Abstract
Volume overload in chronic severe mitral regurgitation (MR) causes left atrial (LA) remodeling. Volume overload generally diminishes after mitral valve surgery and LA size and shape are expected to recover. The recovery of LA functions named as reverse remodeling is said to be related with prognosis and mortality. A few clinical and echocardiographic parameters have been reported to be associated with LA reverse remodeling. In this study, we investigated the relationship between LA peak longitudinal strain (reservoir strain) assessed with 2-dimensional speckle tracking echocardiography (2D STE) and LA reverse remodeling. 53 patients (24 females and 29 males, mean age: 45.7 ± 13.5 years) with severe MR and preserved left ventricular systolic function were included in the study. All patients had normal sinus rhythm. The etiology of MR was mitral valve prolapse (MVP) in 37 patients and rheumatic valvular disease in 16 patients. Mitral valve repair was performed in 30 patients while 23 underwent mitral valve replacement. Echocardiography was performed before the surgery and 6 months later. LA peak atrial longitudinal strain (PALS) was assessed with speckle tracking imaging. LA reverse remodeling was defined as a percent of decrease in LA volume index (LAVI). Left atrial volume index significantly decreased after surgery (58.2 ± 16.6 vs. 43.9 ± 17.2 ml/m2, p ≤ 0.001). Mean LAVI reduction was 22.5 ± 27.2 %. There was no significant difference in LAVI reduction between mitral repair and replacement groups (22.1 ± 22.6 vs. 23.1 ± 32.8 %, p = 0.9). Although the decrease in LAVI was higher in MVP group than rheumatic group, it was not statistically significant (24.4 ± 26.8 vs. 18.2 ± 28.9 %, p = 0.4). Correlates of LAVI reduction were preoperative LAVI (r 0.28, p = 0.039), PALS (r 0.36, p = 0.001) and age (r −0.36, p = 0.007). Furthermore, in multivariate linear regression analysis (entering models), preoperative LAVI, age and PALS were all significant predictors of LA reverse remodeling (p ≤ 0.001, p = 0.04, p = 0.001 respectively). Left atrial peak longitudinal strain measured by 2D STE, in conjunction with preoperative LAVI and age is a predictor of LA reverse remodeling in patients undergoing surgery for severe MR. We suggest that in this patient population, PALS may also be used as a preoperative prognostic marker.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Volume overload caused by mitral regurgitation (MR) is one of the reasons of cardiac remodeling. With the remodeling, the size, shape and function of cardiac chambers change. When exposed to volume overload, cellular atrophy and interstitial fibrosis occur in the left atrium (LA) resulting in increased volume and decreased function of the LA [1, 2]. Previous studies have shown that LA dilatation was predictive of stroke, new onset atrial fibrillation, heart failure, mortality after acute myocardial infarction, severity of diastolic dysfunction and death [3–7]. LA size and volume have generally been used to evaluate LA functions [8, 9]. Speckle tracking echocardiography (STE), a novel angle-independent method based on frame-to-frame tracking of acoustic speckles is used for evaluation of myocardial deformation has been applied recently for evaluation of LA functions. Atrial myocardial deformation has also been evaluated using this novel method in normal population and a number of cardiac pathologies [10–14]. STE based peak atrial longitudinal strain (PALS) was found to be inversely correlated with atrial fibrosis assessed histopathologically during cardiac surgery [15]. Volume overload generally diminishes after mitral valve surgery and LA size and shape are expected to recover. The recovery of LA functions named as reverse remodeling is said to be related with prognosis and mortality [16]. However, only few clinical and echocardiographic parameters were suggested to be associated with LA reverse remodeling. In this study we investigated the relationship between LA peak longitudinal strain (reservoir strain) assessed with 2-dimensional STE and LA reverse remodeling.
Methods
Study population
53 Patients with severe mitral insufficiency, defined according to the American Society of Echocardiography (ASE) guidelines [17], in sinus rhythm with left ventricular ejection fraction (EF) ≥50 % were included in the study during the period between February 2009 and March 2011. Mitral replacement was performed in 23 patients, and the valve was repaired in 30 patients. Patients with previous AF, mitral pathologies other than rheumatic or mitral valve prolapse, mitral stenosis of more than a trivial degree (mitral valve area <2 cm2), aortic valve disease of more than a trivial degree, coronary artery disease, previous cardiac operation and congenital heart diseases were excluded from the study. The use of medications, in particular, the preoperative uses of beta blockers, angiotensin-converting enzyme inhibitors, calcium channel blockers, statins, and diuretics were documented. Informed written consent was obtained from all study subjects, and the study protocol was approved by the institutional ethics committee.
Echocardiography
Two-dimensional and Doppler echocardiography
All echocardiographic studies were done 2 days before and 6 months after the surgery. Echocardiographic studies were performed using a Vivid 7 machine (GE Vingmed Ultrasound AS, Horten, Norway), equipped with a 3.5 MHz transducer. A total of 3 cardiac cycles was recorded at the end of expiration. All data were transferred to a workstation for further offline analysis (EchoPAC PC; GE Vingmed Ultrasound AS).
Left ventricle (LV) end-systolic diameter, end-diastolic diameter, septal and posterior wall thickness at end-diastole were measured in the long-axis view using M-mode echocardiography according to the recommendations of the American Society of Echocardiography [18]. LV end-systolic and end-diastolic volumes and ejection fraction (EF) were determined from the apical four and two-chamber views using the modified bi-plane Simpson’s method. LA anteroposterior diameter was measured by 2D-guided M-mode echocardiography from parasternal long axis view at end-systole. LA volume was determined using the biplane area length method from the apical four and two-chamber views at end-systole. LA volume was indexed to body surface area as recommended [19, 20]. The postoperative percent change in LAVi was calculated as follows:
Percentage change in LAVi = (preoperative LAVi–postoperative LAVi)/preoperative LAVi × 100. LA remodeling was defined as a percent decrease in LAVi >15 % [16].
Mitral regurgitation was qualified by color Doppler and quantified by vena contracta width and the proximal isovelocity surface area (PISA) method in accordance with the recommendations of the ASE [17]. Color Doppler data were acquired at 15–17 frames/s with a depth of 16 cm. Nyquist level was adjusted 50–60 cm/s for color jet area and vena contracta whereas it was approximately 40 cm/s for PISA. Color gain was adjusted to eliminate random color in areas without flow. Mitral inflow velocities were measured at the tips of the mitral leaflets using pulsed Doppler on end-expiration. The Doppler beam was aligned to produce the narrowest possible angle between the beam and the blood flow vector. The peak velocities during early filling (E) and late filling from atrial contraction (A) were measured. Pulsed-wave tissue Doppler echocardiography was performed to obtain mitral annular peak systolic (S′), peak early (E′), and late (A′) diastolic annular velocities in the apical four-chamber view with the sample volume (2-mm axial length) at the lateral side of the mitral annulus. Systolic pulmonary artery pressure was calculated by adding estimated the right atrial pressure to the systolic right ventricular-right atrial gradient calculated from the peak velocity of systolic transtricuspid regurgitant flow signal [21].
Speckle tracking echocardiography (STE)
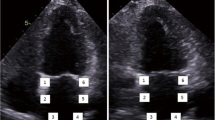
Apical 4-chamber view was used for LA strain measurements. The frame rate for images was adjusted between 5 and 70 frames/s. For 2 dimensional STE analysis, a line was manually drawn along the LA endocardium when the LA was at its minimum volume after contraction. The software then automatically generated additional lines near the atrial epicardium and mid-myocardial line, with a region of interest default width of 15 mm. Before processing, a cine loop preview feature visually confirmed that the internal line follows the LA endocardium throughout the cardiac cycle. If tracking of the LA endocardium was unsatisfactory, manual adjustments or changing software parameters (e.g. region of interest size or smoothing functions) was made. The software divided the LA endocardium into 6 segments. Segments in which no adequate image quality could be obtained were rejected by the software and excluded from the analysis. Overall, 318 segments were analyzed (6 segments for each patient), and a total of 4.8 % segments were excluded. LA peak strain just before mitral valve opening was taken as peak atrial longitudinal strain (PALS) and LA strain just before atrial contraction (onset of the P-wave on electrocardiography) was taken as peak atrial contraction strain (PACS) (Fig. 1).
Left atrial longitudinal strain parameters. Composite figure showing the measurement of peak atrial longitudinal strain (PALS) and of peak atrial contraction strain (PACS) using the speckle tracking echocardiography (STE) from an apical four-chamber view. The dashed curve represents the average atrial longitudinal strain along the cardiac cycle. AVC aortic valve closure
Reproducibility
Inter- and intra-observer reproducibilities were assessed for both the PALS and the PACS values. For intra-observer assessment, the measurements were re-analysed after 2 weeks. The Bland–Altman analysis for inter-observer reproducibility [mean difference-95 % confidence interval (CI)] and intra-observer reproducibility (intra-class correlation coefficient, 95 % CI) were calculated, and the intra-class correlation coefficient showed good inter- and intra-observer agreement [inter-observer and intra-observer agreement were assessed for PALS, −2.7 [7.3–(−1.9)] and 0.90 (0.84–0.95) respectively; and for PACS 1.6 [5.5–(−2.3)] and 0.88 (0.80–0.93) respectively].
Statistical analysis
All values are expressed as the mean ± standard deviation or as percentages. The normal distribution of each variable was assessed using the Kolmogorov–Smirnov test. For the non-normally distributed variables, the comparison of groups was performed using nonparametric tests. Comparisons of the continuous variables in the 2 groups were performed using Student’s t test. Distributions of categorical variables were compared with the χ2 or Fisher’s exact test when appropriate. Pearson correlation analysis was used to compare the relationship of the continuous variables. Multiple linear regression analysis (enter models) was used to determine independent predictors of LA reverse remodeling. All statistical analyses were performed with SPSS version 16.0 (SPSS, Inc, Chicago, IL, USA), and p values of <0.05 were taken to indicate statistical significance.
Results
The mean age was 45.7 ± 13.5 years, and 54.7 % were females. The etiologies of MR in this study were mitral valve prolapse in 37 (69.8 %) patients and rheumatic in 16 (30.2 %) patients. 30 patients (56.6 %) underwent mitral valve repair while mitral valve replacement was done in 23 patients (43.4 %). Tricuspid valve repair (TVR) using annuloplasty ring was done in 13 patients (24.5 %). The mean systolic blood pressure, diastolic blood pressure and heart rate were 129.9 ± 7.3 mmHg, 79.3 ± 5.3 mmHg and 71.3 ± 7.7 bpm respectively. Preoperative PALS was (21.7 ± 8.1 %), and PACS was (10.2 ± 3.5 %). The mean effective regurgitant orifice area preoperatively was 0.58 ± 0.15 cm2), mitral regurgitant volume was (80.4 ± 24.2 ml). The clinical and echocardiographic parameters are listed in Tables 1 and 2.
After surgery, LV end-diastolic volume, LV end-diastolic diameter, LV EF, were significantly decreased while LV systolic volume and LV end-systolic diameter were not significantly reduced. LA volume index and LA antero-posterior diameter were significantly decreased (Table 3). The mean postoperative decrease in LAVI was 22.5 ± 27.2 %. LAVI was ≥40 ml/m2 88.7 % patients and LAVI was ≥60 ml/m2 39.6 % patients.
Left atrial reverse remodeling (as previously defined) was found in 34 patients (64.2 %). 11 patients (20.7 %) showed an increase in LA volume index after surgery, whereas 42 patients showed a decrease in LA volume after surgery. There was no significant difference in LAVİ reduction between mitral repair and replacement groups (22.1 ± 22.6 vs. 23.1 ± 32.8 %, p = 0.9). Besides decrease in LAVI was higher in MVP group than rheumatic group, but it was not statistically significant (24.4 ± 26.8 vs. 18.2 ± 28.9 %, p = 0.4) (Fig. 2). Preoperative LAVI (r 0.28, p = 0.039), age (r −0.36, p = 0.007), and PALS (r 0.36, p = 0.001) were correlated significantly with the percentage of LA volume change (Fig. 3; Table 4). Furthermore, in multivariate linear regression analysis (entering models), preoperative LAVI, age and PALS were all significant predictors of LA reverse remodeling (p ≤ 0.001, p = 0.04, p = 0.001 respectively) (Table 5).
Discussion
In this study, significant cardiac remodeling as indicated by LA and LV volume was seen after MV surgery. Higher PALS and LAVI and younger age were found to be independent predictors of LA reverse remodeling.
Volume overload in MR has a direct effect on the LA. LA tries to compensate volume overload in order to prevent pulmonary congestion [22]. Cellular hypertrophy and interstitial fibrosis occur in the LA when exposed to volume overload, which initiates the remodeling process seen as increase in volume and decrease in function of the LA [1, 2]. Previous studies have shown that LA dilatation is predictive of a number of conditions and mortality [3–7]. In MR patients, LA dilatation was related with AF, cardiac events and survival [23–26].
Left atrial volume and pressure usually drop after surgery or percutaneous valve intervention [27]. The LA functions can recover and is termed reverse remodeling. Reversal of atrial functions can give additional information regarding the prognosis. A study by Laurens et al. [28] has shown that in patients who underwent catheter ablation for atrial fibrillation, left atrial reverse remodeling may be a more robust marker for successful atrial fibrillation ablation. Kim et al. conducted a study on the relationship between LA reverse remodeling and prognosis. They included patients with mitral stenosis who underwent valvuloplasty, and followed them for 11 years. They found that the percentage change in LA volume was related with cardiovascular events and total mortality [29]. But there is no study regarding the left atrial reverse remodeling and clinical outcomes in patients with MR. But it is very likely that LA reverse remodeling will be predictive of better clinical outcomes in these patients as well.
Previous studies have established the relationship between LAVI and LA reverse remodeling. In some studies, LAVI was found to be positively correlated with LA volume change but in others, it was found to be negatively correlated [16, 30–33]. In our study, preoperative left atrial volume index was positively correlated with left atrial volume change. This difference may be due to lower values of preoperative LAVI in our patients. Irreversible structural changes will be less expected when the LAVI is rather low, and reversal of LA size and function is more likely after surgery.
Similar to former studies, we found a reverse correlation between age and LA reverse remodeling [16, 32, 33]. As more fibrosis and hypertrophy occur with increase in age, LA elasticity is reduced and LA remodeling is less probable even if volume overload is released by surgery. Furthermore, older patients may have had MR for a longer duration and more irreversible changes could have occurred. Early surgical intervention can contribute to better reverse atrial remodeling.
Similar to a study by Cho, we could not find the effect of operation type (replacement or repair) on LA reverse remodeling [30]. It must be due to similar preoperative LA volume and LA strain in both groups. As less fibrosis of atrial wall is expected in the degenerative etiology than rheumatic disease, more decrease in volume is also expected but it was not statistically significant. Studies of larger scale are needed.
Left ventricular diastolic dysfunction and filling pressure are expected to have an effect on LA reverse remodeling. It is known that measurements of LV filling in chronic MR exhibit a biphasic pattern depending on the state of LV contractility. Previous studies have suggested that enhanced LV filling and increased chamber compliance in the early stages of chronic MR; as systolic dysfunction sets in, myocardial stiffness increases and filling rates decrease. Filling of the LV is also directly dependent on systolic function; elastic energy stored in the myocardium during systole (restoring forces) is released at the onset of myocardial relaxation, generating a suction force [34]. The subjects in our study have normal ejection fraction, and although the filling pressure is elevated, no correlation was found with the reverse remodeling. LA may also have adaptive changes to volume and pressure overload as in the left ventricle. Moreover, The presence of MR itself is well recognized as a confounding factor in interpreting transmitral flow variables and mitral valve annular E′ as indices of myocardial relaxation [35, 36]. In a study by Diwan et al. [37] in MR patients with normal ejection fraction, no correlation was found between echocardiographic filling parameters and LV filling pressure.
Atrial myocardial deformation has also been evaluated using speckle tracking echocardiography methods in normal population and a number of cardiac pathologies [10–14]. Suman et al. [38] demonstrated that VVI-based LA wall strain and strain rate during ventricular systole was inversely correlated with atrial fibrosis evaluated by three-dimensional-delayed enhancement MRI.
Cameli et al. evaluated atrial longitudinal strain in asymptomatic MR patients of different degrees using STE. In this study, PALS increased in mild MR patients when compared to, control group due to atrial compliance. In patients with moderate and severe MR, PALS decreased because of ultrastructural changes such as interstitial fibrosis [39]. In a recent study by Cameli et al. [15] atrial strain was again evaluated by STE in patients with MR who were undergoing surgery. 46 patients were included in their study. The extent of atrial fibrosis and endocardial thickening were determined from surgery tissue sample. In patients with high PALS, fibrosis was found less. There was a good negative correlation between atrial strain and fibrosis.
Peak atrial longitudinal strain reflects the passive stretching of the LA during LV systole, and is an accurate measurement of LA reservoir function. Volume overload causes atrial remodeling including the increase in interstitial fibrosis, decrease in atrial elasticity, and decline in LA reservoir functions. We also found that higher PALS was an independent predictor of LA reverse remodeling. Higher PALS may indicate that volume overload has caused a lesser fibrosis and a relatively better preserved elasticity of atrial wall. In these patients, volume reversal is more likely.
Study limitation
Although speckle tracking echocardiography is considered angle-independent, echocardiographic image quality should be adequate to perform an optimal analysis. We conducted this study with patients in early postoperative period; therefore, long-term results of the study might be different from what we had observed. Due to the lack of proper national record system, we did not know the exact duration of MR of each patient. Larger studies are needed to confirm the results of the present study, and the prognostic implications of a LA reverse remodeling after MV surgery should be demonstrated with a longer clinical follow-up. Our study patients had preserved EF and normal sinus rhythm. Our results may not reflect all patients with MR.
Conclusion
Left atrial peak longitudinal strain measured by 2D STI, in conjunction with preoperative LAVI and age is a predictor of LA reverse remodeling in patients undergoing surgery for severe MR. We suggest that in this patient population, PALS may also be used as a preoperative prognostic marker.
Abbreviations
- EF:
-
Ejection fraction
- LA:
-
Left atrium
- LAVI:
-
Left atrial volume index
- LV:
-
Left ventricle
- PALS:
-
Peak atrial longitudinal strain
- PACS:
-
Peak atrial contraction strain
- MVP:
-
Mitral valve prolapse
- MVR:
-
Mitral valve replacement
- TVR:
-
Tricuspid valve repair
- PISA:
-
Proximal isovelocity surface area
- STE:
-
Speckle tracking echocardiography
- ASE:
-
American Society of Echocardiography
- MRI:
-
Magnetic resonance imaging
- BNP:
-
Brain natriuretic peptide
- MR:
-
Mitral regurgitation
References
Anne W, Willems R, Roskams T et al (2005) Matrix metalloproteinases and atrial remodeling in patients with mitral valve disease and atrial fibrillation. Cardiovasc Res 67:655–666
Verheule S, Wilson E, Everett T et al (2003) Alterations in atrial electrophysiology and tissue structure in a canine model of chronic atrial dilatation due to mitral regurgitation. Circulation 107:2615–2622
Benjamin EJ, D’Agostino RB, Belanger AJ et al (1995) Left atrial size and the risk of stroke and death. The Framingham Heart Study. Circulation 92:835–841
Tsang TS, Barnes ME, Bailey KR et al (2001) Left atrial volume: important risk marker of incident atrial fibrillation in 1655 older men and women. Mayo Clin Proc 76:467–475
Barnes ME, Miyasaka Y, Seward JB et al (2004) Left atrial volume in the prediction of first ischemic stroke in an elderly cohort without atrial fibrillation. Mayo Clin Proc 79:1008–1014
Moller JE, Hillis GS, Oh JK et al (2003) Left atrial volume: a powerful predictor of survival after acute myocardial infarction. Circulation 107:2207–2212
Pritchett AM, Jacobsen SJ, Mahoney DW et al (2003) Left atrial volume as an index of left atrial size: a population-based study. J Am Coll Cardiol 41:1036–1043
Lester SJ, Ryan EW, Schiller NB et al (1999) Best method in clinical practice and in research studies to determine left atrial size. Am J Cardiol 84:829–832
Rodevan O, Bjornerheim R, Ljosland M et al (1999) Left atrial volumes assessed by three- and two-dimensional echocardiography compared to MRI estimates. Int J Card Imaging 15:397–410
Vianna-Pinton R, Moreno CA, Baxter CM et al (2009) Two dimensional speckle-tracking echocardiography of the left atrium: feasibility and regional contraction and relaxation differences in normal subjects. J Am Soc Echocardiogr 22:299–305
Saraiva RM, Demirkol S, Buakhamsri A et al (2010) Left atrial strain measured by two-dimensional speckle tracking represents a new tool to evaluate left atrial function. J Am Soc Echocardiogr 23:172–180
Cameli M, Caputo M, Mondillo S et al (2009) Feasibility and reference values of left atrial longitudinal strain imaging by two-dimensional speckle tracking. Cardiovasc Ultrasound 7:6
Karabay CY, Zehir R, Güler A et al (2013) Left atrial deformation parameters predict left atrial appendage function and thrombus in patients in sinus rhythm with suspected cardioembolic stroke: a speckle tracking and transesophageal echocardiography study. Echocardiography 30(5):572–581
Candan O, Ozdemir N, Aung SM et al (2013) Left atrial longitudinal strain parameters predict postoperative persistent atrial fibrillation following mitral valve surgery: a speckle tracking echocardiography study. Echocardiography 30(9):1061–1068
Cameli M, Lisi M, Righini FM et al (2013) Usefulness of atrial deformation analysis to predict left atrial fibrosis and endocardial thickness in patients undergoing mitral valve operations for severe mitral regurgitation secondary to mitral valve prolapse. Am J Cardiol 111(4):595–601
Antonini-Canterin F, Beladan CC, Popescu BA et al (2008) Left atrial remodelling early after mitral valve repair for degenerative mitral regurgitation. Heart 94:759–764
Zoghbi WA, Enriquez-Sarano M, Foster E et al (2003) Recommendations for evaluation of the severity of native valvular regurgitation with two dimensional and Doppler echocardiography. J Am Soc Echocardiogr 16:777–802
Lang RM, Bierig M, Devereux RB et al (2006) Recommendations for chamber quantification: a report from the American Society of Echocardiography’s guidelines and Standards Committee and the Chamber Quantification Writing Group. J Am Soc Echocardiogr 35:363–369
Schabelman S, Schiller NB, Silverman NH et al (1981) Left atrial volume estimation by two-dimensional echocardiography. Catheter Cardiovasc Diagn 7:165–178
Schiller NB, Botvinick EH (1986) Noninvasive quantitation of the left heart by echocardiography and scintigraphy. Cardiovasc Clin 17:45–93
Pepi M, Tamborini G, Galli C et al (1994) A new formula for Echo–Doppler estimation of right ventricular systolic pressure. J Am Soc Echocardiogr 7:20–26
Braunwald E, Awe WC (1963) The syndrome of severe mitral regurgitation with normal left atrial pressure. Circulation 27:29–35
Kernis SJ, Nkomo VT, Messika-Zeitoun D et al (2004) Atrial fibrillation after surgical correction of mitral regurgitation in sinus rhythm: incidence, outcome, and determinants. Circulation 110:2320–2325
Tourneau TL, Messika-Zeitoun D, Russo A et al (2010) Impact of left atrial volume on clinical outcome in organic mitral regurgitation. J Am Coll Cardiol 56:570–578
Messika-Zeitoun D, Bellamy M, Avierinos JF et al (2007) Left atrial remodelling in mitral regurgitation-methodologic approach, physiological determinants, and outcome implications: a prospective quantitative Doppler-echocardiographic and electron beam-computed tomographic study. Eur Heart J 28(14):1773–1781
Bitigen A, Bulut M, Tanalp AC, Kirma C, Barutçu I, Pala S, Erkol A, Boztosun B (2007) Left atrial appendage functions in patients with severe rheumatic mitral regurgitation. Int J Cardiovasc Imaging 23(6):693–700
Bitigen A, Türkmen M, Karakaya O, Saglam M, Barutcu I, Esen AM, Türkyilmaz E, Erkol A, Bulut M, Boztosun B, Kirma C (2006) Early effects of percutaneous mitral valvuloplasty on left atrial mechanical functions. Tohoku J Exp Med 209:285–289
Tops LF, Delgado V, Bertini M et al (2011) Left atrial strain predicts reverse remodeling after catheter ablation for atrial fibrillation. J Am Coll Cardiol 57(3):324–331
Kim KH, Kim YJ, Shin DH et al (2010) Left atrial remodelling in patients with successful percutaneous mitral valvuloplasty: determinants and impact on long-term clinical outcome. Heart 96(13):1050–1055
Cho DK, Ha JW, Chang BC et al (2008) Factors determining early left atrial reverse remodeling after mitral valve surgery. Am J Cardiol 101(3):374–377
Marsan NA, Maffessanti F, Tamborini G et al (2011) Left atrial reverse remodeling and functional improvement after mitral valve repair in degenerative mitral regurgitation: a real-time 3-dimensional echocardiography study. Am Heart J 161(2):314–321
Song BG, On YK, Jeon ES et al (2010) Atrioventricular reverse remodeling after valve repair for chronic severe mitral regurgitation: 1-year follow-up. Clin Cardiol 33(10):630–637
Hyllén S, Nozohoor S, Meurling C et al (2013) Determinants of left atrial reverse remodeling after valve surgery for degenerative mitral regurgitation. J Heart Valve Dis 22(1):2–10
Borg AN, Harrison JL, Argyle RA, Pearce KA, Beynon R, Ray SG (2010) Left ventricular filling and diastolic myocardial deformation in chronic primary mitral regurgitation. Eur J Echocardiogr 11:523–529
Rossi A, Cicoira M, Golia G, Anselmi M, Zardini P (2001) Mitral regurgitation and left ventricular diastolic dysfunction similarly affect mitral and pulmonary vein flow Doppler parameters: the advantage of end-diastolic markers. J Am Soc Echocardiogr 14:562–568
Ohte N, Narita H, Akita S, Kurokawa K, Hayano J, Kimura G (2002) Striking effect of left ventricular high filling pressure with mitral regurgitation on mitral annular velocity during early diastole. A study using colour M-mode tissue Doppler imaging. Eur J Echocardiogr 3:52–58
Diwan A, McCulloch M, Lawrie GM, Reardon MJ, Nagueh SF (2005) Doppler estimation of left ventricular filling pressures in patients with mitral valve disease. Circulation 111:3281–3289
Kuppahally SS, Akoum N, Burgon NS et al (2010) Left atrial strain and strain rate in patients with paroxysmal and persistent atrial fibrillation: relationship to left atrial structural remodeling detected by delayed-enhancement MRI. Circ Cardiovasc Imaging 3(3):231–239
Cameli M, Lisi M, Giacomin E et al (2011) Chronic mitral regurgitation: left atrial deformation analysis by two-dimensional speckle tracking echocardiography. Echocardiography 28(3):327–334
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Candan, O., Ozdemir, N., Aung, S.M. et al. Atrial longitudinal strain parameters predict left atrial reverse remodeling after mitral valve surgery: a speckle tracking echocardiography study. Int J Cardiovasc Imaging 30, 1049–1056 (2014). https://doi.org/10.1007/s10554-014-0433-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-014-0433-9