Abstract
Purpose
Pathological cardiac remodeling, characterized by cardiac hypertrophy and fibrosis, is a pathological feature of many cardiac disorders that leads to heart failure and cardiac arrest. Vinpocetine, a derivative of the alkaloid vincamine, has been used for enhancing cerebral blood flow to treat cognitive impairment. However, its role in pathological cardiac remodeling remains unknown. The aim of this study is to examine the effect of vinpocetine on pathological cardiac remodeling induced by chronic stimulation with angiotensin II (Ang II).
Methods
Mice received Ang II infusion via osmotic pumps in the presence of vehicle or vinpocetine. Cardiac hypertrophy and fibrosis were assessed by morphological, histological, and biochemical analyses. Mechanistic studies were carried out in vitro with isolated mouse adult cardiac myocytes and fibroblasts.
Results
We showed that chronic Ang II infusion caused cardiac hypertrophy and fibrosis, which were all significantly attenuated by systemic administration of vinpocetine. In isolated adult mouse cardiomyocytes, vinpocetine suppressed Ang II-stimulated myocyte hypertrophic growth. In cultured cardiac fibroblasts, vinpocetine suppressed TGFβ-induced fibroblast activation and matrix gene expression, consistent with its effect in attenuating cardiac fibrosis. The effects of vinpocetine on cardiac myocyte hypertrophy and fibroblast activation are likely mediated by targeting cyclic nucleotide phosphodiesterase 1 (PDE1).
Conclusions
Our results reveal a novel protective effect of vinpocetine in attenuating pathological cardiac remodeling through suppressing cardiac myocyte hypertrophic growth and fibroblast activation and fibrotic gene expression. These studies may also shed light on developing novel therapeutic agents for antagonizing pathological cardiac remodeling.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In response to chronic mechanical/volume overload and neurohormonal activation due to cardiovascular diseases (such as hypertension, myocardial infarction, and valvular malfunction), hearts undergo pathological remodeling associated with myocardial hypertrophy and fibrosis, which ultimately leads to contractile dysfunction and heart failure. Therefore, it is believed that pathogenic cardiac remodeling represents a major risk factor and a leading predictor of heart failure and mortality. The neurohormonal system renin-angiotensin is overridden in various cardiovascular diseases and plays critical roles in cardiac hypertrophy and remodeling [1]. The importance of the renin-angiotensin system has been demonstrated by many large-scale clinical trials in which angiotensin-converting enzyme (ACE) inhibitors and Ang II type I receptor antagonists significantly improve the survival rates of heart failure patients by decreasing cardiac remodeling [2]. Myocyte hypertrophic growth, characterized by the increased size of individual cardiomyocytes, results from changes in gene transcription, stimulation of protein synthesis, and increased assembly of myofibrils. Cardiac fibroblasts become activated and differentiated to smooth muscle-like myofibroblasts in response to chronic pressure and stress overload, resulting in cell proliferation, migration, and enhanced extracellular matrix (ECM) synthesis. The excessive deposition of ECM by myofibroblasts promotes myocardial stiffness, impairs cardiac function, and contributes to the progression of heart failure [3]. Therefore, developing novel and systemically safe therapeutic agents for antagonizing pathological cardiac remodeling is currently in high demand.
Vinpocetine is produced by slight modification of the vincamine, an alkaloid molecule extracted from the periwinkle plant, Vinca minor [4]. Vinpocetine (trade name Cavinton) was originally marketed in 1978 in Hungary and has been used in many Asian and European countries for treating cognitive disorders, such as stroke, senile dementia, and memory disturbances. Currently, different types of vinpocetine-containing memory pills such as Intelectol (Memory Secret, Miami, FL) and Memolead (Kao Kabushiki Kaisha, Tokyo, Japan) are used worldwide as dietary supplements. There have been no significant side effects and toxicity reported at therapeutic doses of vinpocetine. Vinpocetine has multiple pharmacological effects: improving brain blood flow by acting as a cerebral vasodilator; enhancing cerebral metabolism by increasing oxygen and glucose uptake and stimulating neuronal ATP production [5–7], as well as serving as a potent anti-inflammatory agent in multiple cell types, including epithelial cells, vascular smooth muscle cells, endothelial cells, and macrophages [8]. Vinpocetine also has a number of different cellular targets, such as Ca2+/calmodulin (Ca2+/CAM)-stimulated cyclic nucleotide phosphodiesterase 1 (PDE1), voltage-dependent Na+ channels [4], and IκB kinase (IKK) [8]. We have previously shown that in the peripheral vasculature, vinpocetine promotes vascular relaxation [9] and antagonizes vascular neointimal hyperplasia [10] and atherosclerosis in rodents [11]. In this study, we investigated the novel roles of vinpocetine in cardiac hypertrophy, fibrosis, and pathological cardiac remodeling in vitro and in vivo.
Materials and Methods
Animals
All animals were used in accordance with the guidelines of the National Institutes of Health Institute of Laboratory Animal Resources, 1996) and American Heart Association for the care and use of laboratory animals. The procedures were performed in accordance with experimental protocols that were approved by the University Committee on Animal Resources at the University of Rochester. C57/BL6 male mice were purchased from The Jackson Laboratory (Bar Harbor, ME). Animals were housed under a 12-h light-dark regimen. Cardiac remodeling was induced in vivo by subcutaneous infusion of Ang II (800 ng/min/kg) for 2 weeks or corresponding vehicle using Alzet osmotic mini-pumps (model 1002, Durect Corp, Cupertino, CA) as described previously [12, 13]. Animals at age of 10 weeks were anesthetized with inhaled isoflurane and osmotic mini-pumps were implanted subcutaneously on the back slightly posterior to the scapulae. For systemic vinpocetine treatment, mice were intraperitoneally injected with 5 mg/kg vinpocetine or vehicle every day as described previously [10, 11]. Blood pressure was recorded by tail artery blood pressure measurement. Mouse hearts were excised, excised hearts were washed with saline to remove the blood, and the whole hearts were weighed. Hearts were used for histological and immunoblotting analyses.
Histological Analysis
Excised hearts were fixed in 4% buffered paraformaldehyde and embedded in paraffin. Hearts were transversely sectioned (5 μm), deparaffinized, and stained with Oregon Green 488 or fluorescein isothiocyanate conjugated wheat germ agglutinin (WGA) (Thermo Fisher Scientific), hematoxylin-eosin, or Masson’s trichrome. The cardiomyocytes’ cross-sectional area was measured in more than 200 cardiomyocytes per section for each animal. The fibrosis fraction was defined as the ratio of the Masson’s trichrome-stained blue area to the myocardial area.
Isolation and Culture of Adult Mouse Ventricular Myocytes
Adult mouse cardiomyocytes were isolated from hearts of C57BL/6J mice by enzymatic dissociation using collagenase type II in a Langerdorff perfusion system, according to a previously described protocol with modification [14]. In brief, heparinized and anesthetized mouse heart were rapidly excised, cannulated, and perfused with Ca2+ free isolation buffer (120 mM NaCl, 15 mM KCl, 0.6 mM KH2PO4, 0.6 mM Na2HPO4, 4.6 mM NaHCO3, 1.2 mM MgSO4, 10 mM HEPES, 30 mM taurine, 5.5 mM glucose, 10 mM 2,3-butanedione monoxime (BDM), 10 mM creatine monohydrate, pH 7.4) for 3 min followed by collagenase type II (Worthington) mixed isolation buffer (50 mg in 50 mL isolation buffer, 40 nM CaCl2) until digestion was complete. Myocytes were exposed to stopping buffer (10% FBS and 12.5 μM CaCl2 in isolation buffer) for increasing Ca2+ concentration to 1.4 mM, plated cells in laminin-precoated dishes with plating buffer (2.5% FBS and 1.4 mM CaCl2 in isolation buffer), after 2 h, cells cultured with prewarmed culture medium (0.1% BSA, 10 mM HEPES, 4 mM NaHCO3, 1% penicillin/streptomycin, 10 μM blebbistatin, 0.5% insulin-selenium-transferrin in MEM + Earle’s salts + l-glutamine medium).
Cardiac Myocyte Hypertrophy In Vitro
Adult mouse cardiac myocyte hypertrophy was analyzed as described previously with modification [12]. Myocytes were cultured for 72 h in the presence of blebbistatin, a myosine II inhibitor that prevents myocyte contraction and increases myocyte viability over longer culture durations [15]. Hypertrophy was induced with treatment of either Ang II (100 nM) with indicated treatments. After 72 h, cells were fixed in 4% paraformaldehyde, and microscopic pictures were then taken. The cell area was quantified using Image Pro. Each experimental condition was repeated for at least three times, and 300 or more myocytes were quantified per experiment.
Isolation and Culture of Cardiac Fibroblasts
Fibroblasts were isolated along with cardiac myocytes. When isolating fibroblasts, the supernatant remaining after the initial 10 min settling phase of cardiac myocytes was centrifuged at 2000g for 5 min. This pellet was then resuspended in DMEM + 10% FBS + 2% P/S. Fibroblasts were plated directly into 12-well plates for experimentation, at a density of approximately at 5 × 104 cells/mL. After allowing several hours for attachment, cells were washed several times in PBS, and medium was replaced with DMEM containing 10% FBS and 2% P/S. Cells were then cultured for 2 days and then serum starved (DMEM + 2% P/S) for 24 h before treatment. After serum starvation, cells were stimulated with TGFβ (10 ng/mL) for 24 h in the presence of vehicle, the PDE1 inhibitor IC86340, and/or vinpocetine. Cells were then washed briefly with PBS and flash frozen for RNA extraction.
Real-time PCR
Real-time PCR was used to detect the mRNA expressions of B-type natriuretic peptide (BNP), smooth muscle alpha actin (α-SMA), type 1 collagen (col 1), fibronectine (Fn-1), PDE1A, PDE1C, and TGFβ1 in heart tissue based on manufacture’s instruction of the kit. The primers were BNP: 5′-TCCTAGCCAGTCTCCAGAGCAA-3′ (forward) and 5′-GGTCCTTCAAGAGCTGTCTCTG-3′ (reverse); α-SMA: 5′-GCTTCGCTGGTGATGATGCTC-3′ (forward) and 5′-AGTTGGTGATGATGCCGTGTTC-3′ (reverse); Col1: 5′-CCTCAGGGTATTGCTGGACAAC-3′ (forward) and 5′-CAGAAGGACCTTGTTTGCCAGG-3′ (reverse); Fn 1: 5′- GCTCAGCAAATCGTGCAGC-3′ (forward) and 5′-CTAGGTAGGTCCGTTCCCACT-3′ (reverse); PDE1A:5′-TGAGCACACAGGAACAACAAAC-3′ (forward) and 5′-GTTCCGAAGATCCCTCCAGTC-3′ (reverse); PDE1C:5′-TAGCCATGGTCTTTGCAGCTGC-3′ (forward) and 5′-CAAGCACTGAGCGGTCGTTGTA-3′ (reverse); TGFβ1:5′-ACCATGCCAACTTCTGTCTGGG-3′ (forward) and 5′-TTGTGTTGGTTGTAGAGGGCAA-3′ (reverse); and GAPDH:5′-TCAAGAAGGTGGTGAAGCAG-3′ (forward) and 5′-TGGGAGTTGCTGTTGAAGTC-3′ (reverse). GAPDH was used as a loading control. Total RNA isolation from cardiac tissues and primary cardiomyocytes was performed according to standard methods. 0.5 μg of RNA from each sample was reverse-transcribed by a reverse transcription system (Bio-Rad) following the manufacturer’s protocol. The real-time PCR was performed by Bio-Rad real-time PCR kit and instrument. Briefly, a 20-μL reaction mixture containing 1 μL cDNA template, 10 μL SYBR master mix, 8 μL H2O, and 1 μL of each primer was amplified by the following thermal parameters: denaturing at 95 °C for 3 min and 40 cycles of the amplification step (denaturation at 95 °C for 10 s, annealing and extension at 60 °C for 60 s). Results are expressed as the ratio of BNP, α-SMA, col 1, or Fn-1 normalized to GAPDH mRNA.
Measuring Intracellular cGMP
cGMP levels were measured with the AlphaScreen cGMP Detection Kit (PerkinElmer) according to the manufacturer’s instruction. In brief, the isolated adult mouse cardiomyocytes were plated in MEM without phenol red in 35 mm dishes. Cardiomyocytes were pretreated with vinpocetine for 30 min before Ang II (100 nM) treatment of 15 min. After the treatment, cardiomyocytes were lysed in 20 μL lysis buffer and then stored at −80 °C for future use. The assay was performed in 1/2 area 96-well plate. The cGMP standard curve was prepared by serial dilution in lysis buffer. Ten microliters lysate or cGMP standard solutions were added into each well. Five microliters anti-cGMP acceptor beads were then added to each well and incubated at room temperature in the dark for 1 h. Ten microliters biotinylated cGMP-tracer/streptavidin donor bead mixture were added to each well and incubated at room temperature in the dark for 1 h. The plate was read with the Envision plate reader.
PDE Activity Assay
PDE activity assay was performed as described previously with modification [12, 13]. Frozen heart tissue samples were homogenized in PDE assay lysis buffer [40 mM Tris⋅HCl (pH 7.5), 15 mM β-mercaptoethanol, 20% (vol/vol) glycerol, 1 mM Na3VO4, 100 nM Okadaic acid, Abcam protease inhibitor mixture]. cAMP/cGMP–PDE activities were measured using 1 μM cAMP or cGMP substrate in the presence of 4 μg/mL calmodulin and 0.8 mM CaCl2 (for Ca2+/CaM-dependent PDE assay). Briefly, PDE assay reactions were assembled on ice until addition of cAMP substrate. Reactions were incubated at 25 °C for 10 min and then were terminated at 100 °C for 1 min. After cooling at RT for 10 min, the remaining cAMP or cGMP level in the reaction lysate was measured by AlphaScreen assay kit (PerkinElmer). PDE1 activities were defined as total PDE activity minus PDE activity in the presence of PDE1 inhibitor IC86340 (15 µM). Activities were normalized to protein concentration.
Western Blot
Western blot was performed as previously described [12, 13]. Briefly, lysates were prepared in RIPA buffer with protease inhibitor cocktail (Sigma). The concentrations of total proteins were measured by Bradford protein assay (Bio-Rad). Total lysates were loaded on SDS-PAGE, and electrotransferred onto PVDF membrane. The primary antibodies rabbit anti-p-SMAD2 (Cell signaling), rabbit anti-p-SMAD3 (Cell signaling), and mouse anti-GAPDH (Millipore) were used.
After washing with PBST, the membranes were incubated with horseradish peroxidase coupled secondary antibodies. The signals were visualized using ECL reagents (Amersham).
Immunochemical Staining
Following treatment with vinpocetine for 30 min, fibroblasts were stimulated with TGFβ for 24 h. Cells were fixed in 4% buffered paraformaldehyde and washed with PBS for several times. After blocking with 2% BSA for 1 h, cells were incubated in blocking buffer within anti-α-SMA Cy3 primary antibody (mouse monoclonal, Sigma) overnight at 4 °C. Primary antibody was removed; cells were washed with PBS for three times and then incubated in FITC-secondary antibody for 1 h at room temperature. Following the washing with PBS, cells were labeled with DAPI to show nucleus. The fluorescence microscopy was used to take images.
Statistics
Data are expressed as the mean ± SEM. Comparisons between two groups were evaluated using Student’s t test. One-way ANOVA followed by Tukey’s post hoc test were used for multiple comparisons. P < 0.05 was considered statistically significant.
Results
Vinpocetine Attenuated Heart Enlargement In Vivo in Ang II-Induced Mouse Models
Neurohormonal overactivation of renin-angiotensin plays critical roles in cardiac hypertrophy and remodeling [1]. We performed continuous subcutaneous infusion of Ang II using osmotic mini-pumps, a model well-known to induce cardiac hypertrophy and fibrosis. We found that the heart size assessed by the global morphology (Fig. 1a) as well as the ratio of heart weight versus body weight (Fig. 1b) or tibia length (Fig. 1c) was markedly increased in the Ang II infusion group compared to the vehicle infusion group. There was no significant change of body weight among different groups (Online Resource 1). Interestingly, heart enlargement by Ang II was significantly suppressed by daily vinpocetine treatment at the dose of 5 mg/kg/day. At the same dose, vinpocetine has been previously shown to significantly attenuate lung inflammation [8], injury-induced vascular intimal hyperplasia [10], and atherosclerosis [11] in mouse models. Ang II significantly increased blood pressure as expected and vinpocetine partially attenuated Ang II-induced blood pressure elevation (Online Resource 2a), which is consistent with previously reported vasodilatory effect of vinpocetine [9]. Vinpocetine did not alter the heart rate (Online Resource 2b). Vinpocetine did not have any effect on global heart morphology in normal mice under no disease-related perturbation (Online Resource 3a, b).
Vinpocetine attenuated cardiac hypertrophy induced by Ang II infusion in vivo. a Representative gross heart images showing the effect of vinpocetine on global heart enlargement induced by Ang II. Scale bars 3 mm. Mice were infused with saline (control) or Ang II (800 ng/min/kg) for 14 days and systemically administrated with vehicle or vinpocetine (5 mg/kg/day) daily via IP injection for 17 days (3 days prior and 14 days during Ang II infusion). b, c Quantified results of Ang II-infused model: heart/body weight (HW/BW) ratio (mg/g) or heart weight/tibia length (HW/TL) ratio (mg/mm). Data represent means ± SEM from control (n = 8), Ang II (n = 10), and Ang II + vinpocetine (n = 10). *P < 0.05 vs. control group and # P < 0.05 vs. Ang II group
Vinpocetine Attenuated Ang II-Induced Cardiac Hypertrophy and Fibrosis In Vivo
To investigate the effects of vinpocetine on cardiac structural changes, we measured myocyte cross-sectional area, an indication of myocyte hypertrophy in vivo. We found that Ang II infusion significantly increased cross-sectional area (Fig. 2a, b) and cardiac hypertrophic biochemical marker B-type natriuretic peptide (BNP) expression (Fig. 2c) as anticipated, which were largely suppressed by daily vinpocetine (5 mg/kg/day, IP) treatment. Moreover, we assessed cardiac fibrosis by measuring interstitial collagens. Vinpocetine markedly reduced cardiac fibrosis induced by Ang II (Fig. 3a, b). It is evident that vinpocetine did not exhibit any significant effect on cardiac hypertrophy and fibrosis in normal mice without disease-related perturbation (Online Resource 3c-e).
Vinpocetine inhibited cardiomyocyte hypertrophy induced by Ang II in vivo. a Representative images of heart section stained for cell membrane with oregon green 488 or fluorescein isothiocyanate-conjugated wheat germ agglutinin (WGA). Myocyte cross-sectional area, the area encircled by a yellow dotted line, was calculated. Scale bars 20 μm. b Quantified results of cross-sectional area (averaged from 200 random myocytes per section per animal; n = 8 for the control group, n = 10 for the Ang II, and n = 10 for the Ang II + vinpocetine group. c Hypertrophic marker BNP mRNA expression normalized to GAPDH analyzed by quantitative real-time RT-PCR (n = 5). *P < 0.05 vs. the control group and # P < 0.05 vs. the Ang II group
Vinpocetine inhibited cardiac fibrosis induced by Ang II in vivo. a Representative images showing collagen deposition (blue) in left ventricle of mouse hearts depicted by Masson’s trichrome staining. b Quantification of percentage fibrosis (n = 8 for the control group, n = 10 for the Ang II group, and n = 10 for the Ang II + vinpocetine group). Scale bar 50 μm. *P < 0.05 vs. the control group and # P < 0.05 vs. the Ang II group
Vinpocetine Inhibited Cardiomyocyte Hypertrophy In Vitro
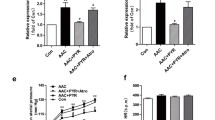
To examine whether vinpocetine has direct effect on cardiomyocytes, we examined the effect of vinpocetine on isolated adult mouse cardiomyocyte hypertrophy by measuring cell area (Fig. 4a). Indeed, vinpocetine reduced Ang II-induced increased cell areas in a dose-dependent manner (Fig. 4b).
Vinpocetine inhibited cardiomyocyte hypertrophy in vitro. a Representative bright-field images of isolated cardiomyocytes treated Ang II (100 nM) or saline (control) for 72 h to induce hypertrophic growth. Scale bar 50 μm. b Vinpocetine dose-dependently inhibited cardiomyocyte hypertrophic growth. Adult mouse myocytes were pre-treated with indicated dose of vinpocetine, followed by Ang II (100 nM) or saline (control) stimulation for 72 h. Quantification of cell area (the area encircled by yellow dotted line in a. Data represent the averages of a minimum of 100 cardiomyocytes from each animal heart preparation and n = 5 independent animal hearts per condition. c IC86340 dose-dependently inhibited cardiomyocyte hypertrophic growth. Adult mouse myocytes were pre-treated with indicated dose of IC86340, followed by Ang II (100 nM) or saline (control) stimulation for 72 h. d Effects of vinpocetine and IC86340 together on cardiomyocyte hypertrophy. Adult mouse myocytes were pre-treated with vehicle or IC86340 (15 μM) with different doses of vinpocetine as indicated, followed by Ang II (100 nM) or saline (control) stimulation for 72 h. e Effects of vinpocetine on intracellular cGMP levels in adult mouse cardiomyocytes (n = 3). *P < 0.05 vs. the control group. *P < 0.05 vs. the control group and # P < 0.05 vs. the Ang II group. ns no significant difference
Among a number of molecular targets of vinpocetine [4, 8], PDE1 represents the first one to be identified. The IC50 values of vinpocetine for inhibiting PDE1 are approximately 10–40 μM, which may vary dependent on different PDE1 isozymes or with distinct experiments [8, 16]. Previous studies have revealed that PDE1 is important for rat cardiac myocyte hypertrophy [12] and fibroblast activation in vitro [13], as well as ISO-induced cardiac remodeling in vivo [12]. Therefore, we hypothesized that the inhibitory effect of vinpocetine on cardiac remodeling may be exerted through inhibiting PDE1. Consistently, a PDE1-selective inhibitor IC86340 also dose-dependently attenuated mouse adult cardiomyocyte hypertrophy (Fig. 4c), similar to vinpocetine. Interestingly, when PDE1 activity was inhibited by IC86340, different doses of vinpocetine exhibited no additional effect (Fig. 4d), suggesting that vinpocetine and IC86340 may share similar mechanism and PDE1 is likely to serve as the pharmacological target for the anti-hypertrophic effect of vinpocetine.
PDE1 family members are encoded by three different genes, PDE1A, PDE1B, and PDE1C. Among them, PDE1A and PDE1C but not PDE1B are expressed in the mouse heart [12]. Indeed, we found that Ang II infusion increased PDE1A and PDE1C expression (Online Resource 4a, b) and PDE1 activity (Online Resource 4c) in the heart, consistent with previously reported upregulation of PDE1A and PDE1C mRNA and/or protein in rodent and human hearts under pathological remodeling [12, 13, 17]. PDE1 family members are Ca2+/CaM-stimulated PDEs, and we have previously shown that PDE1 is responsible for Ang II-mediated reduction of cGMP in cardiomyocytes as assessed by using the PDE1-selective inhibitor IC86340 [12, 13]. Similarly, in the present study, we found that vinpocetine also abolished Ang II-mediated reduction of cGMP in mouse adult cardiomyocytes (Fig. 4e), providing further supporting evidence for the role of vinpocetine in targeting PDE1 in cardiomyocytes.
Vinpocetine Inhibited Cardiac Fibroblast Activation and ECM Synthesis In Vitro
We next evaluated the effects of vinpocetine on adult mouse cardiac fibroblast activation by measuring the induction of smooth muscle alpha-actin (α-SMA), a marker of fibroblast transdifferentiation to myofibroblast. As shown in Fig. 5a, b, α-SMA protein and mRNA were induced by Ang II, which was markedly suppressed by vinpocetine. In addition, vinpocetine also significantly reduced Ang II-induced expression of ECM such as type I collagen (Fig. 5c) and fibronectin (Fig. 5d). Similarly, when PDE1 activity was inhibited by IC86340, vinpocetine had no additional effect (Fig. 5e), suggesting that vinpocetine inhibits cardiac fibroblast activation likely by targeting PDE1.
Vinpocetine inhibited cardiac fibroblast activation and ECM synthesis in vitro. a Representative immunofluorescent microscopic images showing the effects of vinpocetine on α-SMA protein levels in cultured mouse adult cardiac fibroblasts. Cardiac fibroblasts at passage 1 were seeded and cultured overnight in DMEM supplemented with 10% FBS, serum-starved for 48 h, and pretreated with vehicle of vinpocetine (10 μM), then stimulated with 10 ng/mL of TGFβ for 24 h. α-SMA protein was immunostained (green) and nucleus was stained by DAPI (blue). b–d qRT-PCR results showing effects of the vinpocetine on TGFβ stimulated pro-fibrotic marker gene expression, including α-SMA (b), type I collagen (Col1) (c), and fibronectin 1 (Fn 1) (d). e Effects of vinpocetine and PDE1 inhibition together on cardiac fibroblast activation. Adult mouse cardiac fibroblasts were pre-treated with vehicle, IC86340 (15 μM), or both of vinpocetine (10 μM) and IC86340 (15 μM), followed by stimulation with TGFβ (10 ng/mL) for 24 h. The similar results were obtained from at least three independent experiments. *P < 0.05 vs. the control group and # P < 0.05 vs. the Ang II group. ns no significant difference
Discussion
In the present study, we demonstrate that systemically applied vinpocetine inhibits pathological cardiac remodeling including myocardial hypertrophy and fibrosis in mice in response to pathological neurohormonal stress such as chronic Ang II infusion. In addition, using isolated adult cardiac myocytes and fibroblasts, we show that vinpocetine directly attenuated Ang II-stimulated myocyte hypertrophy as well as fibroblast activation and ECM synthesis, likely by targeting PDE1 (Fig. 6). To our knowledge, this is the first study to evaluate the effect of vinpocetine in animal models of cardiac diseases. Our results demonstrated novel and important pharmacological effects of vinpocetine on preventing cardiac hypertrophy, fibrosis, and cardiac remodeling. Pathological cardiac remodeling is involved in the pathogenesis of various cardiovascular disorders that lead to heart failure, such as hypertension, myocardial infarction, heart valve disease, and variety of other causes. Given that vinpocetine has proven to be a safe drug in long-term therapy [18], vinpocetine may represent an attractive therapeutic candidate for treating cardiovascular diseases associated with cardiac remodeling.
Activation of the renin-angiotensin system (RAS) increases blood pressure, which contributes to cardiac hypertrophy and remodeling. Numerous pieces of experimental evidence indicate that Ang II promotes cardiac hypertrophy and fibrosis largely independent of blood pressure elevation [19–22]. Ang II is capable of exerting direct effects in the heart, which involves a number of different cell types, including cardiac myocytes, fibroblasts, and immune cells [23]. Indeed, we found that vinpocetine can directly block cardiac myocyte hypertrophy and fibroblast activation in isolated cells. In addition, we also observed similar results in a different mouse model induced by chronic activation of β-AR through ISO infusion, a model without changing blood pressure (data no shown). These results together support a direct protective effect of vinpocetine in the heart.
Vinpocetine has multiple cellular targets [4, 8], PDE1 was the first molecular target identified for vinpocetine [4]. We have previously shown that both PDE1A and PDE1C are detected in the heart, and the expression of them was significantly upregulated in mouse hearts with pathological remodeling induced by thoracic aorta constriction and/or myocardial infarction, as well as human failing hearts [12, 13, 17]. Consistently, PDE1A and/or PDE1C expression was also upregulated by Ang II (Supplemental Fig. S3) or ISO infusion [12, 13]. Our previous in vitro studies demonstrated that PDE1A plays a critical role in cultured cardiac myocyte hypertrophy and fibroblast activation [12, 13]. For example, PDE1A knockdown with siRNA prevented phenylephrine (PE)-induced myocyte hypertrophy and hypertrophic marker expression in neonatal and adult rat ventricular myocytes [12]. PDE1A knockdown also attenuated TGFβ-stimulated rat cardiac fibroblast activation and ECM protein synthesis, with a Smad2/3-independent and yet unknown mechanism [13]. Interestingly, the effects of vinpocetine on inhibiting cardiac fibroblast activation and matrix protein expression were also independent on Smad2/3 phosphorylation/activation (data not shown), which is consistent with the previous result from PDE1-selective inhibitor IC86340 [13]. Recently, we also reported the role of PDE1C deficiency in antagonizing pathological remodeling induced by thoracic aortic constriction (TAC) [17]. In the current study, we further showed that, when PDE1 activity was inhibited by the selective inhibitor IC86340, vinpocetine exhibited no additional effects on cardiomyocytes hypertrophy (Fig. 4d) and fibroblast activation (Fig. 5e). All these together suggest that PDE1 inhibition may represent one of the major mechanisms for vinpocetine in antagonizing pathological cardiac remodeling. Therefore, this study provides direct evidence demonstrating the protective effect of the therapeutic safe drug vinpocetine on animal models of pathological cardiac remodeling, probably via targeting PDE1 isozymes.
Inflammation also plays a critical role in cardiac hypertrophy and fibrotic response during cardiac remodeling [23]. Activation of IKK/NF-κB signaling has been shown to be involved in cardiac myocyte hypertrophy and ventricular remodeling [24–26]. We have previously shown that vinpocetine is an IKK inhibitor and plays an important role in NF-κB activation in a variety of cell types [8]. Thus, the anti-inflammatory effect of vinpocetine may also contribute to the anti-remodeling effect.
References
Leri A, Liu Y, Li B, Fiordaliso F, Malhotra A, Latini R, et al. Up-regulation of AT(1) and AT(2) receptors in postinfarcted hypertrophied myocytes and stretch-mediated apoptotic cell death. Am J Pathol. 2000;156(5):1663–72.
Klein L, O’Connor CM, Gattis WA, Zampino M, de Luca L, Vitarelli A, et al. Pharmacologic therapy for patients with chronic heart failure and reduced systolic function: review of trials and practical considerations. Am J Cardiol. 2003;91(9A):18F–40F.
Berk BC, Fujiwara K, Lehoux S. ECM remodeling in hypertensive heart disease. J Clin Invest. 2007;117(3):568–75.
Bonoczk P, Gulyas B, Adam-Vizi V, Nemes A, Karpati E, Kiss B, et al. Role of sodium channel inhibition in neuroprotection: effect of vinpocetine. Brain Res Bull. 2000;53(3):245–54.
Szobor A, Klein M. Ethyl apovincaminate therapy in neurovascular diseases. Arzneimittelforschung. 1976;26(10a):1984–9.
Patyar S, Prakash A, Modi M, Medhi B. Role of vinpocetine in cerebrovascular diseases. Pharmacol Rep. 2011;63(3):618–28.
Zhang L, Yang L. Anti-inflammatory effects of vinpocetine in atherosclerosis and ischemic stroke: a review of the literature. Molecules. 2015;20(1):335–47.
Jeon K-I, Xu X, Aizawa T, Lim JH, Jono H, Kwon D-S, et al. Vinpocetine inhibits NF-kappaB-dependent inflammation via an IKK-dependent but PDE-independent mechanism. Proc Natl Acad Sci U S A. 2010;107(21):9795–800.
Kim D, Rybalkin SD, Pi X, Wang Y, Zhang C, Munzel T, et al. Upregulation of phosphodiesterase 1A1 expression is associated with the development of nitrate tolerance. Circulation. 2001;104(19):2338–43.
Cai Y, Knight WE, Guo S, Li JD, Knight PA, Yan C. Vinpocetine suppresses pathological vascular remodeling by inhibiting vascular smooth muscle cell proliferation and migration. J Pharmacol Exp Ther. 2012;343(2):479–88.
Cai Y, Li JD, Yan C. Vinpocetine attenuates lipid accumulation and atherosclerosis formation. Biochem Biophys Res Commun. 2013;434(3):439–43.
Miller CL, Oikawa M, Cai Y, Wojtovich AP, Nagel DJ, Xu X, et al. Role of Ca2+/calmodulin-stimulated cyclic nucleotide phosphodiesterase 1 in mediating cardiomyocyte hypertrophy. Circ Res. 2009;105(10):956–64.
Miller CL, Cai Y, Oikawa M, Thomas T, Dostmann WR, Zaccolo M, et al. Cyclic nucleotide phosphodiesterase 1A: a key regulator of cardiac fibroblast activation and extracellular matrix remodeling in the heart. Basic Res Cardiol. 2011;106(6):1023–39.
Wu MP, Zhang YS, Zhou QM, Xiong J, Dong YR, Yan C. Higenamine protects ischemia/reperfusion induced cardiac injury and myocyte apoptosis through activation of beta2-AR/PI3K/AKT signaling pathway. Pharmacol Res. 2016;104:115–23.
Kabaeva Z, Zhao M, Michele DE. Blebbistatin extends culture life of adult mouse cardiac myocytes and allows efficient and stable transgene expression. Am J Physiol Heart Circ Physiol. 2008;294(4):H1667–1674.
Yan C, Zhao AZ, Bentley JK, Beavo JA. The calmodulin-dependent phosphodiesterase gene PDE1C encodes several functionally different splice variants in a tissue-specific manner. J Biol Chem. 1996;271(41):25699–706.
Knight WE, Chen S, Zhnag Y, Oikawa M, Wu M, Zhou Q, Miller CL, Cai Y, Mickelsen DM, Moravec C, Small EM, Abe J, Yan C. PDE1C deficiency antagonizes pathological cardiac remodeling and dysfunction. Proc Natl Acad Sci U S A. 2016;pii: 201607728:E7116-7125.
Balestreri R, Fontana L, Astengo F. A double-blind placebo controlled evaluation of the safety and efficacy of vinpocetine in the treatment of patients with chronic vascular senile cerebral dysfunction. J Am Geriatr Soc. 1987;35(5):425–30.
Crawford DC, Chobanian AV, Brecher P. Angiotensin II induces fibronectin expression associated with cardiac fibrosis in the rat. Circ Res. 1994;74(4):727–39.
Kim S, Ohta K, Hamaguchi A, Yukimura T, Miura K, Iwao H. Angiotensin II induces cardiac phenotypic modulation and remodeling in vivo in rats. Hypertension. 1995;25(6):1252–9.
Mazzolai L, Nussberger J, Aubert JF, Brunner DB, Gabbiani G, Brunner HR, et al. Blood pressure-independent cardiac hypertrophy induced by locally activated renin-angiotensin system. Hypertension. 1998;31(6):1324–30.
Paradis P, Dali-Youcef N, Paradis FW, Thibault G, Nemer M. Overexpression of angiotensin II type I receptor in cardiomyocytes induces cardiac hypertrophy and remodeling. Proc Natl Acad Sci U S A. 2000;97(2):931–6.
Frieler RA, Mortensen RM. Immune cell and other noncardiomyocyte regulation of cardiac hypertrophy and remodeling. Circulation. 2015;131(11):1019–30.
Purcell NH, Tang G, Yu C, Mercurio F, DiDonato JA, Lin A. Activation of NF-kappa B is required for hypertrophic growth of primary rat neonatal ventricular cardiomyocytes. Proc Natl Acad Sci U S A. 2001;98(12):6668–73.
Li Y, Ha T, Gao X, Kelley J, Williams DL, Browder IW, et al. NF-kappaB activation is required for the development of cardiac hypertrophy in vivo. Am J Physiol Heart Circ Physiol. 2004;287(4):H1712–1720.
Onai Y, Suzuki J, Maejima Y, Haraguchi G, Muto S, Itai A, et al. Inhibition of NF-{kappa}B improves left ventricular remodeling and cardiac dysfunction after myocardial infarction. Am J Physiol Heart Circ Physiol. 2007;292(1):H530–538.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
This study was supported by grants from National Natural Science Foundation of China No.81473379 and No.81673915 (to M. Wu), No. 81570248 (to C. Y.), Shanghai Municipal Bureau of Health Science and Technology Project No. 2011L032B (to M. Wu), the National Institutes of Health National Heart, Lung, and Blood Institute [Grants HL111291; HL088400 (to C.Y.) and DC005843, DC004562, and DC013833 (to J.D. Li)].
Conflict of Interest
The authors declare that they have no conflict of interests.
Ethical Approval
All procedures performed in studies involving animals were in accordance with ethical standards of the institution or practice at which the studies were conducted.
Informed Consent
No human subjects are involved.
Additional information
Mei-ping Wu and Yi-shuai Zhang contributed equally to this work.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 605 kb)
Rights and permissions
About this article
Cite this article
Wu, Mp., Zhang, Ys., Xu, X. et al. Vinpocetine Attenuates Pathological Cardiac Remodeling by Inhibiting Cardiac Hypertrophy and Fibrosis. Cardiovasc Drugs Ther 31, 157–166 (2017). https://doi.org/10.1007/s10557-017-6719-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-017-6719-0