Abstract
Background and purpose
The pathogenesis of primary brain tumors may be related to immune response. Prior studies have shown a strong association between allergy and glioma; however, so far no significant association has been established between allergy and meningioma. Here, we conducted a meta-analysis of published studies to investigate the association of meningioma with the overall and specific allergic conditions.
Methods
PubMed, EMBASE database, and Cochrane Library were searched for pertinent citations published between January 1979 and October 2009. We used the following searching strategy (brain tumor [Text Word] OR meningioma [Text Word]) AND (allergy [Text Word] OR atopy [Text Word] OR asthma [Text Word] OR eczema [Text Word] OR hay fever [Text Word]) to search for relevant studies. Random effect model was used to estimate the association between allergic conditions and meningioma.
Result
A total of 7 studies (5 case–control studies and 2 cohort studies) were included in the analysis, involving 54,391 participants, of whom 2,656 had meningioma. When compared to non-allergic conditions, the pooled odds ratio (OR) with any allergic conditions for meningioma was 0.91 (95% CI: 0.79–1.04, p = 0.163), suggesting that there is no significant association between meningioma and allergic conditions. Interestingly, further analysis showed that eczema had significantly inverse association with meningioma (OR, 0.75; 95% CI: 0.65–0.87, p < 0.05). On the other hand, no significant association was found for asthma (OR, 0.88; 95% CI: 0.75–1.04, p = 0.126) and hay fever (OR, 0.90; 95% CI: 0.79–1.03, p = 0.137).
Conclusion
In conclusion, there was a significantly negative association between eczema and meningioma, suggesting that eczema may reduce the risk of meningioma. Although it was not statistically significant association between meningioma and other specific allergic conditions such as asthma and hay fever, further studies with large sample size may be needed to determine its relationship.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Meningioma is the secondly most common primary neoplasm of the central nervous system, constituting between 13 and 26% of all intracranial tumors [1]. The incidence of meningioma is rising with age, and the peak comes at sixth and seventh decade of life. In contrast to glioma, meningioma affects women more commonly than men. The annual incidence per 100,000 people ranges from 2 to 7 for women and from 1 to 5 for men [2]. A magnetic resonance imaging study conducted among general population reported a prevalence of meningioma to be 0.9% [3]. In an autopsy series, the incidence of meningioma was 1.4% [4], due to the inclusion of asymptomatic meningioma during life time. Meningiomas arise from the arachnoid cap cells embedded in the arachnoid villi, at any site, most commonly the skull vault and the skull base. Although meningiomas usually exhibit benign histological features, atypical or anaplastic tumors can be found in 6% of cases.
According to early studies, risk factors for meningiomas may include hereditary syndromes, high-dose radiation, woman vs man sex, exogenous hormones, and so on [1, 5–9]. The association between allergy and meningioma has been investigated by many studies [10–17]. So far, it has not been conclusive. In a previous metal-analysis study on atopy and the risk of brain tumors performed in 2007, no significant relationship was found between atopy and meningioma (RR, 0.93; 95% CI: 0.72–1.19); however, the study was limited by heterogeneous reporting on meningioma and allergic conditions among included studies [18].
Gliomas, as the most common primary neoplasm of the central nervous system, are strongly related to allergy. Recently, our meta-analysis [19] including 10 case–control and 2 cohort studies showed a significantly inverse association between the risk of glioma and allergy, with the pooled odds ratio (OR) of 0.60 (95% CI: 0.52–0.69, p < 0.001). In addition, this study reported that significant risk reduction was observed in several specific allergic conditions including asthma (OR = 0.70, 95% CI: 0.62–0.79, p < 0.001) as well as eczema (OR = 0.69, 95% CI: 0.62–0.78, p < 0.001) and hay fever (OR = 0.78, 95% CI: 0.70–0.87, p < 0.001). We hypothesized that meningioma may be similar to glioma in its association with allergic conditions. Therefore, we conducted a systematic literature review and meta-analysis of published studies to determine the association between atopic diseases including specific allergic conditions and meningioma.
Materials and methods
Literature search
We searched databases including Pub Med, EMBASE database, and Cochrane Library to identify pertinent citations published between January 1979 and October 2009. We used the following searching strategy: (brain tumor [Text Word] OR meningioma [Text Word]) AND (allergy [Text Word] OR atopy [Text Word] OR asthma [Text Word] OR eczema [Text Word] OR hay fever [Text Word]). We also reviewed the bibliographies from citations for related articles.
Selection criteria and process
We have selected studies based on the following criteria: (1) case–control or cohort studies investigating the association between allergic conditions and meningioma; (2) cases were medically confirmed of meningioma; (3) reported outcome measures with odds ratio (OR) or relative risk (RR) with 95% confidence interval. In cases where there were more than one published study on the same population of patients, the most recent article was selected for analysis. All the potentially relevant papers were reviewed and extracted independently by two investigators (Mei Wang and Chao Chen), and disagreements were resolved by discussion and consensus.
Assessment of methodological quality
We assessed the methodological quality of included studies based on Newcastle–Ottawa Scale (NOS) for quality of case–control and cohort studies [20]. A star system of the NOS (range, 0–9 stars) has been developed for the evaluation. The highest value for quality assessment was 9 stars (Table 1a, b).
Statistical analysis
STATA 10.1 (StataCorp, College Station, TX, USA) was used for statistical analysis. Random effect model was used to estimate the association between allergic conditions and meningioma.
Result
Identification of relevant studies
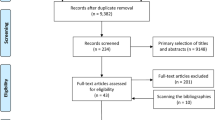
A total of 703 articles were identified. 48 duplicated articles were excluded. 646 articles that did not meet the selection criteria (1) were excluded. One paper [21] did not provide raw data, odds ratio (OR), or relative risk (RR), and it was excluded according to the selection criterion (3). Figure 1 showed a flow diagram of the selection process for relevant studies. Three studies were considered to be ineligible for inclusion in the main analysis for the following reasons: (1) one cohort study expanded the definition of exposures (included immune-related diseases and Type I diabetes) [13]; (2) two case–control studies [17, 22] were excluded as they were contained in another study [15] that was included. The data of the study conducted in the Southeast England were shared in two case–control studies. As the number of shared cases was not large (225 cases and 630 controls were shared, that was 8.7 and 8.2% among the total meningioma cases and controls), we included both the studies [11, 12].
Characteristics of studies included in the main analysis
Characteristics of the studies are presented in Table 2. A total of 7 studies [10–15] included in the meta-analysis, comprised of 5 case–control studies [10–12, 14, 15] and 2 cohort series [13]. The two cohort studies were contained in one article and included 2 different age groups of participants. The analysis of case–control studies included 10,276 participants, of whom 2,589 were meningioma cases, while the analysis of cohort studies included 44,115 participants, of whom 67 were meningioma cases. Studies were conducted in United States, Sweden, Germany, France, United Kingdom, Australia, Denmark, Finland, and Norway. The overall quality of the included studies was adequate. The mean value was 6.8 out of 9 stars (Table 1a, b).
Overall association of allergic conditions with the risk of meningioma
An assessment of heterogeneity of 7 studies [10–15] included for the analysis indicated that the hypothesis of homogeneity could be accepted (I 2 = 31.3%, p = 0.189). And we used random effect model to calculate the summary odds ratio. The odds ratios ranged from 0.76 to 2.44 among these studies. In the pooled analysis (Fig. 2), the summary odds ratio associated with allergic conditions in comparison with non-allergic conditions was 0.91 (95% CI: 0.79–1.04, p = 0.163), suggesting that there was no significant association between meningioma and allergic conditions.
Overall association between allergic conditions and the risk of meningioma. The summary odds ratios were calculated using a random effects model for case–control, cohort, and overall studies. The odds ratio and 95% CI for each study and the final combined results are displayed numerically on the right and graphically as a forest plot on the left. Filled square odds ratio for individual study. Line 95% CI, filled diamond combined result
Subgroup analysis showed that the estimated odds ratios were 0.89 (95% CI: 0.81–0.99, p = 0.030) in the case–control studies [10–12, 14, 15] and 1.40 (95% CI: 0.49–3.98, p = 0.528) in the cohort studies [13]. Two studies [11, 12] contained the data of a study conducted in the Southeast England, and the pooled analysis after excluding either of them did not change the result substantially (excluded Schoemaker, OR = 0.95, 95% CI: 0.83–1.08, p = 0.425; excluded Wigertz A, OR = 0.90, 95% CI: 0.75–1.08, p = 0.268). These results indicated that there is no significant relationship between allergy and meningioma.
Specific allergic conditions and meningioma
Specific allergic conditions and the risk of meningioma were also investigated, which included 5 studies [10–12, 14, 15] for asthma, 6 studies [10–15] for eczema, and 5 studies [10–14] for hay fever, respectively. No statistically significant heterogeneity was detected among those studies for asthma (Chi2 = 0.62, I 2 = 0.0%, p = 0.961), eczema (Chi2 = 0.77, I 2 = 0.0%, p = 0.979), or hay fever (Chi2 = 0.95, I 2 = 0.0%, p = 0.917).
Asthma
For asthma, five case–control studies included 10,285 participants, of whom 2,592 were meningioma cases [10–12, 14, 15]. The pooled OR was 0.88 (95% CI: 0.75–1.04, p = 0.126). No significant association was detected (Fig. 3a).
Association of specific allergic conditions with the risk of meningioma. The summary odds ratios were calculated using a random effects model for asthma (a), eczema (b), and hay fever (c) separately. The odds ratio and 95% CI for each study and the final combined results are displayed numerically on the right and graphically as a forest plot on the left. Filled square odds ratio for individual study. Line 95% CI, filled diamond combined result
Eczema
Six studies included in the analysis of eczema comprised of one cohort study and five case–control studies [10–15]. A total of 24,820 participants was included, of whom 2,633 were meningioma cases. The pooled OR was 0.75 (95% CI: 0.65–0.87, p < 0.05), suggesting a significant inverse association of eczema and the occurrence of meningioma (Fig. 3b).
Hay fever
No significant association was detected for hay fever. The analysis included one cohort study and four case–control studies [10–14]. A total of 23,364 participants was included, of whom 2,300 were meningioma cases. The pooled OR was 0.90 (95% CI: 0.79–1.03, p = 0.137; Fig. 3c).
Publication bias
The funnel plot of the included studies was largely symmetric (Fig. 4). These results indicated a possibility that publication bias may have played a role in the observed effect, but it was unlikely to explain the full magnitude of the association (Begg’s test: p = 0.293, Egger’s test: p = 0.426).
Discussion
We have shown a non-significant association between overall allergic condition and meningioma based on 5 case–control studies and 2 cohort studies [10–15]; this result is consistent with a previous meta-analysis results (RR = 0.93, 95% CI = 0.72–1.19) [18]. Furthermore, our study showed non-significant association for certain specific allergic conditions such as asthma (OR = 0.88, 95% CI: 0.75–1.04) and hay fever (OR = 0.90, 95% CI: 0.79–1.03). Most importantly, our study has demonstrated for the first time a significantly inverse association (OR = 0.75, 95% CI: 0.65–0.87, p < 0.05) between the particular allergic condition eczema and meningioma, suggesting that eczema may negatively influence the development of meningioma, in a fashion similar to the effect of allergy on glioma.
Cohort study offers prospectively a relatively accurate and clear definition of the temporal relation between exposure and disease. However, it may be difficult to recruit adequate cases because meningioma is a rare disease, and the cohort may be limited by the selection bias. The two cohort studies [13] included in our analysis all consisted of individuals from the Swedish Twin Registry birth cohort of same-gender twins. One cohort study consisted twins born between 1886 and 1925, and all participants received a questionnaire mailed in 1967; 14,535 answered all questions relating to allergic conditions (69.85% response rate) [13]. These participants were followed from 1968 until death, loss to follow-up, diagnosis of first glioma or meningioma, or 2000, whichever occurred first, resulting in 41 patients diagnosed with meningioma. In another cohort study of twins born between 1926 and 1958, the questionnaires were mailed to individuals in 1973. 29,573 answered the question concerning allergic conditions (80.94% response rate) [13]. These participants were followed from 1974 until death, loss to follow-up, diagnosis of first glioma or meningioma, or 2000, whichever occurred first, resulting in 26 patients developed meningioma. The incidence of meningioma in the first cohort study is much higher than the second one, most likely because of the longer follow-up time and the older average age.
The recall bias may be substantial in case–control study, because the disease may influence cases’ physical and mental state and affects. And these studies can not be performed in a blinded fashion. Four of the five case–control studies involved in our research are population based [10–12, 15], and only one study is hospital based [14]. Given the consistency of results from case–control studies of allergies and meningioma in population-based and hospital-based studies, the selection and reporting bias of the hospital-based study seems to be minimal. In all of these case–control studies, the majority of interviews were conducted by trained person using a computer-assisted interview program. Only a small part of the data was collected by questionnaire or telephone [11, 12, 15]. Two case–control studies only included allergic diseases occurring at least 2 years before diagnosis of the brain tumor or before the interview for controls [10, 15]. In another study, meningioma risk in relation to allergies was evaluated for allergies diagnosed at least 1 year prior to the first diagnosis with meningioma, and for controls, a censoring date was based on the year when interviews were conducted [12]. The last two studies recorded the detailed age or year diagnosed with the condition of interest [11, 14].
The pathophysiological mechanism for the inverse association between allergic diseases and meningioma is unknown [9]. Genetic influence on immune reactions may play a considerable role in the association between allergic conditions and brain tumor. Preetha et al. reported single nucleotide polymorphisms (SNPs) in innate immune genes and their surrounding regions were significantly different from meningiomas to non-neoplastic controls [23]. Sixty-eight SNPs in 36 genetic regions were significantly associated with the risk of meningioma at p < 0.05, and 17 SNPs in 12 genetic regions were associated p < 0.01. After correcting for multiple testing, gene region–based tests reported seven regions as statistically significant: TNFRSF18, NFKB1, FCER1G, CD14, C1D, CCR6, and VCAM1. NFKB1 and FCER1G were given the relationship between T-cell regulation and chronic inflammation, and IgE and allergic diseases [24, 25]. Functional polymorphisms in FCER1A, which also coded for a subunit of the IgE receptor, were strongly associated with serum IgE levels in a genome-wide association study [25]. Compared with a previous examination of the same SNPs for glioma risk, NFKB1 was significant in both studies (p < 0.01). However, gene region–based hits (p < 0.05) differed between glioma (ALOX5, SELP, and SOD) and meningioma (TNFSRF18, FCER1G, VCAM1, NFKB1, CD14, C1D, and CCR6) [26].
Tumor immune response is mainly based on cellular mechanisms that are controlled by Th1-CD4-positive cells. These cells enhance cytotoxic T cell and natural killer cell activation through interferon and suppress T-helper 2 (Th2)-CD4-positive T cells, which are critical for enhancing allergic phenotypes [27]. In our study, eczema shows a significantly inverse association with meningioma (OR = 0.75, 95% CI: 0.65–0.87, p < 0.05). In fact, the course of eczema or called atopic dermatitis is biphasic: an initial Th2 phase precedes a chronic phase in which Th0 cells (cells that share some activities of both Th1 and Th2 cells) and Th1 cells are predominant [28]. The Th2 cytokines IL-4, IL-5, IL10, and IL-13 predominate in the acute phase of the lesions, and in chronic lesions, there is an increase of interferon-γ, IL-12, IL-5, and GM-CSF72 [29]. Atopic dermatitis frequently starts in early infancy, and the incidence of meningioma is rising with age, and the peak comes at sixth and seventh decade. It is possible that continued chronic phase of eczema can reduce the risk of meningioma. This was supported by a previous study, in which the risk was more strongly reduced in subjects who still had eczema 1 year prior to the reference date than in those who reported their eczema had stopped prior to this time [12]. It was reported people stopped eczema more than 10 years have a significant increased risk of meningioma compared to those whom have current eczema [11].
Our studies may have the following limitations. The heterogeneity of the studies included in this meta-analysis contained the following several aspects: (1) The study designs were different. While five studies were case–control studies, two were cohort studies [10–15]. (2) The studies used different methods to collect information of the participants’ history of allergic conditions. Most interviews were conducted by trained person or through the use of a computer-assisted program. Other studies were done through questionnaire and telephone [11, 12, 15]. With different methods, the participants may have different attitude and may not understand the questions in an identical way. All these would affect the accuracy of the data collection. (3) The definition of exposure varied among these studies. Mostly, the exposure included asthma and eczema. In several studies, allergies to food, medicine, insects, pollens, and chemicals were involved in the exposure [14]. Some studies did not explain the definition of exposure clearly. Different definition of exposure may lead to different incidence. (iv) The questions offered to participants are different. In some studies, measurement of allergic conditions was based on several basic questions concerning the respondent ever or never had the interested conditions [13, 14]; and in other studies, additional questions about the age of first diagnosis, the duration, current or past disease, and comorbidities [10–12]. The prevalence of allergic conditions in Schoemaker’s study [12] was 46.7% in case groups and 46.1% in control groups, while the prevalence in Schlehofer’s study [15] was 26.8% and 28.4% for cases and controls, respectively, suggesting a variation in exposure. Publication bias is another major problem. It arises from the design or execution of single study or from the tendency of journals to reject negative studies. Although we include all of the studies that meet our criteria, publication bias is hard to avoid because that no unpublished studies are included, and we may reach misleading conclusion. In our research, publication bias about eczema and meningioma may affect the meta-analysis results, potentially producing overstated conclusion.
In summary, our study demonstrated that eczema is significantly associated with the occurrence of meningioma. The results may suggest a unique role of eczema in the pathogenesis of meningioma. Prospective cohort studies with adequate numbers of cases are needed to further confirm the interesting association. Although it was not statistically significant association between meningioma and other specific allergic conditions such as asthma and hay fever, further studies with large sample size may be needed to determine its relationship. Measurements of biomarkers may provide biological bases for the role of immune function in meningioma development.
References
Bondy M, Ligon BL (1996) Epidemiology and etiology of intracranial meningiomas: a review. J Neurooncol 29:197–205
Longstreth WT Jr, Dennis LK, McGuire VM, Drangsholt MT, Koepsell TD (1993) Epidemiology of intracranial meningioma. Cancer 72:639–648
Vernooij MW, Ikram MA, Tanghe HL, Vincent AJ, Hofman A, Krestin GP, Niessen WJ et al (2007) Incidental findings on brain MRI in the general population. N Engl J Med 357:1821–1828
Rausing A, Ybo W, Stenflo J (1970) Intracranial meningioma—a population study of ten years. Acta Neurol Scand 46:102–110
Rajaraman P, Wang SS, Rothman N, Brown MM, Black PM, Fine HA et al (2007) Polymorphisms in apoptosis and cell cycle control genes and risk of brain tumors in adults. Cancer Epidemiol Biomarkers Prev 16:1655–1661
Bethke L, Murray A, Webb E, Schoemaker M, Muir K, McKinney P et al (2008) Comprehensive analysis of DNA repair gene variants and risk of meningioma. J Natl Cancer Inst 100:270–276
Sadetzki S, Flint-Richter P, Starinsky S, Novikov I, Lerman Y, Goldman B et al (2005) Genotyping of patients with sporadic and radiation-associated meningiomas. Cancer Epidemiol Biomarkers Prev 14:969–976
Wrensch M, Bondy ML, Wiencke J, Yost M (1993) Environmental risk factors for primary malignant brain tumors: a review. J Neurooncol 17:47–64
Wrensch M, Minn Y, Chew T, Bondy M, Berger MS (2002) Epidemiology of primary brain tumors: current concepts and review of the literature. Neuro Oncol 4:278–299
Berg-Beckhoff G, Schuz J, Blettner M, Munster E, Schlaefer K, Wahrendorf J et al (2009) History of allergic disease and epilepsy and risk of glioma and meningioma (INTERPHONE study group, Germany). Eur J Epidemiol 24:433–440
Wigertz A, Lonn S, Schwartzbaum J, Hall P, Auvinen A, Christensen HC et al (2007) Allergic conditions and brain tumor risk. Am J Epidemiol 166:941–950
Schoemaker MJ, Swerdlow AJ, Hepworth SJ, van Tongeren M, Muir KR, McKinney PA (2007) History of allergic disease and risk of meningioma. Am J Epidemiol 165:477–485
Schwartzbaum J, Jonsson F, Ahlbom A, Preston-Martin S, Lonn S, Soderberg KC et al (2003) Cohort studies of association between self-reported allergic conditions, immune-related diagnoses and glioma and meningioma risk. Int J Cancer 106:423–428
Brenner AV, Linet MS, Fine HA, Shapiro WR, Selker RG, Black PM et al (2002) History of allergies and autoimmune diseases and risk of brain tumors in adults. Int J Cancer 99:252–259
Schlehofer B, Blettner M, Preston-Martin S, Niehoff D, Wahrendorf J, Arslan A et al (1999) Role of medical history in brain tumour development. Results from the international adult brain tumour study. Int J Cancer 82:155–160
Wiemels J, Wrensch M, Claus EB (2010) Epidemiology and etiology of meningioma. J Neurooncol 99:307–314
Ryan P, Lee MW, North B, McMichael AJ (1992) Risk factors for tumors of the brain and meninges: results from the Adelaide Adult Brain Tumor Study. Int J Cancer 51:20–27
Linos E, Raine T, Alonso A, Michaud D (2007) Atopy and risk of brain tumors: a meta-analysis. J Natl Cancer Inst 99:1544–1550
Chen C, Xu T, Chen J, Zhou J, Yan Y, Lu Y et al (2010) Allergy and risk of glioma: a meta-analysis. Eur J Neurol 18:387–395
Wells GA, Shea B, O’Connell D et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. http://www.ohri.ca/programs/clinical_epidemiology/oxford.htm
Schwartzbaum J, Ahlbom A, Malmer B, Lonn S, Brookes AJ, Doss H et al (2005) Polymorphisms associated with asthma are inversely related to glioblastoma multiforme. Cancer Res 65:6459–6465
Cicuttini FM, Hurley SF, Forbes A, Donnan GA, Salzberg M, Giles GG et al (1997) Association of adult glioma with medical conditions, family and reproductive history. Int J Cancer 71:203–207
Rajaraman P, Brenner AV, Neta G, Pfeiffer R, Wang SS, Yeager M et al (2010) Risk of meningioma and common variation in genes related to innate immunity. Cancer Epidemiol Biomarkers Prev 19:1356–1361
Chang M, Lee AJ, Fitzpatrick L, Zhang M, Sun SC (2009) NF-kappa B1 p105 regulates T cell homeostasis and prevents chronic inflammation. J Immunol 182:3131–3138
Weidinger S, Gieger C, Rodriguez E, Baurecht H, Mempel M, Klopp N et al (2008) Genome-wide scan on total serum IgE levels identifies FCER1A as novel susceptibility locus. PLoS Genet 4:e1000166
Rajaraman P, Brenner AV, Butler MA, Wang SS, Pfeiffer RM, Ruder AM et al (2009) Common variation in genes related to innate immunity and risk of adult glioma. Cancer Epidemiol Biomarkers Prev 18:1651–1658
Ellyard JI, Simson L, Parish CR (2007) Th2-mediated anti-tumour immunity: friend or foe? Tissue Antigens 70:1–11
Grewe M, Walther S, Gyufko K, Czech W, Schopf E, Krutmann J (1995) Analysis of the cytokine pattern expressed in situ in inhalant allergen patch test reactions of atopic dermatitis patients. J Invest Dermatol 105:407–410
Taha RA, Leung DY, Ghaffar O, Boguniewicz M, Hamid Q (1998) In vivo expression of cytokine receptor mRNA in atopic dermatitis. J Allergy Clin Immunol 102:245–250
Author information
Authors and Affiliations
Corresponding author
Additional information
Mei Wang and Chao Chen contributed equally to the work.
Rights and permissions
About this article
Cite this article
Wang, M., Chen, C., Qu, J. et al. Inverse association between eczema and meningioma: a meta-analysis. Cancer Causes Control 22, 1355–1363 (2011). https://doi.org/10.1007/s10552-011-9808-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10552-011-9808-6