Abstract
Cellular interactions with extracellular matrix play essential roles in tumor initiation, progression and metastasis. Focal adhesion kinase (FAK) is a cytoplasmic tyrosine kinase identified as a key mediator of signaling by integrins, a major family of cell surface receptors for extracellular matrix, as well as other receptors in both normal and cancer cells. FAK is activated by integrins through disruption of an auto-inhibitory intra-molecular interaction between its kinase domain and the amino terminal FERM domain. The activated FAK forms a binary complex with Src family kinases which can phosphorylate other substrates and trigger multiple intracellular signaling pathways to regulate various cellular functions. Subcellular localization of FAK in focal adhesions is essential for FAK signaling, which is another distinguishing feature of the kinase. Integrin-FAK signaling has been shown to activate a number of signaling pathways through phosphorylation and protein-protein interactions to promote tumorigenesis. FAK also plays a prominent role in tumor progression and metastasis through its regulation of both cancer cells and their microenvironments including cancer cell migration, invasion, epithelial to mesenchymal transition, and angiogenesis. More recently, a role for FAK in tumor initiation and progression has been demonstrated directly using xenograft as well as conditional knockout mouse models. In agreement with these experimental data, overexpression and activation of FAK have been found in a variety of human cancers. A number of small molecule inhibitors for FAK have been developed and in various phases of testing for cancer treatments. Overall, the intensive research on FAK signaling in cancer have yielded a wealth of information on this pivotal kinase and these and future studies are leading to potentially novel therapies for cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Cellular interactions with extracellular matrix (ECM) play crucial roles in many aspects of cancer initiation and progression. Integrin family cell adhesion receptors are the major mediators of cell adhesion to ECM, which link ECM to actin cytoskeleton at the cellular structure called focal adhesions (or focal contacts)[1]. Besides clustered integrins themselves, multiple structural and signaling molecules have been localized to focal adhesions, which highlight the importance of focal adhesions in the regulation of cellular structure and functions. Chief among these proteins is focal adhesion kinase (FAK), a non-receptor tyrosine kinase, that is the earliest identified and one of the most prominent signaling molecules in focal adhesions (hence its namesake).
FAK was discovered by two converging lines of research both aimed at understanding of the molecular mechanisms of cancer. In the first, researchers searched for proteins that were tyrosine phosphorylated in an integrin-dependent manner based on the premise that integrin-mediated cell adhesion could provide anchorage-dependent growth of normal cells through triggering tyrosine phosphorylation of key signaling molecules and thus abnormalities in their phosphorylation might be responsible for anchorage-independent growth, one of the hallmarks of cancer cells. These studies revealed several proteins in the platelets [2, 3] and one major protein at approximately 120 kDa in fibroblasts whose phosphorylations are induced by integrins [4, 5]. In the second set of studies, key substrates of the v-Src oncoprotein were identified and cloned through generation of monoclonal antibodies against a mixture of proteins in the v-Src transformed cells in order to understand the mechanism of transformation by the oncogenic tyrosine kinase [6, 7]. It was soon found that one of these substrates was a tyrosine kinase itself and identical to the 120 kDa protein whose phosphorylation was triggered by integrin-mediated cell adhesion [7, 8]. This protein was named as FAK based on its prominent localization in the focal adhesions [7] and FAK activation and phosphorylation stimulated by integrins as well as oncogenic transformation provided a plausible mechanism for anchorage-independent growth of cancer cells [8]. Since these initial findings in the early 1990s, numerous studies have linked FAK-mediated signaling pathways to cancer and a variety of other biological and disease processes. This review will focus primarily on the roles of FAK signaling pathways in cancer development and progression, although it is clear that FAK also plays important roles in many other biological and disease processes [9, 10].
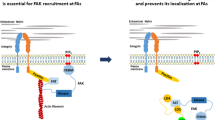
Human fak gene (known as ptk2) is localized at chromosome 8q24.3 (GeneBank or FASTA: NW_923984.1; Entez ID: 5747 and Nucleotide Access Number: AC100860) [11]. Although transcript variants encoding two slightly different protein isoforms have been found for this gene (isoform α has 1,052 aa and β has additional 22 aa to the N-terminus of α), FAK protein (also known as PTK2, FAK1, pp125FAK and FADK) shares >95% amino acid identity and structure across different species [12]. FAK is composed of an N-terminal FERM (protein 4.1, ezrin, radixin and moesin homology) domain, a central kinase domain, and a C-terminal domain that includes the focal adhesion targeting (FAT) sequence responsible for FAK's localization to focal adhesions (Fig. 1). Both the N-terminal and C-terminal domains have been shown to mediate FAK interaction with a variety of other proteins critical for activation of FAK by integrins or other cell surface receptors as well as FAK regulation of different cellular functions (Fig. 2).
FAK structure, phosphorylation sites, and its associated proteins. FAK is composed of a central kinase domain flanked by an N-terminal FERM domain and a C-terminal region containing two proline-rich (PR1 and PR2) motifs and a FAT domain. Several known phosphorylation sites as well as proteins that interact with FAK are also highlighted in the diagram
FAK signaling pathways in normal cellular functions and cancer. FAK is activated by ECM binding to integrins (or other stimuli) by disruption of an auto-inhibitory interaction of the kinase and amino terminal FERM domains. The activated FAK then binds to Src family kinases and other intracellular signaling molecules to trigger multiple downstream pathways to regulate cell survival, apoptosis, cell cycle progression and proliferation, and focal adhesion dynamics and cell migration. Changes in these functions are involved in epithelial to mesenchymal transition, tumor angiogenesis, and ECM remodeling during both normal embryonic development and tumor initiation, progression and metastasis
Integrin-mediated cell adhesion is the major upstream activator of FAK, and increased activation and tyrosine phosphorylation of FAK have been observed in essentially all adherent cells analyzed so far. The amino-terminal FERM domain of FAK plays a critical role in the regulation of FAK activation through an intra-molecular interaction with the kinase domain of FAK based on the recently solved crystal structure of FAK as well as earlier mutational studies [13–16]. When FAK is in the inactive state, direct contact between the FERM domain and kinase domain of FAK blocks access to its catalytic cleft and sequesters its activation loop as well as the key autophosphorylation site Y397. In contrast, in the active state, FERM domain is displaced by an activating protein (e.g. integrin β cytoplasmic domain or other activators), allowing rapid autophosphorylation of Y397 and exposure of the docking site for Src family kinases, which phosphorylates additional sites on FAK leading to its full activation. Such an conformational change of FAK associated with its activation is demonstrated directly in live cells by fluorescence resonance energy transfer (FRET) using probes designed based on the structural information [17]. Besides integrins, the amino terminal domain of FAK also interacts with the activated PDGF, EGF and HGF receptors, and these interactions may be important for cross-talk between integrins and growth factor receptors, and could potentially be involved in regulation of FAK’s activity [18].
Once it is activated, FAK undergoes autophosphorylated at Y397, which creates a binding site for Src via the SH2 domain [19–21]. FAK binding to the SH2 domain of Src displaces Src Y527 binding to it, relieving the auto-inhibitory interaction and leading to activation of Src. Conversely, activated Src phosphorylates additional sites on FAK, including residues Y576 and Y577 in FAK’s kinase activation loop, leading to further increased activity of FAK. The mutually activated FAK/Src complex then initiates a cascade of phosphorylation events and new protein-protein interactions to trigger several signaling pathways. These FAK signaling pathways have been shown to regulate a variety of cellular functions both in normal and cancer cells.
2 FAK regulation of cellular functions in tumorigenesis
A potential role of FAK in tumorigenesis was first suggested by initial studies showing FAK as one converging point by integrin signaling in normal cells and oncogenic transformation by v-Src [8, 22]. FAK activation in transformed cells in the absence of adhesion signal was proposed to be responsible for anchorage-independent growth of these cells, a hallmark of cancerous cells. These studies suggested that FAK may contribute to tumorigenesis through its promotion of cell survival and/or proliferation. It was subsequently found that activation of FAK in cell adhesion protects cells from anoikis, a form of apoptosis induced by cell detachment from the ECM [23]. Expression of an activated, membrane-targeted FAK construct called CD2FAK in epithelial cells prevents anoikis [23, 24]. Conversely, inhibition of FAK activity by a number of different approaches induced apoptosis in attached cells [25–27]. Furthermore, both the catalytic activity and the autophosphorylation site Y397 of FAK are required for protecting cells from anoikis, suggesting a role for the FAK/Src complex and its downstream pathways in cell survival [23, 28, 29].
FAK also binds p85 subunit of PI3K through autophosphorylated Y397 [30]. Phospholipid production stimulated by FAK association and activation of PI3K can activate Akt kinase, which inhibits apoptosis by regulating various cell death machinery proteins [31, 32]. Indeed, FAK activation of PI3K/Akt signaling pathway has been found to protect cells from apoptosis induced by collagen gel contraction or UV irradiation [23, 28, 29]. In human leukemic HL-60 cells, FAK activation of PI3K/Akt pathway increased expression of IAP proteins through NFκB and protected the cells from oxidative-stress induced apoptosis [29]. Besides PI3K/Akt signaling pathways, FAK has also been suggested to regulate cell survival in a more direct manner through its binding and sequestering the death domain protein RIP [33]. Several recent studies also suggested that FAK can promote cell survival via its interactions with p53 through the FERM domain in a kinase-independent manner [34]. This interaction suppresses transcriptional activation of a number of p53 target genes including p21, Mdm2 and Bax that are important mediators of p53-induced apoptosis, which would account for inhibition of apoptosis by FAK in a p53 dependent manner. In addition, FAK/p53 complex is also detected in the nucleus where FAK promotes ubiquitin-proteasome mediated degradation of p53 by Mdm2 through interactions with both proteins [34]. Therefore, FAK may promote cell survival by multiple pathways through both kinase-dependent and kinase-independent mechanisms.
In addition to a positive role for cell survival, FAK signaling pathways can promote tumorigenesis through stimulation of cell cycle progression. Several early studies provided first support for such a role for FAK in cell proliferation. Microinjection of a dominant negative fragment of FAK into either fibroblasts or endothelial cells (ECs) decreased their DNA synthesis [35]. Disruption of FN matrix assembly by treating cells with FN fragments suppressed FAK tyrosine phosphorylation and resulted in the delay of the G1 to S transition [36]. Using a tetracycline-regulated expression system, we showed that expression of wild type FAK accelerated G1 to S transition whereas expression of a dominant negative FAK mutant inhibited cell cycle progression at G1 phase [37]. These were further supported by many additional studies in different systems including tumor cells. One notable exception is that MEFs isolated from FAK total knockout mice did not exhibit defects in cell proliferation although a significant decrease in migration was observed [38]. It should be noted, however, that the MEFs were isolated in a p53-null background that could bypass the requirement for FAK signaling in cell proliferation, while FAK-null MEFs in wild type p53 background can not be obtained due to deficiency in cell survival (see discussion above). In addition, up-regulation of the FAK related kinase Pyk2 has been found in some knockout studies [39], although not in others [40], which could provide a compensation for FAK in promoting cell proliferation or other cellular activities.
Several FAK signaling pathways have been shown to play an important role in cell proliferation. The formation of FAK/Src complex allows Src to phosphorylate Y925 on FAK to mediate its interaction with Grb2 leading to the activation of RAS-Erk signaling pathway [41]. FAK interaction with another adaptor molecule SHC also contributes to the activation of this pathway [42]. Moreover, FAK has been shown to increase expression of cyclin D1 and decrease expression of Cdk inhibitor, p21 [37]. FAK regulation of cyclin D1 is primarily at the transcriptional level and mediated by Erk signaling, which leads to consequent activation of transcription factor Ets and its binding to the EtsB site in the cyclin D1 promoter [43]. JNK signaling pathway has also been shown to mediate FAK regulation of cell cycle progression, which could presumably also affect cyclin D1 expression via the phosphorylation of transcription factor Jun [44]. Lastly, the transcription factor KLF8 has also been identified to mediate FAK regulation of cyclin D1 through its binding to the GT box A as well as its repression of a potential inhibitory regulator of cyclin D1 promoter [45]. Interestingly, transcriptional activation of cyclin D1 by FAK requires both Ets family and KLF8 factors in a temporally differential manner. Besides cyclin D1 and p21, several other cell cycle regulators including cyclin E, cdk inhibitor p27 and S-phase kinase associated protein2 (Skp2) have been shown to mediate FAK regulation of cell cycle progression in different cells including tumor cells [46–49].
3 Role of FAK signaling in promoting tumor progression and metastasis
Tumor progression and metastasis are complex disease processes that involve multiple changes in tumor cells as well as their microenvironments. Promotion of cell survival and proliferation by FAK signaling pathways contributes to a number of these processes such as tumor growth and the survival of metastatic cells in new sites. Ample evidence has also indicated that FAK signaling pathways can stimulate tumor progression and metastasis through their regulation of cell migration, invasion, EMT and angiogenesis.
A role for FAK in cell migration was first suggested by observations of increased expression or activation of FAK in the migrating keratinocytes in epidermal wound healing or ECs migrating into the wounded monolayer in vitro, respectively [50, 51]. Furthermore, FAK-/- fibroblasts derived from FAK KO mouse embryo showed a significant decrease in cell migration compared with the cells from wild-type mice. The impaired cell migration may be an explanation for the general mesodermal deficiency and embryonic lethal phenotype of the KO mice [38]. Similarly, inhibition of FAK by microinjection of the FAK C-terminal recombinant protein (i.e. FRNK) decreased migration of both fibroblasts and ECs [35]. Conversely, overexpression of FAK in a number of cell lines including the FAK-/- cells promotes their migration on FN [52–54].
A number of downstream signaling pathways have been identified to mediate FAK-stimulated cell migration. One well characterized pathway involves FAK complex formation with Src and subsequent phosphorylation of the adaptor molecule Cas by the FAK/Src complex [52–56]. Numerous studies have shown that autophosphorylation of FAK at Y397 and its association with Src at the site is essential for FAK’s ability to promote cell migration, as mutation of Y397 to F both disrupted FAK association with Src and its stimulation of cell migration in many cell types including FAK-/- cells [52, 54, 55]. Inhibition of cell spreading by blocking FAK functions with FRNK (C-terminal FAK fragment functioning as a dominant negative FAK) could be partially rescued by overexpression of Src [57]. Similarly, disruption of Cas binding to FAK by either FAK mutation or dominant negative Cas constructs abolish both Cas phosphorylation by FAK/Src complex and stimulation of cell migration by FAK [55]. Tyrosine phosphorylated Cas has been shown to associate with several SH2-containing proteins including Crk. The Cas/Crk complex formation has been shown to regulate membrane ruffling and cell migration through DOCK180 and Rac [58, 59]. Paxillin, another cytoskeletal adaptor molecule, is also phosphorylated by FAK/Src complex at Y31 and Y118 residues, which can then recruit Crk in a similar manner as Cas [60]. In addition, paxillin is associated with a complex containing an adaptor molecule PKL, a guanine nucleotide exchange factor Pix/Cool, and the Cdc42/Rac target-effector PAK, which may link Cdc42 and Rac to PAK and its downstream targets, such as LIMK and MLCK [61, 62] in the regulation of cell migration.
A second mechanism of FAK promotion of cell migration involves its interactions with PI3K and an adaptor molecule Grb7 [63, 64]. A FAK mutant that selectively disrupted its binding to PI3K and Grb7, but retained binding to Src and induction of Cas phosphorylation, failed to promote cell migration [30]. Inhibition of PI3K by wortmannin or LY294002 prevented FAK-stimulated cell migration [30]. Rac is a downstream effector of PI3K that plays a major role in the regulation of cell migration. In addition, the increased production of D3-phosphoinositides upon FAK/PI3K complex formation and activation of PI3K can increase Grb7 association with phosphatidylinositol phosphates in the plasma membrane through the PH domain of Grb7. Although FAK interaction with Grb7 is independent of FAK/PI3K association [63], the phosphatidylinositol phosphates binding to the PH domain of Grb7 can induce a conformational change in Grb7, leading to increased phosphorylation of Grb7 by FAK [65]. Grb7 association with FAK through its SH2 domain as well as its phosphorylation by FAK are both crucial for Grb7 stimulation of cell migration [64]. These data suggest a cooperation of FAK/PI3K signaling pathway with FAK/Grb7 complex in the regulation of cell migration.
FAK has also been shown to regulate cell migration by modulating the assembly and disassembly of actin cytoskeleton through its effects on the Rho subfamily of small GTPases. In FAK-null cells, RhoA shows increased activity which is correlated with increased spreading and reduced migration of these cells. Re-expression of FAK in FAK-null cells decreased the RhoA activity and restored cell migration [66–68]. Furthermore, inhibition of ROCK, a downstream effecter of RhoA, also rescued the migration defect of FAK-null cells [68]. In contrast to these results in FAK-null cells, other studies showed that FAK can stimulate RhoA activation by induction of phosphorylation and activation of p190RhoGEF [69]. It is not clear why both FAK and loss of FAK could increase RhoA activation, but it is possible that FAK may regulate RhoA via multiple mechanisms perhaps in a spatially regulated manner in cell migration. FAK has also been shown to interact with GRAF [70], which contains GAP activity for RhoA and Cdc42, as well as ASAP1 [71], which is a GAP protein for Arf subfamily small GTPases. These interactions could also contribute to FAK regulation of actin cytoskeleton in cell migration. Recently, N-WASP, a key Cdc42 downstream effector, has been identified as a direct substrate of FAK. N-WASP phosphorylation by FAK maintains its cytoplasmic localization and thereby facilitates its stimulation of actin polymerization through the Arp2/3 complex in cell migration [72].
Besides its well-characterized role in cell migration, FAK signaling has recently been shown to promote invasion of both normal and transformed cells by a number of mechanisms. Inhibition of FAK by dominant negative mutant reduced v-Src stimulated cell invasion and blocked experimental metastasis in nude mice without significant change in migration of these cells [73]. Similarly, v-Src transformation failed to promote invasion of FAK-null fibroblasts in contrast to the wild type control cells, while cell migration for both cells was stimulated to similar extent [67]. In v-Src transformed cells, FAK was found to promote the formation of the v-Src-Cas-Crk-Dock180 complex which activates Rac1 and JNK, leading to increased expression of MMP2 and MMP9 [67]. Furthermore, inhibition of FAK has also been shown to reduce secretion of MMP9 in carcinoma cells [74]. Lastly, FAK has been shown recently to interact with endophilin A2 and mediate its phosphorylation by FAK/Src complex [75]. Tyrosine phosphorylation of endophilin A2 reduces its interaction with dynamin and decreases endocytosis of MT1-MMP, leading to increased accumulation of MT1-MMP on the cell surface responsible for the increased invasion of the v-Src transformed cells.
FAK is also shown to cooperate with Src in the disruption of E-cadherin-based intercellular adheren junctions [76], a critical event for epithelial to mesenchymal transition (EMT) that is associated with the initiation of invasive phenotype of metastatic carcinomas through increased cell migration and remodeling of the extracellular matrix microenvironment [77–79]. Expression of FAK mutants resistant to Src phosphorylation significantly decreased Src-mediated disruption of E-cadherin-based cell contacts in colon cancer cells [80]. TGF-β induced EMT mediated by Src or integrin-dependent FAK activation results in E-cadherin downregulation in mouse epithelial cells or hepatocytes [81–85]. Examination of patient specimens demonstrated that increased expression of FAK was correlated with the loss of E-cadherin in nodal metastases of laryngeal tumors [86]. Importantly, the KLF8 transcription factor has been identified to strongly induce EMT by directly repressing the transcription of E-cadherin [87], suggesting a potentially important role for KLF8 in the progression of metastasis. Indeed, increased expression of KLF8 has been found in several types of invasive human cancer [46, 87–89] and is well correlated with the loss of E-cadherin and invasive potential [87].
Tumor angiogenesis is an integral part of cancer progression and metastasis. FAK signaling has been shown to promote angiogenesis in embryonic development as well as various physiological and disease processes in adult organism, including tumor angiogenesis. A role of FAK in angiogenesis was first suggested by the observation that during the mouse embryo development, FAK expression became increasingly restricted to the blood vessels [90]. Furthermore, increased EC migration into wounded monolayer was correlated with increased tyrosine phosphorylation and kinase activity of FAK [50]. Activation of VEGF receptor-2 by VEGF-A induces the association of FAK with PI3K, which is required for the stimulation of migration of porcine aortic ECs [91]. Angiopoietin-1, another angiogenesis stimulator, also increased FAK phosphorylation during angiogenesis in vitro [92]. Additional support for a role of FAK in tumor angiogenesis comes from studies of the involvement of integrins, the major upstream activators of FAK in the process. It has been shown that blockade of integrins αvβ3 with monoclonal antibodies or small molecules inhibited tumor angiogenesis in a variety of animal models [93–95]. Moreover, a recent report showed that the formation of a signaling complex containing FAK and integrin αvβ5 in a Src-dependent manner is essential for VEGF stimulated angiogenesis in a mouse model [96]. Studies using both total and conditional KO of FAK in mice have provided direct evidence for a role of FAK in angiogenesis in vivo. Total KO of FAK results in early embryonic lethality associated with severely impaired vessel development [97]. EC-specific KO of FAK has been reported by two groups [40, 98], both showing that FAK is essential for embryonic angiogenesis in vivo. Furthermore, both studies showed that reduced EC survival upon FAK inactivation contributed to the defective vascular phenotype in the developing embryos. However, while one group found that deletion of FAK in ECs reduced their migration in response to FN and VEGF in wound closure assays, which can be rescued by re-expression of FAK [40], the other group did not observe any decrease in migration of mutant ECs by tracking individual cells using time-lapse microscopy [98]. Future studies will be necessary to resolve the discrepancy, but it is likely that defective EC migration also contributes to the phenotype given the well characterized function of FAK in the regulation of cell migration in many cell types including ECs. Consistent with these KO studies, transgenic mice with overexpression of FAK in ECs increased angiogenesis [99]. Nevertheless, direct demonstration of FAK in tumor angiogenesis in mouse models is still lacking due to the embryonic lethality, albeit late, of the EC-conditional KO mice. A recent study established inducible inactivation of FAK in ECs, but it was found that the FAK related kinase Pyk2 was up-regulated and compensated for the loss of FAK in this mouse model [39].
4 Analysis of the role of FAK in cancer using animal models
Recent studies using both xenograft mouse models and conditional KO mice have provided more direct evidence for a causative role for FAK in cancer. Such in vivo data was initially obtained in a xenograft mouse model in which a constitutively active recombinant FAK (CD2-FAK) was found to transform MDCK cells and induce tumor formation in immunodeficient mice [23]. Several subsequent studies provided additional support for the requirement of FAK in both tumor formation and progression in xenograft mouse models of tumors [10, 100–103]. Overexpression of FAK in U-215MG human malignant astrocytoma cells leads to an increase in tumor volume and cell number that are attributed to increased tumor cell proliferation [104]. Stable expression of FRNK in v-Src-transformed NIH-3T3 fibroblasts results in an decrease in experimental lung metastasis and FAK appears to mediates MMP-dependent cell invasion rather than cell migration in this model [73]. Tail vein injection of B16-F10 mouse melanoma cell stably expressing FRNK results in a 50% reduction in the lung metastases [100]. Although expression of FRNK often leads to cell death, results of the these studies do not seem to be due to selection of a rare fraction of FRNK-expressing clones that are somehow resistant to FRNK-induced death regulation, because tetracycline-regulated inducible FRNK expression leads to consistent inhibition of primary tumor growth and experimental lung metastasis in a rat breast cancer model [102]. These results are further confirmed by using FAK null or knockdown cells showing that the catalytic activity of FAK is required for metastatic breast cancer progression via the regulation of uPA expression [103] and that FAK is required for ErbB-2/3-induced breast cancer tumor formation and lung metastasis [101].
FAK gene knockout in mice resulted in an embryonic lethal phenotype due to extensive defects in cell migration [38]. Although these studies validate a critical role of FAK in embryonic development and implicate its involvement in cancer metastasis due to its regulation of cell migration, the embryonic lethality prevented direct analysis of the role of FAK in cancer development and progression using the total knockout model. Interestingly, however, McLean et al showed that reduced FAK expression in the heterozygous mice suppressed 7, 12-dimethylbenz[a]anthracene/12-O-tetradecanoylphorbol-13-acetate (DMBA/TPA) induction of papilloma formation [105]. However, the rate of conversion to malignant carcinoma in this two stage carcinogenesis model was not changed, possibly due to the residual expression of FAK. Indeed, conditional specific deletion of FAK gene from the skin epidermal cells and hair follicles results in a significant reduction of papilloma formation and progression to squamous-cell carcinomas as well [106]. These results provide compelling evidence that FAK contributes to both tumor formation and malignant progression. Close examination of the FAK-null keratinocytes demonstrates a decrease in the cell cycle progression and an increase in the cell apoptosis [106], indicating that FAK-dependent cell cycle progression and survival signaling is required for the tumor formation and progression in the skin carcinogenesis model. Mammary epithelium-specific FAK knockout blocks transition from premalignant hyperplasia to carcinoma and subsequent metastasis in a mammary conditional polyoma virus middle T transgenic mouse model of spontaneous breast cancer by inhibiting the hyperplastic cell proliferation but not survival. Interestingly, the FAK deletion does not affect the initial outgrowth of normal mammary ducts. These results suggest that the FAK-regulated aberrant proliferation is required for the formation of hyperlastic lesions and metastatic tumors [107].
5 Increased expression and activation of FAK in various human cancers
Several initial studies have correlated FAK expression with diverse cancer types in humans. Using paired normal and neoplastic tissue samples and Northern and Western blot analyses of tumor homogenates, increased levels of FAK mRNA and protein were found in vast majority of invasive and metastatic tumors, whereas FAK was barely detectable in normal tissues or non-invasive benign tumors [108–110]. Increased FAK gene dosage was correlated with increased protein expression during the transition from noninvasive adenoma to invasive carcinoma in a colon cancer model [111]. One of the studies also showed that increased FAK protein levels were often associated with advanced disease of colon as well as breast cancers [108]. In metastatic prostate tumors, both FAK mRNA and protein levels were elevated relative to normal tissue [112]. These studies suggested a potentially critical role for FAK in malignant and metastatic progression of human cancers, and promoted more extensive studies of FAK expression and activation in various tumors.
Since the publication of the initial studies, a large number of reports have documented similar findings in a wide range of human cancers by Western blot and immunohistochemical (IHC) analysis (Table 1). Newly developed FAK antibodies combined with IHC analysis allowed not only direct examination of cellular FAK protein in tumor versus adjacent normal tissue but also analysis of cellular FAK phosphorylation in situ. The broad range of tumor types investigated include astrocytic [113–117], breast [110, 118–121], cervical [122], colorectal [108, 110, 119, 121, 123–127], endometrial [128], esophageal [129], gastric [121], head and neck [111, 130], hepatocellular [108, 120, 131–134], laryngeal [86, 135, 136], lung [137–141], oral [137–143], ovarian [144–146], prostate [112, 147] and thyroid [109, 148] carcinomas as well as sarcomas [108]. Consistent observations have also been obtained in cell lines derived from tumors and normal tissue origins (reviewed in [149]). Interestingly, these studies have correlated increased FAK expression and activity with not only malignant or metastatic disease but also poor prognosis [108, 119, 146, 150, 151]. These studies indicate that FAK could be used as a prognostic marker. However, the increased FAK expression in colon adenocarcinoma tissue was not correlated with tumor grade or patient survival rate [126], and there is no correlation between FAK expression in pancreatic tumor tissue with tumor grade, metastasis or patient survival rate despite a strong correlation with tumor volume [152]. Although FAK overexpression has been correlated with tumor invasiveness and metastasis for many tumor types, exceptions exist. For example, FAK expression is reduced in liver metastases of colorectal adenocarcinoma as compared to the matched primary tumors [124]. These data suggest a role for FAK also in pre-metastatic tumors. Indeed, weak expression of FAK in cervical cancer is correlated with nodal metastasis, recurrent disease, and poor disease outcome [122]. Another report has correlated FAK elevation with the malignant transformation of cervical squamous cells and breast epithelial cells [153]. In agreement with this, activation of FAK in breast carcinoma is correlated with malignant transformation but not with invasiveness [154]. Taken together, these studies suggest that FAK may play a role at different stages of tumor progression depending on tumor types.
The molecular mechanisms responsible for the increased FAK expression in cancer remain largely absent. The fak gene amplification or copy number increase has been observed in a variety of cancer types. However it is not always correlated with increased FAK expression [111, 130, 155], suggesting the presence of tumor type-dependent mechanisms. Recent analysis of human fak gene promoter has identified p53 and NFκB as potentially direct repressor and activator, respectively of the promoter [156, 157], suggesting an intriguing possibility that aberrant expression or mutation of p53 and/or NFκB could play a role in increased FAK expression in cancer. Nevertheless, all the studies support an important role for FAK in tumor formation and metastasis.
6 Targeting FAK using specific small molecule inhibitors
The compelling evidence, both clinical and experimental, for a role of FAK in cancer has prompted the screening for and validation of small-molecule inhibitors specific for FAK. Several such inhibitors have recently been described and some of them have already moved to phase I clinical trial.
PF-573,228–Initial effort identified this molecule as a prototype ATP-competitive inhibitor of FAK [158]. In test tube, this compound inhibited catalytic activity of recombinant FAK with an IC50 of 4 nmol/L whereas its IC50 for the FAK related kinase Pyk2 was greater than 1,000 nmol/L. In cultured cells, it blocked FAK autophosphorylation at Y397 with an IC50 of 30–100 nM in both normal fibroblasts and human tumor cells. Interestingly, the compound inhibited cell migration by blocking adhesion turnover but has no effect on the cell growth or survival. There is no report that further evaluates this compound in pre-clinical or clinical trials.
PF-562,271—This compound is a potent ATP-competitive dual inhibitor of both FAK and Pyk2 [159]. In test tube, it exhibits >100-fold selectivity for FAK and Pyk2 (with an IC50 of 1.5 and 14 nmol/L, respectively) versus a panel of non-target kinases. It also displays robust inhibition of FAK phosphorylation in a dose-dependent fashion both in cultured cells that correlates with inhibited migration and in mouse tumors after orally administered. Although this compound, like PF-573,228, does not affect growth or apoptosis in adherent culture of several human cancer cell lines including PC-3M (prostate), BT474 (breast), BxPc3 (pancreatic), LoVo (colon), U87MG (glioblastoma), and H460 (lung), it strongly inhibits anchorage independent growth in soft agar of these cells and their xenografted tumors in vivo without observation of obvious undesired weight loss, morbidity, or mortality. A recent report also demonstrated that when orally administered, this compound not only inhibits MDA-MB-231 tumor growth in the bone but also safely promotes the normal bone formation in a rat bone model of human cancer [160]. These results strongly urged the use of this compound for clinical trials in human cancer patients.
A preliminary report of phase I clinical trials [161] describes treatment of 32 patients with colorectal, breast, neuroendocrine, lung, gastric, small-cell, ovary and other cancers. In this study, the patients received twice daily oral doses (5 mg up to 105 mg) for 21 days. Side-effects including nausea, vomiting, fatigue, anorexia, abdominal pain, diarrhea, headache, sensory neuropathy, rash, constipation, and dizziness of Common Toxicity Criteria grades 1-2 were observed in >10% of the patients. Doses >15 mg produced steady state plasma concentrations greater than the levels required for maximum inhibition of FAK. On day 14, one patient with metastatic ovarian cancer showed a significant (46%) decline in tumor volume and symptoms. Nine patients tolerated over 3 cycles and another four patients had stable disease after 6 cycles of treatment. Dose-limiting toxicities including nausea, vomiting and diarrhea were observed at the dose of 105 mg. These studies provide evidence that the compound is well tolerated in cancer patients on extended oral administration. Continuous dosing of 6–12 months at 105 mg allowed the patients to exhibit prolonged stable disease as measured by FDG-PET imaging (http://www.imakenews.com/barnesreport/e_article000913743.cfm?x=b11,0,w). Similar phase I trials are also going on in patients with positive PET scans due to advanced non hematologic malignancies, including pancreatic, head and neck, and prostatic neoplasms, and patients with other malignancies appropriate for serial biopsy (http://clinicaltrials.gov/ct2/show/NCT00666926?term=aurora+kinase&rank=14). Further clinical trials appear to be in progress.
TAE226—This compound is another dual inhibitor specific for both FAK and insulin-like growth factor I receptor (IGF-IR). The activity of TAE226 has been assessed in human glioma [161, 162], neuroblastoma [163], breast [164], ovarian [165] and esophageal [166] cancer cell lines. In glioma cell lines [162, 167], TAE226 inhibited ECM–induced autophosphorylation of FAK and IGF-I–induced phosphorylation of IGF-IR and activity of the downstream MAPK, Akt or S6K. Unlike the above described two inhibitors, TAE226 not only inhibits the glioma cell adhesion and migration, but also inhibits their growth and induces cell cycle arrest and apoptosis. TAE226 treatment also inhibited the glioma cell invasion on Matrigel. Importantly, TAE226 treatment significantly increased the animal survival in an intracranial glioma xenograft model. In cultured human neuroblastoma cell lines [163], TAE226 treatment resulted in a dose-dependent decrease in FAK phosphorylation and cell viability, and induced cell cycle arrest and apoptosis. In cultured breast cancer cell lines [164], TAE226 treatment induced a dose-dependent cell detachment and apoptosis. Interestingly, the immortalized normal breast MCF-10A cell line was resistant to the TAE226 treatment. In cultured ovarian cancer cells [165], TAE226 inhibited FAK phosphorylation at Y397 and Y861 in a time- and dose-dependent fashion. TAE226 inhibited cell growth in vitro and tumor growth in vivo. Interestingly, combined treatment with TAE226 and docetaxel achieved a significantly synergized inhibition of the cell growth both in vitro and in vivo. The combined chemotherapy also induced tumor regression and significantly prolonged animal survival. Close examination suggested that reduced pericyte coverage, EC apoptosis and reduced microvessel density and tumor cell proliferation appeared to be the cellular responses responsible for the therapeutic effects. TAE226 treatment also inhibited esophageal cancer cell migration and proliferation and induced apoptosis in vitro and inhibited tumor growth in vivo [166]. Phase I clinical trials of TAE226 seem to be underway by Novartis (http://www.imakenews.com/barnesreport/e_article000913743.cfm?x=b11,0,w).
Taken together, these studies highlight the significance of dual inhibition of both FAK and IGF-IR signaling by TAE226 and combined use of TAE226 with existing chemotherapeutic drugs for effective treatment of cancer.
7 Conclusion and perspectives
The large body of evidence has clearly identified FAK as one of the key molecules that promotes tumor initiation, growth and metastasis by providing abnormal signals for cell survival, proliferation, migration, EMT, invasion, and angiogenesis. As increased expression and activation of FAK is the cause of the aberrant FAK signaling in a wide array of cancer, FAK has emerged as a promising molecular target for therapeutic strategies that specifically inhibit FAK activity. It also opens an opportunity to identify FAK downstream factors as diagnostic and/or prognostic biomarkers for tumors. Although it is relatively easy to achieve specific inhibition of FAK, tumor-specific targeting remains a problem to solve given that FAK is ubiquitously expressed, albeit at relatively low levels, in normal tissues as well. Tumor-specific delivery of FAK inhibitors or partially down-regulating FAK expression or activity to normal (rather than ‘zero’) levels in combination with the use of other tumor-specific drugs that are otherwise less effective individually appears to be a sound strategy to consider during clinical trials to both maximize tumor-specificity and minimize undesired toxicity to normal tissues.
A major conceptual advance in cancer research in recent years is the proposed role of cancer stem cells in the initiation and progression of various cancers [168, 169]. Although recent studies have suggested that a number of developmental signaling pathways such as Hedgehog, Wnt and Notch pathways play important roles in the regulation of various cancer stem cells, our understanding of the mechanisms of signal transduction in cancer stems cells are still incomplete at present. The importance of FAK signaling as a potential therapeutic target for cancer also lies in the possibility of its involvement in the regulation of cancer stem cells. In this regard, integrins α6 (i.e. CD49f), β1 (i.e. CD29) and β3 (i.e. CD61) have been identified as markers for breast cancer stem cells in recent studies [170–172], raising the exciting possibility for a role of FAK in mammary cancer stem cells given its important function in mediating integrin signaling. Future studies will clarify whether and how FAK signaling is involved in the regulation of tissue stem cells and corresponding cancer stem cells and whether FAK may promote tumorigenesis and/or metastatic through its effects on self-renewal as well as other functions such as migration and invasion of cancer stem cells. It will also be interesting to determine the potential cross-talks between integrin-FAK signaling and other signaling pathways involved in the regulation of cancer stem cells. These studies may suggest that the use of a combination of inhibitors for multiple signaling pathways might be more effective than blockade of a single pathway to eradicate cancer stem cells.
References
Hynes, R. (2002). Integrins: bidirectional, allosteric signaling machines. Cell, 110, 673–687.
Golden, A., Brugge, J. S., & Shattil, S. J. (1990). Role of platelet membrane glycoprotein IIb-IIIa in agonist-induced tyrosine phosphorylation of platelet proteins. Journal of Cell Biology, 111, 3117–3127.
Ferrell Jr., J. E., & Martin, G. S. (1989). Tyrosine-specific protein phosphorylation is regulated by glycoprotein IIb-IIIa in platelets. Proceedings of the National Academy of Sciences of the United States of America, 86, 2234–2238.
Kornberg, L. J., Earp, H. S., Turner, C. E., Prockop, C., & Juliano, R. L. (1991). Signal transduction by integrins: increased protein tyrosine phosphorylation caused by clustering of beta 1 integrins. Proceedings of the National Academy of Sciences of the United States of America, 88, 8392–8396.
Guan, J. L., Trevithick, J. E., & Hynes, R. O. (1991). Fibronectin/integrin interaction induces tyrosine phosphorylation of a 120-kDa protein. Cell Regulation, 2, 951–964.
Kanner, S. B., Reynolds, A. B., Vines, R. R., & Parsons, J. T. (1990). Monoclonal antibodies to individual tyrosine-phosphorylated protein substrates of oncogene-encoded tyrosine kinases. Proceedings of the National Academy of Sciences of the United States of America, 87, 3328–3332.
Schaller, M. D., Borgman, C. A., Cobb, B. S., Vines, R. R., Reynolds, A. B., & Parsons, J. T. (1992). pp125FAK a structurally distinctive protein-tyrosine kinase associated with focal adhesions. Proceedings of the National Academy of Sciences of the United States of America, 89, 5192–5196.
Guan, J. L., & Shalloway, D. (1992). Regulation of focal adhesion-associated protein tyrosine kinase by both cellular adhesion and oncogenic transformation. Nature, 358, 690–692.
Parsons, J. T. (2003). Focal adhesion kinase: the first ten years. Journal of Cell Science, 116, 1409–1416.
Mitra, S. K., & Schlaepfer, D. D. (2006). Integrin-regulated FAK-Src signaling in normal and cancer cells. Current Opinion in Cell Biology, 18, 516–523.
Fiedorek Jr., F. T., & Kay, E. S. (1995). Mapping of the focal adhesion kinase (Fadk) gene to mouse chromosome 15 and human chromosome 8. Mammalian Genome, 6, 123–126.
Whitney, G. S., Chan, P. Y., Blake, J., Cosand, W. L., Neubauer, M. G., Aruffo, A., et al. (1993). Human T and B lymphocytes express a structurally conserved focal adhesion kinase, pp125FAK. DNA and Cell Biology, 12, 823–830.
Lietha, D., Cai, X., Ceccarelli, D. F., Li, Y., Schaller, M. D., & Eck, M. J. (2007). Structural basis for the autoinhibition of focal adhesion kinase. Cell, 129, 1177–1187.
Dunty, J. M., Gabarra-Niecko, V., King, M. L., Ceccarelli, D. F., Eck, M. J., & Schaller, M. D. (2004). FERM domain interaction promotes FAK signaling. Molecular and Cellular Biology, 24, 5353–5368.
Cooper, L. A., Shen, T. L., & Guan, J. L. (2003). Regulation of focal adhesion kinase by its amino-terminal domain through an autoinhibitory interaction. Molecular and Cellular Biology, 23, 8030–8041.
Cohen, L. A., & Guan, J. L. (2005). Residues within the first subdomain of the FERM-like domain in focal adhesion kinase are important in its regulation. Journal of Biological Chemistry, 280, 8197–8207.
Cai, X., Lietha, D., Ceccarelli, D. F., Karginov, A. V., Rajfur, Z., Jacobson, K., et al. (2008). Spatial and temporal regulation of focal adhesion kinase activity in living cells. Molecular and Cellular Biology, 28, 201–214.
Sieg, D. J., Hauck, C. R., Ilic, D., Klingbeil, C. K., Schaefer, E., Damsky, C. H., et al. (2000). FAK integrates growth-factor and integrin signals to promote cell migration. Nature Cell Biology, 2, 249–256.
Cary, L. A., & Guan, J. L. (1999). Focal adhesion kinase in integrin-mediated signaling. Frontiers in Bioscience, 4, D102–113.
Schaller, M. D., Hildebrand, J. D., Shannon, J. D., Fox, J. W., Vines, R. R., & Parsons, J. T. (1994). Autophosphorylation of the focal adhesion kinase, pp125FAK, directs SH2-dependent binding of pp60src. Molecular and Cellular Biology, 14, 1680–1688.
Xing, Z., Chen, H. C., Nowlen, J. K., Taylor, S. J., Shalloway, D., & Guan, J. L. (1994). Direct interaction of v-Src with the focal adhesion kinase mediated by the Src SH2 domain. Molecular Biology of the Cell, 5, 413–421.
Zachary, I., Sinnett-Smith, J., & Rozengurt, E. (1992). Bombesin, vasopressin, and endothelin stimulation of tyrosine phosphorylation in Swiss 3T3 cells. Identification of a novel tyrosine kinase as a major substrate. Journal of Biological Chemistry, 267, 19031–19034.
Frisch, S. M., Vuori, K., Ruoslahti, E., & Chan-Hui, P. Y. (1996). Control of adhesion-dependent cell survival by focal adhesion kinase. Journal of Cell Biology, 134, 793–799.
Chan, P. Y., Kanner, S. B., Whitney, G., & Aruffo, A. (1994). A transmembrane-anchored chimeric focal adhesion kinase is constitutively activated and phosphorylated at tyrosine residues identical to pp125FAK. Journal of Biological Chemistry, 269, 20567–20574.
Hungerford, J. E., Compton, M. T., Matter, M. L., Hoffstrom, B. G., & Otey, C. A. (1996). Inhibition of pp125FAK in cultured fibroblasts results in apoptosis. Journal of Cell Biology, 135, 1383–1390.
Xu, L. H., Owens, L. V., Sturge, G. C., Yang, X., Liu, E. T., Craven, R. J., et al. (1996). Attenuation of the expression of the focal adhesion kinase induces apoptosis in tumor cells. Cell Growth & Differentiation, 7, 413–418.
Ilic, D., Almeida, E. A., Schlaepfer, D. D., Dazin, P., Aizawa, S., & Damsky, C. H. (1998). Extracellular matrix survival signals transduced by focal adhesion kinase suppress p53-mediated apoptosis. Journal of Cell Biology, 143, 547–560.
Chan, P. C., Lai, J. F., Cheng, C. H., Tang, M. J., Chiu, C. C., & Chen, H. C. (1999). Suppression of ultraviolet irradiation-induced apoptosis by overexpression of focal adhesion kinase in Madin-Darby canine kidney cells. Journal of Biological Chemistry, 274, 26901–26906.
Sonoda, Y., Matsumoto, Y., Funakoshi, M., Yamamoto, D., Hanks, S. K., & Kasahara, T. (2000). Anti-apoptotic role of focal adhesion kinase (FAK). Induction of inhibitor-of-apoptosis proteins and apoptosis suppression by the overexpression of FAK in a human leukemic cell line, HL-60. Journal of Biological Chemistry, 275, 16309–16315.
Reiske, H. R., Kao, S. C., Cary, L. A., Guan, J. L., Lai, J. F., & Chen, H. C. (1999). Requirement of phosphatidylinositol 3-kinase in focal adhesion kinase-promoted cell migration. Journal of Biological Chemistry, 274, 12361–1236.
Hennessy, B. T., Smith, D. L., Ram, P. T., Lu, Y., & Mills, G. B. (2005). Exploiting the PI3K/AKT pathway for cancer drug discovery. Nature Reviews. Drug discovery, 4, 988–1004.
Luo, J., Manning, B. D., & Cantley, L. C. (2003). Targeting the PI3K-Akt pathway in human cancer: rationale and promise. Cancer Cell, 4, 257–262.
Kurenova, E., Xu, L. H., Yang, X., Baldwin Jr., A. S., Craven, R. J., Hanks, S. K., et al. (2004). Focal adhesion kinase suppresses apoptosis by binding to the death domain of receptor-interacting protein. Molecular and Cellular Biology, 24, 4361–4371.
Lim, S. T., Chen, X. L., Lim, Y., Hanson, D. A., Vo, T. T., Howerton, K., et al. (2008). Nuclear FAK promotes cell proliferation and survival through FERM-enhanced p53 degradation. Molecular Cell, 29, 9–22.
Gilmore, A. P., & Romer, L. H. (1996). Inhibition of focal adhesion kinase (FAK) signaling in focal adhesions decreases cell motility and proliferation. Molecular Biology of the Cell, 7, 1209–1224.
Sechler, J. L., & Schwarzbauer, J. E. (1998). Control of cell cycle progression by fibronectin matrix architecture. Journal of Biological Chemistry, 273, 25533–25536.
Zhao, J. H., Reiske, H., & Guan, J. L. (1998). Regulation of the cell cycle by focal adhesion kinase. Journal of Cell Biology, 143, 1997–2008.
Ilic, D., Furuta, Y., Kanazawa, S., Takeda, N., Sobue, K., Nakatsuji, N., et al. (1995). Reduced cell motility and enhanced focal adhesion contact formation in cells from FAK-deficient mice. Nature, 377, 539–544.
Weis, S. M., Lim, S. T., Lutu-Fuga, K. M., Barnes, L. A., Chen, X. L., Gothert, J. R., et al. (2008). Compensatory role for Pyk2 during angiogenesis in adult mice lacking endothelial cell FAK. Journal of Cell Biology, 181, 43–50.
Shen, T. L., Park, A. Y., Alcaraz, A., Peng, X., Jang, I., Koni, P., et al. (2005). Conditional knockout of focal adhesion kinase in endothelial cells reveals its role in angiogenesis and vascular development in late embryogenesis. Journal of Cell Biology, 169, 941–952.
Schlaepfer, D. D., Hanks, S. K., Hunter, T., & van der Geer, P. (1994). Integrin-mediated signal transduction linked to Ras pathway by GRB2 binding to focal adhesion kinase. Nature, 372, 786–791.
Schlaepfer, D. D., Jones, K. C., & Hunter, T. (1998). Multiple Grb2-mediated integrin-stimulated signaling pathways to ERK2/mitogen-activated protein kinase: summation of both c-Src—and focal adhesion kinase-initiated tyrosine phosphorylation events. Molecular and Cellular Biology, 18, 2571–2585.
Zhao, J., Pestell, R., & Guan, J. L. (2001). Transcriptional activation of cyclin D1 promoter by FAK contributes to cell cycle progression. Molecular Biology of the Cell, 12, 4066–4077.
Oktay, M., Wary, K. K., Dans, M., Birge, R. B., & Giancotti, F. G. (1999). Integrin-mediated activation of focal adhesion kinase is required for signaling to Jun NH2-terminal kinase and progression through the G1 phase of the cell cycle. Journal of Cell Biology, 145, 1461–1469.
Zhao, J., Bian, Z. C., Yee, K., Chen, B. P., Chien, S., & Guan, J. L. (2003). Identification of transcription factor KLF8 as a downstream target of focal adhesion kinase in its regulation of cyclin D1 and cell cycle progression. Molecular Cell, 11, 1503–1515.
Ding, Q., Grammer, J. R., Nelson, M. A., Guan, J. L., Stewart Jr., J. E., & Gladson, C. L. (2005). p27Kip1 and cyclin D1 are necessary for focal adhesion kinase regulation of cell cycle progression in glioblastoma cells propagated in vitro and in vivo in the scid mouse brain. Journal of Biological Chemistry, 280, 6802–6815.
Bond, M., Sala-Newby, G. B., & Newby, A. C. (2004). Focal adhesion kinase (FAK)-dependent regulation of S-phase kinase-associated protein-2 (Skp-2) stability. A novel mechanism regulating smooth muscle cell proliferation. Journal of Biological Chemistry, 279, 37304–37310.
Carrano, A. C., Eytan, E., Hershko, A., & Pagano, M. (1999). SKP2 is required for ubiquitin-mediated degradation of the CDK inhibitor p27. Nature Cell Biology, 1, 193–199.
Bryant, P., Zheng, Q., & Pumiglia, K. (2006) Focal adhesion kinase controls cellular levels of p27/Kip1 and p21/Cip1 through Skp2-dependent and independent mechanisms. Molecular and Cellular Biology, 26, 4201–4213.
Romer, L. H., McLean, N., Turner, C. E., & Burridge, K. (1994). Tyrosine kinase activity, cytoskeletal organization, and motility in human vascular endothelial cells. Molecular Biology of the Cell, 5, 349–361.
Gates, R. E., King Jr., L. E., Hanks, S. K., & Nanney, L. B. (1994). Potential role for focal adhesion kinase in migrating and proliferating keratinocytes near epidermal wounds and in culture. Cell Growth & Differentiation, 5, 891–899.
Cary, L. A., Chang, J. F., & Guan, J. L. (1996). Stimulation of cell migration by overexpression of focal adhesion kinase and its association with Src and Fyn. Journal of Cell Science, 109, 1787–1794.
Sieg, D. J., Hauck, C. R., & Schlaepfer, D. D. (1999). Required role of focal adhesion kinase (FAK) for integrin-stimulated cell migration. Journal of Cell Science, 112, 2677–2691.
Owen, J. D., Ruest, P. J., Fry, D. W., & Hanks, S. K. (1999). Induced focal adhesion kinase (FAK) expression in FAK-null cells enhances cell spreading and migration requiring both auto—and activation loop phosphorylation sites and inhibits adhesion-dependent tyrosine phosphorylation of Pyk2. Molecular and Cellular Biology, 19, 4806–4818.
Cary, L. A., Han, D. C., Polte, T. R., Hanks, S. K., & Guan, J. L. (1998). Identification of p130Cas as a mediator of focal adhesion kinase-promoted cell migration. Journal of Cell Biology, 140, 211–221.
Klemke, R. L., Leng, J., Molander, R., Brooks, P. C., Vuori, K., & Cheresh, D. A. (1998). CAS/Crk coupling serves as a "molecular switch" for induction of cell migration. Journal of Cell Biology, 140, 961–972.
Richardson, A., & Parsons, T. (1996). A mechanism for regulation of the adhesion-associated proteintyrosine kinase pp125FAK [published erratum appears in Nature 1996 Jun 27;381(6585):810]. Nature, 380, 538–540.
Cho, S. Y., & Klemke, R. L. (2000). Extracellular-regulated kinase activation and CAS/Crk coupling regulate cell migration and suppress apoptosis during invasion of the extracellular matrix. Journal of Cell Biology, 149, 223–236.
Cheresh, D. A., Leng, J., & Klemke, R. L. (1999). Regulation of cell contraction and membrane ruffling by distinct signals in migratory cells. Journal of Cell Biology, 146, 1107–1116.
Turner, C. E. (2000). Paxillin interactions. Journal of Cell Science, 113, 4139–4140.
Turner, C. E., Brown, M. C., Perrotta, J. A., Riedy, M. C., Nikolopoulos, S. N., McDonald, A. R., et al. (1999). Paxillin LD4 motif binds PAK and PIX through a novel 95-kD ankyrin repeat, ARF-GAP protein: A role in cytoskeletal remodeling. Journal of Cell Biology, 145, 851–863.
West, K. A., Zhang, H., Brown, M. C., Nikolopoulos, S. N., Riedy, M. C., Horwitz, A. F., et al. (2001). The LD4 motif of paxillin regulates cell spreading and motility through an interaction with paxillin kinase linker (PKL). Journal of Cell Biology, 154, 161–176.
Han, D. C., & Guan, J. L. (1999). Association of focal adhesion kinase with Grb7 and its role in cell migration. Journal of Biological Chemistry, 274, 24425–24430.
Han, D. C., Shen, T. L., & Guan, J. L. (2000). Role of Grb7 targeting to focal contacts and its phosphorylation by focal adhesion kinase in regulation of cell migration. Journal of Biological Chemistry, 275, 28911–28917.
Shen, T. L., Han, D. C., & Guan, J. L. (2002). Association of Grb7 with phosphoinositides and its role in the regulation of cell migration. Journal of Biological Chemistry, 277, 29069–29077.
Ren, X. D., Kiosses, W. B., Sieg, D. J., Otey, C. A., Schlaepfer, D. D., & Schwartz, M. A. (2000). Focal adhesion kinase suppresses Rho activity to promote focal adhesion turnover. Journal of Cell Science, 113, 3673–3678.
Hsia, D. A., Mitra, S. K., Hauck, C. R., Streblow, D. N., Nelson, J. A., Ilic, D., et al. (2003). Differential regulation of cell motility and invasion by FAK. Journal of Cell Biology, 160, 753–767.
Chen, B. H., Tzen, J. T., Bresnick, A. R., & Chen, H. C. (2002). Roles of Rho-associated kinase and myosin light chain kinase in morphological and migratory defects of focal adhesion kinase-null cells. Journal of Biological Chemistry, 277, 33857–33863.
Zhai, J., Lin, H., Nie, Z., Wu, J., Canete-Soler, R., Schlaepfer, W. W., et al. (2003). Direct interaction of focal adhesion kinase with p190RhoGEF. Journal of Biological Chemistry, 278, 24865–24873.
Hildebrand, J. D., Taylor, J. M., & Parsons, J. T. (1996). An SH3 domain-containing GTPase-activating protein for Rho and Cdc42 associates with focal adhesion kinase. Molecular and Cellular Biology, 16, 3169–3178.
Liu, Y., Loijens, J. C., Martin, K. H., Karginov, A. V., & Parsons, J. T. (2002). The association of ASAP1, an ADP ribosylation factor-GTPase activating protein, with focal adhesion kinase contributes to the process of focal adhesion assembly. Molecular Biology of the Cell, 13, 2147–2156.
Wu, X., Suetsugu, S., Cooper, L. A., Takenawa, T., & Guan, J. L. (2004). Focal adhesion kinase regulation of N-WASP subcellular localization and function. Journal of Biological Chemistry, 279, 9565–9576.
Hauck, C. R., Hsia, D. A., Puente, X. S., Cheresh, D. A., & Schlaepfer, D. D. (2002). FRNK blocks v-Src-stimulated invasion and experimental metastases without effects on cell motility or growth. Embo Journal, 21, 6289–6302.
Shibata, K., Kikkawa, F., Nawa, A., Thant, A. A., Naruse, K., Mizutani, S., et al. (1998). Both focal adhesion kinase and c-Ras are required for the enhanced matrix metalloproteinase 9 secretion by fibronectin in ovarian cancer cells. Cancer Research, 58, 900–903.
Wu, X., Gan, B., Yoo, Y., & Guan, J. L. (2005). FAK-mediated src phosphorylation of endophilin A2 inhibits endocytosis of MT1-MMP and promotes ECM degradation. Developmental Cell, 9, 185–196.
Irby, R. B., & Yeatman, T. J. (2002). Increased Src activity disrupts cadherin/catenin-mediated homotypic adhesion in human colon cancer and transformed rodent cells. Cancer Research, 62, 2669–2674.
Avizienyte, E., & Frame, M. C. (2005). Src and FAK signalling controls adhesion fate and the epithelial-to-mesenchymal transition. Current Opinion in Cell Biology, 17, 542–547.
McLean, G. W., Carragher, N. O., Avizienyte, E., Evans, J., Brunton, V. G., & Frame, M. C. (2005). The role of focal-adhesion kinase in cancer—a new therapeutic opportunity. Nature Reviews. Cancer, 5, 505–515.
Thiery, J. P., & Sleeman, J. P. (2006). Complex networks orchestrate epithelial-mesenchymal transitions. Nature Reviews. Molecular and Cellular Biology, 7, 131–142.
Avizienyte, E., Wyke, A. W., Jones, R. J., McLean, G. W., Westhoff, M. A., Brunton, V. G., et al. (2002). Src-induced de-regulation of E-cadherin in colon cancer cells requires integrin signalling. Nature Cell Biology, 4, 632–638.
Bailey, K. M., & Liu, J. (2008). Caveolin-1 up-regulation during epithelial to mesenchymal transition is mediated by focal adhesion kinase. Journal of Biological Chemistry, 283, 13714–13724.
Cicchini, C., Laudadio, I., Citarella, F., Corazzari, M., Steindler, C., Conigliaro, A., et al. (2008). TGFbeta-induced EMT requires focal adhesion kinase (FAK) signaling. Experimental Cell Research, 314, 143–152.
Nakamura, K., Yano, H., Schaefer, E., & Sabe, H. (2001). Different modes and qualities of tyrosine phosphorylation of Fak and Pyk2 during epithelial-mesenchymal transdifferentiation and cell migration: analysis of specific phosphorylation events using site-directed antibodies. Oncogene, 20, 2626–2635.
Prunier, C., & Howe, P. H. (2005). Disabled-2 (Dab2) is required for transforming growth factor beta-induced epithelial to mesenchymal transition (EMT). Journal of Biological Chemistry, 280, 17540–17548.
Strizzi, L., Bianco, C., Normanno, N., Seno, M., Wechselberger, C., Wallace-Jones, B., et al. (2004). Epithelial mesenchymal transition is a characteristic of hyperplasias and tumors in mammary gland from MMTV-Cripto-1 transgenic mice. Journal of Cellular Physiology, 201, 266–276.
Rodrigo, J. P., Dominguez, F., Suarez, V., Canel, M., Secades, P., & Chiara, M. D. (2007). Focal adhesion kinase and E-cadherin as markers for nodal metastasis in laryngeal cancer. Archives of Otolaryngology-head & Neck Surgery, 133, 145–150.
Wang, X., Zheng, M., Liu, G., Xia, W., McKeown-Longo, P. J., Hung, M. C., et al. (2007). Kruppel-like factor 8 induces epithelial to mesenchymal transition and epithelial cell invasion. Cancer Research, 67, 7184–7193.
Wang, X., Urvalek, A. M., Liu, J., & Zhao, J. (2008). Activation of KLF8 transcription by focal adhesion kinase in human ovarian epithelial and cancer cells. Journal of Biological Chemistry, 283, 13934–13942.
Wang, X., & Zhao, J. (2007). KLF8 transcription factor participates in oncogenic transformation. Oncogene, 26, 456–461.
Polte, T. R., Naftilan, A. J., & Hanks, S. K. (1994). Focal adhesion kinase is abundant in developing blood vessels and elevation of its phosphotyrosine content in vascular smooth muscle cells is a rapid response to angiotensin II. Journal of Cellular Biochemistry, 55, 106–119.
Qi, J. H., & Claesson-Welsh, L. (2001). VEGF-induced activation of phosphoinositide 3-kinase is dependent on focal adhesion kinase. Experimental Cell Research, 263, 173–182.
Kim, I., Kim, H. G., Moon, S. O., Chae, S. W., So, J. N., Koh, K. N., et al. (2000). Angiopoietin-1 induces endothelial cell sprouting through the activation of focal adhesion kinase and plasmin secretion. Circulation Research, 86, 952–959.
Brooks, P. C., Montgomery, A. M., Rosenfeld, M., Reisfeld, R. A., Hu, T., Klier, G., et al. (1994). Integrin alpha v beta 3 antagonists promote tumor regression by inducing apoptosis of angiogenic blood vessels. Cell, 79, 1157–1164.
Brooks, P. C., Stromblad, S., Klemke, R., Visscher, D., Sarkar, F. H., & Cheresh, D. A. (1995). Antiintegrin alpha v beta 3 blocks human breast cancer growth and angiogenesis in human skin. Journal of Clinical Investigation, 96, 1815–1822.
Eliceiri, B. P., & Cheresh, D. A. (1999). The role of alphav integrins during angiogenesis: insights into potential mechanisms of action and clinical development. Journal of Clinical Investigation, 103, 1227–1230.
Eliceiri, B. P., Puente, X. S., Hood, J. D., Stupack, D. G., Schlaepfer, D. D., Huang, X. Z., et al. (2002). Src-mediated coupling of focal adhesion kinase to integrin alpha(v)beta5 in vascular endothelial growth factor signaling. Journal of Cell Biology, 157, 149–160.
Ilic, D., Kovacic, B., McDonagh, S., Jin, F., Baumbusch, C., Gardner, D. G., et al. (2003). Focal adhesion kinase is required for blood vessel morphogenesis. Circulation Research, 92, 300–307.
Braren, R., Hu, H., Kim, Y. H., Beggs, H. E., Reichardt, L. F., & Wang, R. (2006). Endothelial FAK is essential for vascular network stability, cell survival, and lamellipodial formation. Journal of Cell Biology, 172, 151–162.
Peng, X., Ueda, H., Zhou, H., Stokol, T., Shen, T. L., Alcaraz, A., et al. (2004). Overexpression of focal adhesion kinase in vascular endothelial cells promotes angiogenesis in transgenic mice. Cardiovascular Research, 64, 421–430.
Abdel-Ghany, M., Cheng, H. C., Elble, R. C., & Pauli, B. U. (2002). Focal adhesion kinase activated by beta(4) integrin ligation to mCLCA1 mediates early metastatic growth. Journal of Biological Chemistry, 277, 34391–34400.
Benlimame, N., He, Q., Jie, S., Xiao, D., Xu, Y. J., Loignon, M., et al. (2005). FAK signaling is critical for ErbB-2/ErbB-3 receptor cooperation for oncogenic transformation and invasion. Journal of Cell Biology, 171, 505–516.
van Nimwegen, M. J., Verkoeijen, S., van Buren, L., Burg, D., & van de Water, B. (2005). Requirement for focal adhesion kinase in the early phase of mammary adenocarcinoma lung metastasis formation. Cancer Research, 65, 4698–4706.
Mitra, S. K., Lim, S. T., Chi, A., & Schlaepfer, D. D. (2006). Intrinsic focal adhesion kinase activity controls orthotopic breast carcinoma metastasis via the regulation of urokinase plasminogen activator expression in a syngeneic tumor model. Oncogene, 25, 4429–4440.
Wang, D., Grammer, J. R., Cobbs, C. S., Stewart Jr., J. E., Liu, Z., Rhoden, R., et al. (2000). p125 focal adhesion kinase promotes malignant astrocytoma cell proliferation in vivo. Journal of Cell Science, 113, 4221–4230.
McLean, G. W., Brown, K., Arbuckle, M. I., Wyke, A. W., Pikkarainen, T., Ruoslahti, E., et al. (2001). Decreased focal adhesion kinase suppresses papilloma formation during experimental mouse skin carcinogenesis. Cancer Research, 61, 8385–8389.
McLean, G. W., Komiyama, N. H., Serrels, B., Asano, H., Reynolds, L., Conti, F., et al. (2004). Specific deletion of focal adhesion kinase suppresses tumor formation and blocks malignant progression. Genes & Development, 18, 2998–3003.
Lahlou, H., Sanguin-Gendreau, V., Zuo, D., Cardiff, R. D., McLean, G. W., Frame, M. C., et al. (2007). Mammary epithelial-specific disruption of the focal adhesion kinase blocks mammary tumor progression. Proceedings of the National Academy of Sciences of the United States of America, 104, 20302–20307.
Owens, L. V., Xu, L., Craven, R. J., Dent, G. A., Weiner, T. M., Kornberg, L., et al. (1995). Overexpression of the focal adhesion kinase (p125FAK) in invasive human tumors. Cancer Research, 55, 2752–2755.
Owens, L. V., Xu, L., Dent, G. A., Yang, X., Sturge, G. C., Craven, R. J., et al. (1996). Focal adhesion kinase as a marker of invasive potential in differentiated human thyroid cancer. Annals of Surgical Oncology, 3, 100–105.
Weiner, T. M., Liu, E. T., Craven, R. J., & Cance, W. G. (1993). Expression of focal adhesion kinase gene and invasive cancer. Lancet, 342, 1024–1025.
Agochiya, M., Brunton, V. G., Owens, D. W., Parkinson, E. K., Paraskeva, C., Keith, W. N., et al. (1999). Increased dosage and amplification of the focal adhesion kinase gene in human cancer cells. Oncogene, 18, 5646–5653.
Tremblay, L., Hauck, W., Aprikian, A. G., Begin, L. R., Chapdelaine, A., & Chevalier, S. (1996). Focal adhesion kinase (pp125FAK) expression, activation and association with paxillin and p50CSK in human metastatic prostate carcinoma. International Journal of Cancer, 68, 164–171.
Zagzag, D., Friedlander, D. R., Margolis, B., Grumet, M., Semenza, G. L., Zhong, H., et al. (2000). Molecular events implicated in brain tumor angiogenesis and invasion. Pediatric Neurosurgery, 33, 49–55.
Wang, J. F., Park, I. W., & Groopman, J. E. (2000). Stromal cell-derived factor-1alpha stimulates tyrosine phosphorylation of multiple focal adhesion proteins and induces migration of hematopoietic progenitor cells: roles of phosphoinositide-3 kinase and protein kinase C. Blood, 95, 2505–2513.
Gutenberg, A., Bruck, W., Buchfelder, M., & Ludwig, H. C. (2004). Expression of tyrosine kinases FAK and Pyk2 in 331 human astrocytomas. Acta Neuropathologica, 108, 224–230.
Hecker, T. P., Grammer, J. R., Gillespie, G. Y., Stewart Jr., J., & Gladson, C. L. (2002). Focal adhesion kinase enhances signaling through the Shc/extracellular signal-regulated kinase pathway in anaplastic astrocytoma tumor biopsy samples. Cancer Research, 62, 2699–2707.
Jones, G., Machado Jr., J., & Merlo, A. (2001). Loss of focal adhesion kinase (FAK) inhibits epidermal growth factor receptor-dependent migration and induces aggregation of nh(2)-terminal FAK in the nuclei of apoptotic glioblastoma cells. Cancer Research, 61, 4978–4981.
Garcia, S., Dales, J. P., Charafe-Jauffret, E., Carpentier-Meunier, S., Andrac-Meyer, L., Jacquemier, J., et al. (2007). Overexpression of c-Met and of the transducers PI3K, FAK and JAK in breast carcinomas correlates with shorter survival and neoangiogenesis. International Journal of Oncology, 31, 49–58.
Cance, W. G., Harris, J. E., Iacocca, M. V., Roche, E., Yang, X., Chang, J., et al. (2000). Immunohistochemical analyses of focal adhesion kinase expression in benign and malignant human breast and colon tissues: correlation with preinvasive and invasive phenotypes. Clinical Cancer Research, 6, 2417–2423.
Lark, A. L., Livasy, C. A., Dressler, L., Moore, D. T., Millikan, R. C., Geradts, J., et al. (2005). High focal adhesion kinase expression in invasive breast carcinomas is associated with an aggressive phenotype. Modern Pathology, 18, 1289–1294.
Su, J. M., Gui, L., Zhou, Y. P., & Zha, X. L. (2002). Expression of focal adhesion kinase and alpha5 and beta1 integrins in carcinomas and its clinical significance. World Journal of Gastroenterology, 8, 613–618.
Gabriel, B., zur Hausen, A., Stickeler, E., Dietz, C., Gitsch, G., Fischer, D. C., et al. (2006). Weak expression of focal adhesion kinase (pp125FAK) in patients with cervical cancer is associated with poor disease outcome. Clinical Cancer Research, 12, 2476–2483.
Han, N. M., Fleming, R. Y., Curley, S. A., & Gallick, G. E. (1997). Overexpression of focal adhesion kinase (p125FAK) in human colorectal carcinoma liver metastases: independence from c-src or c-yes activation. Annals of Surgical Oncology, 4, 264–268.
Ayaki, M., Komatsu, K., Mukai, M., Murata, K., Kameyama, M., Ishiguro, S., et al. (2001). Reduced expression of focal adhesion kinase in liver metastases compared with matched primary human colorectal adenocarcinomas. Clinical Cancer Research, 7, 3106–3112.
Lark, A. L., Livasy, C. A., Calvo, B., Caskey, L., Moore, D. T., Yang, X., et al. (2003). Overexpression of focal adhesion kinase in primary colorectal carcinomas and colorectal liver metastases: immunohistochemistry and real-time PCR analyses. Clinical Cancer Research, 9, 215–222.
Theocharis, S. E., Kouraklis, G. P., Kakisis, J. D., Kanelli, H. G., Apostolakou, F. E., Karatzas, G. M., et al. (2003). Focal adhesion kinase expression is not a prognostic predictor in colon adenocarcinoma patients. European Journal of Surgical Oncology, 29, 571–574.
Yu, H. G., Tong, S. L., Ding, Y. M., Ding, J., Fang, X. M., Zhang, X. F., et al. (2006). Enhanced expression of cholecystokinin-2 receptor promotes the progression of colon cancer through activation of focal adhesion kinase. International Journal of Cancer, 119, 2724–2732.
Livasy, C. A., Moore, D., Cance, W. G., & Lininger, R. A. (2004). Focal adhesion kinase overexpression in endometrial neoplasia. Applied Immunohistochemistry & Molecular Morphology, 12, 342–345.
Miyazaki, T., Kato, H., Nakajima, M., Sohda, M., Fukai, Y., Masuda, N., et al. (2003). FAK overexpression is correlated with tumour invasiveness and lymph node metastasis in oesophageal squamous cell carcinoma. British Journal of Cancer, 89, 140–145.
Canel, M., Secades, P., Rodrigo, J. P., Cabanillas, R., Herrero, A., Suarez, C., et al. (2006). Overexpression of focal adhesion kinase in head and neck squamous cell carcinoma is independent of fak gene copy number. Clinical Cancer Research, 12, 3272–3279.
Fujii, T., Koshikawa, K., Nomoto, S., Okochi, O., Kaneko, T., Inoue, S., et al. (2004). Focal adhesion kinase is overexpressed in hepatocellular carcinoma and can be served as an independent prognostic factor. Journal of Hepatology, 41, 104–111.
Itoh, S., Maeda, T., Shimada, M., Aishima, S., Shirabe, K., Tanaka, S., et al. (2004). Role of expression of focal adhesion kinase in progression of hepatocellular carcinoma. Clinical Cancer Research, 10, 2812–2817.
Miyasaka, Y., Enomoto, N., Nagayama, K., Izumi, N., Marumo, F., Watanabe, M., et al. (2001). Analysis of differentially expressed genes in human hepatocellular carcinoma using suppression subtractive hybridization. British Journal of Cancer, 85, 228–234.
Yuan, Z., Fan, J., Wu, Z. Q., Zhou, J., & Qiu, S. J. (2007). [Focal adhesion kinase mRNA overexpression in hepatocellular carcinoma HCC) and correlation thereof with prognosis of HCC]. Zhonghua Yi Xue Za Zhi, 87, 1256–1259.
Aronsohn, M. S., Brown, H. M., Hauptman, G., & Kornberg, L. J. (2003). Expression of focal adhesion kinase and phosphorylated focal adhesion kinase in squamous cell carcinoma of the larynx. Laryngoscope, 113, 1944–1948.
Yu, H. G., Schrader, H., Otte, J. M., Schmidt, W. E., & Schmitz, F. (2004). Rapid tyrosine phosphorylation of focal adhesion kinase, paxillin, and p130Cas by gastrin in human colon cancer cells. Biochemical Pharmacology, 67, 135–146.
Carelli, S., Zadra, G., Vaira, V., Falleni, M., Bottiglieri, L., Nosotti, M., et al. (2006). Up-regulation of focal adhesion kinase in non-small cell lung cancer. Lung Cancer, 53, 263–271.
Hsu, N. Y., Chen, C. Y., Hsu, C. P., Lin, T. Y., Chou, M. C., Chiou, S. H., et al. (2007). Prognostic significance of expression of nm23-H1 and focal adhesion kinase in non-small cell lung cancer. Oncology Reports, 18, 81–85.
Imaizumi, M., Nishimura, M., Takeuchi, S., Murase, M., & Hamaguchi, M. (1997). Role of tyrosine specific phosphorylation of cellular proteins, especially EGF receptor and p125FAK in human lung cancer cells. Lung Cancer, 17, 69–84.
Nishimura, M., Machida, K., Imaizumi, M., Abe, T., Umeda, T., Takeshima, E., et al. (1996). Tyrosine phosphorylation of 100–130 kDa proteins in lung cancer correlates with poor prognosis. British Journal of Cancer, 74, 780–787.
Wang, X. Y., Liu, T., Zhu, C. Z., Li, Y., Sun, R., Sun, C. Y., et al. (2005). Expression of KAI1, MRP-1, and FAK proteins in lung cancer detected by high-density tissue microarray. Ai Zheng, 24, 1091–1095.
He, Z. X., He, H. W., Wang, D., & Fang, M. X. (2006). Expression and clinical significance of focal adhesion kinase in oral squamous cell carcinoma. Sichuan Da Xue Xue Bao Yi Xue Ban, 37, 876–878.
Kornberg, L. J. (1998). Focal adhesion kinase expression in oral cancers. Head Neck, 20, 634–639.
Grisaru-Granovsky, S., Salah, Z., Maoz, M., Pruss, D., Beller, U., & Bar-Shavit, R. (2005). Differential expression of protease activated receptor 1 (Par1) and pY397FAK in benign and malignant human ovarian tissue samples. International Journal of Cancer, 113, 372–378.
Judson, P. L., He, X., Cance, W. G., & Van Le, L. (1999). Overexpression of focal adhesion kinase, a protein tyrosine kinase, in ovarian carcinoma. Cancer, 86, 1551–1556.
Sood, A. K., Coffin, J. E., Schneider, G. B., Fletcher, M. S., DeYoung, B. R., Gruman, L. M., et al. (2004). Biological significance of focal adhesion kinase in ovarian cancer: role in migration and invasion. American Journal of Pathology, 165, 1087–1095.
Rovin, J. D., Frierson Jr., H. F., Ledinh, W., Parsons, J. T., & Adams, R. B. (2002). Expression of focal adhesion kinase in normal and pathologic human prostate tissues. Prostate, 53, 124–132.
Kim, S. J., Park, J. W., Yoon, J. S., Mok, J. O., Kim, Y. J., Park, H. K., et al. (2004). Increased expression of focal adhesion kinase in thyroid cancer: immunohistochemical study. Journal of Korean Medical Science, 19, 710–715.
Gabarra-Niecko, V., Schaller, M. D., & Dunty, J. M. (2003). FAK regulates biological processes important for the pathogenesis of cancer. Cancer Metastasis Reviews, 22, 359–374.
Recher, C., Ysebaert, L., Beyne-Rauzy, O., Mansat-De Mas, V., Ruidavets, J. B., Cariven, P., et al. (2004). Expression of focal adhesion kinase in acute myeloid leukemia is associated with enhanced blast migration, increased cellularity, and poor prognosis. Cancer Research, 64, 3191–3197.
Schlaepfer, D. D., Mitra, S. K., & Ilic, D. (2004). Control of motile and invasive cell phenotypes by focal adhesion kinase. Biochimica et Biophysica Acta, 1692, 77–102.
Furuyama, K., Doi, R., Mori, T., Toyoda, E., Ito, D., Kami, K., et al. (2006). Clinical significance of focal adhesion kinase in resectable pancreatic cancer. World Journal of Surgery, 30, 219–226.
Oktay, M. H., Oktay, K., Hamele-Bena, D., Buyuk, A., & Koss, L. G. (2003). Focal adhesion kinase as a marker of malignant phenotype in breast and cervical carcinomas. Human Pathology, 34, 240–245.
Madan, R., Smolkin, M. B., Cocker, R., Fayyad, R., & Oktay, M. H. (2006). Focal adhesion proteins as markers of malignant transformation and prognostic indicators in breast carcinoma. Human Pathology, 37, 9–15.
Okamoto, H., Yasui, K., Zhao, C., Arii, S., & Inazawa, J. (2003). PTK2 and EIF3S3 genes may be amplification targets at 8q23-q24 and are associated with large hepatocellular carcinomas. Hepatology, 38, 1242–1249.
Golubovskaya, V., Kaur, A., & Cance, W. (2004). Cloning and characterization of the promoter region of human focal adhesion kinase gene: nuclear factor kappa B and p53 binding sites. Biochimica et Biophysica Acta, 1678, 111–125.
Golubovskaya, V. M., & Cance, W. G. (2007). Focal adhesion kinase and p53 signaling in cancer cells. International Review of Cytology, 263, 103–153.
Slack-Davis, J. K., Martin, K. H., Tilghman, R. W., Iwanicki, M., Ung, E. J., Autry, C., et al. (2007). Cellular characterization of a novel focal adhesion kinase inhibitor. Journal of Biological Chemistry, 282, 14845–14852.
Roberts, W. G., Ung, E., Whalen, P., Cooper, B., Hulford, C., Autry, C., et al. (2008). Antitumor activity and pharmacology of a selective focal adhesion kinase inhibitor, PF-562,271. Cancer Research, 68, 1935–1944.
Bagi, C. M., Roberts, G. W., & Andresen, C. J. (2008). Dual focal adhesion kinase/Pyk2 inhibitor has positive effects on bone tumors: implications for bone metastases. Cancer, 112, 2313–2321.
Siu, L. L., Burris, H. A., Mileshkin, L., Camidge, D., Rischin, D. R., Chen, E. X., et al. (2007). Phase 1 study of a focal adhesion kinase (FAK) inhibitor PF-00562271 in patients (pts) with advanced solid tumors. Journal of Clinical Oncology, 25, 3527.
Liu, T. J., LaFortune, T., Honda, T., Ohmori, O., Hatakeyama, S., Meyer, T., et al. (2007). Inhibition of both focal adhesion kinase and insulin-like growth factor-I receptor kinase suppresses glioma proliferation in vitro and in vivo. Molecular Cancer Therapeutics, 6, 1357–1367.
Beierle, E. A., Trujillo, A., Nagaram, A., Golubovskaya, V. M., Cance, W. G., & Kurenova, E. V. (2008). TAE226 inhibits human neuroblastoma cell survival. Cancer Investigation, 26, 145–151.
Golubovskaya, V. M., Virnig, C., & Cance, W. G. (2008). TAE226-induced apoptosis in breast cancer cells with overexpressed Src or EGFR. Molecular Carcinogenesis, 47, 222–234.
Halder, J., Lin, Y. G., Merritt, W. M., Spannuth, W. A., Nick, A. M., Honda, T., et al. (2007). Therapeutic efficacy of a novel focal adhesion kinase inhibitor TAE226 in ovarian carcinoma. Cancer Research, 67, 10976–10983.
Watanabe, N., Takaoka, M., Sakurama, K., Tomono, Y., Hatakeyama, S., Ohmori, O., et al. (2008). Dual tyrosine kinase inhibitor for focal adhesion kinase and insulin-like growth factor-I receptor exhibits anticancer effect in esophageal adenocarcinoma in vitro and in vivo. Clinical Cancer Research, 14, 4631–4639.
Shi, Q., Hjelmeland, A. B., Keir, S. T., Song, L., Wickman, S., Jackson, D., et al. (2007). A novel low-molecular weight inhibitor of focal adhesion kinase, TAE226, inhibits glioma growth. Molecular Carcinogenesis, 46, 488–496.
Dalerba, P., Cho, R. W., & Clarke, M. F. (2007). Cancer stem cells: models and concepts. Annual Review of Medicine, 58, 267–284.
Wicha, M. S., Liu, S., & Dontu, G. (2006). Cancer stem cells: an old idea—a paradigm shift. Cancer Research, 66, 1883–1890 discussion 1895–1886.
Shackleton, M., Vaillant, F., Simpson, K. J., Stingl, J., Smyth, G. K., Asselin-Labat, M-L., et al. (2006). Generation of a functional mammary gland from a single stem cell. Nature, 439, 84–88.
Stingl, J., Eirew, P., Ricketson, I., Shackleton, M., Vaillant, F., Choi, D., et al. (2006). Purification and unique properties of mammary epithelial stem cells. Nature, 439, 993–997.
Kouros-Mehr, H., Bechis, S. K., Slorach, E. M., Littlepage, L. E., Egeblad, M., Ewald, A. J., et al. (2008). GATA-3 links tumor differentiation and dissemination in a luminal breast cancer model. Cancer Cell, 13, 141–152.
Acknowledgments
We thank Dr. Ming Luo for critical reading of the manuscript and helpful suggestions. This work is supported by American Cancer Society grant (RSG CCG-111381) to J Zhao and NIH grant GM48050 to JL Guan.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhao, J., Guan, JL. Signal transduction by focal adhesion kinase in cancer. Cancer Metastasis Rev 28, 35–49 (2009). https://doi.org/10.1007/s10555-008-9165-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10555-008-9165-4