Abstract
The diagnostic performance of in-stent restenosis (ISR) by 64-slice multidetector CT coronary angiography (CTCA) has been reported to be influenced by multiple factors. We evaluated individual factors (stent diameter, material and strut thickness) and therefore determined the proper population for follow-up by using this modality. A total of 171 stents were evaluated in 83 consecutive patients with stents imaged with CTCA and conventional coronary angiography. The stent diameter ranged from 2.25 mm to 4.5 mm. 2 models of stainless steel (Taxus Liberte (Boston Scientific, US), 56 stents and Cypher Select (Cordis, US), 34 stents) and 2 models of cobalt alloy (Endeavor (Medtronic, US), 33 stents and Firebird2 (MicroPort, China), 48 stents) were included. By comparing to conventional coronary angiography, the image quality and diagnostic accuracy for ISR were evaluated. The image quality of Taxus, Endeavor and Firebird are markedly better than Cypher in large caliber group (≧3.0 mm) (P < 0.001). Except for Cypher, all other stents with diameter ≧3.0 mm showed excellent diagnostic accuracy (sensitivity 100%, specificity 94.4–96% whereas stents with diameter <3.0 mm had poor diagnostic accuracy (sensitivity 100%, specificity 33.3–70%). Cypher is the stent with thickest strut in our study, and showed reduced image quality and diagnostic accuracy in all stent size, due to large number of unassessable stents. Among 16 binary ISR, 12 lesions were correctly diagnosed by CTCA while the other 4 lesions were unassessable. The main reason for low specificity in small caliber group is the large number of unassessable stents. CTCA has high diagnostic accuracy to identify ISR in selected stents with a diameter of ≧3.0 mm.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Coronary artery stenting is currently the predominant strategy of myocardial revascularization in patients with obstructive coronary artery disease [1, 2]. Even with the wide application of drug-eluting stent (DES), in-stent restenosis (ISR) and stent occlusion (SO) remains the major complication of this procedure [3, 4]. Early detection of ISR is of clinical significance to avoid recurrent ischemic progress, prevent myocardial infarction, and thereby improve long term prognosis.
Invasive coronary angiography (ICA) is currently the gold standard procedure for assessment of ISR. However, ICA is both costly and invasive, and is associated with potentially serious complications [5, 6]. CT coronary angiography (CTCA) has been proved to be a reliable non-invasive alternative to ICA for detection of coronary artery stenosis since the advent of 64-slice CT [7]. Nevertheless, the value of CTCA for evaluation of stent patency remains uncertain and needs to be validated in the recent guideline on CTCA [8]. We aim to evaluate the value of CTCA for detection of ISR, and further identify the proper population for CTCA follow-up based on stent models (materials and strut thickness) and calibers.
Methods
Patient population
Between January 2008 and July 2010, consecutive clinically-ordered patients with prior coronary stent implantation in native vessels were prospectively enrolled in our study. 4 different DES with varied diameters (2.25–4.5 mm) were included: Taxus Liberte (Boston Scientific, US), Cypher Select (Cordis, US), Endeavor (Medtronic, US) and Firebird2 (MicroPort, China). Follow-up CTCA and repeat ICA were performed in all cases within an interval of 2 weeks. Exclusion criteria for CTCA include renal insufficiency (serum creatinine > 1.5 mg/dl), allergy to contrast media, clinical history of uncontrolled hyperthyroidism or multiple myeloma, atrial fibrillation or other rhythm irregularity, and inability to perform breath hold. All patients gave written informed consent, and the study protocol was approved by the hospital ethics committee.
Scan protocol of CTCA
A 64-slice multidetector CT (Lightspeed VCT 64, GE, Milwaukee, US) was employed for scanning. β-blocker (25–50 mg) was administrated orally in patients with heart rate >65 bpm. A bolus of contrast media (Iopamidol, 370 mg iodine/ml, Schering AG, Berlin, German) was injected into antecubital vein at the rate of 4.5–5 ml/s, followed by a 20–40 ml saline flush by using dual-barrel power injector (Tyco, Cincinnati, US). The amount of the contrast media was determined according to the patient’s body weight and scan time. A test bolus was firstly injected and the region of interest was placed within ascending aorta to determine a proper delay time, which was defined as 4 s plus the peak time of ascending aorta. Retrospective ECG-gated CTA was performed with collimation = 64 × 0.625 mm, slice thickness = 0.625 mm, rotation time = 350 ms, pitch and current were ECG modified (ECG-dependent dose modulation technique was applied, full dose during the R-R interval of 40–80%), and tube voltage = 120 Kvp.
Image reconstruction and analysis
For better delineation of both vessel wall and stent lumen [9], two sets of axial images were reconstructed with different kernels: smooth kernel (soft, GE) and sharp kernel (bone, GE) with slice thickness of 0.625 mm. Data was transferred to an offline workstation (ADW4.3, GE) for further analysis. Axial images, curved planar reformation (CPR), multiplanar reformation (MPR) as well as volume rendering (VR) images were available for evaluation. All images were evaluated independently by two radiologists experienced in CTCA who were blinded to the number, location, diameter, and type of stents, to the clinical history of patients. Disagreements between the two readers for any image set were resolved by consensus, and the consensus findings were used in all assessments of diagnostic performance.
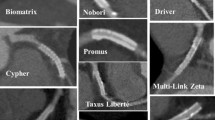
A standard American Heart Association (AHA) segmentation was employed for evaluation of all segments [10]. Image quality of stent was assessed by using a 3-point semi-quantitative scale: 3 = excellent (absence of strut artifact), 2 = acceptable (presence of less strut artifact, but still diagnostic), 1 = poor (presence of severe strut artifact, non-diagnostic) (Fig. 1). The presence and extent of ISR were visually and semi-quantitatively classified into 3 grades (Fig. 2): grade 1 none or slight neointimal proliferation with absence of ISR, grade 2 mild neointimal proliferation with ISR < 50%, grade 3 neointimal proliferation with ISR ≧ 50% or occlusion. Grade 3 was considered as binary ISR on CTCA.
Comparison of image quality of different stents by using 3-point semi-quantitative scale. a Firebird2, (3.0 × 18 mm, 3.0 × 23 mm), score 3. b Endeavor, (3.0 × 24 mm, 3.0 × 24 mm), score 3. c Taxus Liberte, (3.0 × 28 mm), score 3. d Cypher Select, (3.0 × 23 mm), score 2. e Cypher Select, (3.0 × 18 mm, 3.0 × 23 mm, 2.75 × 13 mm, 2.75 × 18 mm), score 1
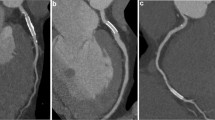
ISR Grading: CTCA compared to ICA. The white lines on the figures show the sites of stents. A to c are Grade 1 (show absence of ISR). a The long-axis view of one Endeavor stent placed in proximal left anterior descending artery (LAD) (3.5 × 16 mm) on CTCA. b The short-axis view of stent on CTCA. c The Invasive coronary angiography (ICA) image. D to f are Grade 2 (show ISR < 50%). d The long-axis view of one Taxus stent placed in proximal left circumflex artery (LCx) (3.0 × 8 mm) on CTCA. e The short-axis view of stent on CTCA. f The Invasive coronary angiography (ICA) image. G to I are Grade 3 (show ISR > 50%). g The long-axis view of one Taxus stent placed in middle right coronary artery (RCA) (3.0 × 20 mm) on CTCA. h The short-axis view of stent on CTCA. i The Invasive coronary angiography (ICA) image. J to l are Grade 3 (show total occlusion). j The long-axis view of two Firebird stents placed in proximal LAD (3.0 × 18 mm) and proximal-middle LAD (2.75 × 18 mm) on CTCA. k The short-axis view of stent on CTCA. l The Invasive coronary angiography (ICA) image
ICA procedure and analysis
The ICA was performed with standard techniques, and at least 2 different views were obtained for each main vessel. All segments were evaluated by 2 skilled observers who were blinded to the results of CTCA. Disagreements between the two readers were resolved by consensus. The percent diameter stenosis (%DS) was calculated by quantitative coronary angiography, employing the same grading system as of CTCA. Binary ISR or significant stenosis (>50%) in non-stented segment was considered for further interventions.
Statistical analysis
Sensitivity, specificity, positive (PPV) and negative predictive values (NPV) of diagnostic accuracy of CTCA were calculated on patient-based, segment-based and stent-based analysis respectively, using ICA as the gold standard. A further comparison of diagnostic accuracy of ISR on CTCA between stents of different models and calibers was carried out. Unassessable stents and non-stented segments were regarded as having binary ISR and significant coronary artery stenosis (>50%) for further analysis, as ISR and significant stenosis could not be excluded in those segments. Comparisons of quantitative variables were performed by one-way analysis of variance for normally distributed variables. SPSS 13.0 (IBM, US) was used for data analysis. A probability value of 0.05 was considered to be statistically significant.
Results
Patient characteristics and stent parameters
83 patients (mean age: 66.66 ± 9.42, range 46–83 years, 62 males and 21 females) with 171 stents (stent per patient: 2.1 ± 1.22, range 1–6 stents) were finally included in our study. The average interval between CTCA and last stenting procedure was 16.92 ± 6.27 months (range 4–31 months). The dose length product (DLP) of CTCA was 550.36 ± 49.27 mGy cm (range 441–660 mGy cm). An ICA was performed in all patients with an interval of 6.93 ± 2.63 days (range 1–14 days) of the CTCA. Detailed stent parameters were given in Table 1 and Table 2.
Comparison of image quality between different stent groups
According to the semi-quantitative score, the image quality of Firebird, Endeavor and Taxus were markedly better than Cypher in large caliber group (≧3 mm) (P < 0.001) (Table 2). Firebird, Endeavor and Taxus had no unassessable stents in those subsets while Cypher had 33.3% (7/21) unassessable stents. For small caliber group, the image quality dropped significantly and the unassessable stents of Firebird, Endeavor, Taxus and Cypher were 31.8% (7/22), 69.2% (9/13), 50% (14/28) and 92.3% (12/13), respectively. Firebird has the lowest unassessable rate and best semi-quantitative score in small caliber group. Hence, the thinnest stent (Firebird) has the best image quality while the thickest stent (Cypher) has the worst. As Taxus and Endeavor have similar strut thickness and cell-design, comparison of image quality between those two was carried out in both large and small caliber groups, to determine the influence of stainless steel and cobalt alloy on image quality of CTCA. The results showed there is no statistical significance between image quality of Taxus and Endeavor in both subgroups (P > 0.05).
Binary ISR and non-stented segment stenosis: compare CTCA with ICA
16 binary ISR (≧50% or occlusion, grade 3) were identified on ICA within 16 stents (9.36%) in 13 patients. Among those ICA-proved binary ISR, 12 stents were correctly diagnosed by CTCA while another 4 stents were unassessable on CTCA. The diagnostic performance of each stent group was described in detail in Table 3.
Firebird, Endeavor and Taxus showed excellent diagnostic accuracy in their large caliber group (≧3 mm) and all binary ISR were correctly detected by CTCA in those subsets. All stents in this population were found to be assessable. Nevertheless, Low PPV and specificity were observed in small caliber group of Firebird, Endeavor and Taxus, mainly due to increased number of unassessable stents. Cypher, however, was revealed to be inferior to other 3 counterparts in both large and small caliber groups.
Other than Grade 3 ISR, ICA revealed 14 stents of Grade 2 ISR and 141 stents of Grade 1 (Table 4). The diagnostic accuracy of CTCA grading were 68.8% (97/141), 50% (7/14) and 75% (12/16) for Grade 1, 2 and 3 groups. When excluding unassessable stents, the values were dramatically raising to 98% (97/99), 70% (7/10) and 100% (12/12) respectively.
In addition to stent based analysis, a further patient based and segment based analysis were carried out (Table 5). Binary ISR or significant coronary stenosis (>50%) in non-stented segments could be reliably excluded with high NPV and sensitivity per patient as well as per segment.
Discussion
Since ISR is a major complication of coronary intervention procedures, an early diagnosis is of clinical significance to prevent secondary myocardial infarction, and therefore improve long term prognosis. ICA is currently the most applied and reliable modality for ISR diagnosis. However, the incidence of ISR has been decreased significantly since the use of drug-eluting stents [11, 12] and negative ICA findings in symptomatic post-stenting patients are not unusual. Thus, an alternative non-invasive imaging modality to ICA may markedly reduce the number of negative ICA as well as undesired complications.
64-slice CT has no doubt played an irreplaceable role in non-invasive diagnosis of coronary artery disease with high sensitivity and specificity when comparing to ICA [7]. Previous studies have also shown that CTCA has a high sensitivity and specificity for diagnosis of ISR, which ranged from 75 to 95% and 74–98% respectively [9, 13–17]. However, all those studies did not include enough number of stents with diameter less than 3 mm and did not compare the performance between stents of different materials and strut thickness. Thus, those results might overestimate the diagnostic performance of CTCA. In our experience, the number of unassessable stents increases remarkably as nominal diameter decreases, and there is for sure varied extent of strut artifact between stents of same caliber but different strut thickness. For this reason, our study mostly focused on the impact of those factors on the diagnostic performance of CTCA.
Stent-related factors affecting CTCA imaging
The results of our study showed that stent diameter is the most important factor affecting image quality. Stents with nominal diameter ≥3 mm have best image quality score, lumen visibility and excellent diagnostic performance comparing to ICA, except for Cypher stent. On the other hand, stents with nominal diameter less than 3 mm were revealed to have low PPV and specificity for diagnosis of ISR, mainly due to much more severe strut artifact, resulting in large number of unassessable stents.
Firebird, Taxus and Endeavor are both having open-cell design and with metal strut thickness less than 100 μm, which is thought to be another main factor affecting strut artifact. Those 3 stents showed similar diagnostic performance between large caliber groups, despite different stent materials. For small caliber group, Firebird showed best image quality and diagnostic accuracy, mainly thanks to its thinnest metal strut thickness. Considering the large sample number of each subgroup, the results of our study are believed to be reliable and we are convinced that there is no significant difference between stents of stainless steel and cobalt alloy in terms of strut artifact as well as diagnostic performance for ISR.
Cypher, however, showed reduced diagnostic performance even in large caliber group. Better performance is only acquired when diameter is ≥3.5 mm. The underlying reason is thought to be thicker metal strut with closed-cell system. Cypher has much thicker metal strut (140 μm) than its stainless steel countpart Taxus (97 μm), and is the only stent with closed-cell design, all making it have the most severe strut artifact. Therefore, CTCA evaluation of Cypher stent with nominal diameter less than 3.5 mm seems to be unpractical and is not recommended for the use of post-stenting follow-up.
Proper population for CTCA follow-up
Based on the above findings of our study, stents with nominal diameter ≥3 mm (except for Cypher stent) have excellent diagnostic performance for ISR, and may be useful for CTCA follow-up. On the other hand, Cypher stents and other stents with nominal diameter less than 3 mm yield impaired diagnostic performance and are not recommended for CTCA follow-up. Notably, although there is a large number of unassessable stents in small caliber group, the overall NPV and sensitivity of remain perfect (100%). That means a clinically-significant binary ISR can be confidently excluded by a negative CTCA examination.
Therefore, the best candidates for this examination are patients with atypical symptoms and stents’ property meeting the above criteria. CTCA in those patients can reliably exclude binary ISR as well as significant stenosis in non-stented segments with high probability, and save patients from unnecessary ICA. Asymptomatic patients shall be followed up only clinically, and patients with typical symptoms should be referred directly for angiography.
Challenges in CTCA and tips for improvement
The image quality of CTCA is sensitive to various factors, which include stent-related artifacts and patient-related artifacts. The key for a successful study is to reduce those artifacts as much as we can.
For stent-related artifacts, as has been introduced by many other authors, a proper convolution kernel (bone, sharp) of reconstruction can effectively reduce beam hardening artifacts [13, 18], which is the major type of artifact that complicates the imaging of coronary stents. Hence, pair images of 2 reconstruction kernels (soft or smooth, bone or sharp) shall be routinely obtained for delineation of both soft tissue structures (vessel wall, vessel lumen) and objects next to high-density obstacles (stent lumen).
For patient-related artifacts, thorough explanation of the examination and good training of breath-holding are the initial steps to ensure good image quality. In addition, heart rate shall be controlled with administration of beta-blocker.
Finally, combination of proper reformation images can help to estimate the lesion extent more precisely. As ISR secondary to a neointimal proliferation might be eccentric, an improper angle of CPR may under-or overestimate the extent of ISR. In our experience, a perpendicular view (short axis view of stent) is the most reliable view for the assessment of ISR extent because both eccentric and concentric lesion can be clearly visualized in it.
Study limitations
The number of ISR in our study is relatively small and additional studies in larger populations are warranted before widespread clinical adoption of CTCA. Furthermore, there are far more other types of stent widely used in clinical application. Previous in vitro studies have revealed varied performance of 29–68 different types of stents [19, 20]. Further in vivo studies have to be carried out to cover the whole stent catalogue in order to establish a complete profile of each individual.
Conclusions
Stent diameter and strut thickness are 2 underlying stent-related factors affecting diagnostic performance of CTCA. Binary ISR can be correctly diagnosed and excluded in large caliber groups of Firebird, Taxus and Endeavor, with stent diameter ≥3 mm. Consequently, CTCA follow-up in symptomatic patients is promising, although additional research is warranted.
References
Sousa JE, Serruys PW, Costa MA (2003) New frontiers in cardiology: drug-eluting stents: Part I. Circulation 107:2274–2279
Sousa JE, Serruys PW, Costa MA (2003) New frontiers in cardiology: drug-eluting stents: Part II. Circulation 107:2283–2289
Moses JW, Leon MB, Popma JJ et al (2003) Sirolimus-eluting stents versus standard stents in patients with stenosis in a native coronary artery. N Engl J Med 349:1315–1323
Stone GW, Ellis SG, Cox DA et al (2004) A polymer-based, paclitaxel-eluting stent in patients with coronary artery disease. N Engl J Med 350:221–231
De Bono D, The Joint Audit Committee of the British Cardiac Society, Royal College of Physicians of London (1993) Complications of diagnostic cardiac catheterisation: results from 34,041 patients in the United Kingdom confidential enquiry into cardiac catheter complications. Br Heart J 70:297–300
Young N, Chi KK, Ajaka J et al (2002) Complications with outpatient angiography and interventional procedures. Cardiovasc Intervent Radiol 25:123–126
Stein PD, Yaekoub AY, Matta F et al (2008) 64-slice CT for diagnosis of coronary artery disease: a systematic review. Am J Med 121:715–725
Daniel BM, Daniel SB, Matthew JB et al (2010) ACCF/ACR/AHA/NASCI/SAIP/SCAI/SCCT 2010 Expert consensus document on coronary computed tomographic angiography: a report of the American College of Cardiology Foundation Task Force on Expert Consensus Documents. Circulation 121:2509–2543
Ehara M, Surmely JF, Kawai M et al (2006) Diagnostic accuracy of 64-slice computed tomography for detecting angiographically significant coronary artery stenosis in an unselected consecutive patient population: comparison with conventional invasive angiography. Circ J 70:564–571
Austen WG, Edwards JE, Frye RL et al (1975) A reporting system on patients evaluated for coronary artery disease. Report of the Ad Hoc Committee for Grading of Coronary Artery Disease, Council on Cardiovascular Surgery, American Heart Association. Circulation 51:5–40
Tanabe K, Serruys PW, Grube E et al (2003) TAXUS III Trial: in-stent restenosis treated with stent-based delivery of paclitaxel incorporated in a slow-release polymer formulation. Circulation 107:559–564
Fajadet J, Morice MC, Bode C et al (2005) Maintenance of long-term clinical benefit with sirolimus-eluting coronary stents: three-year results of the RAVEL trial. Circulation 111:1040–1044
Ehara M, Kawai M, Surmely JF et al (2007) Diagnostic accuracy of coronary in-stent restenosis using 64-slice computed tomography: comparison with invasive coronary angiography. JACC 49:951–959
Rist C, von Ziegler F, Nikolaou K et al (2006) Assessment of coronary artery stent patency and restenosis using 64-slice computed tomography. Acad Radiol 13:1465–1473
Rixe J, Achenbach S, Ropers D et al (2006) Assessment of coronary artery stent restenosis by 64-slice multi-detector computed tomography. Eur Heart J 27:2567–2572
Cademartiri F, Schuijf JD, Pugliese F et al (2007) Usefulness of 64-slice multislice computed tomography coronary angiography to assess in-stent restenosis. J Am Coll Cardiol 49:2204–2210
Oncel D, Oncel G, Karaca M (2007) Coronary stent patency and in-stent restenosis: determination with 64-section multidetector CT coronary angiography-initial experience. Radiology 242:403–408
Gaspar T, Halon D, Lewis B et al (2005) Diagnosis of coronary in-stent restenosis with multidetector row spiral computed tomography. JACC 46:1573–1579
Maintz D, Seifarth H, Raupach R et al (2006) 64-slice multidetector coronary CT angiography: in vitro evaluation of 68 different stents. Eur Radiol 16:818–826
Maintz D, Burg MC, Seifarth H et al (2009) Update on multidetector coronary CT angiography of coronary stents: in vitro evaluation of 29 different stent types with dual-source CT. Eur Radiol 19:42–49
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, J., Li, M., Lu, Z. et al. In vivo evaluation of stent patency by 64-slice multidetector CT coronary angiography: shall we do it or not?. Int J Cardiovasc Imaging 28, 651–658 (2012). https://doi.org/10.1007/s10554-011-9842-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-011-9842-1