7 (1.2 ± 0.06).
Experimental data shows temporal coincidence among maximal apoptotic values, minimal cellularities, low weight, and deep thymic structural changes.
Effect of 5-FU on DNA damage
Oligonucleosomal cleavage accompanies apoptosis in most of the systems. In this study, agarose gel electrophoresis was used to assess the degradation pattern of nuclear DNA in 5-FU induced apoptosis.
DNA damage was evident in the form of discernible fragmentation ladders in agarose gels (Fig. 4). Fragmented DNA was undetectable in the thymus control sample. Nonetheless, DNA obtained from 48 h post-5-FU-treated mice has revealed characteristic fragments composed of approximately 200 bp. On day 7, DNA degradation almost disappeared. The cleavage of DNA corroborates the previous morphological and biochemical analyses, indicating that in vivo 5-FU-induced apoptosis was maximal during the first 48 h.
Induction of DNA fragmentation in the thymus of mice after 5-FU administration. Samples from the control and the 5-FU-treated mice were obtained at each day of the experimental protocol (n = 2 mice/group). DNA molecules were extracted from 4 × 107 thymic cells and were electrophoretically separated on 1.5% agarose gels containing ethidium bromide. Internucleosomal DNA cleavage was visualized by UV transillumination. The figure shows the results of one representative experiment from three independent determinations at 0, 2, and 7 days post-5-FU
FAS expression
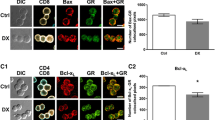
It has been reported that the major proportion of thymic chemotherapeutic drug-induced-apoptosis in vitro is mediated by the CD95/CD95L system (Eichhorst et al. 2001). To determine if 5-FU-induced apoptosis is accompanied by FAS upregulation, immunoblottings were performed (Fig. 5).
Upregulation of FAS induced by 5-FU. Thymi were harvested from 5-FU-treated mice at the scheduled times; cell suspensions were obtained and lysed. Western Blotting analyses were performed as described in “Material and methods .” A representative blot of three independent experiments is shown. FAS expression was noticeable along the study, showing an upregulation in the acute period of 5-FU injury until the fifth day. Thereafter, it returned to the control levels from day 7 onward. Results represent mean ± SEM of three mice per group; **P < 0.01 indicates significant differences between protein expressions from the control group (day 0) and 5-FU-treated group
FAS expression showed an abrupt increase from the sixth hour (8.8 ± 0.74) until the end of the study (8.3 ± 1.4), reaching approximately three times over control values from the 12th hour to the fifth day post-5-FU dosing (11.1 ± 0.2 and 17.6 ± 0.4, respectively, p < 0.01). Interestingly, FAS upregulation was in agreement with the maximal apoptotic values. A direct and significant linear correlation was obtained between FAS overexpression and apoptotic indexes (r = 0.8490, p = 0.007). These data allow us to postulate that FAS is linked to 5-FU-mediated apoptosis in the thymus.
Bax expression
It has been shown in vivo that, blocking the CD95 system in the thymus, the apoptotic process takes place to a lesser extent (Eichhorst et al. 2001). It suggests that another potential mechanism might be involved in cell death, such as the Bax apoptotic pathway. To analyze this assumption, Bax immunoblottings were performed at each scheduled time (Fig. 6). Bax was overexpressed from the sixth hour (34.36 ± 3.8, p < 0.01) to the fifth day (31.07 ± 1.85, p < 0.01) post-5-FU, showing a total increase of two times over control values (39.28 ± 1.03). This fact was almost coincident with the significant increase of the apoptotic values, the cleavage of DNA and FAS upregulation. More interestingly, Bax and FAS overexpressions showed a direct and significant correlation (r = 0.8197, p = 0.0127).
Overexpression of Bax induced by 5-FU. Thymi were harvested from 5-FU-treated mice at the indicated time points; the cell suspensions obtained were lysed and subject to Western blotting analyses as described in “Material and methods.” A representative blot of three independent experiments is shown. Bax expression was noticeable throughout the study, showing an overexpression in the acute period of 5-FU injury. Thereafter, it returned to control levels from day 5 onward. Results represent mean ± SEM of three mice per group; *P < 0.05 and **P < 0.01 indicate significant differences between protein expressions from the control group (day 0) and 5-FU-treated group
These results gave a novel approach to the 5-FU-induced-apoptosis mechanism involved in vivo model, which is consistent with previous in vitro studies.
Caspase 3 expression
To confirm whether pro-caspase 3 was activated during 5-FU treatment, the cleavages of this precursor were detected by immunoblottings. The activation of pro-caspase 3 was indicated by the disappearance of the 32 kd pro-enzyme form (Zhang et al. 2000a, b). Indeed, control values of the inactive caspase 3 (11.6 ± 0.5) showed a remarkable decrease from the 24th hours (5.2 ± 0.4, p < 0.01) to the fifth day (2.8 ± 0.1, p < 0.01) post-5-FU. Moreover, the cleaved active forms of Caspase 3 (20 and 17 Kd) were overexpressed from the first day (11.14 ± 1.6, p < 0.05) to the fifth day (19.34 ± 0.47, p < 0.01) after 5-FU treatment (Fig. 7).
Cleavage of the inactive pro-caspase 3 induced by 5-FU. Thymi were harvested from 5-FU-treated mice at the scheduled times; cell suspensions were obtained and lysed. Western blotting analyses were performed as described in “Material and methods.” A representative blot of three independent experiments is shown. Cleavages of the inactive pro-caspase 3 were noticed from 12 h until the seventh day, indicating the presence of the active caspase 3 enzymes. Results represent mean ± SEM of three mice per group; **P < 0.01 indicates significant differences between protein expressions from the control group (day 0) and 5-FU-treated group
In addition, this pattern is coincident with the increase of the apoptotic values and DNA fragmentation. Furthermore, this fact is also concomitant with FAS and Bax overexpressions, which might reflect the interaction between these proteins during the apoptotic process.
Discussion
The regulation of cell survival and death is critical for the development and function of the mammalian immune system. The importance of apoptosis for the cell number maintenance, the deletion of useless or autoreactive cells, and the removal of expanded lymphocyte clones that are generated during an immune response have already been reported by several authors (Marsden and Strassen 2003; Newton et al. 1998; Smith et al. 1996; Ogasawara et al. 1995).
Although very little is known about apoptosis in thymus upon a chemotherapeutic insult. Severe side effects are caused by the fact that most of cytotoxic drugs are not able to distinguish between normal and tumoral cells (Guest and Uetrecht 2000).
It has been reported that 5-FU causes a reduction of the thymus weight during the first hours of treatment (Eichhorst et al. 2001). However, little is known about the thymus’ behaviour in a longer period.
Experimental data from this in vivo model upon basic parameters of the thymus, taken together with other experimental approaches for evaluating apoptosis, have provided us with a novel scope of the response against 5-FU injury.
In this study, 5-FU induces loss of thymic weight, viability, and cellularity. Even more, thymic alterations of the structural organization, as well as the apoptotic percentages, were seriously altered, along the first 5 days post-5-FU. The aforementioned morphological thymic microenvironmental changes are linked to the enhancement of apoptosis. Thus, it is assumed that 5-FU might trigger the overexpression of key regulatory and executor molecules involved in apoptosis.
Nevertheless, the way in which this drug induces apoptosis is controversial (Fisher 1994). A possible mechanism may be the induction of apoptosis via death receptor, such as FAS (Fulda et al. 2000; Kasibhatla et al. 1998; Herr et al. 1997; Friesen et al. 1996; Krammer 1999).
This report shows that 5-FU induces an upregulation of FAS from the sixth hour onwards. Moreover, the chronological coincidence of this fact with maximal apoptosis and DNA fragmentation seemed to be strongly associated. These results are in agreement with other groups that have studied the mechanism involved during in vivo chemotherapy-induced apoptosis (Eichhorst et al. 2001). However, it has been reported that, when the CD95 system is blocked, apoptosis values are diminished, although not totally abrogated. Therefore, other potential mechanism might be involved in cell death. A possible protein committed in this alternative apoptotic event seemed to be the pro-apoptotic protein Bax (Takemura et al. 2005; Gottlieb and Oren 1998; Friedlander et al. 1996; Miyashita et al. 1995).
This study reveals that 5-FU induces an overexpression of Bax and FAS, both proteins share a similar pattern. Like FAS, Bax upregulation was coincident to maximal apoptotic indexes and the presence of intranucleosomal DNA degradation. These data provide a new insight regarding the expression of Bax during chemotherapy-induced apoptosis, which is consistent with the previously in vitro reported studies (Kobayashi et al. 2000; Zhang et al. 2000a, b; Fincucane et al. 1999; Nita et al. 1998; Martin and Green 1995).
The execution of the apoptotic program appears to be uniformly mediated through consecutive activation of the members of the caspase family (Marsden and Strassen 2003). Previous studies have suggested that caspase 3 is implicated in thymocyte cell death during T-cell development (Jiang et al. 1999; Zhang et al. 2000a, b). Nevertheless, data of caspase 3 activity during thymocyte-induced apoptosis in vivo post-chemotherapy are unavailable.
The current in vivo study provides a first approach about the role of caspase 3 during 5-FU treatment. Experimental data show an increase expression of cleaved caspase 3 from 24 h to 5 days post-5-FU. Interestingly, the occurrence of the active form of caspase 3 was in concordance with maximal apoptotic values, cleavage of DNA, and overexpressions of FAS and Bax.
In summary, in vivo effects of 5-FU showed that the morphological structure of the thymus was deeply altered, leading to hypocellularity, weight loss, and architectural changes. Therefore, this report revealed that 5-FU-induced apoptosis is related to the upregulation of the pro-apoptotic proteins FAS and Bax, and it is also linked to the overexpression of the active form of caspase 3.
Even more, this in vivo study offers a new insight about the pro-apoptotic proteins involved during 5-FU treatment upon the thymus and the possible mechanism involved in the lymphopenia associated with this chemotherapeutic agent.
References
Alnemri ES. Mammalian cell death proteases: a family of highly conserved aspartate specific cysteine proteases. J Cell Biochem. 1997;64:33–42.
Anderson NC, Moore JJT, Jenkinson O, Jenkinson E. Cellular interactions in thymocyte development. Annu Rev Immunol. 1996;14:73–99.
Asaga T, Inaba M, Nagano A, Yanoma S. Induction of apoptosis in breast cancer cells by preoperative oral administration of 5-fluorouracil. Gan To Kagaku Ryoho. 2001;70:46–51.
Ashkenazi A, Dixit VM. Death receptors: signaling and modulation. Science. 1998;281:1305–8.
Chinnaiyan AM, Dixit VW. The cell-death machine. Curr Biol. 1996;6:555–62.
Eichhorst S, Muerkoster S, Weigand M, Kramer P. The chemotherapeutic drug 5-fluorouracil induces apoptosis in mouse thymocytes in vivo via activation of the CD95 (APO-1/Fas) system. Cancer Res. 2001;61:243–8.
Fincucane MD, Bossy-Wetzel E, Waterhouse NJ, Cotter TG, Green DR. Bax-induced caspase activation and apoptosis via cytochrome c release from mitochondria in inhibitable by Bcl-xL. J Biol Chem. 1999;74:2225–33.
Fisher DE. Apoptosis in cancer therapy: crossing the threshold. Cell. 1994;9:91–6.
Friedlander P, Haupt Y, Prives C, Oren MA. Mutant p53 that discriminates between p53-responsive genes cannot induce apoptosis. Mol Cell Biol. 1996;16:4961–71.
Friesen C, Herr I, Krammer PH, Debatin KM. Involvement of the CD95 (APO-1/Fas) receptor/ligand system in drug-induced apoptosis in leukemia cells. Nat Med. 1996;2:574–7.
Fulda S, Strauss G, Meyer E, Debatin KM. Functional CD95 ligand and CD95 death inducing signaling complex in activation-induced cell death and doxorubicin-induced apoptosis in leukemic T cells. Blood. 2000;95:301–8.
Gottlieb TM, Oren M. p53 and apoptosis. Semin Cancer Biol. 1998;8:359–68.
Guest L, Uetrecht J. Drugs toxic to the bone marrow the target stroma cells. Immunopharmacol. 2000;46:103–12.
Henkart PA. ICE family proteases: mediators of all apoptotic cell death? Immunity. 1996;4:195–201.
Herr I, Withelm D, Bohler T, Angel P, Debatin KM. Activation of CD95 (APO-1/Fas) signaling by ceramide mediates cancer therapy-induced apoptosis. EMBO J. 1997;16:6200–8.
Horowitz RW, Heerdt BG, Hu X, Schwartz EL, Walder S. Combination therapy with 5-Fluorouracil and INF-alfa2 induces a nonrandom increase in DNA fragments of less than 3 megabases in HT29 colon carcinoma cells. Clin Cancer Res. 1997;3:1317–22.
Ijiri K, Potten CS. Further studies on the response of intestinal cells of different hierarchical status to eighteen different cytotoxic agents. B J Cancer. 1987;55:113–23.
Jacobson MD. Programmed cell death: a missing key is found. Trends Cell Biol. 1997;7:467–9.
Jiang D, Zheng L, Lenardo MJ. Caspases in T-cell receptor-induced thymocyte apoptosis. Cell Death Differ. 1999;6:402–11.
Jones KH, Senft JA. An improved method to determine cell viability by simultaneous staining with fluorescein diacetate-propidium iodide. J Histochem Cytochem. 1985;33:77–9.
Juaristi AJ, Aguirre MV, Todaro JS, Alvarez MA, Brandan NC. EPO receptor, Bax and Bcl-xL expressions in murine erythropoiesis after cyclophospamide treatment. Toxicology. 2007;231:188–99.
Kasibhatla S, Brunner T, Genestier L, Echeverri F, Mahboubi A, Green DR. DNA damaging agents induce expression of Fas ligand and subsequent apoptosis in T lymphocytes via activation of NF-ĸB and AP-1. Mol Cell. 1998;1:543–51.
Kerr JF. History of the events leading to the formulation of the apoptosis concept. Toxicology. 2002;27:181–2.
Kerr JF, Winterford CM, Harmon BV. Apoptosis. Its significance in cancer and cancer therapy. Cancer. 1994;35:796–807.
Kobayashi T, Sawa H, Morikawa J, Zhang W, Shiku H. Bax induction activates apoptotic cascade via mitochondrial cytochrome c release and bax overespression enhances apoptosis induced by chemotherapeutic agents in DLD-1 colon cancer cells. Jpn J Cancer Res. 2000;91:1264–8.
Krammer PH. CD95 (APO-1/Fas)-mediated apoptosis: live and let die. Adv Immunol. 1999;71:163–210.
Lee H, Kim YJ, Kim HW, Lee DH, Sung M-K, Park T. Induction of apoptosis by cordycep militaris through activation of caspase-3 in leukemia HL-60 cells. Biol Pharm Bull. 2006;29:670–4.
Marsden VS, Strassen A. Control of apoptosis in the immune system: Bcl-2, BH3-only proteins and more. Annu Rev Immunol. 2003;21:71–105.
Martin SJ, Green DR. Protease activation during apoptosis: death by a thousand cuts. Cell. 1995;82:349–52.
Miyashita T, Krajewski S, Krajewska M, Wang HG, Lin HK, Liebermann DA, Hoffman B, Reed JC. Tumor suppressor p53 is a regulator of bcl-2 and bax gene expression in vitro and in vivo. Oncogene. 1995;9:293–9.
Newton K, Harris AW, Bath ML, Smith KGC, Strasser A. A dominant interfering mutant of FADD/Mort1 enhances deletion of autorreactive thymocytes and inhibitions of proliferation of mature T lymphocytes. EMBO J. 1998;17:706–18.
Nita ME, Nagawa H, Tominaga O, Tsuno N, Fuji S, Sasaki S, Fu CG, Takenoue T, Tsuruo T, Muto T. 5-Fluorouracil induces apoptosis in human colon cancer cell lines with modulation of Bcl-2 family proteins. Br J Cancer. 1998;78:986–92.
Ogasawara J, Suda T, Nagata S. Selective apoptosis of CD4 + CD8 + thymocytes by the anti-Fas antibody. J Exp Med. 1995;181:485–91.
Parker WB, Cheng YC. Metabolism and mechanism of action of 5-fluorouracil. Pharmacol Ther. 1990;48:381–95.
Pritchard DM, Watson AJM, Potten CS, Jackman AL, Hickmann JA. Inhibition by uridine but not thymidine of p53-dependent intestinal apoptosis initiated by 5-fluorouracil: evidence for the involvement of RNA perturbation. Proc Nat Acad Sci U S A. 1997;94:1795–9.
Ricci S, Zong W. Chemotherapeutic approaches for targeting cell death pathways. Oncologist. 2006;11:342–57.
Rocha B, Vassalli P, Guy-Grand D. Thymic and extrathymic origins of gut intraepithelial lymphocyte population in mice. J Exp Med. 1994;180:681–6.
Romero Benitez MM, Aguirre MV, Juaristi JA, Alvarez MA, Trifaro JM, Brandan NC. In vitro erythroid recovery following paclitaxel injury: Correlation between GATA-1, c-MYB, NF-E2, Epo receptor expressions and apoptosis. Toxicol Appl Pharmacol. 2004;194:230–8.
Samali A, Cai J, Zhivotovsky B, Jones DP, Orrenius S. Presence of a pre-apoptotic complex of pro-caspase-3, Hsp60 and Hsp10 in the mitochondrial fraction of Jurkat cells. EMBO J. 1999;18:2040–8.
Savill J, Fadok V. Corpse clearance defines the meaning of cell death. Nature. 2000;12,407:784–8.
Smith KGC, Strassen A, Vaux DL. CrmA expression in T lymphocytes of transgenic mice inhibits CD95 (Fas/APO-1)-transducing apoptosis, but does not cause lymphadenophaty on autoimmune disease. EMBO J. 1996;15:5167–76.
Takemura K, Noguchi M, Ogi K, Tokino T, Kubota H, Miyazaki A, Kohama G, Hiratsuka H. Enhanced Bax in oral SCC in relation to antitumor effects of chemotherapy. J Oral Pathol Med. 2005;34:93–9.
Thornberry NA, Lazebnik Y. Caspases: enemies within. Science. 1998;281:1312–6.
Werner JM, Eger K, Jurgen Steinfelder H. Comparison of the rapid pro-apoptotic effect of trans-beta-nitrostyrenes with delayed apoptosis induced by the standard agent 5-fluorouracil in colon cancer cells. Apoptosis. 2007;12:235–46.
Yang MY, Serrine S, Monks L, Monks TJ. 2,3,5-Tris(Glutation-S-yl)hydroquinone (TGHQ)-mediate apoptosis of human promyelocytic leukemia cells is preceded by mitochondrial cytochrome c release in the absence of a decrease in the mitochondrial membrane potential. Tox Sci. 2005;86:92–100.
Yeager AM, Levin J, Levin FC. The effects of 5-fluoruracil on hematopoiesis: studies of murine megakaryocite-CFC, granulocyte-macrophage-CFC, and peripheral blood cell levels. Exp Hematol. 1983;11:944–52.
Zhang J, Mikecz K, Finnega A, Glant TT. Spontaneous thymocyte apoptosis is regulated by a mitochondrion-mediated signaling pathway. J Immunol. 2000a;165:2970–4.
Zhang L, Yu J, Park BH, Kinzler KW, Volgestein B. Role of Bax in the apoptotic response to anticancer agents. Science. 2000b;290:989–92.
Acknowledgment
This work was supported by grants from SEGCyT-UNNE and CONICET, Argentina. We Thank Dr. M. Teresa De Jesus Saavedra and Dr. M. Barbara De Biasio for their expert help with DNA fragmentation. We also thank Professor Milagros Delfino for the English revision.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aquino Esperanza, J.A., Aguirre, M.V., Aispuru, G.R. et al. In vivo 5-flourouracil-induced apoptosis on murine thymocytes: involvement of FAS, Bax and Caspase3. Cell Biol Toxicol 24, 411–422 (2008). https://doi.org/10.1007/s10565-008-9056-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-008-9056-z