Abstract
Purpose
We conducted a meta-analysis of randomized controlled trials (RCTs) and quasi-RCTs to synthesize evidence about the efficacy and safety of alternate-day vs daily dosing of statins.
Methods
We searched selected databases through January 2, 2017 to identify relevant RCTs and quasi-RCTs. The primary outcome was change in low-density lipoprotein cholesterol (LDL-C), total cholesterol (TC), and triglycerides (TG), while secondary outcomes included adverse events and adherence.
Results
Twelve RCTs and 1 quasi-RCT (n = 1023 patients) were included in the analysis. Pooled analysis revealed no statistically significant difference between alternate-day and daily regimens of atorvastatin and rosuvastatin in terms of change in LDL-C (mean difference [MD] 6.79 mg/dL, 95% confidence interval [CI] −1.59, 15.17, p = 0.11, and 10.51 mg/dL, 95%CI −0.23, 21.26, p = 0.06, respectively) and TG (p > 0.05). Daily regimens of atorvastatin and rosuvastatin were superior to alternate-day regimes in term of change in TC (MD 12.45 mg/L, 95%CI 8.14, 16.76, p < 0.00001, and 15.80 mg/dL, 95%CI 5.66, 25.95, p = 0.002, respectively). For all outcomes, there was no statistically significant difference between alternate-day and daily regimens for both fluvastatin and pravastatin (p > 0.05). Both regimens of statins were generally well tolerated with good adherence.
Conclusions
Alternate-day dosing of individual statins (especially atorvastatin and rosuvastatin) is as efficacious as daily dosing on LDL-C and TG.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Statins are some of the most widely prescribed medications worldwide [1,2,3]. Statins are effective in lowering low-density lipoprotein cholesterol (LDL-C) by up to 52% and triglycerides (TG) by up to 44% [4, 5]. Additionally, they increase high-density lipoprotein cholesterol (HDL-C) by up to 10% [6,7,8]. For every 1.0 mmol/L (38.7 mg/dL) reduction in LDL-C, there is a corresponding 20–25% drop in cardiovascular (CV) disease (CVD) mortality [9]. Therefore, their key role in primary and secondary prevention of CVD is now well established [10,11,12,13,14,15].
In general, statins have a favorable safety profile [16,17,18,19]. However, adverse events (AEs) are considered the most common cause for statin therapy discontinuation [20,21,22,23]. The most common AEs associated with statin therapy are muscle-related [24,25,26,27]. Statin-associated adverse muscle symptoms (SAMSs) may affect 10–15% of patients receiving statins [25, 28, 29]. Fortunately, statin-associated fatal rhabdomyolysis is a rare event that develops in only about 2–3 cases per 100,000 patients treated per year [30, 31]. Alternate-day dosing of statins has been proposed for patients who experience side effects with daily dosing [27]. Therefore, several studies investigated the effectiveness of this approach compared with daily dosing of statins [32,33,34,35,36]. However, most of these studies are limited by small sample size. Therefore, we conducted a systematic review and meta-analysis of randomized controlled trials (RCTs) and quasi-RCTs to provide more definitive evidence about the efficacy and safety of alternate-day dosing of statins compared with daily dosing.
Methods
We followed preferred reporting items for systematic reviews and meta-analyses (PRISMA) statement guidelines for the preparation of this meta-analysis (Supplementary File 1: Table S1) [37]. All steps were conducted in accordance with the “Cochrane handbook for systematic reviews of interventions.” This study was prospectively registered in PROSPERO, University of York (CRD42017054519).
Literature Search Strategy
We performed a computerized literature search of PubMed, SCOPUS, Web of Science, and Embase from inception until January 2, 2017 using the following keywords: (atorvastatin OR fluvastatin OR lovastatin OR pitavastatin OR pravastatin OR rosuvastatin OR simvastatin OR cerivastatin OR mevinolin OR statin OR statins) and (alternate day OR alternate-day OR every other day OR every-other-day OR non-every day OR non-daily OR twice a week OR twice-weekly OR twice weekly OR once a week OR once-weekly OR once weekly). To ensure that no relevant studies were missed, we conducted manual searches for potential trials that were included in the reference lists of review articles on that topic and the abstracts from selected congresses: scientific sessions of the European Society of Cardiology (ESC), the American Heart Association (AHA), American College of Cardiology (ACC), European Society of Atherosclerosis (EAS), and National Lipid Association (NLA). The wild-card term “*” was used to increase the sensitivity of the search strategy. The literature search was restricted to articles published in English and conducted on human subjects.
After removal of duplicate articles by Endnote X7 (Thompson Reuter, CA, USA), two independent authors screened the retrieved articles in two steps: the first step was to screen the titles and abstracts for eligibility, and the second step was to screen the full text of the eligible abstracts according to the inclusion and exclusion criteria. Disagreement was resolved by the opinion of a third author (MB).
Study Selection
Original studies were included if they met the following criteria: (і) a RCT or a quasi-RCT (i.e., the method of allocation is known but is not considered strictly random); (іі) comparison of the efficacy of alternate-day dosing of statin therapy vs daily dosing on one or more of the following lipid profile parameters: total cholesterol (TC), LDL-C, or TG for a duration ≥6 weeks; and (ііі) reporting complete data on serum lipid concentrations after treatment.
Exclusion criteria included (і) non-randomized trials, observational studies, experimental studies, reviews, book chapters, and theses; (іі) studies whose full texts were not available; (ііі) studies that contained fabricated data or were retracted by the journal; and (іv) studies whose data were not reliable for extraction (e.g., reported graphically only). Exclusion of an article for the last reason was applied only after contacting the authors for details and not receiving a response.
Data Extraction
Eligible studies were reviewed and the following data were extracted: (1) first author’s name; (2) year of publication; (3) study location; (4) study design; (5) interventions doses and durations; (6) study population characteristics; (7) concentrations of TC, LDL-C, and TG; and (8) incidence of adverse events.
If there were multiple groups with different treatment doses of any or both regimens (alternate-day and daily), we extracted the data of the group with the same dose as the comparator group. Moreover, if there were multiple follow-up time points, we extracted the data from the last time point. Data extraction was performed independently by two reviewers. Disagreements were resolved by a third reviewer (MB).
Outcomes of Interest
The primary outcome was the mean change in one of the following lipid parameters: TC, LDL-C, and TG. Secondary outcomes included the incidence of AEs and the adherence of patients with statin therapy.
Quantitative Data Synthesis
Lipid concentrations were collated in mg/dL in an Excel spreadsheet. To convert from mmol/L to mg/dL, cholesterol (TC and LDL-C) levels were multiplied by 38.67, and TG levels were multiplied by 88.57. If the changes in lipid levels during the trial were not directly reported, they were calculated as follows: (measure at end of follow-up) − (measure at baseline). Standard deviations (SDs) of the change scores were calculated using the following formula: SD = square root [(SDpre-treatment)2 + (SDpost treatment)2 − (2R × SDpre treatment × SDpost-treatment)], assuming a correlation coefficient (R) = 0.5 [38,39,40]. If the outcome measures were reported as median and range, mean and SD values were estimated using the method described by Hozo et al. [41] and if reported as mean and standard error (SE) or confidence interval (CI), mean and SD values were estimated using the method described by Altman et al. [42].
The change scores in lipid concentrations between randomization groups (i.e., alternate day vs daily dosing) were pooled as mean differences (MDs) with a 95% CI in a meta-analysis model. RevMan version 5.3 (The Cochrane Collaboration, Oxford, UK) was used to conduct this analysis.
Subgroup Analysis
To investigate the effect of each type of statin on the lipid concentrations, data were presented into four subgroups as follows: (і) atorvastatin, (іі) rosuvastatin, (ііі) fluvastatin, and (іv) pravastatin.
Assessment of Heterogeneity
Heterogeneity was assessed by visual inspection of the forest plots and measured by I-square and Chi-square tests. We interpreted heterogeneity according to the recommendations of Cochrane handbook for systematic reviews of interventions, in which an alpha level (for Chi-square test) below 0.1 represents the presence of a significant heterogeneity, and I-square test is interpreted as follows: 0–40%: heterogeneity might not be important; 30–60%: may represent a moderate heterogeneity; 50–90%: may represent a substantial heterogeneity. In the case of a significant heterogeneity, a random-effect model was used. Otherwise, a fixed-effect model was employed.
Sensitivity Analysis
In order to evaluate the effect of each study on the combined effect estimate, we performed a sensitivity analysis using the “leave-one-out approach,” i.e., removing one study each time and repeating the analysis. Also, the same approach was conducted to resolve the presence of any significant heterogeneity. Moreover, we conducted an additional sensitivity analysis that excluded studies that compared the same overall doses in both alternate-day and daily groups (e.g., 10 mg daily vs 20 mg every other day).
Meta-Regression
We conducted a random-effect meta-regression using the unrestricted maximum likelihood method to evaluate the association between the changes in the lipid parameters and the duration of statin therapy.
Quality Assessment
We used “The Cochrane Collaboration tool” for assessing the risk of bias in the included trials. This tool includes the following domains: sequence generation (selection bias), allocation sequence concealment (selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective outcome reporting (reporting bias), and other potential sources of bias. The authors’ judgment is classified as “low risk,” “high risk,” or “unclear risk” of bias. This assessment was performed independently by two reviewers. Disagreements were resolved by a third reviewer (MB).
Publication Bias
Potential publication bias was evaluated by visual inception of Begg’s funnel plot asymmetry and was statistically confirmed by Egger’s weighted regression test [43]. Additionally, the “trim-and-fill” approach was conducted to correct the funnel plot asymmetry resulting from the publication bias by imputing the potentially missing number of studies [44]. We used Comprehensive Meta-Analysis (CMA) version 2 (Biostat, NJ, USA) to conduct the publication bias analyses.
Results
Flow and Characteristics of Included Studies
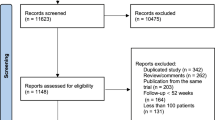
Our search resulted in 2398 citations. Following removal of duplicates and title/abstract screening, only 19 articles were eligible for full-text screening. Of these 19 articles, 6 were excluded for the following reasons: non-randomized trials (n = 3), observational study (n = 1), insufficient data for analysis (n = 1), and daily dosing was compared with three-times-weekly dosing not with alternate-day dosing (n = 1). Finally, 12 RCTs [33,34,35, 45,46,47,48,49,50,51,52,53] and 1 quasi-RCT [32] met our inclusion criteria and were included in our analysis (see PRISMA flow diagram; Fig. 1).
In total, 1023 patients were included in our analysis. Of these patients, 505 received alternate-day dosing of statin therapy and 518 received daily dosing. The sample size of the included trials ranged from 37 to 284 patients. Only 3 trials used the same overall doses of statins in both alternate-day and daily groups (e.g., 10 mg daily vs 20 mg every other day), while the remaining trials used different overall doses of statins but with the same doses given per day (e.g., 10 mg daily vs 10 mg every other day). Included trials were published between 1998 and 2014, and they were conducted in the USA (n = 3), India (n = 3), Iran (n = 2), Egypt, China, Turkey, Canada, and Thailand. The duration of the treatment ranged from 6 to 24 weeks. The summary of the included studies and their main results are shown in Table 1; the baseline characteristics of their populations are shown in Table 2.
Quality of the Included Studies
According to The Cochrane Collaboration tool, the included trials ranged from low to moderate quality. The summary of quality assessment domains of the included studies is shown in Fig. 2.
Efficacy Analysis (Primary Outcome)
Overall Pooled Analysis
The overall pooled analysis of 13 RCTs comparing the impact of alternate-day dosing vs daily dosing of statin therapy on TG (MD 6.56 mg/dL, 95%CI −1.76, 14.88, p = 0.12, Fig. 3) was not statistically significant. However, TC and LDL-C lowering was significantly greater in the daily dosing group (MD 12.08 mg/dL, 95%CI 8.58, 15.58, p < 0.00001 (Fig. 4) and 7.95 mg/dL, 95%CI 2.62, 13.29, p = 0.003 (Fig. 5), respectively). A statistically significant heterogeneity was present for LDL-C (Chi2 p < 0.0001 and TG (Chi2 p = 0.01). This was resolved by sensitivity analysis that excluded the study by Rifaie et al. [51] (Chi2 p = 0.31) for TG. However, sensitivity analysis failed to resolve the heterogeneity for LDL-C. For TC, no heterogeneity was present (Chi2 p = 0.24).
Subgroup Analysis
Subgroup pooled analysis of 8 RCTs compared the impact of alternate-day dosing vs daily dosing of atorvastatin on LDL-C (MD 6.79 mg/dL, 95%CI −1.59, 15.17, p = 0.11, Fig. 5) and TG (MD: 6.43 mg/dL, 95%CI −5.75, 18.61, p = 0.30, Fig. 3) was not statistically significant. The lowering effect of atorvastatin on TC was statistically greater in the daily dosing group (MD 12.45 mg/dL, 95%CI 8.14, 16.76, p < 0.00001, Fig. 4). A statistically significant heterogeneity was present for LDL-C (Chi2 p < 0.00001) and TG (Chi2 p = 0.002). This was resolved by sensitivity analysis that excluded the study by Rifaie et al. [51] (Chi2 p = 0.11) for TG. However, sensitivity analysis failed to resolve the heterogeneity for LDL-C. In case of TC, no heterogeneity was present (Chi2 p = 0.27).
Subgroup pooled analysis of 3 trials compared the impact of alternate-day dosing vs daily dosing of rosuvastatin on LDL-C (MD 10.51 mg/dL, 95%CI −0.23, 21.26, p = 0.06, Fig. 5) and TG (MD 9.20 mg/dL, 95%CI −2.78, 21.19, p = 0.13, Fig. 3) was not statistically significant. However, TC lowering was statistically greater in the daily dosing group (MD 15.80 mg/dL, 95%CI 5.66, 25.95, p = 0.002, Fig. 4). No statistically significant heterogeneity was observed for all outcomes (Chi2 p > 0.1) except for LDL-C, and it was best resolved by sensitivity analysis that excluded the study by Wongwiwatthananukit et al. [53] (Chi2 p = 0.24).
For all outcomes, subgroup analysis showed insignificant difference between alternate-day dosing vs. daily dosing of both fluvastatin and pravastatin (p > 0.05, Figs. 3, 4, and 5).
Secondary Outcomes
Adverse Events
Most of the included studies did not report the incidence of AEs in each group separately. Therefore, we could not pool the AEs in a meta-analysis model. However, both regimens were generally well tolerated. The descriptive summary of incidence of AEs in the included studies is shown in Table 3.
Compliance with Both Regimens
Of the 13 trials that were included in this meta-analysis, 7 trials [32, 35, 47, 48, 50,51,52] did not report adherence in any of the treatment groups, 5 trials [33, 45, 46, 49, 53] reported no difference in adherence between both groups, and 1 trial [34] suggested that the alternate-day regimen resulted in a worse adherence when compared with the daily dosing.
Sensitivity Analysis
The leave-one-out sensitivity analysis revealed that the overall combined analyses were robust for all outcomes except for TG, which was sensitive to the study by Rifaie et al. [51] (p = 0.002). As for the “atorvastatin” subgroup, the combined effect estimate on TC was robust. However, the combined effect estimates on LDL-C and TG were sensitive to the studies by Aghasadeghi et al. [45] (p = 0.01) and Rifaie et al. [51] (p = 0.02), respectively. In case of the “rosuvastatin” subgroup, the combined effect estimate on TG was robust. However, the combined effect estimates on TC and LDL-C were sensitive to the studies by Dulay et al. [32] (p = 0.06) and Li et al. [48] (p = 0.006), respectively. Removing any of the mentioned studies from the analysis resulted in favoring the daily dosing over the alternate-day dosing except for the study by Dulay et al. [32], which caused an insignificant difference between the two groups. The summary of leave-one-out sensitivity analyses is shown in Supplementary File 2: Tables S2a–c. The overall combined effect estimates on TC, LDL-C, and TG were robust in case of sensitivity analysis that excluded the three trials that used the same overall doses of statins in both regimens (e.g., 10 mg daily vs 20 mg every other day).
Meta-Regression
The random-effect meta-regression using the unrestricted maximum likelihood method did not indicate any statistically significant association between the changes in any of the lipid parameters and the duration of statin therapy (LDL-C [slope −0.1961; 95% CI −1.218 to 0.8257; p = 0.7068]; TC [slope −0.4631; 95% CI −1.0548 to 0.1286; p = 0.125]; TG [slope 1.2882; 95% CI −0.1099 to 2.6863; p = 0.0709]). Meta-regression scatter plots are shown in Supplementary File 3: Figs. S1–S3.
Publication Bias
Visual inception of funnel plots suggested a potential publication bias for TG. The trim and fill approach corrected the asymmetry of the funnel plot of TG by imputing 4 studies (corrected MD 0.28354 mg/dL, 95%CI −8.51685, 9.08393, Supplementary File 3: Fig. S4). However, the funnel plots were symmetric for TC and LDL-C (Supplementary File 3: Fig. S5, S6). Egger’s test excluded the presence of publication bias in all outcomes (two-tailed p > 0.05).
Discussion
To our knowledge, this is the first meta-analysis to evaluate the efficacy and safety of alternate-day dosing of statin therapy compared with daily dosing. The results of this meta-analysis suggest that there was no statistically significant difference between alternate-day and daily regimens of atorvastatin and rosuvastatin in terms of LDL-C and TG. However, the effect of daily regimens of atorvastatin and rosuvastatin were superior to alternate-day regimes on TC.
Atorvastatin and rosuvastatin are considered high-potency statins [54]. Besides the long half-life of atorvastatin which is about 14 h, its long-lasting active metabolites prolong its inhibiting effect on HMG-CoA reductase up to 20–30 h [54,55,56,57]. The half-life of rosuvastatin is 19 h [58]. This prolonged action of both atorvastatin and rosuvastatin could explain the effectiveness of their alternate-day dosing on LDL-C compared with daily dosing. In contrast, our meta-analysis did not show any difference between alternate-day dosing of either fluvastatin or pravastatin versus daily dosing for all outcomes despite the short half-lives of these agents (1.2 and 1.8 h, respectively) [16]. The effectiveness of the alternate-day dosing with fluvastatin and pravastatin is more dependent upon their prolonged action at the cellular level than plasma half-life [52].
Statin intolerance has become a critical challenge to the widespread use of guideline-directed LDL-C lowering therapy [59]. Among myocardial infarction survivors who experience statin intolerance, the risk of recurrent myocardial infarction, CV events, and hospitalizations for CV events is 50% higher than patients who adhere to high-intensity statin therapy [60]. Several factors improve adherence to high-intensity statin therapy; these include interaction with the prescriber, participation in cardiac rehabilitation, and elimination of economic barriers [31]. A change in statin agent and/or dosage is a recommended approach by many consensus documents and the practice of lipid specialists worldwide [61, 62]. Some patients can tolerate alternate-day statins but not daily dose statins. In this meta-analysis, the use of alternate-day dosing of statins (particularly the long-acting statins, atorvastatin, and rosuvastatin) is a reasonable treatment option in patients who are intolerant to more than one statin.
The issue of compliance with the alternate-day dosing has been a limitation to this regimen [52]. According to the current meta-analysis, only 1 RCT reported poor compliance with alternate-day regimen [34]. However, the durations of most of the included trials were relatively short; therefore, long-term compliance with alternate-day regimen compared with daily regimen has not been established.
It has been suggested in many large studies that statins are associated with an increase in the risk of new-onset diabetes mellitus (NODM) [63, 64]. Moreover, Preiss et al. concluded, in a large meta-analysis of RCTs, that high-dose statins are associated with a larger risk of NODM compared with moderate-dose statins [65]. From this, we could say that, theoretically, alternate-day statins may have a reduced risk of NODM. However, this hypothesis needs to be tested in large RCTs.
This meta-analysis has several limitations: (1) the sample size of most of included trials was relatively small [33, 45, 47, 48, 52]; (2) heterogeneity was observed between studies especially in term of change in LDL-C. This heterogeneity may result from many factors including the population characteristics, hypersynthesis vs decreased catabolism of LDL (with possibly different half-lives of LDL particles), and statin doses or the duration of treatment. However, this significant heterogeneity was addressed by using a random-effect model in the analysis. It was also explored by subgroup analysis and meta-regression; (3) we excluded an important RCT because the data were insufficient for analysis [36]; (4) alternate-day regimens with fluvastatin and pravastatin were investigated in only 1 RCT for each statin [46, 52]; (5) most of the included trials were of short duration [33, 45, 48, 51]; (6) the included studies did not use a standardized tool to assess SAMS [66]; (7) we could not pool the AEs of both groups in a meta-analysis model because most of the included studies did not report the AEs in each group separately [32, 46, 49, 50, 52, 53]; and (8) none of the included studies reported any data on non-HDL-C, a secondary target of therapy in several recent lipid guideline documents [67, 68].
Based on the results of this meta-analysis, further large-scale RCTs with long-term treatment are recommended to confirm these findings and investigate the effects of alternate-day dosing of statins vs daily dosing on patient compliance particularly in patients with SAMS, risk of NODM and CV events. More RCTs are needed to investigate the efficacy of other statin regimens (e.g., twice or once weekly) compared with daily and alternate-day regimens. Also, there is a need to investigate the effectiveness of combining other statin regimens (e.g., twice or once weekly) with other lipid-lowering agents, especially since some of these options have demonstrated favorable results in a few small studies [69,70,71].
In conclusion, this meta-analysis shows that alternate-day dosing of individual statins (especially atorvastatin and rosuvastatin) is as efficacious as daily dosing on LDL-C and TG. Moreover, this regimen is well-tolerated with good adherence. Therefore, alternate-day dosing is a reasonable treatment option in patients with statin intolerance. Further large-scale RCTs are recommended to confirm and extend these findings.
References
Ramkumar S, Raghunath A, Raghunath S. Statin therapy: review of safety and potential side effects. Acta Cardiol Sin. 2016;32:631–9.
Farnier M, Davignon J. Current and future treatment of hyperlipidemia: the role of statins. Am J Cardiol. 1998;82:3J–10J.
Banach M, Mikhailidis DP, Kjeldsen SE, Rysz J. Time for new indications for statins? Med Sci Monit. 2009;15:MS1–5.
Bakker-Arkema RG, Davidson MH, Goldstein RJ, Davignon J, Isaacsohn JL, Weiss SR, et al. Efficacy and safety of a new HMG-CoA reductase inhibitor, atorvastatin, in patients with hypertriglyceridemia. JAMA. 1996;275:128–33.
Hunninghake DB, Stein EA, Bays HE, Rader DJ, Chitra RR, Simonson SG, et al. Rosuvastatin improves the atherogenic and atheroprotective lipid profiles in patients with hypertriglyceridemia. Coron Artery Dis. 2004;15:115–23.
Huang W-C, Lin T-W, Chiou K-R, Cheng C-C, Kuo F-Y, Chiang C-H, et al. The effect of intensified low density lipoprotein cholesterol reduction on recurrent myocardial infarction and cardiovascular mortality. Acta Cardiol Sin. 2013;29:404–12.
Taylor F, Huffman MD, Macedo AF, Moore TH, Burke M, Davey Smith G, et al. Statins for the primary prevention of cardiovascular disease. Cochrane Database Syst Rev. 2013; doi:10.1002/14651858.CD004816.pub5.
Odden MC, Pletcher MJ, Coxson PG, Thekkethala D, Guzman D, Heller D, et al. Cost-effectiveness and population impact of statins for primary prevention in adults aged 75 years or older in the United States. Ann Intern Med. 2015;162:533–41.
Perk J, De Backer G, Gohlke H, Graham I, Reiner Ž, Verschuren WMM, et al. European guidelines on cardiovascular disease prevention in clinical practice (version 2012). Atherosclerosis. 2012;223:1–68.
Pedersen TR, Olsson AG, Faergeman O, Kjekshus J, Wedel H, Berg K, et al. Lipoprotein changes and reduction in the incidence of major coronary heart disease events in the scandinavian simvastatin survival study (4S). Circulation. 1998;97:1453–60.
Mabuchi H, Kita T, Matsuzaki M, Matsuzawa Y, Nakaya N, Oikawa S, et al. Large scale cohort study of the relationship between serum cholesterol concentration and coronary events with low-dose simvastatin therapy in Japanese patients with hypercholesterolemia and coronary heart disease. Circ J. 2002;66:1096–100.
Sacks FM, Pfeffer MA, Moye LA, Rouleau JL, Rutherford JD, Cole TG, et al. The effect of pravastatin on coronary events after myocardial infarction in patients with average cholesterol levels. N Engl J Med. 1996;335:1001–9.
Cheung BMY, Lauder IJ, Lau C-P, Kumana CR. Meta-analysis of large randomized controlled trials to evaluate the impact of statins on cardiovascular outcomes. Br J Clin Pharmacol. 2004;57:640–51.
Pyörälä K, Ballantyne CM, Gumbiner B, Lee MW, Shah A, Davies MJ, et al. Reduction of cardiovascular events by simvastatin in nondiabetic coronary heart disease patients with and without the metabolic syndrome: subgroup analyses of the scandinavian simvastatin survival study (4S). Diabetes Care. 2004;27:1735–40.
Shepherd J, Cobbe SM, Ford I, Isles CG, Lorimer AR, Macfarlane PW, et al. Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. N Engl J Med. 1995;333:1301–8.
Schachter M. Chemical, pharmacokinetic and pharmacodynamic properties of statins: an update. Fundam Clin Pharmacol. 2005;19:117–25.
De Angelis G. The influence of statin characteristics on their safety and tolerability. Int J Clin Pract. 2004;58:945–55.
Black DM. A general assessment of the safety of HMG CoA reductase inhibitors (statins). Curr Atheroscler Rep. 2002;4:34–41.
Golomb BA, Evans MA. Statin adverse effects: a review of the literature and evidence for a mitochondrial mechanism. Am J Cardiovasc Drugs. 2008;8:373–418.
Maningat P, Gordon BR, Breslow JL. How do we improve patient compliance and adherence to long-term statin therapy? Curr Atheroscler Rep. 2013;15:291.
Banach M, Serban M-C. Discussion around statin discontinuation in older adults and patients with wasting diseases. J Cachexia Sarcopenia Muscle. 2016;7:396–9.
Grundy SM. Statin discontinuation and intolerance: the challenge of lifelong therapy. Ann Intern Med. 2013;158:562.
Mikhailidis DP, Katsiki N, Athyros VG. Adherence to statin treatment: an important issue in clinical practice. Curr Med Res Opin. 2016:1–2.
Banach M, Aronow WS, Serban M-C, Rysz J, Voroneanu L, Covic A. Lipids, blood pressure and kidney update 2015. Lipids Health Dis. 2015;14:167.
Banach M, Rizzo M, Toth PP, Farnier M, Davidson MH, Al-Rasadi K, et al. Statin intolerance - an attempt at a unified definition. Position paper from an international lipid expert panel. Arch Med Sci. 2015;11:1–23.
Banach M, Stulc T, Dent R, Toth PP. Statin non-adherence and residual cardiovascular risk: there is need for substantial improvement. Int J Cardiol. 2016;225:184–96.
Patel J, Martin SS, Banach M. Expert opinion: the therapeutic challenges faced by statin intolerance. Expert Opin Pharmacother. 2016;17:1497–507.
Joy TR, Hegele RA. Narrative review: statin-related myopathy. Ann Intern Med. 2009;150:858–68.
Rallidis LS, Fountoulaki K, Anastasiou-Nana M. Managing the underestimated risk of statin-associated myopathy. Int J Cardiol. 2012;159:169–76.
Collins R, Reith C, Emberson J, Armitage J, Baigent C, Blackwell L, et al. Interpretation of the evidence for the efficacy and safety of statin therapy. Lancet. 2016;388:2532–61.
Colantonio LD, Huang L, Monda KL, Bittner V, Serban M-C, Taylor B, et al. Adherence to high-intensity statins following a myocardial infarction hospitalization among medicare beneficiaries. JAMA Cardiol. 2017;16:1886–97.
Dulay D, LaHaye SA, Lahey KA, Day AG. Efficacy of alternate day versus daily dosing of rosuvastatin. Can J Cardiol. 2009;25:e28–31.
Jafari M, Ebrahimi R, Ahmadi-Kashani M, Balian H, Bashir M. Efficacy of alternate-day dosing versus daily dosing of atorvastatin. J Cardiovasc Pharmacol Ther. 2003;8:123–6.
Ghia CJ, Panda AS, Khobragade LR, Jha RK, Rambhad GS. Alternate day versus once daily atorvastatin for primary prevention of (CHD) in naïve patients of dyslipidemia. J Clin Diagn Res. 2014;8:27–31.
Keleş T, Akar Bayram N, Kayhan T, Canbay A, Sahin D, Durmaz T, et al. The comparison of the effects of standard 20 mg atorvastatin daily and 20 mg atorvastatin every other day on serum LDL-cholesterol and high sensitive C-reactive protein levels. Anadolu Kardiyol Derg. 2008;8:407–12.
Matalka MS, Ravnan MC, Deedwania PC. Is alternate daily dose of atorvastatin effective in treating patients with hyperlipidemia? The alternate day versus daily dosing of atorvastatin study (ADDAS). Am Heart J. 2002;144:674–7.
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009;339:b2535.
Balk EM, Earley A, Patel K, Trikalinos TA, Dahabreh IJ. Empirical assessment of within-arm correlation imputation in trials of continuous outcomes. Agency for Healthcare Research and Quality (US); 2012. Report No.: 12(13)-EHC141-EF.
Follmann D, Elliott P, Suh I, Cutler J. Variance imputation for overviews of clinical trials with continuous response. J Clin Epidemiol. 1992;45:769–73.
Methods guide for effectiveness and comparative effectiveness reviews. Agency for Healthcare Research and Quality (US); 2008. PMID: 21433403.
Hozo SP, Djulbegovic B, Hozo I, Hogg R, Craig A, Mood A, et al. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. 2005;5:13.
Altman DG, Bland JM. Standard deviations and standard errors. BMJ. 2005;331:903.
Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–34.
Duval S, Tweedie R. Trim and fill: a simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics. 2000;56:455–63.
Aghasadeghi K, Zare D. Efficacy of alternate day dosing of atorvastatin. Cent Eur J Med. 2008;3:163–6.
Graham MR, Lindsey CC, Kennedy JA. Maintenance of low-density lipoprotein goal with step-down pravastatin therapy. Pharmacotherapy. 2002;22:21–6.
Hadjibabaie M, Vosough-Ghanbari S, Radfar M. Comparative efficacy of daily versus alternate-day dosing of atorvastatin in type 2 diabetic patients. Therapy. 2007;5:541–5.
Li J-J, Yang P, Liu J, Jia Y-J, Li Z-C, Guo Y-L, et al. Impact of 10mg rosuvastatin daily or alternate-day on lipid profile and inflammatory markers. Clin Chim Acta. 2012;413:139–42.
Pattanaik S, Malhotra S, Sharma YP, Pandhi P. Comparison of alternate-day atorvastatin treatment to daily treatment in maintaining LDL-cholesterol targets in patients with variable coronary risk profile. J Cardiovasc Pharmacol. 2012;59:479–84.
Pramanik S, Das AK, Chakrabarty M, Bandyopadhyay SK, Ghosh M, Dalai CK. Efficacy of alternate-day versus everyday dosing of atorvastatin. Indian J Pharmacol. 2012;44:362–5.
Rifaie O, Zahran A, Nammas W. Alternate-day versus daily atorvastatin in coronary artery disease: a randomized study. Anadolu Kardiyol Derg. 2012;12:90–6.
Rindone JP, Hiller D, Arriola G. A comparison of fluvastatin 40 mg every other day versus 20 mg every day in patients with hypercholesterolemia. Pharmacotherapy. 1998;18:836–9.
Wongwiwatthananukit S, Sansanayudh N, Dhummauppakorn R, Kitiyadisai C. Efficacy and safety of rosuvastatin every other day compared with once daily in patients with hypercholesterolemia. Ann Pharmacother. 2006;40:1917–23.
Elis A, Lishner M. Non-every day statin administration—a literature review. Eur J Intern Med. 2012;23:474–8.
Reindl EK, Wright BM, Wargo KA. Alternate-day statin therapy for the treatment of hyperlipidemia. Ann Pharmacother. 2010;44:1459–70.
Stern RH, Yang BB, Hounslow NJ, MacMahon M, Abel RB, Olson SC. Pharmacodynamics and pharmacokinetic-pharmacodynamic relationships of atorvastatin, an HMG-CoA reductase inhibitor. J Clin Pharmacol. 2000;40:616–23.
Lea AP, McTavish D. Atorvastatin. A review of its pharmacology and therapeutic potential in the management of hyperlipidaemias. Drugs. 1997;53:828–47.
Martin PD, Warwick MJ, Dane AL, Hill SJ, Giles PB, Phillips PJ, et al. Metabolism, excretion, and pharmacokinetics of rosuvastatin in healthy adult male volunteers. Clin Ther. 2003;25:2822–35.
Degreef LE, Opdam FL, Teepe-Twiss IM, Jukema JW, Guchelaar HJ, Tamsma JT. The tolerability and efficacy of low-dose simvastatin in statin-intolerant patients. Eur J Intern Med. 2010;21:293–6.
Serban M-C, Colantonio LD, Manthripragada AD, Monda KL, Bittner VA, Banach M, et al. Statin intolerance and risk of coronary heart events and all-cause mortality following myocardial infarction. J Am Coll Cardiol. 2017;69:1386–95.
Hovingh GK, Gandra SR, McKendrick J, Dent R, Wieffer H, Catapano AL, et al. Identification and management of patients with statin-associated symptoms in clinical practice: a clinician survey. Atherosclerosis. 2016;245:111–7.
Rosenson RS, Gandra SR, McKendrick J, Dent R, Wieffer H, Cheng L-I, et al. Identification and management of statin-associated symptoms in clinical practice: extension of a clinician survey to 12 further countries. Cardiovasc Drugs Ther. 2017;31:187–95.
Sattar N, Preiss D, Murray HM, Welsh P, Buckley BM, de Craen AJM, et al. Statins and risk of incident diabetes: a collaborative meta-analysis of randomised statin trials. Lancet. 2010;375:735–42.
Preiss D, Sattar N. Statins and the risk of new-onset diabetes. Curr Opin Lipidol. 2011;22:460–6.
Preiss D, Seshasai SRK, Welsh P, Murphy SA, Ho JE, Waters DD, et al. Risk of incident diabetes with intensive-dose compared with moderate-dose statin therapy. JAMA. 2011;305:2556.
Colantonio LD, Kent ST, Huang L, Chen L, Monda KL, Serban M-C, et al. Algorithms to identify statin intolerance in medicare administrative claim data. Cardiovasc Drugs Ther. 2016;30:525–33.
Sniderman AD, Toth PP, Thanassoulis G, Furberg CD. An evidence-based analysis of the national lipid association recommendations concerning non-HDL-C and apoB. J Clin Lipidol. 2016;10:1248–58.
Banach M, Jankowski P, Jóźwiak J, Cybulska B, Windak A, Guzik T, et al. PoLA/CFPiP/PCS guidelines for the management of dyslipidaemias for family physicians 2016. Arch Med Sci. 2017;1:1–45.
Reddy KJ, Singh M, Batsell RR, Bangit JR, Zaheer MS, John S, et al. Efficacy of combination drug pulse therapy in maintaining lipid levels in patients intolerant of daily statin use. J Clin Hypertens. 2009;11:766–8.
Athyros VG, Tziomalos K, Kakafika AI, Koumaras H, Karagiannis A, Mikhailidis DP. Effectiveness of ezetimibe alone or in combination with twice a week atorvastatin (10 mg) for statin intolerant high-risk patients. Am J Cardiol. 2008;101:483–5.
Ruisinger JF, Backes JM, Gibson CA, Moriarty PM. Once-a-week rosuvastatin (2.5 to 20 mg) in patients with a previous statin intolerance. Am J Cardiol. 2009;103:393–4.
Acknowledgements
We thank Stephen A LaHaye (MD, FRCPC) and Andrew G Day (MSc) for providing data from their trial [32].
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Funding
None to declare.
Conflict of Interest
Dimitri P. Mikhailidis has given talks and attended conferences sponsored by MSD, AstraZeneca and Libytec; Peter P. Toth consults for Abbvie, Amarin, AstraZeneca, Amgen, Gemphire, Kowa, Merck, Regeneron, and Sanofi and serves on the Speakers Bureau for Amarin, Amgen, Kowa, Merck, Regeneron, and Sanofi; Patrick Moriarty has research funding from Regeneron, Sanofi-Aventis, Pfizer, Novartis, Amgen, Ionis, and Catabasis, and he serves as a consultant for Genzyme, Kowa, Duke Clinical Research Institute, Eliaz Therapeutics, Aegerion, Alexion, and Esperion; Paul Muntner received grant support and honoraria from Amgen; Alberico L. Catapano has received research grants to his institution from Amgen, Astra-Zeneca, Merck, Regeneron/Sanofi, and Sigma Tau and honoraria for lectures, advisory boards, or as a steering committee member from Aegerion, Akcea, Amgen, Sanofi-Regeneron, Pfizer, AstraZeneca, ISIS Pharma, Kowa, Lilly, Boehringer Ingelheim, MSD,Sigma Tau, Recordati; Michael J. Pencina declares research grants from Sanofi-Regeneron; Robert S. Rosenson declares research funding to his institution from Akcea, Amgen, Astra Zeneca, Eli Lilly, Esperion, Medicines Company, Regneron, and Sanofi; advisory board fees from Akcea, Amgen, Easy Vitals, Eli Lilly, Regneron and Sanofi; honoraria from Kowa; royalties from UpToDate, Inc.; and stock holdings in MediMergent LLC; Maciej Banach declares advisory boards fees from Abbott Vascular, Amgen, Daichi Sankyo, Esperion, Lilly, MSD, Resverlogix, Sanofi-Aventis, Speakers Bureau from Abbott/Mylan, Abbott Vascular, Actavis, Akcea, Amgen, Biofarm, KRKA, MSD, Sanofi-Aventis, and Valeant and grants from Valeant, Sanofi-Aventis.
Ethical Approval
This article does not include any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Rights and permissions
About this article
Cite this article
Awad, K., Mikhailidis, D.P., Toth, P.P. et al. Efficacy and Safety of Alternate-Day Versus Daily Dosing of Statins: a Systematic Review and Meta-Analysis. Cardiovasc Drugs Ther 31, 419–431 (2017). https://doi.org/10.1007/s10557-017-6743-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-017-6743-0