Abstract
To evaluate the accuracy of coronary artery calcium (CAC) scoring at various tube voltages and different monoenergetic image reconstructions on a first-generation dual-source photon-counting detector CT (PCD-CT). A commercially available anthropomorphic chest phantom with calcium inserts was scanned at different tube voltages (90 kV, Sn100kV, 120 kV, and Sn140kV) on a first-generation dual-source PCD-CT system with quantum technology using automatic exposure control with an image quality (IQ) level of 20. The same phantom was also scanned on a conventional energy-integrating detector CT (120 kV; weighted filtered back projection) for reference. Extension rings were used to emulate different patient sizes. Virtual monoenergetic images at 65 keV and 70 keV applying different levels of quantum iterative reconstruction (QIR) were reconstructed from the PCD-CT data sets. CAC scores were determined and compared to the reference. Radiation doses were noted. At an IQ level of 20, radiation doses ranged between 1.18 mGy and 4.64 mGy, depending on the tube voltage and phantom size. Imaging at 90 kV or Sn100kV was associated with a size-dependent radiation dose reduction between 23% and 48% compared to 120 kV. Tube voltage adapted image reconstructions with 65 keV and QIR 3 at 90 kV and with 70 keV and QIR 1 at Sn100kV allowed to calculate CAC scores comparable to conventional EID-CT scans with a percentage deviation of ≤ 5% for all phantom sizes. Our phantom study indicates that CAC scoring with dual-source PCD-CT is accurate at various tube voltages, offering the possibility of substantial radiation dose reduction.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Coronary calcifications carry prognostic information as the presence and amount of calcification predict the likelihood of future cardiovascular events, hereby playing a pivotal role in patient therapy and management [1,2,3,4]. Coronary artery calcium (CAC) scoring with computed tomography (CT) has been established as a valuable tool for further refinement of risk stratification in asymptomatic patients at intermediate risk for future cardiovascular events [5,6,7]. Usually, standardized and reproducible quantification of CAC is performed using electrocardiography (ECG)-gated non-contrast enhanced CT at a tube voltage of 120 kV with an average slice thickness of 2.5–3 mm [2].
Photon-counting detector computed tomography (PCD-CT) is an emerging technology capable of directly converting incident photons in an electrical signal. This is in contrast to energy-integrating detectors (EID) in which an indirect conversion technology is used [8]. These PCD-generated signals include the energy information of every individual photon. With this technology, virtual monoenergetic image (VMI) reconstructions have become the standard for image evaluation and analysis, hereby substituting conventional polychromatic EID-CT image reconstructions [9]. VMI are reconstructed independently of the tube voltage, e.g., VMI-70 keV images can be computed from data sets acquired with tube voltages other than 120 kV, such as 90 kV or 100 kV. Image reconstruction is further performed using a new iterative reconstruction algorithm entitled quantum iterative reconstruction (QIR), which is optimized to handle data from PCD-CT systems, and which can be adjusted to 4 different strength levels (QIR1-4, or QIR-off) [10,11,12].
A recent study showed accurate CAC scores using a first-generation PCD-CT when using VMI reconstructions at 70 keV and applying QIR-off, when using 65 keV and applying QIR 3; or when using 65 keV and QIR 4 [11]. This study, however, applied only a tube voltage of 120 kV for their data acquisition, so that the dose reduction potential of the PCD-CT system was not fully evaluated. This issue is especially important considering the fact that CAC scoring is intended for screening and risk stratification in asymptomatic patients [4, 13,14,15].
The purpose of our study was to evaluate the accuracy of coronary artery calcium (CAC) scoring at various tube voltages and different monoenergetic image reconstructions on a first-generation dual-source photon-counting detector CT (PCD-CT).
Materials and methods
Anthropomorphic phantom
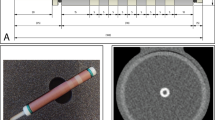
We used a commercially available anthropomorphic chest phantom with a 10 cm diameter insert for calcium scoring (QRM, Moehrendorf, Germany) [16]. The phantom contains nine cylindrical inserts varying in size (1 mm, 3 mm and 5 mm) and density (hydroxyapatite at 200 mg/cm3, 400 mg/cm3 and 800 mg/cm3). The external diameter of the phantom was adapted using different extension rings resulting in total diameters of 30 × 20 cm (small), 35 × 25 cm (medium) and 40 × 30 cm (large) to emulate different body sizes (Fig. 1). The phantom was optimized by the manufacturer for spectral imaging by ensuring a constant background material value of 35 Hounsfield Units (HU) at all energy thresholds acquired with the PCD-CT system.
Scan acquisition
ECG-gated sequential acquisitions were performed on a first-generation PCD-CT system with quantum technology (NAEOTOM Alpha; Siemens Healthcare GmbH; Forchheim, Germany) equipped with two photon-counting detectors (cadmium telluride), each with a 144 × 0.4 mm collimation. Each phantom size was scanned at 90 kV, 100 kV using a tin filter (Sn100kV), 120 kV, and 140 kV using a tin filter (Sn140kV), respectively. On this novel photon-counting system tin filtration is currently only available for scans at a tube voltage of 100 kV or 140 kV. Gantry rotation time was 0.25 s for every acquisition. For each tube voltage, the tube current was automatically adjusted to achieve an identical image quality level (IQ level) of 20 for each scan. IQ level represents a novel parameter to obtain a system and reconstruction-independent image quality definition. This derives the quality reference mAs, denoting effective mAs applied for the protocol specific reference water-equivalent diameter with a CT geometry correction [11]. The tube current adjustment was based on the standard automatic exposure control functionality of the PCD-CT system utilizing the attenuation information derived from the topogram, which was performed individually for each phantom size. The adjustment takes into account actual size of the phantom as well as the tube voltage, which modifies the expected material contrast measured in the raw data. IQ level 20 was applied for all scans, as higher IQ levels (i.e. 40, 60, 80) did not improve accuracy of CAC scoring in a previous study [11]. Table 1 provides further information about the scan protocol.
For reference, the identical chest phantom was scanned without and with the same extension rings in an ECG-gated sequential mode on a third-generation dual-source CT system with energy-integrating detector (SOMATOM Force; Siemens Healthcare GmbH; Forchheim, Germany) at 120 kV. Automatic tube current modulation was applied with a quality reference mAs of 80 mAs/rot. It has to be noted that the reference points defined for each system are comparable and were determined after consultation with the CT manufacturer. The value differences are due to a change in the reference point definition. The gantry rotation time was 0.25 s.
Image reconstruction
VMI reconstructions were computed at 65 keV and 70 keV. These two keV levels previously showed least deviation of CAC scores in comparison with EID-CT derived CAC scores [11]. Images were computed with a field-of-view of 200 mm, slice thickness of 3 mm, increment of 1.5 mm, and using a smooth-tissue reconstruction kernel (Qr36). For both keV-energies and all three phantom sizes, images were reconstructed with QIR-off and with QIR at strength levels of 1 to 4. In contrast to EID-CT, standard FBP algorithms are not applicable during the reconstruction process of monoenergetic images, since retrieval of monoenergetic information from the threshold data may cause image noise amplification. For spectral results, “QIR-off” provides minimally possible statistical optimization compared with naive weighted FBP.
The reference scan images acquired on the EID-CT were reconstructed by applying weighted filtered back-projection (wFBP), a calcium-aware reconstruction kernel (Sa36), 3 mm slice thickness, 1.5 mm increment, and a field-of-view of 200 mm. WFBP represents the standard reconstruction for scans dedicated to calcium scoring acquired on EID-CTs with a tube voltage of 120 kV and therefore was chosen for image reconstruction of EID-CT scans. In contrast, iterative image reconstruction have been identified to cause substantial reduction of CAC scores on 120 kV scans and should therefore not be applied [17]. The Sa36 kernel is a specific calcium-aware kernel developed for polychromatic images acquired with an EID-CT providing robust CAC scores even at patient-specific tube voltages [18]. According to the manufacturer, the Sa36-kernel is not only the recommended kernel for coronary calcium scoring CT scans at reduced tube voltages, but also the recommended kernel for CT calcium scoring at 120 kV to provide comparable results between various CT scanners due to an ex-factory based internal calibration.
Coronary artery calcium scoring
One reader (with two years of experience in cardiovascular radiology) quantified the CAC score using a dedicated, commercially available software (CaScore, Syngo.via VB60, Siemens). This software detects and highlights all voxels above the threshold of ≥ 130 HU. Highlighted voxels are then assigned to represent coronary calcium by the reader. The Agatston convention was applied and calcifications were defined as a plaque with an area of at least 1.03 mm2 and with an attenuation threshold of 130 HU [1, 19].
Statistical analysis
The Friedman test with Bonferroni correction was used to compare the Agatston scores between tube voltages, phantom sizes, and QIR strength levels. Statistical analysis was performed using a commercially available program (IBM SPSS Statistics, version 26, Chicago, IL, USA). A two-sided p-value < 0.05 was considered to infer statistical significance.
Results
Radiation dose
PCD-CT data acquisition with standard tube voltage of 120 kV and an IQ level of 20 resulted in radiation doses (i.e. CTDIvol) of 1.76 mGy for the small size phantom, 2.96 mGy for the medium size phantom and 4.64 mGy for the large size phantom.
Compared to the PCD-CT scan at 120 kV, modifying the tube voltage to Sn100kV resulted in a 23% lower radiation dose in the small phantom size, 26% in the medium phantom size, and 48% in the large phantom size, while keeping a constant IQ level of 20 for all scans. Scanning with a tube voltage of 90 kV resulted in a 33% lower radiation dose in the small phantom, 29% in the medium phantom, and 28% in the large phantom. In contrast to PCD-CT with 90 kV or Sn100kV, PCD-CT with Sn140kV was associated with a higher (small and medium phantom size) or similar radiation dose compared to the standard image acquisition at 120 kV. Dose levels are presented in Table 1.
CAC scoring
Figure 2 demonstrates the measured CAC scores for the three phantom sizes, varying tube voltages, QIR strength levels, and VMI reconstructions. Supplementary Table 1 provides the corresponding absolute numbers.
Bar chart. This bar chart demonstrates the measured coronary artery calcium (CAC) scores for the three phantom sizes, varying tube voltages, quantum iterative reconstruction (QIR) strength levels, and virtual monoenergetic reconstruction (VMI) reconstructions on a dual-source photon-counting detector CT (red and blue) as well as the CAC score of the reference scans on an energy-integrating detector CT (green)
CAC scores of the small phantom size ranged between 600 (90 kV, QIR 4) and 706 (Sn140kV, QIR-off) at 65 keV and between 547 (90 kV, QIR 4) and 663 (Sn140kV, QIR-off) at 70 keV.
CAC scores of the medium phantom size ranged between 621 (120 kV, QIR 4) and 700 (120 kV, QIR-off) at 65 keV and between 560 (90 kV, QIR 4) and 672 (Sn100kV, QIR-off) at 70 keV.
CAC scores of the large phantom size ranged between 584 (Sn140kV, QIR 4) and 737 (120 kV, QIR-off) at 65 keV and between 531 (Sn140kV, QIR 4) and 697 (120 kV, QIR-off) at 70 keV.
Overall, CAC scores at 65 keV were higher compared to scores at 70 keV in the small and medium phantom size. This was also found in the large phantom size except for two measurements both acquired with Sn100kV; CAC scores determined at 70 keV QIR-off and QIR 1 were higher compared to values derived from the respective 65 keV reconstructions (QIR-off: 697 vs. 657 and QIR 1: 653 vs. 650). CAC scores determined on scans acquired with a tube voltage of 90 kV were significantly lower (p < 0.001) compared to scores derived at 120 kV, when pooling all phantom sizes and VMI reconstructions. CAC scores acquired with tubes voltages of Sn100kV and Sn140kV were similar compared to CAC scores derived at 120 kV (p > 0.05). Overall, CAC scores were lower for smaller phantom sizes (p < 0.001) and at higher QIR strength levels (p < 0.001). The reference CAC score of the small, medium, and large phantom were 616, 663, and 661, respectively.
The percentage deviation of the CAC scores derived from the PCD-CT scans with various tube voltages compared to the reference EID-CT scan is shown in Fig. 3. Corresponding percentage deviation values are provided in Supplementary Table 2. For the small and large phantom, the highest deviation was found at Sn140kV. For scans at 90 kV, 65 keV and QIR 3 resulted in the lowest deviation (small phantom size, − 0.6%; medium phantom size, − 4.2%; and large phantom size, − 1.3%) compared to the reference EID-CT scan. For Sn100kV, image reconstruction with 70 keV and QIR 1 resulted in the lowest deviation (small phantom size, − 0.2%; medium phantom size, − 5.2%; and large phantom size, − 1.3%) compared to the reference EID-CT scan.
Discussion
Precise and reproducible CAC scoring is important for risk stratification in patients suspected of having coronary artery disease [1,2,3,4,5,6, 15]. In this study, we showed that CAC scoring is feasible with low tube voltage scans on a first-generation dual-source PCD-CT system. Highest accuracy was found at 90 kV and Sn100kV. Low tube voltage scanning was associated with a substantial, size-dependent radiation dose reduction between 23 and 48%. Tube voltage adapted image reconstructions with 65 keV and QIR 3 (at 90 kV) and with 70 keV and QIR 1 (at Sn100kV) allowed to calculate CAC scores comparable to conventional EID-CT scans with a percentage deviation of ≤ 5%.
Lowering the tube voltage is one of the most efficient ways to reduce radiation dose [18,19,20,21,22]. As the maximum attenuation of calcified plaques is directly dependent of the applied tube voltage, reducing the tube voltage also has a direct impact on quantification of the CAC score. Recent studies showed the feasibility of kV-independent CAC scoring on EID-CTs using artificial, dedicated, calcium-aware kernels at tube potentials other than 120 kV [18,19,20,21]. Booij et al. found consistent CAC scores with a maximal deviation of 8% when utilizing a calcium specific reconstruction kernel with varying tube voltage using an EID-CT leading to a radiation dose reduction of up to 22% [21]. In a study of Vingiani et al., the Sn100 kV EID-CT protocol allowed for a radiation dose reduction of 77% compared to the 120 kV standard protocol with consistent Agatston scores when reconstructing with a kV-independent iterative algorithm [19]. However, Apfaltrer et al. depicted a significant systematic difference in CAC scores between Sn100kV and 120 kV EID-CT scans [23]. In addition, van der Werf et al. reported relevant Agatston score deviations up to 22% in the presence of low-density CAC depending on the surrounding material when performing the scan at a reduced tube voltage of 100 kV and applying a calcium-aware reconstruction kernel. In our study, low tube voltage PCD-CT CAC scoring scan protocols were associated with a substantial radiation dose reduction between 23 and 48%, depending on the phantom size.
Specific reconstruction kernels are necessary to obtain accurate CAC scores at low kV in EID-CTs. In contrast, VMI reconstruction on PCD-CT obviates the need for this intermediate step. It was shown that VMI on PCD-CT scans acquired with 120 kV allow for CAC scoring with a deviation < 1% to the reference when using reconstructions of 65 keV/QIR 3, 65 keV/QIR 4, or 70 keV/QIR-off [11]. Symons et al. [24] investigated the performance of PCD-CT for CAC scoring when applying a reduced tube current and a fix tube voltage of 120 kV compared to an EID-CT. The authors found a similar accuracy at all dose settings and a significantly higher reproducibility of CAC scores at the lowest dose settings for PCD-CT scans [24]. Sandstedt et al. previously described in a cadaver study that accurate CAC quantification at low noise levels is feasible using PCD-CT [25]. Our study indicates that tube voltage-adapted image reconstructions with 65 keV and QIR 3 (at 90 kV) and with 70 keV and QIR 1 (at Sn100kV) allow for CAC score quantification that is comparable to conventional EID-CT scans with a deviation of below 5%. In addition, image acquisition at Sn100kV and 90 kV reduced the radiation dose by up to 48% and 33%, respectively.
The following study limitations merit consideration. First, the presented findings are phantom based and further patient studies are necessary to validate our ex-vivo results. Second, limited reproducibility and interscan-variability of the CAC score are well-known shortcomings [23, 24, 26,27,28,29]. Interscan-variability was not assessed in the present study. Third, we did not define the lowest possible IQ-level enabling accurate CAC score quantification. Finally, we did only analyze three different phantom sizes.
In conclusion, low tube voltage scanning with first-generation dual-source photon-counting detector CT for coronary artery calcium scoring is feasible, with highest accuracy at 90 kV and Sn100kV. This allows for considerable radiation dose reduction ranging between 23% and 48% depending on phantom size.
Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.
Code availability
Not applicable.
Abbreviations
- CAC:
-
Coronary artery calcium
- EID-CT:
-
Energy-integrating detector computed tomography
- QIR:
-
Quantum iterative reconstruction
- PCD-CT:
-
Photon-counting detector computed tomography
- VMI:
-
Virtual monoenergetic image
References
Agatston AS et al (1990) Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol 15(4):827–832
Hecht HS et al (2018) CAC-DRS: coronary artery calcium data and reporting system. An expert consensus document of the society of cardiovascular computed tomography (SCCT). J Cardiovasc Comput Tomogr 12(3):185–191
Knuuti J et al (2020) 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes: the Task Force for the diagnosis and management of chronic coronary syndromes of the European Society of Cardiology (ESC). Eur Heart J 41(3):407–477
Zimmermann GS et al (2019) Predictive value of coronary calcifications for future cardiac events in asymptomatic patients: underestimation of risk in asymptomatic smokers. Int J Cardiovasc Imaging 35(7):1387–1393
Vliegenthart R, Morris PB (2012) Computed tomography coronary artery calcium scoring: review of evidence base and cost-effectiveness in cardiovascular risk prediction. J Thorac Imaging 27(5):296–303
van’t Klooster CC et al (2021) Added value of cardiovascular calcifications for prediction of recurrent cardiovascular events and cardiovascular interventions in patients with established cardiovascular disease. Int J Card Imaging. https://doi.org/10.1007/s10554-021-02164-9
Eberhard M et al (2021) Incremental prognostic value of coronary artery calcium score for predicting all-cause mortality after transcatheter aortic valve replacement. Radiology 301(1):105–112
Flohr T et al (2020) Photon-counting CT review. Phys Med 79:126–136
Ferda J et al (2021) Computed tomography with a full FOV photon-counting detector in a clinical setting, the first experience. Eur J Radiol 137:109614
Flohr T et al (2020) Basic principles and clinical potential of photon-counting detector CT. Chin J Acad Radiol 3(1):19–34
Eberhard M et al (2021) Coronary calcium scoring with first generation dual-source photon-counting CT-first evidence from phantom and in-vivo scans. Diagnostics (Basel) 11(9):1708
Euler A et al (2021) High-pitch photon-counting detector computed tomography angiography of the aorta: intraindividual comparison to energy-integrating detector computed tomography at equal radiation dose. Invest Radiol. https://doi.org/10.1097/RLI.0000000000000816
Zhang D et al (2021) Coronary artery calcium scoring at lower tube voltages–Dose determination and scoring mechanism. Eur J Radiol 139:109680
Adelhoefer S et al (2020) Coronary artery calcium scoring: new insights into clinical interpretation—lessons from the CAC consortium. Radiol Cardiothoracic Imaging 2(6):e200281
Greenland P et al (2018) Coronary calcium score and cardiovascular risk. J Am Coll Cardiol 72(4):434–447
Ulzheimer S, Kalender WA (2003) Assessment of calcium scoring performance in cardiac computed tomography. Eur Radiol 13(3):484–497
Messerli M et al (2016) Impact of advanced modeled iterative reconstruction on coronary artery calcium quantification. Acad Radiol 23(12):1506–1512
van der Werf NR et al (2021) Evaluating a calcium-aware kernel for CT CAC scoring with varying surrounding materials and heart rates: a dynamic phantom study. Eur Radiol. https://doi.org/10.1007/s00330-021-08076-5
Vingiani V et al (2020) Low-kV coronary artery calcium scoring with tin filtration using a kV-independent reconstruction algorithm. J Cardiovasc Comput Tomogr 14(3):246–250
Tao S et al (2021) Technical Note: kV-independent coronary calcium scoring: a phantom evaluation of score accuracy and potential radiation dose reduction. Med Phys 48(3):1307–1314
Booij R et al (2020) Dose reduction for CT coronary calcium scoring with a calcium-aware image reconstruction technique: a phantom study. Eur Radiol 30(6):3346–3355
Marwan M et al (2013) Very low-dose coronary artery calcium scanning with high-pitch spiral acquisition mode: comparison between 120-kV and 100-kV tube voltage protocols. J Cardiovasc Comput Tomogr 7(1):32–38
Apfaltrer G et al (2018) High-pitch low-voltage CT coronary artery calcium scoring with tin filtration: accuracy and radiation dose reduction. Eur Radiol 28(7):3097–3104
Symons R et al (2019) Coronary artery calcium scoring with photon-counting CT: first in vivo human experience. Int J Cardiovasc Imaging 35(4):733–739
Sandstedt M et al (2021) Improved coronary calcification quantification using photon-counting-detector CT: an ex vivo study in cadaveric specimens. Eur Radiol. https://doi.org/10.1007/s00330-021-07780-6
Budoff MJ et al (2009) Reproducibility of coronary artery calcified plaque with cardiac 64-MDCT: the Multi-Ethnic Study of Atherosclerosis. AJR Am J Roentgenol 192(3):613–617
Rutten A, Isgum I, Prokop M (2008) Coronary calcification: effect of small variation of scan starting position on Agatston, volume, and mass scores. Radiology 246(1):90–98
Willemink MJ et al (2014) Coronary artery calcification scoring with state-of-the-art CT scanners from different vendors has substantial effect on risk classification. Radiology 273(3):695–702
Hoffmann U et al (2006) Evidence for lower variability of coronary artery calcium mineral mass measurements by multi-detector computed tomography in a community-based cohort–consequences for progression studies. Eur J Radiol 57(3):396–402
Funding
V.M. is funded by the research grant “Young Talents in Clinical Research” of the Swiss Academy of Medical Sciences (SAMS) and the G. & J. Bangerter-Rhyner Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
T.A. is employee of Siemens Healthineers. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mergen, V., Higashigaito, K., Allmendinger, T. et al. Tube voltage-independent coronary calcium scoring on a first-generation dual-source photon-counting CT—a proof-of-principle phantom study. Int J Cardiovasc Imaging 38, 905–912 (2022). https://doi.org/10.1007/s10554-021-02466-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-021-02466-y