Abstract
Speckle tracking echocardiography (STE) or two-dimensional (2D) strain imaging is a novel ultrasound method to assess myocardial deformation. Peak systolic longitudinal strain (PSLS) of the basal septum (IVS) and the opposite lateral (LVFW) wall were measured in addition to standard echocardiography in 88 consecutive patients (pts) with obstructive hypertrophic cardiomyopathy (HOCM) who underwent a septal ablation procedure (PTSMA) and who were re-evaluated 12 ± 12 after months. At baseline, PSLS was substantially reduced both in basal regions. While PSLS remained unchanged in the basal IVS, i.e. the target region for PTSMA (baseline: −5.3 ± 4.1%; follow-up: −6.0 ± 4.3%; P = 0.06), it improved in the opposite LVFW (from −9.4 ± 4.7 to −12.4 ± 4.8%; P < 0.0001). Wall thickness decreased in both regions (Septum: from 20 ± 4 to 17 ± 4 mm; P < 0.0001; LV free wall: from 13 ± 2 to 12 ± 2 mm; P = 0.001). PSLS correlated significantly with wall thickness, both at baseline and at follow-up. NYHA functional class (from 2.9 ± 0.4 to 1.6 ± 0.6; P < 0.0001) and objective exercise capacity (from 96 ± 42 to 114 ± 42 W; P = 0.001) improved together with the reduction of outflow obstruction (LVOTO: from 62 ± 30 to 11 ± 19 mm Hg at rest, from 121 ± 26 to 43 ± 40 mm Hg with provocation; P < 0.0001). During the 12 months of observation, no patient had a severe adverse event. Regional myocardial deformation can be assessed quantitatively by STE. Reduction of LV afterload by elimination of the outflow gradient following a successful PTSMA with low doses of alcohol results in improvement of systolic lateral longitudinal function.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Hypertrophic cardiomyopathy (HCM) is a genetic disorder characterized by left ventricular (LV) wall hypertrophy, diastolic LV dysfunction, and myocardial disarray [1, 2]. Up to 70% of the patients additionally have manifest or latent obstruction to LV outflow (LVOTO; 3); this variant is denominated also obstructive HCM (HOCM). Patients with HOCM and symptoms limiting daily activities despite adequate medical therapy may be candidates for surgical myectomy or catheter-based septal ablation [4–19]. Both procedures aim at reducing outflow obstruction together with the distorted movement of the mitral valve (“SAM” = systolic anterior movement) that often results in obstruction-associated mitral regurgitation. Comparable short- and mid-term treatment results have been reported [2, 4, 20–22] with both interventions concerning clinical improvement, reduction of outflow obstruction, and preservation of global LV function [4–24].
Noninvasive evaluation of global and regional systolic LV function in HCM is routinely done by calculation of LV ejection fraction (EF) and visual judgement of segmental function from 2-dimensional echocardiographic images [1, 2, 25]. Quantitative ultrasound assessment of regional LV function can be performed by measuring myocardial strain or deformation [26, 27]. Usually obtained from tissue Doppler-derived velocity measurements, strain imaging has been proposed as a sensitive tool to detect early systolic function abnormalities in HCM. Recently, a novel method to measure strain from standard 2-dimensional images (speckle tracking echocardiography: STE, or 2D strain analysis) has been developed [28–33]. STE analyzes routine B-mode grey-scale loops for frame-by-frame movement of stable patterns of natural acoustic markers, or speckles, present in ultrasound images over the cardiac cycle.
The objective of the present study was to characterize regional systolic longitudinal LV function using STE in addition to standard echocardiographic assessment in patients with symptomatic HOCM before and after a septal ablation procedure (PTSMA). We hypothesized that longitudinal systolic strain was reduced already at baseline in both septum and lateral wall, that it would further deteriorate in the septal target region, and improve in the opposite lateral wall.
Patients and methods
Patients
Criteria for selection of patients to undergo septal ablation have been described previously [9, 12–14], and follow largely those established for septal myectomy [1, 2, 15–19]. Septal ablation was considered in patients with symptoms limiting daily activities (Functional class >II, exercise-induced syncope) despite adequate medical treatment, or if medical treatment was not tolerated. A substantial degree of outflow obstruction (pressure drop >50 mm Hg at rest or >100 mm Hg with provocation) was required as well as a suitable left ventricular and coronary morphology. Routine pre-interventional workup included noninvasive risk assessment with respect to the need for implantation of an automatic defibrillator [2]. Between January 2000 and December 2004, in 103 consecutive patients these inclusion criteria were met, and septal ablation was attempted. In 88 patients (86%) the procedure was completed; in 15 patients (14%) it had to be aborted, mainly because echo monitoring showed involvement of myocardium outside the septal target region [13]. Peri-and post-interventional mortality during the observation period was 0%. The baseline characteristics of our patient cohort are displayed in Table 1. All 88 patients underwent clinical and non-invasive follow-up after 12 ± 12 [3–30] months.
Septal ablation procedure
Detailed descriptions of the technique used by our group and its evolution since 1996 have been repeatedly published [6, 9, 12]. A temporary pacemaker lead was inserted in all patients. An over-the-wire balloon was introduced into the target septal branch presumed to be responsible for blood supply to the septal area involved in obstruction. The balloon was inflated, and an angiogram of the vessel performed to exclude dye reflux into the main vessel. If the target region was correctly marked by injection of the echo contrast agent (Levovist®, Schering, Berlin, Germany; 350 mg/ml), 1 ml of absolute alcohol per 1 cm of septal thickness were slowly injected under analgesic medication (5–10 mg of morphine). Ten minutes after the last alcohol injection the balloon was deflated and removed, ensuring that no alcohol backwash occured into the left anterior descending artery. A final angiogram excluded LAD damage and verified septal branch occlusion, and a final haemodynamic measurement was performed.
Standard echocardiographic measurements
Standard echocardiography examinations were performed in all patients using Vivid Five and Seven digital ultrasound system (GE Medical Systems, Horten, Norway). Two or three cardiac cycles were stored for each imaging plane in a digital format. Left ventricular and left atrial dimensions were measured according to the recommendations of the American Society for Echocardiography [25]. Left ventricular ejection fraction was measured using Simpson’s method. Diastolic LV function was evaluated by analysis of mitral Doppler inflow. The presence of an intraventricular or subaortic gradient was sought at rest and with routine provocation by a Valsalva maneuver, in selected cases by dynamic stress echocardiography. A dedicated four chamber view was archived in addition to the standard views, looking specifically at the SAM-septum contact with a frame rate > 40 Hz. Mitral regurgitation (MR) was evaluated according to the recommendations of the American Society for Echocardiography.
Speckle-tracking or 2D-strain analysis
In each patient, baseline and follow-up four-chamber views of maximum image quality, clearly showing the area of septal-mitral apposition, were retrieved from the database of our echo laboratory for further analysis. After manual tracing of the endocardial contour on an end-systolic frame, a dedicated software (GE Medical Systems, Horten, Norway) automatically tracked the contour on the other frames of that cineloop. Adequate tracking was verified in real-time and corrected by adjusting the region of interest or manually correcting the contour. With these adjustments, the 2D-strain software finally tracked the basal myocardial segments adequately in all patients. Peak systolic longitudinal strain was measured at end-systole in the basal septum with the region of mitral-septal apposition (“SAM-septal-contact”) as a reference point, and in the opposite LV free wall.
Statistics
Patient data were collected in a relational database (Filemaker 3.0, Claris Corp.) and analyzed with the Statview 5 (SAS Inc.) statistical software package. Continuous data are expressed as mean ± standard deviation after checking for normality of distribution (otherwise: median and range). Student’s t-test for paired and unpaired samples, or their non-parametric analogues (Wilcoxon signed-rank and Mann–Whitney test), or the chi2-test were used for group comparisons as appropriate. A regression analysis was performed for regional wall thickness and %strain as measured by speckle tracking. A two-tailed P value of <0.05 was considered to indicate statistical significance.
Results
Clinical and haemodynamic and echocardiographic results of septal ablation
Baseline characteristics of our patients are displayed in Table 1. On average, 5 years had elapsed since the initial diagnosis of HOCM. All but two were taking beta-blockers, Verapamil or other drugs in adequate doses, and other interventions (Myectomy, DDD-pacing) directed against outflow obstruction had been attempted in six. The mean amount of aethanol injected during the ablation session was 2.0 ± 0.4 (range: 1–3) ml resulting in a CK peak of 473 ± 184 U/l (range: 158–1,230) with an MB-fraction of 55 ± 23 U/l (Normal values: <80 and <15 U/l, resp.). PTSMA-induced atrioventricular conduction damage requiring permanent AV-sequential pacing occurred in 7% (6 pts). In-hospital mortality was 0%, follow-up was 100% complete.
At follow-up, symptoms and exercise capacity were found substantially improved. Standard echo-Doppler examination showed reduction or elimination of the LVOT gradient, reduction of left atrial size, and reduction of LV septal and free wall thickness (Table 2). Global systolic LV function a expressed by fractional shortening and ejection fraction remained within normal limits.
Regional systolic longitudinal strain
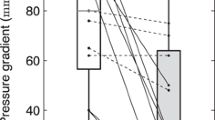
Strain data from the basal septum and the opposite LV free wall could be obtained in all patients before and after PTSMA. At baseline, and compared to normals, peak systolic longitudinal strain (PSLS) was substantially reduced in both segments analyzed (Table 2). A significant inverse correlation was found between wall thickness and PSLS (Fig. 1) both at baseline and follow-up.
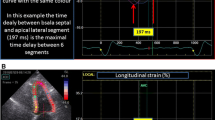
While on average PSLS remained unchanged in the basal IVS, i.e. the target region for PTSMA, 36 patients (41%, group A) showed positive strain values (i.e. systolic lengthening or “stretching”), 52 (49%, group B) had maintained or improved septal shortening. Group A was characterized by a better septal function at baseline (Table 3). Longitudinal shortening uniformly and significantly improved in the opposite lateral wall. Figures 2 and 3 show two cases with elimination of SAM and LVOTO after PTSMA, and the strain curves derived from the basal septum and the basal free wall.
HOCM before (A, B) and after (C, D) a successful septal ablation: 2D imaging shows SAM (green arrow) at baseline (A), and absence of SAM together with septal thinning (C). Systolic longitudinal strain (B) is reduced both on the septal side (yellow curve) and on the lateral side (red). After gradient elimination, lateral strain is markedly improved, septal strain unchanged (D). (Color figure online)
Discussion
Septal ablation and myectomy for symptomatic HOCM
Manifest or latent (i.e. provocable) intraventricular gradients are found in up to 70% of patients with HCM, and seem to have a negative impact on symptoms and prognosis [1–3]. For patients with HOCM who remain symptomatic despite adequate medical treatment, septal ablation as a catheter-based alternative to surgical myectomy has emerged [2, 4–14] since 1995. Both septal reduction interventions aim at removing parts of the protruding myocardium, have a comparable peri-interventional mortality rate in experienced institutions, and effectively reduce heart failure symptoms and outflow obstruction as shown by several single-center series [7, 9–14], a multi-center registry [24], and some non-randomized comparisons [20–22]. Moreover, CMR studies after septal ablation and one echocardiographic follow-up study after myectomy [34–36] have suggested that reduction of systolic intraventricular pressure, i.e. LV afterload, seems to reduce LV free wall thickness and global LV mass, supporting the concept that even in the genetically driven hypertrophic process of HCM LV mass is afterload-sensitive.
The present data summarize our current experience with septal ablation after several modifications of the interventional protocol until 1999. Clinical and haemodynamic results are consistent with our and others’ previous reports [9–14, 20–22]. Notably, in-hospital mortality was 0% in this series. The rate of aborted procedures, however, was higher than previously reported. Standard echocardiographic measurements once again documented preservation of global LV dimensions and function, and a reduction of myocardial thickness both in the targeted septum and in remote posterolateral wall segments. Furthermore, since all echocardiographic studies during this period were archived digitally in a raw-data format, and since follow-up was 100%, we took the chance to further analyze regional deformation characteristics after a successful septal ablation.
Echocardiographic methods for the assessment of HCM
Echocardiography is the primary diagnostic tool in HCM to assess LV hypertrophy, systolic and diastolic LV function, left atrial dilatation, and outflow obstruction [1–3, 25]. Echocardiographic monitoring is successfully used for monitoring both surgical and catheter-based septal reduction interventions [9, 11, 13, 18, 35, 36]. Tissue Doppler- (TDE-) based methods allow further insight into global and regional LV mechanics [37]. Diastolic LV dysfunction appears to be uniformly present in HCM. TDE-based studies have shown that systolic LV function is also often depressed despite a normal global EF [27, 30]. However, all Doppler-based methods are angle-dependent, and tissue Doppler-based deformation imaging requires specific machine settings and a time-consuming image analysis. Speckle tracking or 2D strain imaging is a new echo technique that is able to derive quantitative deformation data from standard sector images, obviating some of the problems associated with TDE [28–33]. Furthermore, STE can be used to retrospectively analyze archived digital 2D cineloops if certain quality criteria (grey scale, frame rate) are met. STE was found to accurately measure LV deformation as compared to the reference methods of sonomicrometry, TDE, and magnetic resonance imaging [28, 31] in animals, healthy probands, and a variety of cardiac conditions. Normal values for longitudinal peak LV strain around −20% have been reported, with an increase from base to apex.
In all our patients regional LV deformation parameters from basal IVS and lateral wall could be obtained within a reasonable analysis time, somewhat different from previous publications with around 5% of LV segments being rejected by the analysis software [29]. The thickened, rather echogenic basal LV walls in our HOCM cohort may have facilitated this result. At baseline, and compared to the normal values reported, longitudinal strain was severely reduced in our patients both in the septal and lateral wall. This finding is consistent with the results of Serri [30] and coworkers: In a series of 26 patients with HNCM analyzed with 2D strain they found global longitudinal strain to be reduced to −15%, with the basal segments, and especially the basal septum, most severely affected (to −9%). Kato and colleagues described a negative correlation between TDE-derived strain values and haemodynamic parameters [27]. As compared to these studies, our patients were older and had a more pronounced LA dilatation, findings which may explain their even more pronounced reduction of basal longitudinal LV strain.
At follow-up, together with relief from obstruction and a slight regression of wall thickness of the respective segments, peak systolic longitudinal strain significantly improved in the basal free wall. The correlation between wall thickness and longitudinal strain was still present. This improvement was consistent with our expectations and with results in other situations of systolic LV unloading, e.g. aortic valve replacement for aortic stenosis [38]. The findings with respect to the basal septum are more difficult to interpret. Probably, creating a limited necrosis [23, 37] by injecting a rather low amount of aethanol in a region that is relatively stiff already at baseline does not create much of additional, measurable dysfunction. In the subgroup of patients, however, who had better septal function at baseline, systolic “stretching” of the ablation region tended to develop until follow-up (Fig. 3). Interestingly, Van Dockum et al. [35] saw virtually identical results in their CMR study using tissue tagging in 9 patients at baseline and 6 months after septal ablation: Longitudinal septal shortening did not change (from 7.6 ± 4.4 to 8.7 ± 3.8%; P = 0.17) while the lateral wall improved (from 11.2 ± 3.3 to 13.5 ± 4.5%: P = 0.04). Reduction of regional wall thickness by 2 mm (septum) and 1 mm (lateral wall) was also noted. The authors concluded that a beneficial structural and functional LV remodeling process occured in their patients after elimination of outflow obstruction. Our present data seem to support this conclusion; however, the subgroup with septal “stretching” needs a close long-term follow-up.
Limitations
Limitations of this report arise for one from the retrospective nature of our study. Whether a baseline or follow-up study was analyzed, was clearly visible from the time markers and the presence or absence of the septal ablation lesion. Since our analysis was limited to only 2 out of 16 LV wall segments and to longitudinal deformation, the effect of outflow gradient reduction on strain measurements in other LV regions and other dimensions of myocardial deformation (i.e. radial and circumferential), or on global systolic LV strain, remains to be assessed. Finally, changes in medication may have interfered with our observation.
Conclusion
Speckle tracking echo imaging is a promising new tool to analyze regional systolic function in hypertrophic obstructive cardiomyopathy. A negative correlation seems to exist between regional wall segment thickness and longitudinal systolic function, with the septum already most severely compromised at baseline. Reduction of LV afterload by elimination of the outflow gradient following a successful septal ablation results in regression of lateral wall thickness and in improvement of systolic lateral shortening. Furthermore, the reported findings support the concept of afterload dependency of myocardial mass and function in obstructive HCM. The prospective use of speckle tracking in larger patient groups and different clinical settings, and a comparison against CMR tissue tagging, is warranted.
References
Wigle DE, Rakowski H, Kimball BP, Williams WG (1995) Hypertrophic cardiomyopathy: clinical spectrum and treatment. Circulation 92:1680–1692
Maron BJ, McKenna WJ, Danielson GK, Kappenberger LJ, Kuhn HJ, Seidman CE, Shah PM, Spencer WH III, Spirito P, Ten Cate FJ, Wigle ED, Vogel RA, Abrams J, Bates ER, Brodie BR, Danias PG, Gregoratos G, Hlatky MA, Hochman JS, Kaul S, Lichtenberg RC, Lindner JR, O’Rourke RA, Pohost GM, Schofield RS, Tracy CM, Winters WL Jr, Klein WW, Priori SG, Alonso-Garcia A, Blomstrom-Lundqvist C, De Backer G, Deckers J, Flather M, Hradec J, Oto A, Parkhomenko A, Silber S, Torbicki A (2003) American College of Cardiology/European Society of Cardiology clinical expert consensus document on hypertrophic cardiomyopathy. A report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus Documents and the European Society of Cardiology Committee for Practice Guidelines. J Am Coll Cardiol 42:1687–1713
Maron MS, Olivotto I, Zenovich AG, Link MS, Pandian NG, Kuvin JT, Cecchi F, Udelson JE, Maon BJ (2006) Hypertrophic cardiomyopathy is predominantly a disease of left ventricular outflow tract obstruction. Circulation 114:2232–2239
Sigwart U (1995) Non-surgical myocardial reduction for hypertrophic obstructive cardiomyopathy. Lancet 346:211–214
Kimmelstiel CD, Maron BJ (2004) Role of percutaneous septal ablation in hypertrophic obstructive cardiomyopathy. Circulation 109:452–456
Knight CJ, Kurbaan AS, Seggewiss H, Henein M, Gunning M, Harrington D, Fassbender D, Gleichmann U, Sigwart U (1997) Non-surgical septal reduction for hypertrophic obstructive cardiomyopathy: outcome in the first series of patients. Circulation 95:2075–2081
Kuhn H, Gietzen F, Leuner C, Gerenkamp T (1997) Induction of subaortic septal ischemia to reduce obstruction in hypertrophic obstructive cardiomyopathy. Eur Heart J 18:846–851
Braunwald E (1997) Induced septal infarction: a new strategy for hypertrophic obstructive cardiomyopathy. Circulation 95:1981
Faber L, Seggewiss H, Gleichmann U (1998) Percutaneous transluminal septal myocardial ablation in hypertrophic obstructive cardiomyopathy: results with respect to intra-procedural myocardial contrast echocardiography. Circulation 98:2415–2421
Flores-Ramirez R, Lakkis NM, Middleton KJ, Killip D, Spencer WH 3rd, Nagueh SF (2001) Echocardiographic insights into the mechanisms of relief of left ventricular outflow tract obstruction after nonsurgical septal reduction therapy in patients with hypertrophic obstructive cardiomyopathy. J Am Coll Cardiol 37:208–214
Lakkis NM, Nagueh SF, Kleiman NS, Killip DM, He ZX, Verani M, Roberts R, Spencer WH III (1998) Echocardiography-guided ethanol septal reduction for hypertrophic obstructive cardiomyopathy. Circulation 98:1750–1755
Seggewiss H, Gleichmann U, Faber L, Fassbender D, Schmidt HK, Strick S (1998) Percutaneous transluminal septal myocardial ablation in hypertrophic obstructive cardiomyopathy: acute results and 3-months follow-up in 25 patients. J Am Coll Cardiol 31:252–258
Faber L, Seggewiss H, Ziemssen P (2000) Targeting percutaneous transluminal septal ablation for HOCM by intra-procedural echocardiographic monitoring. J Am Soc Echocardiogr 13:1074–1079
Faber L, Meissner A, Ziemssen P, Seggewiss H (2000) Percutaneous transluminal septal myocardial ablation for HOCM: long-term follow-up in the first series of 25 patients. Heart 83:326–331
Heric B, Lytle BW, Miller OP (1995) Surgical management of hypertrophic obstructive cardiomyopathy. J Thor Cardiovasc Surg 110:195–208
Schulte HD, Bircks W, Lösse B (1987) Techniques and complications of transaortic subvalvular myectomy in patients with hypertrophic obstructive cardiomyopathy (HOCM). Z Kardiol 76(suppl 3):145–151
Robbins RC, Stinson EB, Daily PO (1996) Long-term results of left ventricular myotomy and myectomy for obstructive hypertrophic cardiomyopathy. J Thorac Cardiovasc Surg 111:586–594
Ralph-Edwards A, Woo A, McCrindle BW, Shapero JL, Schwartz L, Rakowski H, Wigle DE, Williams WG (2005) Hypertrophic obstructive cardiomyopathy: comparison of outcomes after myectomy or alcohol ablation adjusted by propensity score. J Thorac Cardiovasc Surg 129:351–358
Minami K, Boethig D, Woltersdorf H, Seifert D, Koerfer R (2002) Long-term follow-up of surgical treatment of HOCM. Eur J Cardiothorac Surg 22:206–210
Nagueh SF, Ommen SR, Lakkis NM, Killip D, Zoghbi WA, Schaff HV, Danielson GK, Quinones MA, Tajik JA, Spencer WH (2001) Comparison of ethanol septal reduction therapy with surgical myectomy for the treatment of hypertrophic obstructive cardiomyopathy. J Am Coll Cardiol 38:1707–1710
Qin JX, Shiota T, Lever HM, Kapadia SR, Sitges M, Rubin DN, Bauer F, Greenberg NL, Agler DA, Drinko JK, Martin M, Tuzcu EM, Smedira NG, Lytle B, Thomas JD (2001) Outcome of patients with hypertrophic obstructive cardiomyopathy after percutaneous transluminal septal myocardial ablation and septal myectomy surgery. J Am Coll Cardiol 38:1994–2000
Qin JX, Shiota T, Lever HM, Asher CR, Popovic ZB, Greenberg NL, Agler DA, Drinko JK, Smedira NG, Tuzcu EM, Lytle BW, Thomas JD (2004) Conduction system abnormalities in patients with obstructive hypertrophic cardiomyopathy following septal reduction interventions. Am J Cardiol 93:171–175
Veselka J, Duchonova R, Prochazkova S, Palenickova J, Sorajja P, Tesar D (2005) Effects of varying dosing in percutaneous septal ablation for obstructive HCM on early hemodynamic changes. Am J Cardiol 95:675–678
Faber L, Seggewiss H, Gietzen FH, Kuhn H, Boekstegers P, Neuhaus L, Seipel L, Horstkotte D (2005) Catheter-based septal ablation for symptomatic hypertrophic obstructive cardiomyopathy: follow-up results of the “TASH”-registry of the German Cardiac Society. Z Kardiol 94:516–523
Cheitlin MD, For the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (ACC/AHA/ASE Committee to Update the 1997 Guidelines for the Clinical Application of Echocardiography) (2003) ACC/AHA/ASE 2003 guideline update for the clinical application of echocardiography. J Am Soc Echocardiogr 16:1091–1110
Yang H, Sun JP, Lever HM et al (2003) Use of strain imaging in detecting segmental dysfunction in patients with hypertrophic cardiomyopathy. J Am Soc Echocardiogr 16:233–239
Kato TS, Noda A, Izawa H et al (2004) Discrimination of nonobstructive hypertrophic cardiomyopathy from hypertensive left ventricular hypertrophy on the basis of strain rate imaging by tissue Doppler ultrasonography. Circulation 110:3808–3814
Amundsen BH, Helle-Valle T, Edvardsen T, Torp H, Crosby J, Lyseggen E, Stoylen A, Ihlen H, Lima JA, Smiseth OA, Slordahl SA (2006) Noninvasive myocardial strain measurement by speckle tracking echocardiography: validation against sonomicrometry and tagged magnetic resonance imaging. J Am Coll Cardiol 47:789–793
Leitman M, Lysyansky P, Sidenko S et al (2004) Two-dimensional strain—a novel software for real-time quantitative echocardiographic assessment of myocardial function. J Am Soc Echocardiogr 17:1021–1029
Serri K, Reant P, Lafitte M, Berhouet M, Le Bouffos V, Roudaut R, Lafitte S (2006) Global and regional myocardial function quantification by two-dimensional strain: application in hypertrophic cardiomyopathy. J Am Coll Cardiol 47:1175–1181
Cho GY, Chan J, Leano R, Strudwick M, Marwick TH (2006) Comparison of two-dimensional speckle and tissue velocity based strain and validation with harmonic phase magnetic resonance imaging. Am J Cardiol 97:1661–1666
Helle-Valle T, Crosby J, Edvardsen T, Lyseggen E, Amundsen BH, Smith HJ, Rosen BD, Lima JA, Torp H, Ihlen H, Smiseth OA (2005) New noninvasive method for assessment of left ventricular rotation: speckle tracking echocardiography. Circulation 112:3149–3156
Notomi Y, Lysyansky P, Setser RM, Shiota T, Popovic ZB, Martin-Miklovic MG, Weaver JA, Oryszak SJ, Greenberg NL, White RD, Thomas JD (2005) Related measurement of ventricular torsion by two-dimensional ultrasound speckle tracking imaging. J Am Coll Cardiol 45:2034–2041
Van Dockum WG, Beek AM, Ten Cate FJ, Ten Berg JM, Bondarenko O, Gotte MJ, Twisk JW, Hofman MB, Visser CA, van Rossum AC (2005) Early onset and progression of left ventricular remodeling after alcohol septal ablation in hypertrophic obstructive cardiomyopathy. Circulation 111:2503–2508
Van Dockum WG, Kuijer JPA, Goette MJW, Ten Cate FJ, Ten Berg JM, Beek AM, Twisk JWR, Marcus JT, Visser CA, van Rossum AC (2006) Septal ablation in hypertrophic cardiomyopathy improves systolic myocardial function in the lateral (free) wall: a follow-up study using CMR tissue tagging and 3D strain analysis. Eur Heart J 27:2833–2839
Deb SJ, Schaff HV, Dearani JA, Nishimura RA, Ommen SR (2004) Septal myectomy results in regression of LV hypertrophy in patients with HOCM. Ann Thorac Surg 78:2118–2122
D’Andrea A, D’Andrea L, Caso P, Scherillo M, Zeppillio P, Calabro R (2006) The usefulness of Doppler myocardial imaging in the study of the athlete’s heart and in the differential diagnosis between physiological and pathological ventricular hypertrophy. Echocardiography 23:149–157
Otto CM (2006) Valvular aortic stenosis: disease severity and timing of intervention. J Am Coll Cardiol 47:2141–2151
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Faber, L., Prinz, C., Welge, D. et al. Peak systolic longitudinal strain of the lateral left ventricular wall improves after septal ablation for symptomatic hypertrophic obstructive cardiomyopathy: a follow-up study using speckle tracking echocardiography. Int J Cardiovasc Imaging 27, 325–333 (2011). https://doi.org/10.1007/s10554-010-9678-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-010-9678-0