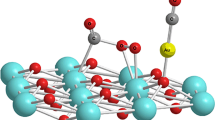
We used density functional theory to examine whether doping oxides makes them better oxidation catalysts. We studied in detail titania doped with Au and used CO oxidation as a test of the oxidizing power of the system. We show that doping with Au, Ag, Cu, Pt, Pd, Ni reduces dramatically the bond of surface oxygen to titania or ceria, making them better oxidation catalysts. These calculations suggest that it is worthwhile to explore doped oxides as oxidation catalysts.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Q. Fu H. Saltsburg M. Flytzani-Stephanopoulos (2003) Science 301 935 Occurrence Handle10.1126/science.1085721 Occurrence Handle1:CAS:528:DC%2BD3sXmt1els7s%3D
Q. Fu W. Deng H. Saltsburg M. Flytzani-Stephanopoulos (2005) Appl. Catal. B 56 57 Occurrence Handle1:CAS:528:DC%2BD2MXhtVKnsLg%3D
J. Guzman S. Carrettin A. Corma (2005) J. Am. Chem. Soc. 127 3286 Occurrence Handle10.1021/ja043752s Occurrence Handle1:CAS:528:DC%2BD2MXhtlCgtLo%3D
A.M. Venezia G. Pantaleo A. Longo G. Di Carlo M.P. Casaletto F.L. Liotta G. Deganello (2005) J. Phys. Chem. B 109 2821 Occurrence Handle10.1021/jp045928i Occurrence Handle1:CAS:528:DC%2BD2MXmslKktA%3D%3D
J. Guzman B.C. Gates (2004) J. Am. Chem. Soc. 126 2672 Occurrence Handle10.1021/ja039426e Occurrence Handle1:CAS:528:DC%2BD2cXht1Wisbk%3D
J.T. Calla R.J. Davis (2005) Catal. Lett. 99 21 Occurrence Handle10.1007/s10562-004-0771-7 Occurrence Handle1:CAS:528:DC%2BD2MXotFKj
N.A. Hodge C.J. Kiely R. Whyman M.R.H. Siddiqui G.J. Hutchings Q.A. Pankhurst F.E. Wagner R.R. Rajaram S.E. Golunski (2002) Catal. Today 72 133 Occurrence Handle10.1016/S0920-5861(01)00487-4 Occurrence Handle1:CAS:528:DC%2BD38XisV2jtbk%3D
Y. Nishihata J. Mizuki T. Akao H. Tanaka M. Uenishi M. Kimura T. Okamoto N. Hamada (2002) Nature 418 164 Occurrence Handle10.1038/nature00893 Occurrence Handle1:CAS:528:DC%2BD38XltFGls7o%3D
H. Tanaka N. Mizuno M. Misono (2003) Appl. Catal. A 244 371 Occurrence Handle1:CAS:528:DC%2BD3sXjtFegur8%3D
H. Tanaka I. Tan M. Uenishi M. Kimura K. Dohmae (2001) Top. Catal. 16/17 63 Occurrence Handle10.1023/A:1016626713430 Occurrence Handle1:CAS:528:DC%2BD3MXns1GgsL0%3D
H. Tanaka M. Taniguchi N. Kajita M. Uenishi I. Tan N. Sato K. Narita M. Kimura (2004) Top. Catal. 30/31 389 Occurrence Handle10.1023/B:TOCA.0000029780.70319.36 Occurrence Handle1:CAS:528:DC%2BD2cXmtVens7k%3D
G.C. Bond (2002) Catal. Today 72 5 Occurrence Handle10.1016/S0920-5861(01)00522-3 Occurrence Handle1:CAS:528:DC%2BD38XisV2jtLw%3D
H. Haruta M. Date (2001) Appl. Cat. A 222 427 Occurrence Handle1:CAS:528:DC%2BD38XjvFai
M. Haruta (2004) Gold Bull. 37 27 Occurrence Handle1:CAS:528:DC%2BD2cXnt1emsL8%3D
R. Meyer C. Lemire S.K. Shaikhutdinor H.-J. Freund (2004) Gold Bull. 37 72 Occurrence Handle1:CAS:528:DC%2BD2cXnt1emsLg%3D
J.P. Perdew J.A. Chevary S.H. Vosko K.A. Jackson M.R. Pederson D.J. Singh C. Fiolhais (1992) Phys. Rev. B 46 6671 Occurrence Handle1:CAS:528:DyaK38XlvFyks7c%3D
J.P. Perdew K. Burke Y. Wang (1996) Phys. Rev. B 54 16533 Occurrence Handle10.1103/PhysRevB.54.16533 Occurrence Handle1:CAS:528:DyaK2sXntFSk
G. Kresse J. Hafner (1993) Phys. Rev. B 47 558 Occurrence Handle10.1103/PhysRevB.47.558 Occurrence Handle1:CAS:528:DyaK3sXlt1Gnsr0%3D
G. Kresse J. Hafner (1994) Phys. Rev. B 49 14251 Occurrence Handle10.1103/PhysRevB.49.14251 Occurrence Handle1:CAS:528:DyaK2cXkvFKrtL4%3D
G. Kresse J. Furthmuller (1996) Phys. Rev. B 54 11169 Occurrence Handle10.1103/PhysRevB.54.11169 Occurrence Handle1:CAS:528:DyaK28Xms1Whu7Y%3D
G. Kresse J. Furthmuller (1996) Comput. Mater. Sci. 6 15 Occurrence Handle10.1016/0927-0256(96)00008-0 Occurrence Handle1:CAS:528:DyaK28XmtFWgsrk%3D
D. Vanderbilt (1990) Phys. Rev. B 41 7892 Occurrence Handle10.1103/PhysRevB.41.7892
G. Makov M.C. Payne (1995) Phys. Rev. B 51 4014 Occurrence Handle10.1103/PhysRevB.51.4014 Occurrence Handle1:CAS:528:DyaK2MXjvVGksbk%3D
W.H. Press S.A. Teukolsky W.T. Flannery B.P. Vetterling (1992) Numerical Recipes in Fortran: The Art of Scientific Computing Cambridge University Press Cambridge
H. Jónsson, G. Mills and K.W. Jacobsen, in Classical and Quantum Dynamics in Condensed Phase Simulations: Proceedings of the International School of Physics “Computer Simulation of Rare Events and the Dynamics of Classical and Quantum Condensed-Phase Systems”, B.J. Berne, G. Cicotti and D.F. eds, Coker (World Scientific Publishing Company, Singapore, 1998) ch. 18.
The results report for the remaining of the paper were obtained on a slab composed of 12 layers (4 triple-layers) in order to reduce the computational cost associated with the 15 layers slab. The energy to form a bridging oxygen vacancy in AuxTi1-xO2(110) oscillates with the slab thickness, as it does for the undoped oxide (see Ref. 28). However, regardless of slab thickness doping produces a dramatic lowering of the energy to form a vacancy and variations due to slab thickness do not alter our qualitative conclusions.
V. Shapovalov and H. Metiu, In preparation (2005).
X.Y. Wu A. Selloni S.K. Nayak (2004) J. Chem. Phys. 120 4512 Occurrence Handle1:CAS:528:DC%2BD2cXhslaku7o%3D
G. Pacchioni F. Frigoli D. Ricci J.A. Weil (2001) Phys. Rev. B 63 054102 Occurrence Handle10.1103/PhysRevB.63.054102
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chrétien, S., Metiu, H. Density Functional Study of the CO Oxidation on a Doped Rutile TiO2(110): Effect of Ionic Au in Catalysis. Catal Lett 107, 143–147 (2006). https://doi.org/10.1007/s10562-005-0014-6
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10562-005-0014-6