Abstract
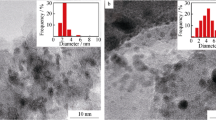
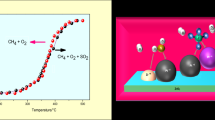
The partial oxidation of methane was studio on Pt/Al2O3, Pt/ZrO2, Pt/CeO2 and Pt/Y2O3 catalysts. For Pt/Al2O3, Pt/ZrO2 and Pt/CeO2, temperature programmed surface reaction (TPSR) studies showed partial oxidation of methane comprehends two steps: combustion of methane followed by CO2 and steam reforming of unreacted methane, while for Pt/Y2O3 a direct mechanism was observed. Oxygen Storage Capacity (OSC) evaluated the reducibility and oxygen transfer capacity of the catalysts. Pt/CeO2 catalyst showed the highest stability on partial oxidation. The results were explained by the higher reducibility and oxygen storage/release capacity which allowed a continuous removal of carbonaceous deposits from the active sites, favoring the stability of the catalyst. For Pt/Al2O3 and Pt/ZrO2 catalysts the increase of carbon deposits around or near the metal particle inhibits the CO2 dissociation on CO2 reforming of methane. Pt/Y2O3 was active and stable for partial oxidation of methane and its behaviour was explained by a change in the reaction mechanism.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
J.H. Lunsford (2000) Catal. Today 63 165 Occurrence Handle1:CAS:528:DC%2BD3cXotlKnur4%3D Occurrence Handle10.1016/S0920-5861(00)00456-9
J.R. Rostrup-Nielsen J. Sehested (2002) Adv. Catal. 47 65 Occurrence Handle1:CAS:528:DC%2BD3sXhtVyhtb4%3D
S. Freni G. Calogero S. Cavallaro (2000) J. Power Sources 87 28 Occurrence Handle1:CAS:528:DC%2BD3cXitFyqtbo%3D Occurrence Handle10.1016/S0378-7753(99)00357-2
P.D.F. Vernon M.L.H. Green A.K. Cheetham A.T. Ashcroft (1990) Catal. Lett. 6 181 Occurrence Handle1:CAS:528:DyaK3MXktlWmsLw%3D Occurrence Handle10.1007/BF00774718
P.M. Torniainen X. Chu L.D. Schmidt (1994) J. Catal. 146 1 Occurrence Handle1:CAS:528:DyaK2cXit1ajs7g%3D Occurrence Handle10.1016/0021-9517(94)90002-7
Y. Boucouvalas Z. Zhang X.E. Verykios (1996) Catal. Lett. 40 189 Occurrence Handle1:CAS:528:DyaK28Xlt1KgtrY%3D Occurrence Handle10.1007/BF00815281
A.A. Lemonidou A.E. Stambouli G.J. Tjatjopoulos I.A. Vasalos (1997) Catal. Lett. 43 235 Occurrence Handle1:CAS:528:DyaK2sXht1Cktro%3D Occurrence Handle10.1023/A:1018975630498
A.G. Steghuis J.G. Ommen ParticleVan J.A. Lercher (1998) Catal. Today 46 91 Occurrence Handle1:CAS:528:DyaK1cXmvVyrsbw%3D Occurrence Handle10.1016/S0920-5861(98)00330-7
L.V. Mattos, E.R. de Oliveira, P.D. Resende, F.B. Noronha and F.B. Passos, Catal. Today 77 (2002) 245.
L.V. Mattos E. Rodino D.E. Resasco F.B. Passos F.B. Noronha (2003) Fuel Process. Technol. 1677 1
C.E. Hori H. Permana K.Y.S. Ng A. Brenner K. More K.M. Rahmoeller D. Belton (1998) Appl. Catal. Environ. B 16 105 Occurrence Handle1:CAS:528:DyaK1cXhvVyhur0%3D Occurrence Handle10.1016/S0926-3373(97)00060-X
F.B. Passos M. Schmal R. Frety (1992) Catal. Lett. 14 57 Occurrence Handle1:CAS:528:DyaK38Xltlymtrc%3D Occurrence Handle10.1007/BF00764219
E. Rogemond N. Essayem R. Frety V. Perrichon M. Primet F. Mathis (1997) J. Catal. 166 229 Occurrence Handle1:CAS:528:DyaK2sXhs1Gntrg%3D Occurrence Handle10.1006/jcat.1997.1493
M. Pettre Ch. Eichner M. Perrin (1946) Trans. Faraday Soc. 43 335 Occurrence Handle10.1039/tf946420335b
D. Dissanayake M.P. Rosynek K.C.C. Kharas J.H. Lunsford (1991) J. Catal. 132 117 Occurrence Handle1:CAS:528:DyaK3MXmtFWmurw%3D Occurrence Handle10.1016/0021-9517(91)90252-Y
M.E.S. Hegarty A.M. O’Connor J.R.H. Ross (1998) Catal. Today 42 225 Occurrence Handle1:CAS:528:DyaK1cXkt1eltbs%3D Occurrence Handle10.1016/S0920-5861(98)00096-0
M.M.V.M. Souza M. Schmal (2005) Appl. Catal. A: Gen. 281 19 Occurrence Handle1:CAS:528:DC%2BD2MXhsFSitrs%3D Occurrence Handle10.1016/j.apcata.2004.11.007
W.J.M. Vermeiren E. Blomsma P.A. Jacobs (1992) Catal. Today 13 421 Occurrence Handle10.1016/0920-5861(92)80168-M
E.P.J. Mallens J.H.B.J. Hoebink C.B. Marin (1995) Catal. Lett. 33 291 Occurrence Handle1:CAS:528:DyaK2MXnt1Grt7s%3D Occurrence Handle10.1007/BF00814232
Y.H. Hu E. Ruckenstein (1998) J. Phys. Chem. A. 102 10568 Occurrence Handle1:CAS:528:DyaK1cXnsF2ns7c%3D Occurrence Handle10.1021/jp9837535
W.Z. Wheng M.S. Chen Q.G. Yan T.H. Wu Z.S. Chao Y.Y. Liao H.L. Wan (2000) Catal. Today 63 317 Occurrence Handle10.1016/S0920-5861(00)00475-2
D. Wang O. Dewaele A.M. Groote Particlede G.F. Froment (1996) J. Catal. 159 418 Occurrence Handle1:CAS:528:DyaK28XivFCgtLg%3D Occurrence Handle10.1006/jcat.1996.0105
L.V. Mattos, E.R. Oliveira, P.D. Resende, P. D., F. B. Noronha and F. B. Passos, Catal. Today 101 (2005) 23.
E. Ruckenstein H. Wang (2000) J. Catal. 190 32 Occurrence Handle1:CAS:528:DC%2BD3cXnsVertA%3D%3D Occurrence Handle10.1006/jcat.1999.2736
H. Nishimoto K. Nakagawa N. Ikenaga T. Suzuki (2002) Catal. Lett. 82 161 Occurrence Handle1:CAS:528:DC%2BD38Xns1Cmtbk%3D Occurrence Handle10.1023/A:1020554309639
J. Wei, E.J. Iglesia, J. Phys. Chem. B 108 (2004) 4094.
M.H. Yao R.J. Baird F.W. Kunz T.E. Hoost (1997) J. Catal. 166 67 Occurrence Handle1:CAS:528:DyaK2sXhtVKgsrk%3D Occurrence Handle10.1006/jcat.1997.1504
J. Kaspar P. Fornasiero M. Graziani (1999) Catal. Today 50 285 Occurrence Handle1:CAS:528:DyaK1MXitVKgu7s%3D Occurrence Handle10.1016/S0920-5861(98)00510-0
D.L. Trimm (1999) Catal. Today 49 3 Occurrence Handle1:CAS:528:DyaK1MXps1Khsw%3D%3D Occurrence Handle10.1016/S0920-5861(98)00401-5
J.R. Rostrup-Nielsen I. Alstrup (1999) Catal. Today 53 311 Occurrence Handle1:CAS:528:DyaK1MXnsFeqsLs%3D Occurrence Handle10.1016/S0920-5861(99)00125-X
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Passos, F.B., Oliveira, E.R., Mattos, L.V. et al. Effect of the support on the mechanism of partial oxidation of methane on platinum catalysts. Catal Lett 110, 161–167 (2006). https://doi.org/10.1007/s10562-006-0105-z
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10562-006-0105-z