Abstract
Objective
We conducted a systematic review of studies addressing the relation between cigarette smoking and gastric cancer to estimate the magnitude of the association for different levels of exposure and cancer locations.
Methods
Published cohort, case–cohort, and nested case–control studies were identified through PubMed, Scopus, and Web of Science searches, from inception to July 2007. Relative risk (RR) estimates referring to the comparison of two categories of exposure (e.g., current smokers vs. never smokers) were combined using a random effects model. Generalized least squares regression was used for trend estimation. Heterogeneity was quantified using the I 2 statistic.
Results
Forty-two articles were considered for the systematic review. Comparing current smokers with never smokers: the summary RR estimates were 1.62 in males (95% CI: 1.50–1.75; I 2 = 46.0%; 18 studies) and 1.20 in females (95% CI: 1.01–1.43; I 2 = 49.8%; nine studies); the RR increased from 1.3 for the lowest consumptions to 1.7 for the smoking of approximately 30 cigarettes per day in the trend estimation analysis; smoking was significantly associated with both cardia (RR = 1.87; 95% CI: 1.31–2.67; I 2 = 73.2%; nine studies) and non-cardia (RR = 1.60; 95% CI: 1.41–1.80; I 2 = 18.9%; nine studies) cancers.
Conclusion
Our study provides solid evidence to classify smoking as the most important behavioral risk factor for gastric cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Tobacco smoking was recently included in the list of environmental factors that increase the risk of gastric cancer [1, 2], after low fruit and vegetables intake, high salt consumption [3, 4] and H. pylori infection [5].
A meta-analysis was published in 1997 [1], showing a 44% increase in the risk of gastric cancer among ever smokers compared to never smokers. The association was stronger in males and the summary RR weighted on the inverse of the variance from three cohort studies was 2.55, compared to 1.34 from 17 case–control studies. In the last decade, a large number of cohort, case–cohort, and nested case–control analyses became available, allowing a more accurate quantification of the association between smoking and gastric cancer and, in some cases, according to cancer location and histological type. A systematic review and meta-analysis published in 2006 [6] showed a significant 79% and 22% increased risk of gastric cancer in male and female smokers, respectively, but it only included 10 cohort studies conducted in Japanese populations. The evidence for a dose–response relationship has not been systematically explored and quantified [1, 6], and the role of tobacco consumption in the occurrence of stomach cancers of distinct topographic and histological subtypes remains unclear.
To update the previous meta-analyses, we conducted a systematic review of cohort studies addressing the relation between cigarette smoking and gastric cancer, including a larger number of studies and estimating the magnitude of the association for different levels of exposure and cancer locations.
Methods
Literature search and selection of studies for the systematic review
Published cohort, case–cohort, and nested case–control studies presenting results on the association between smoking and gastric cancer were identified through PubMed (http://www.ncbi.nlm.nih.gov/entrez/), Scopus (http://www.scopus.com), and Web of Science® (http://portal.isiknowledge.com) searches, from inception to July 2007, under the expression [(smoke OR smoking OR nicotine OR tobacco OR lifestyle OR lifestyles OR cigarette OR diet OR alcohol) AND (gastric cancer OR stomach cancer OR cardia cancer)]. Articles in English, French, Italian, Spanish or Portuguese, published as a full paper or letter to the editor, were considered in the review.
Two researchers from each of two independent groups of reviewers (group 1: RLL, TPT, RSP; group 2: AKP, AN, IP) independently screened the list of references (full articles were retrieved for further analysis whenever necessary) to exclude reports not referring to cohort investigations or not assessing the association between cigarette smoking and gastric cancer. In the systematic review, we included all the studies evaluating the association between cigarette smoking (all measures of exposure were considered), and gastric cancer occurrence or mortality (all cancer locations and histological types were considered). Within each group, the disagreements between the reviewers were discussed with a third reviewer from the same group and resolved by consensus. Disagreements between the two groups of reviewers were discussed with a third researcher (NL) and resolved by consensus.
Additionally, the reference lists provided by the papers previously identified and in the two meta-analyses published in English [1, 6] addressing the association between smoking and gastric cancer were screened using the same criteria and methodology.
Data extraction
Articles included in the systematic review were reviewed independently by two researchers from each of the two independent groups of reviewers and data were extracted using a predefined questionnaire form. Discrepancies in the evaluation of the articles were resolved following the same method used in the screening of reference lists.
Each study was characterized according to: year of publication; country of origin and population evaluated; length of follow-up (when the mean or median follow-up time was not available, we present the midpoint of the follow-up period); number, age and gender of the participants; outcome (incidence of gastric cancer or mortality); number of cancer cases or deaths, number of non-cases (when cumulative incidence ratios were presented), number of person-years at risk (when incidence rate ratios were presented), number of controls (in nested case–control or case–cohort analyses); control for confounding; RR estimates (as provided in the original articles or computed for this review with the available information) for the association between different measures of smoking exposure (current smoking status, frequency of consumption, or other measures, whenever available) and gastric cancer with different locations or histological types (regardless of the cancer locations and histological type, and specifically for each location and histological type, whenever available).
Only data regarding cigarette smoking or “smoking” not further specified (assumed to represent mostly cigarette consumption), were extracted and analyzed.
When studies had results published for different follow-up times, only the articles referring to the analyses of data with the longest follow-up are described and used in meta-analysis, except when the necessary data regarding risk estimates or respective precision was available only on the studies reporting results for a shorter follow-up.
When a study provided risk estimates with different degrees of adjustment for confounders, the one adjusted for the largest number of possible confounding variables was selected. Sex-specific RR estimates were extracted whenever available.
Meta-analysis
Meta-analyses were conducted to obtain summary RR estimates for the association between cigarette smoking and gastric cancer considering different exposures, as follows: (1) current smokers vs. never smokers; (2) current smokers, highest category of exposure vs. never smokers; (3) current smokers, category closest to 20 cigarettes per day vs. never smokers; (4) former smokers vs. never smokers; (5) trend estimation. The summary RR estimates for current smokers vs. never smokers was also estimated for cardia and non-cardia cancers.
Relative risks (RRs) (cumulative incidence ratios or incidence density ratios), hazard ratios, and odds ratios were treated the same and are referred to as RRs.
When sex-specific estimates were available, they were considered separately as if obtained from different studies.
Each category of current cigarette consumption was assumed to correspond to an exposure equal to the midpoint of the respective category range. For this purpose, we assumed that the open-ended upper category had the amplitude of the preceding stratum. This information was used for trend estimation and to identify the category corresponding to the exposure closest to 20 cigarettes per day. If the midpoint of two categories was equally distant from 20, we opted for the one with a larger number of gastric cancer cases or deaths.
When both gastric cancer incidence and mortality data was provided, preference was given to incidence, unless the necessary information was available only for mortality data.
For the purpose of analysis, results referring to “cardia”, “upper third” or “proximal” stomach cancers were taken as equivalent to cancer of the gastric cardia, and “distal” stomach cancers were taken as equivalent to cancers not located in the cardia. When site-specific results were presented only according to the tumor histological type we opted for the results referring to the cancer of the intestinal type (more frequent than those of the diffuse type).
Studies that did not provide information for all cancer locations together were only included in the analysis for cardia and non-cardia cancers. Data referring to the distal stomach were also used for trend estimation, and to compute summary estimates for the highest and for the category of exposure closest to 20 cigarettes, due to the smaller number of studies available for these analyses, because distal cancers represent most of the gastric tumors. If non-cardia cancer were divided in body and antrum cancer, only antrum cancer was considered for analysis, under the assumption that it represents most of the non-cardia cancers.
Only studies presenting risk estimates considering “never smokers” or “never regular smokers” as the reference category were considered for the meta-analyses.
We used two different approaches to obtain summary RR estimates: (1) RR estimates referring to the comparison of two categories of exposure (current smokers vs. never smokers; former smokers vs. never smokers; current smokers, highest category of exposure vs. never smokers; current smokers, category closest to 20 cigarettes per day vs. never smokers) were combined using a random effects model, regardless of the homogeneity of effects across studies; (2) generalized least squares regression was used for trend estimation according to the method proposed by Greenland and Longnecker [7], considering both linear and non-linear relationships between smoking and gastric cancer. The latter can be used in studies presenting risk estimates for three or more categories of exposure, and reporting, for each level of exposure, the amount of cigarettes smoked, the RRs and respective variances, the number of cases and the number of controls for nested case–control data, or the number of cases for incidence–rate data, or the number of cases and non-cases for cumulative incidence data.
Heterogeneity was quantified using the I 2 statistic [8], and both stratified and meta-regression analyses considering the study characteristics (region, outcome, duration of follow-up, and adjustment for confounding) were used to explore heterogeneity.
Sensitivity analyses were conducted considering broader inclusion criteria for the meta-analyses (e.g., including studies in which the reference class includes former smokers or current smokers, and if the different categories of consumption include both current and former smokers; assuming the category of exposure with a higher number of subjects as equivalent to the group of “current smokers”; including in the analysis the risk estimates for distal stomach when the studies did not provide information for all cancer locations together). The dose–response relationship was also estimated using a weighted least squares regression, for trend estimation without correction for covariance of RRs, allowing the inclusion of a larger number of studies (the only information required from each category is the level of exposure, the respective RRs and their variances).
Publication bias was examined through funnel plot visual analysis, the Begg adjusted rank correlation test [9], and the Egger’s regression asymmetry test [10]. The analysis was conducted separately in studies performed in males and females (since different RR estimates are expected to differ with gender [1, 6]) and using RR estimates for current vs. never smokers. A 0.1 level of significance was used in the statistical tests to improve sensitivity of detecting publication bias.
All statistical analyses were conducted with STATA®, version 9.2. All tests of statistical significance are two sided.
Results
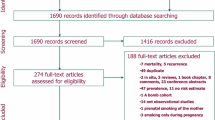
The electronic database search yielded 3,550 articles, published from 1958 to July 2007, from which 44 were considered eligible for the systematic review. The screening of reference lists yielded 10 additional reports. Twenty articles [11–30] were multiple publications of eight studies and only those [17–21, 24, 26, 30] referring to the longest follow-up periods and with more data available are presented. Therefore, 42 articles were considered for the systematic review.
Ten articles were excluded from the meta-analyses because did not provide RRs referring to the exposures eligible for meta-analysis [31–34], or its precision estimates [21, 35–39]. Thus, 32 studies (27 cohort and five nested case–control) were included in at least one of the meta-analyses (Fig. 1).
From the 42 articles included in the systematic review, 20 were from Asian countries [17, 18, 20, 26, 30, 32, 33, 37, 40–51], 12 from Europe [21, 34, 38, 52–60], and 10 from the United States [19, 24, 31, 35, 36, 39, 61–64]. Among the latter, two were conducted in participants of Asian origin [19, 63]. Concerning the outcome, 24 studies evaluated incident cases of gastric cancer [17–19, 30, 32–34, 37, 39, 41, 43–46, 52–57, 59–61, 63] and 18 evaluated gastric cancer deaths [20, 21, 24, 26, 31, 35, 36, 38, 40, 42, 47–51, 58, 62, 64]. Nine [17, 30, 43, 44, 52–54, 60, 61] studies presented results for specific anatomical locations, and two [43, 44] addressed the risk of gastric cancer of different histological subtypes. Regarding confounding control, 31 studies [17–21, 26, 30, 34–44, 46–49, 51, 53, 55, 56, 58, 61–64] provided results adjusted for age, and 11 [34, 41–43, 46, 51, 52, 54, 61–63] for socioeconomic status (using information on educational level or type of health insurance). The mean follow-up time was 8.5 years (range: 1.5−25 years).
Current smokers vs. never smokers (all stomach cancers)
For current smokers compared to never smokers, when all stomach cancers were considered, the summary RR was 1.53 (95% CI: 1.42–1.65; I 2 = 57.6%; 32 estimates from 23 studies). The sex specific summary RRs were 1.62 (95% CI: 1.50–1.75; I 2 = 46.0%; 18 studies) for males and 1.20 (95% CI: 1.01–1.43; I 2 = 49.8%; nine studies) for females (Fig. 2).
Among males, I 2 was 0% among studies in which the potential confounding effect of fruit and vegetables was controlled in analysis (RRsummary = 1.95; 95% CI: 1.68–2.25; five studies), and close to 20% in those conducted in Asian populations (RRsummary = 1.57; 95% CI: 1.47–1.68; 12 studies) (Table 1). In a meta-regression analysis including the variables sex (males, females or males and females), population (Asian, European or American), and adjustment for the confounding effect of fruit or vegetables, the τ 2 estimate was brought to 0. The summary RR estimates are independently associated with the participants’ sex, being lower in females compared with males (coefficient = −0.32, p < 0.001) but similar for studies evaluating males and females together compared to males alone (coefficient = 0.02, p = 0.740), and with the adjustments for fruit and vegetables consumption, with higher estimates in studies controlling this confounding effect (coefficient = 0.28, p < 0.001). No significant differences were observed according to the origin of the population evaluated (Europe vs. Asia: coefficient = 0.09, p = 0.244; America vs. Asia: coefficient = −0.01, p = 0.816).
Current smokers (category closest to 20 cigarettes per day) vs. never smokers (all stomach cancers)
Compared to never smokers, the consumers of approximately 20 cigarettes per day had an increased risk of gastric cancer (RRsummary = 1.53, 95% CI: 1.41–1.67; I 2 = 46.1%; 24 estimates from 21 studies), more pronounced in males (RRsummary = 1.59, 95% CI: 1.43–1.77; I 2 = 49.6%; 14 studies) than in females (RRsummary = 1.28, 95% CI: 1.11–1.47; I 2 = 0%; four studies) (Fig. 3).
In studies conducted in males, the stratification according to adjustment for the consumption of fruit and/or vegetables yielded two homogeneous groups of studies, with higher RRs observed in the three studies in which adjustments were performed (RRsummary = 1.48, 95% CI: 1.41–1.55; I 2 = 0% vs. RRsummary = 2.20, 95% CI: 1.83–2.64; I 2 = 0%) (Table 1).
Current smokers (highest category of exposure) vs. never smokers (all stomach cancers)
The overall summary RR estimate for current smokers in the highest categories of consumption compared to never smokers was 1.66 (95% CI: 1.46–1.88; I 2 = 41.8%; 25 estimates from 22 studies). The summary estimates were 1.60 (95% CI: 1.32–1.96; I 2 = 0%) for seven studies presenting results for males and females together. Considering the gender-specific estimates, the summary RRs were, respectively, 1.76 (95% CI: 1.49–2.08; I 2 = 49.2%, 14 studies) and 1.27 (95% CI: 0.70–2.31; I 2 = 63.7%, four studies) for males and females.
Former smokers vs. never smokers (all stomach cancers)
Considering never smokers as the reference category, male former smokers had a significantly increased risk of gastric cancer (RRsummary = 1.34, 95% CI: 1.22–1.47; I 2 = 35.8%; 16 studies), but no significant differences were observed in females (RRsummary = 1.16, 95% CI: 0.92–1.46; I 2 = 29.9%; eight studies) (Fig. 4).
Among males, heterogeneity was very low among the studies having incidence as the outcome (RRsummary = 1.32, 95% CI: 1.21–1.45; I 2 = 0%), conducted in Asian (RRsummary = 1.36, 95% CI: 1.27–1.46; I 2 = 0%) or European (RRsummary = 1.20, 95% CI: 0.90–1.61; I 2 = 0%) populations, or providing RR estimates adjusted for fruit and/or vegetables consumption (RRsummary = 1.55, 95% CI: 1.34–1.79; I 2 = 0%). Both the studies with mean/median follow-up time below 10 years (RRsummary = 1.39, 95% CI: 1.30–1.49; I 2 = 3.1%) and 10 or more years (RRsummary = 1.09, 95% CI: 0.95–1.25; I 2 = 0%) were homogeneous in their results, but in the latter there was a small and non statistically significant increase in the risk among the former smokers. Stratification according to the year of publication also yielded more homogeneous groups (Table 1).
Trend estimation (all stomach cancers)
The regression models including the number of cigarettes consumed per day (continuous) alone or also including, one at the time, the variables sex of the participants (males, females or males and females), study outcome (incidence, mortality), population origin (Asian, European or American), duration of follow-up (<10 years, ≥10 years), or confounding control (socioeconomic status, fruit/vegetables) showed a statistically significant association between the dose and the occurrence of gastric cancer, but heterogeneity was substantial (I 2 ≈ 75%). In the model including the number of cigarettes and year of publication (<1998, ≥1998), heterogeneity was moderate (I 2 = 30.7%). The above variables were then added to the latter model, one at the time, to achieve a better explanation of heterogeneity, including the variables significantly associated with the RR at a p < 0.10 level.
The model with the lowest heterogeneity (I 2 = 14.0%) included the number of cigarettes consumed per day (β = 0.00237, p = 0.252), sex (females vs. males: β = −0.59396, p = 0.083; males and females vs. males: β = −0.01045, p = 0.893), population origin (European vs. Asian: β = 0.29302, p = 0.076; American vs. Asian: β = 0.97569, p = 0.025), adjustment for fruit or vegetables consumption (β = 0.21393, p = 0.030), and year of publication (≥1998 vs. <1998: β = 0.36146, p < 0.001). When the square of the number of cigarettes consumed per day was added to the previous model, no substantial improvement in the homogeneity was observed (I 2 = 11.3%) and no statistical significance was achieved by neither the number of cigarettes consumed (β = 0.01122, p < 0.076) nor its square (β = −0.00020, p < 0.138).
Current smokers vs. never smokers (cardia and non-cardia cancers)
Comparing current smokers with never smokers, both cardia (RRsummary = 1.87, 95% CI: 1.31–2.67; I 2 = 73.2%; nine estimates from nine studies) and non-cardia (RRsummary = 1.60, 95% CI: 1.41–1.80; I 2 = 18.9%; nine estimates from nine studies) gastric cancers were significantly associated with smoking, despite the substantial heterogeneity observed for cardia cancer (Fig. 5).
Only two studies [54, 61] provided a clear definition of cardia cancer (ICD: C16.0), yielding a combined RR estimate of 3.14 (95% CI: 2.04–4.83; I 2 = 0%) for current smokers vs. never smokers, males and females together.
Current smokers vs. never smokers (cancers of the intestinal and diffuse histological type)
Two studies [43, 44] presented RR estimates for the association between smoking and stomach cancers of the intestinal and diffuse histological types, in males. The RRs were 1.61 (95% CI: 1.11–2.32) and 2.1 (95% CI: 1.2–3.6) for the cancer of the intestinal type, and 0.6 (95% CI: 0.3–1.1) and 2.1 (95% CI: 1.08–4.08) for the cancer of the diffuse type.
Sensitivity analysis
Relaxing the inclusion criteria to accommodate the studies otherwise not considered in the analyses, as described in the Methods section, the summary RR estimates and the magnitude of heterogeneity not explained by random variation were similar to the observed when using the original criteria. Twelve additional studies [17, 18, 32–34, 37, 45, 46, 49, 51, 59, 61] were available for the analysis of current vs. never smokers (RRsummary = 1.54, 95% CI: 1.44–1.65; I 2 = 54.9%; 44 estimates from 35 studies). RR estimates from two studies [49, 63] were added to the comparison of consumers of approximately 20 cigarettes per day vs. never smokers (RRsummary = 1.53, 95% CI: 1.41–1.66; I 2 = 43.4%; 26 estimates from 23 studies), and highest category of exposure vs. never smokers (RRsummary = 1.64, 95% CI: 1.45–1.85; I 2 = 39.6%; 27 estimates from 24 studies). One additional study [61] was considered in the meta-analysis for the comparison of former and never smokers (RRsummary = 1.30, 95% CI: 1.21–1.40; I 2 = 26.3%; 31 estimates from 24 studies).
An analysis of the dose–response relationship was conducted through weighted least squares regression, allowing the inclusion of 24 studies. Following a strategy similar to the one described above for the analysis using the method proposed by Greenland and Longnecker, a model including the variables number of cigarettes consumed per day (β = 0.01588, p < 0.001), square of the number of cigarettes consumed per day (β = −0.00024, p = 0.007), sex (females vs. males: β = −0.16690, p = 0.007; males and females vs. males: β = −0.01262, p = 0.831), outcome (mortality vs. incidence: β = 0.18378, p < 0.001), population origin (European vs. Asian: β = 0.24631, p = 0.003; American vs. Asian: β = 0.00519, p = 0.922), adjustment for fruit or vegetables consumption (β = 0.12204, p = 0.022), and year of publication (≥1998 vs. <1998: β = 0.19899, p < 0.001). The RRs predicted with this model increase from 1.3 for the lowest consumptions to 1.7 for the smoking of approximately 30 cigarettes per day, and then a slight decrease is observed to a RR of 1.5 for smokers of 50 cigarettes per day.
Funnel plots
Publication bias is suggested for the studies conducted in males both by visual inspection of the funnel plot and according to the Egger’s regression asymmetry test (p = 0.048), but not the Begg adjusted rank correlation test (p = 0.17). Regarding the studies in females, neither visual inspection nor tests of statistical significance resulted in funnel plot asymmetry (Fig. 6).
Funnel plot of cohort studies evaluating the association between smoking and gastric cancer (current smokers vs. never smokers) in males (top) and females (bottom). *Begg adjusted rank correlation test (males: p = 0.17; females: p > 0.99), Egger’s regression asymmetry test (males: p = 0.048; females: p = 0.96)
Discussion
This is the most comprehensive systematic review and meta-analysis of cohort studies addressing the association between cigarette smoking and gastric cancer. It shows that the risk of gastric cancer is increased by 60% in male smokers and 20% in female smokers, compared to never smokers, and the associations are weaker in former smokers. The trend estimation analysis suggests an increase in the RR estimates from 1.3, for the lowest consumptions, to 1.7 for the smoking of 30 cigarettes per day. The meta-analysis also supports the classification of smoking as a risk factor for gastric cancer regardless of the anatomical location of the tumor within the stomach.
Heterogeneity was moderate to high in most of the analyses conducted, but the large majority of the studies found a positive association between smoking and gastric cancer. Although the conclusion of a positive association between smoking and gastric cancer is not compromised by heterogeneity, the methodological characteristics of the studies and the populations evaluated are related to the magnitude of association. In addition to the gender differences in the summary estimates, especially for current vs. never smokers, both the subgroup and the meta-regression analyses show that studies controlling for the confounding effect of fruit or vegetables are more homogeneous and yield higher summary RR estimates. The latter can be explained by the strong association between fruit and vegetables consumption and both smoking habits [65, 66] and gastric cancer risk [4]. Studies with shorter follow-up periods yielded stronger associations between former cigarette smoking habits and cancer, suggesting that the risk of cancer is higher in subjects quitting smoking more recently (since the information on the smoking status refers to the baseline evaluation of the cohorts), and therefore that smoking plays a predominant role in the phases of carcinogenesis closer to cancer. This hypothesis is in accordance with the accumulated evidence on the role of smoking in the occurrence of gastric intestinal meta-plasia and its progression to cancer [67–69].
Only one [55] of the studies included in our meta-analysis took the effect of H. pylori infection into account in the analysis, and no conclusions are possible regarding its potential role as an effect modifier, or as an explanation for the interstudy heterogeneity. Similarly, the contribution of the interindividual difference in the bioactivation of procarcinogens and detoxification of carcinogens [70, 71] was not explored in the studies included in the analyses.
The analysis of the association between smoking and gastric cancer across different definitions of exposure and criteria for inclusion of the studies yielded consistent and robust conclusions. Our results favor a dose–response relationship, but the trend estimation following the Greenland and Longnecker method [7] failed to support such an association. However, the results from this analysis must be interpreted with caution, because it could be conducted with only 12 studies (with small variability both regarding the levels of exposure and the methodological characteristics of the studies). The method used in the sensitivity analysis is responsible for spurious precision, but receives the input from a larger number of studies and therefore is likely to yield conclusions less prone to bias. The trend estimation conducted in the sensitivity analysis supported a dose–response relationship between cigarette smoking and gastric cancer. It also provided valuable information for the understanding of the gender differences in the magnitude of the association between smoking and gastric cancer. The male–female differences were independent from the level of exposure suggesting that other factors may contribute to explain the higher summary RR estimates observed in males. On the one hand, since the proportion of gastric carcinomas of the intestinal type is higher in males, whereas the diffuse type tends to occur more frequently in females [72], if a weaker association between smoking and the gastric cancer of the diffuse type could be demonstrated, this would contribute to explain the stronger association observed in males when no distinction is made regarding the histological types of gastric cancer. On the other hand, socioeconomic status is strongly associated with both the smoking habits and the risk of gastric cancer, and the structure of confounding may differ with gender and across settings [73]. If an association between high socioeconomic status and smoking is observed more frequently, or has a higher magnitude among females [74, 75], the lack of adjustment for these factors or residual confounding may account for lower RR in studies conducted in women. Similar phenomena may justify the lower summary RR estimates for studies conducted in populations of Asian origin.
Despite these plausible explanations for the male–female differences in the magnitude of the association between smoking and gastric cancer, it should be taken into account that the results supporting this claim (both the trend estimation and the summary RR estimates for the category of exposure closest to 20 cigarettes per day vs. never smokers) are based in the observation made in only four studies. The IARC Monograph on the Carcinogenic Risks to Humans, vol 83 [2], concluded that the RRs for men and women were similar in studies evaluating adequate numbers of women.
The distinct results with different study endpoints could be explained if smoking was associated with a poorer prognosis strongly than with the occurrence of cancer. However, this hypothesis is not supported by studies evaluating the effect of smoking on the prognosis of gastric cancer, which found no significant differences in survival according to smoking status [76, 77].
The visual inspection of the funnel plots and the corresponding statistical tests for asymmetry show that negative studies are underrepresented among those conducted in males. It is unlikely that the search strategy is responsible for this bias. The systematic review included searches in PubMed and in other two important electronic databases. In addition to articles including the terms “tobacco” or “smoking”, we also reviewed reports having other topics as the main interest, identified in a broad search for articles addressing different lifestyles and through backward citation tracking. It is likely that most of the cohort studies addressing this topic have been included in the analyses since the study of a rare disease with a cohort design usually implies large investigations, likely to be published in high circulation international journals. Both publication and outcome reporting bias may have accounted for the funnel plot asymmetry, resulting in an overestimation of the association between smoking and gastric cancer in males. However, given the large number of studies available for analysis it is unlikely that those eventually missing (expectedly small studies and in low number) could have influence our main conclusions.
We observed no differences in the effect of cigarette smoking according to cancer location. It was quite clear that the harmful effect of smoking applies to any anatomical location. From a public health point of view, the benefits from tobacco control activities may be expected for all cancer sites, which are important, given the decline in cancers of the distal stomach and the increase in those located in the cardia [78]. For the cardia cancer, the heterogeneity across individual studies was very high and the true magnitude of the association remains to be clarified, even if all studies showed an increase in the risk of cardia cancer. Caution is required for the interpretation of differences in the specification of cancer subsites across studies. The classification criteria are often not described [17, 43, 53, 55], and some studies included in the group of cardia cancer the tumors located in the upper third of the stomach [30, 44]. However, the two studies [54, 61] defining cardia cancers as those pertaining to the ICD code C16.0 provided a stronger and homogeneous summary RR estimate.
Unfortunately, the small number of studies providing results for cancers of each main histological type and the heterogeneity of the results regarding the cancers of the diffuse type (one study suggests an increased risk among smokers and the other a protective effect of smoking) preclude a meta-analysis, and we cannot draw conclusions from the available evidence on this topic. Future research in gastric cancer aetiology should assess the association between environmental factors and gastric cancer according to histological type, both in males and in females.
In conclusion, the present study is the most comprehensive assessment of the association between smoking and gastric cancer, adding to previous knowledge an updated quantification of the dose–response relationship, a clearer understanding of the factors contributing to the heterogeneity in the results, and the role of smoking as a risk factor for cardia and non-cardia gastric cancer. Our results show that smoking is the behavioral factor for which the evidence regarding the association with gastric cancer is more consistent.
References
Tredaniel J, Boffetta P, Buiatti E et al (1997) Tobacco smoking and gastric cancer: review and meta-analysis. Int J Cancer 72:565–573
International Agency for Research on Cancer (2004) Tobacco smoke and involuntary smoking. In: IARC monographs on the evaluation of carcinogenic risks to humans, vol. 83. IARC, Lyon
World Cancer Research Fund (1997) Food, nutrition and the prevention of cancer: a global perspective. American Institute for Cancer Research, Washington, DC
Lunet N, Lacerda-Vieira A, Barros H (2005) Fruit and vegetables consumption and gastric cancer: a systematic review and meta-analysis of cohort studies. Nutr Cancer 53:1–10
International Agency for Research on Cancer. Schistosomes, liver flukes and Helicobacter pylori. In: IARC monographs on the evaluation of carcinogenic risks to humans, vol. 61. IARC, Lyon
Nishino Y, Inoue M, Tsuji I et al (2006) Tobacco smoking and gastric cancer risk: an evaluation based on a systematic review of epidemiologic evidence among the Japanese population. Jpn J Clin Oncol 36:800–807
Greenland S, Longnecker M (1992) Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am J Epidemiol 135:1301–1309
Higgins J, Thompson S (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21:1539–1558
Begg CB, Mazumdar M (1994) Operating characteristics of a rank correlation test for publication bias. Biometrics 50:1088–1101
Egger M, Smith GD, Schieder M, Minder C (1997) Bias in meta-analysis detected by a simple graphical test. BMJ 315:629–634
Guo W, Blot WJ, Li JY et al (1994) A nested case–control study of oesophageal and stomach cancers in the Linxian nutrition intervention trial. Int J Epidemiol 23:444–450
Kato I, Tominaga S, Ito Y et al (1992) A prospective study of atrophic gastritis and stomach cancer risk. Jpn J Cancer Res 83:1137–1142
Nomura A, Grove JS, Stemmermann GN, Severson RK (1990) A prospective study of stomach cancer and its relation to diet, cigarettes, and alcohol consumption. Cancer Res 50:627–631
Abnet CC, Qiao YL, Mark SD et al (2001) Prospective study of tooth loss and incident esophageal and gastric cancers in China. Cancer Causes Control 12:847–854
Yun YH, Jung KW, Bae JM et al (2005) Cigarette smoking and cancer incidence risk in adult men: National Health Insurance Corporation Study. Cancer Detect Prev 29:15–24
Liaw KM, Chen CJ (1998) Mortality attributable to cigarette smoking in Taiwan: a 12-year follow-up study. Tob Control 7:141–148
Tran GD, Sun XD, Abnet CC et al (2005) Prospective study of risk factors for esophageal and gastric cancers in the Linxian general population trial cohort in China. Int J Cancer 113:456–463
Inoue M, Tajima K, Kobayashi S et al (1996) Protective factor against progression from atrophic gastritis to gastric cancer—data from a cohort study in Japan. Int J Cancer 66:309–314
Nomura AM, Stemmermann GN, Chyou PH (1995) Gastric cancer among the Japanese in Hawaii. Jpn J Cancer Res 86:916–923
Wen CP, Tsai SP, Chen CJ, Cheng TY (2004) The mortality risks of smokers in Taiwan—Part I: cause-specific mortality. Prev Med 39:528–535
Doll R, Peto R, Boreham J, Sutherland I (2005) Mortality from cancer in relation to smoking: 50 years observations on British doctors. Br J Cancer 92:426–429
Doll R, Peto R (1976) Mortality in relation to smoking: 20 years’ observations on male British doctors. Br Med J 2:1525–1536
Doll R, Peto R, Wheatley K et al (1994) Mortality in relation to smoking: 40 years’ observations on male British doctors. Br Med J 309:901–911
McLaughlin JK, Hrubec Z, Blot WJ, Fraumeni JF Jr (1990) Stomach cancer and cigarette smoking among U.S. veterans, 1954–1980. Cancer Res 50:3804
Kahn HA (1966) The Dorn study of smoking and mortality among U.S. veterans: report on eight and one-half years of observation. Natl Cancer Inst Monogr 19:1–125
Mizoue T, Tokui N, Nishisaka K et al (2000) Prospective study on the relation of cigarette smoking with cancer of the liver and stomach in an endemic region. Int J Epidemiol 29:232–237
Ngoan LT, Mizoue T, Fujino Y et al (2002) Dietary factors and stomach cancer mortality. Br J Cancer 87:37–42
McLaughlin JK, Hrubec Z, Blot WJ, Fraumeni JF Jr (1995) Smoking and cancer mortality among U.S. veterans: a 26-year follow-up. Int J Cancer 60:190–193
Blair A, Walrath J, Rogot E (1985) Mortality patterns among U.S. veterans by occupation. I. Cancer. J Natl Cancer Inst 75:1039–1047
Sung NY, Choi KS, Park EC et al (2007) Smoking, alcohol and gastric cancer risk in Korean men: the National Health Insurance Corporation Study. Br J Cancer 97:700–704
Hammond EC, Horn D (1958) Smoking and death rates: report on forty-four months of follow-up of 187,783 men. 2. Death rates by cause. J Am Med Assoc 166:1294–1308
Freedman ND, Chow WH, Gao YT et al (2007) Menstrual and reproductive factors and gastric cancer risk in a large prospective study of women. Gut 56:1671–1677
Wong BC, Lam SK, Wong WM et al (2004) Helicobacter pylori eradication to prevent gastric cancer in a high-risk region of China: a randomized controlled trial. JAMA 291:187–194
Terry P, Nyren O, Yuen J (1998) Protective effect of fruits and vegetables on stomach cancer in a cohort of Swedish twins. Int J Cancer 76:35–37
Hammond EC (1966) Smoking in relation to the death rates of one million men and women. Natl Cancer Inst Monogr 19:127–204
Weir JM, Dunn JE Jr (1970) Smoking and mortality: a prospective study. Cancer 25:105–112
Yuan JM, Ross RK, Wang XL et al (1996) Morbidity and mortality in relation to cigarette smoking in Shanghai, China. A prospective male cohort study. JAMA 275:1646–1650
Carstensen JM, Pershagen G, Eklund G (1987) Mortality in relation to cigarette and pipe smoking: 16 years’ observation of 25,000 Swedish men. J Epidemiol Community Health 41:166–172
Kasum CM, Jacobs Jr DR, Nicodemus K, Folsom AR (2002) Dietary risk factors for upper aerodigestive tract cancers. Int J Cancer 99:267–272
Kurosawa M, Kikuchi S, Xu J, Inaba Y (2006) Highly salted food and mountain herbs elevate the risk for stomach cancer death in a rural area of Japan. J Gastroenterol Hepatol 21:1681–1686
Sauvaget C, Lagarde F, Nagano J et al (2005) Lifestyle factors, radiation and gastric cancer in atomic-bomb survivors (Japan). Cancer Causes Control 16:773–780
Fujino Y, Mizoue T, Tokui N et al (2005) Cigarette smoking and mortality due to stomach cancer: findings from the JACC Study. J Epidemiol 15(Suppl 2):S113–119
Koizumi Y, Tsubono Y, Nakaya N et al (2004) Cigarette smoking and the risk of gastric cancer: a pooled analysis of two prospective studies in Japan. Int J Cancer 112:1049–1055
Sasazuki S, Sasaki S, Tsugane S (2002) Cigarette smoking, alcohol consumption and subsequent gastric cancer risk by subsite and histologic type. Int J Cancer 101:560–566
Murata M, Takayama K, Choi BC, Pak AW (1996) A nested case–control study on alcohol drinking, tobacco smoking, and cancer. Cancer Detect Prev 20:557–565
Xu Z, Brown LM, Pan GW et al (1996) Cancer risks among iron and steel workers in Anshan, China, Part II: case–control studies of lung and stomach cancer. Am J Ind Med 30:7–15
Kato I, Tominaga S, Matsumoto K (1992) A prospective study of stomach cancer among a rural Japanese population: a 6-year survey. Jpn J Cancer Res 83:568–575
Akiba S, Hirayama T (1990) Cigarette smoking and cancer mortality risk in Japanese men and women—results from reanalysis of the six-prefecture cohort study data. Environ Health Perspect 87:19–26
Kono S, Ikeda M, Tokudome S et al (1987) Cigarette smoking, alcohol and cancer mortality: a cohort study of male Japanese physicians. Jpn J Cancer Res 78:1323–1328
Jee SH, Samet JM, Ohrr H et al (2004) Smoking and cancer risk in Korean men and women. Cancer Causes Control 15:341–348
Ho SY, Lam TH, Jiang CQ et al (2002) Smoking, occupational exposure and mortality in workers in Guangzhou, China. Ann Epidemiol 12:370–377
Sjodahl K, Lu Y, Nilsen TI et al (2007) Smoking and alcohol drinking in relation to risk of gastric cancer: a population-based, prospective cohort study. Int J Cancer 120:128–132
Lindblad M, Rodriguez LA, Lagergren J (2005) Body mass, tobacco and alcohol and risk of esophageal, gastric cardia, and gastric non-cardia adenocarcinoma among men and women in a nested case–control study. Cancer Causes Control 16:285–294
Gonzalez CA, Pera G, Agudo A et al (2003) Smoking and the risk of gastric cancer in the European prospective investigation into cancer and nutrition (EPIC). Int J Cancer 107:629–634
Siman JH, Forsgren A, Berglund G, Floren CH (2001) Tobacco smoking increases the risk for gastric adenocarcinoma among Helicobacter pylori-infected individuals. Scand J Gastroenterol 36:208–213
Nordlund L, Carstensen J, Pershagen G (1997) Cancer incidence in female smokers: a 26-year follow-up. Int J Cancer 73:625–628
Engeland A, Andersen A, Haldorsen T, Tretli S (1996) Smoking habits and risk of cancers other than lung cancer: 28 years’ follow-up of 26,000 Norwegian men and women. Cancer Causes Control 7:497–506
Tverdal A, Thelle D, Stensvold I et al (1993) Mortality in relation to smoking history: 13 years’ follow-up of 68,000 Norwegian men and women 35–49 years. J Clin Epidemiol 46:475–487
Hansen S, Melby KK, Aase S et al (1999) Helicobacter pylori infection and risk of cardia cancer and non-cardia gastric cancer. A nested case–control study. Scand J Gastroenterol 34:353–360
Jansson C, Johansson AL, Bergdahl IA et al (2005) Occupational exposures and risk of esophageal and gastric cardia cancers among male Swedish construction workers. Cancer Causes Control 16:755–764
Freedman ND, Abnet CC, Leitzmann MF et al (2007) A prospective study of tobacco, alcohol, and the risk of esophageal and gastric cancer subtypes. Am J Epidemiol 165:1424–1433
Chao A, Thun MJ, Henley SJ et al (2002) Cigarette smoking, use of other tobacco products and stomach cancer mortality in US adults: the Cancer Prevention Study II. Int J Cancer 101:380–389
Galanis DJ, Kolonel LN, Lee J, Nomura A (1998) Intakes of selected foods and beverages and the incidence of gastric cancer among the Japanese residents of Hawaii: a prospective study. Int J Epidemiol 27:173–180
Kneller RW, McLaughlin JK, Bjelke E et al (1991) A cohort study of stomach cancer in a high-risk American population. Cancer 68:672–678
Padrão P, Lunet N, Santos AC, Barros H (2007) Smoking, alcohol, and dietary choices: evidence from the Portuguese National Health Survey. BMC Public Health 7:138
Dallongeville J, Marécaux N, Fruchart JC, Amouyel P (1998) Cigarette smoking is associated with unhealthy patterns of nutrient intake: a meta-analysis. J Nutr 128:1450–1457
Peleteiro B, Bastos J, Barros H, Lunet N (2008) Systematic review of gastric intestinal metaplasia prevalence and its area level association with smoking. Gac Sanit (in press)
Peleteiro B, Lunet N, Figueiredo C et al (2007) Smoking, Helicobacter pylori virulence, and type of intestinal metaplasia in Portuguese males. Cancer Epidemiol Biomarkers Prev 16:322–326
Mesquita P, Almeida R, Lunet N et al (2006) Metaplasia—a transdifferentiation process that facilitates cancer development. The model of gastric intestinal metaplasia. Crit Rev Oncog 12:3–26
Agudo A, Sala N, Pera G et al (2006) Polymorphisms in metabolic genes related to tobacco smoke and the risk of gastric cancer in the European prospective investigation into cancer and nutrition. Cancer Epidemiol Biomarkers Prev 15:2427–2434
La Torre G, Boccia S, Ricciardi G (2005) Glutathione S-transferase M1 status and gastric cancer risk: a meta-analysis. Cancer Lett 217:53–60
Sipponen P, Correa P (2002) Delayed rise in incidence of gastric cancer in females results in unique sex ratio (M/F) pattern: etiologic hypothesis. Gastric Cancer 5:213–219
Lopez A, Hollinshaw N, Piha T (1994) A descriptive model of the cigarette epidemic in developed countries. Tob Control 3:242–47
Santos AC, Barros H (2004) Smoking patterns in a community sample of Portuguese adults, 1999–2000. Prev Med 38:114–9
Idris BI, Giskes K, Borrell C et al (2007) Higher smoking prevalence in urban compared to non-urban areas: time trends in six European countries. Health Place 13:702–712
Huang XE, Tajima K, Hamajima N et al (2000) Effects of dietary, drinking, and smoking habits on the prognosis of gastric cancer. Nutr Cancer 38:30–36
Palli D, Russo A, Saieva C et al (2000) Dietary and familial determinants of 10-year survival among patients with gastric carcinoma. Cancer 89:1205–1213
Lunet N (2006) Gastric cancer frequency and trends. Arq Med 20:79–85
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ladeiras-Lopes, R., Pereira, A.K., Nogueira, A. et al. Smoking and gastric cancer: systematic review and meta-analysis of cohort studies. Cancer Causes Control 19, 689–701 (2008). https://doi.org/10.1007/s10552-008-9132-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10552-008-9132-y