Abstract
During the past three decades, the social/natural environment and lifestyle of people in China have undergone a marked transformation to westernization. However, age-standardized cancer rates have not been determined to any great extent in China. In this study, we tracked the cancer incidence between 1981 and 2000 in Tianjin, to identify the changes in incidence associated with social and economic changes. Cancer incidence data were collected by the Tianjin Cancer Registry. Sex, age, and organ site-specific incidence trends were analyzed by the “join-point regression” method. Overall crude cancer incidence increased, but the age-standardized incidence slightly decreased during the study period. The incidence of lung cancer increased between 1981 and 1996 but decreased between 1996 and 2000. The incidences of uterine, esophageal, stomach, and liver cancers decreased. However, the incidences of colorectal, pancreatic, breast, ovarian, and prostate cancers all increased during the study period. There was an aging-related increase in the overall crude cancer incidence and an alteration in the distribution of cancer types in Tianjin. The incidences of cancer types that are more prevalent in developed countries appeared to increase in China, whereas the incidences of cancer types that are more prevalent in developing countries appeared to decline.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Cancer is a major disease that has a great burden on public health worldwide. Cancer has overtaken heart disease as the number one killer in age under 85 in the U.S. [1]. Currently, the overall cancer incidence in developed countries is more than twice that seen in developing countries [2–4]. Epidemiological studies have linked the increase in the cancer incidence in the developed countries to many environmental factors and dietary habits [5, 6]. The main reasons for a greater cancer burden in affluent societies include the earlier timing of the start of the tobacco epidemic, earlier exposure in life to occupational carcinogens, and Western diet and lifestyle [7, 8]. With the globalization of the economy, unprecedented social and environmental changes, and aging of the populations in many countries, conspicuous trends are being seen in the cancer incidence [9]. In Asia alone, cancer incidence is predicted to double in the next 20 years on the basis of current trends [3].
China has undergone great changes in the economy and lifestyle, since its industrialization and modernization programs began in the late 1970s. However, because there are limited cancer registration systems existing in China to date, little is known about the actual incidences of various cancers in the various regions and cities of China, the information that is important in the planning and implementation of cancer prevention strategies and health services as well as in the allocation of available resources. Therefore, updated epidemiological data on the incidence of cancers and their associations with societal/environmental changes, especially exposures to distinct causative environmental factors [10–12] are needed.
In the study reported here, we comprehensively analyzed changes in the incidence and distribution of cancers in the urban Tianjin area that had occurred between 1981 and 2000. As the third largest city located in the east coastal area in China, Tianjin (formerly known as Tientsin) has a population of 11 million (including both urban and surrounding counties) and has experienced remarkable changes in environment and lifestyle as a result of recent economic development. We also provided possible explanations for the observed incidence trends in terms of social and economic changes that had occurred during the study period.
Materials and methods
Sources of data
The data presented were derived from the Tianjin Cancer Registry, in which data on cancer cases of all Tianjin residents since 1981 have been collected and analyzed. The registry covers a population of about 4 million people living in the urban Tianjin area. Information collected on the cancer reporting card includes patient’s name, sex, current age or date of birth, mailing address, reporting hospital or medical institute, time of diagnosis, ICD-X code (X International Classification of Disease), and occupation. The Tianjin Cancer Registry is a member of the International Agency for Research on Cancer (IARC) of the World Health Organization and has contributed cancer incidence data to the IARC publication “Cancer Incidence in Five Continents” since 1981 [13–16]. All death certificates, the second source of cancer registry information, have been registered at both the district public health unit and the local police department of the city. The district public health unit routinely reviews all death certificates and identifies deaths that are directly or indirectly caused by cancer, but the analysis presented here included all patients with reported primary cancer. Population data, along with age- and sex-specific information, were obtained from the police department in Tianjin, where all residents of Tianjin are required by law to be registered for their current residence in the city.
Statistical analysis
Cancer types were classified according to the ICD-X [17]. Cancer types examined included the top 12 in males and top 14 in females (including cancers common to both sexes and sex-specific cancer types, i.e., cancers of the breast, ovary, uterine, and prostate) as listed in Table 4. We first calculated the age-standardized incidence rates (ASIR) and then analyzed sex-, age-, and site-specific trends in these standardized rates by the “join-point regression” method [18] using the available SEER.Stat software [19]. In this statistical method, a year when a change in trend is detected during the study period is called a “join-point,” and join-points that show statistical significance (P < 0.05) are retained in the final site-specific models. The maximum number of join-points allowed in our analysis was two, a number determined on the basis of the duration of the study period. The estimated annual percentage changes (EAPCs) were also determined to show linear trends by time period. The ranks for each cancer site in the year 2000 were also reported. The world standard population in the year 1985 was used as a standard in the comparisons with international rates. The top five cancer sites and their proportions were listed for each selected year. The birth cohort effect on the age-specific incidence was estimated using the SAS software, V9 (SAS Institute, Cary, NC).
Results
Between 1981 and 2000, a total of 140,445 cancer cases were reported to the Tianjin Cancer Registry, of which 53.73% were males (Table 1). The mean age at diagnosis was 61.55 years (standard deviation: 14.19 years), and the mean age was higher for males than for females (62.72 and 60.19 years, respectively). More than half of the patients were diagnosed on the basis of histopathologic findings, and 35% were diagnosed on the basis of clinical signs and symptoms. Diagnosis in <1% was obtained from death certificates (Table 1).
The overall crude and age-adjusted incidences for the entire study period were 214.97/1,00,000 and 198.97/1,00,000 for males and 187.82/1,00,000 and 157.99/1,00,000 for females, respectively. For males, the crude incidence increased throughout the study period by 1.90 percent per year; and for females, the crude incidence increased by 1.14 percent per year between 1981 and 1992 and by 3.62 percent per year between 1992, and 2000 (Table 2). We also calculated age-specific incidences by sex and year of diagnosis. In general, changes in incidences were not consistent across age groups. In particular, for the age group of 0–44 years, the cancer incidence increased in both males (EAPC: 1.59) and females (EAPC: 2.06); for the 55–64 age group, the cancer incidence decreased in both males (EAPC: −1.32) and females (EAPC: −0.84); for the 75 and older age group, the cancer incidence for females decreased by 7.08 percent per year between 1981 and 1985 but then increased by 2.02 percent per year between 1985 and 2000. No significant trends were found in other age groups.
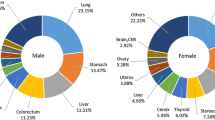
To determine what cancer types were frequently diagnosed in different time periods, we listed the top five cancer sites for the year 1981, 1985, 1990, 1995 and 2000. As shown in Table 3, for all 20 years combined, lung, stomach, liver, esophageal, and colorectal cancers ranked as the top five cancer types in males, whereas lung, breast, colorectal, stomach, and liver cancers ranked as the top five cancer types in females. Lung cancer was the top cancer type in every year studied. Relatively minor changes have been observed for the top five cancer types in male, but the top five cancer types have changed more markedly for female with cervix uteri, and esophageal cancers dropping out of the top five list.
To accurately measure the changes in the incidence of each of the major cancer types during the 20 years of the study period, we performed join-point analyses by sex and cancer type of the age-standardized incidence rates between 1981 and 2000 (Table 4). When all cancer types were combined by sex, however, no significant join-point was found. That is, each year between 1981 and 2000, age-standardized cancer rates decreased by an average of 0.48% in males and 0.31% in females.
When cancer types were analyzed separately, significant join-points were identified for lung and uterine cancers. In particular, the incidence of lung cancer significantly increased between 1981 and 1996 but decreased only slightly between 1996 and 2000. The incidence of uterine cancer drastically declined by 8.96 percent per year between 1981 and 1991, but then leveled off during the following 10 years.
As shown in Table 4, the incidences of esophageal, stomach, and liver cancers showed an approximately linear decrease during the study period, and the trends were statistically significant. Specifically, esophageal cancer decreased by 5.07 percent per year in males and by 6.00 percent per year in females; stomach cancer decreased by about 3.50 percent per year in both sexes; and liver cancer decreased by about 1.15 percent per year in both sexes. In contrast, kidney cancer increased by 4.09 percent per year in males and 2.99 percent per year in females, and brain cancer increased by 1.58 percent per year in males and 2.05 percent per year in females.
Different trends by sex were found for leukemia and colorectal, pancreatic, and laryngeal cancers. The incidence of colorectal cancer increased in males (EAPC = 1.63) but remained unchanged in females; the incidence of pancreatic cancer increased in females (EAPC = 1.20) but remained unchanged in males; and the incidence of laryngeal cancer and leukemia decreased in females (EAPC = −1.95 and −1.50, respectively) but remained unchanged in males.
The incidences of breast, ovarian, and prostate cancers all increased during the study period. In particular, the incidence of breast cancer increased by 1.86 percent per year; the incidence of ovarian cancer increased by 1.95 percent per year; and the incidence of prostate cancer increased by 3.43 percent per year.
Further analyses were performed to determine the birth-cohort cancer incidence rates for both males and females. These results suggested decreasing trends in the ASIR. Specifically, for those born after 1915, age-specific cancer incidence rates were decreased, but the rates were increasing in other younger birth cohorts in every age group, except for those born after 1935 and before 1945. However, males and females showed similar patterns (Fig. 1).
Discussion
In our analyses of the data from the Tianjin Cancer Registry collected between 1981 and 2000, we found that the overall crude cancer incidence increased during this period, whereas the age-standardized incidence for both sexes slightly decreased, although there were some differences in sex-specific and birth-cohort-specific cancer incidence rates in different cancer types in the period.
Although the underlying biological mechanisms remain unknown, there are some social, environmental, and economical changes that may explain these findings. First, the findings suggest that aging of the population is a significant contributor to the increase in the overall crude cancer incidence, in fact, the proportion of the aged people (over 65 years of age) increased from 6.75% in 1981 to 12.62% in 2000, underscoring the need for increased cancer screening for older populations in Tianjin. However, the decreasing ASIR (Age-Standardized Incidence Rate) of cancer suggest that the early stage of modernization in China that led to better health care and social well-being might have reduced the burden of cancer. This is supported by a similar trend of reduced cancer incidence observed in Qidong and Shanghai, other two large cities in China [20, 21]. In contrast, the age-adjusted cancer incidence increased in France and the United States [22, 23] during the same period covered by our analysis. In Italy, although the incidence of cancer among males in certain regions also decreased, the overall incidence increased [24, 25].
However, it is interesting that the overall age-specific cancer incidence had not significantly increased after the early stage of modernization in China, but our analyses revealed changes in specific cancer incidences and patterns that warrant attention. Specifically, significant join-points were found for lung and uterine cancers. For lung cancer, the incidence in both sexes increased between 1981 and 1996 but decreased between 1996 and 2000. For uterine cancer, the incidence decreased between 1981 and 1991 but remained fluctuated in the following 10 years. The incidence of esophageal, stomach, and liver cancers decreased for both sexes during the 20-year study period, which might have contributed to the overall decreasing trend in cancer incidence.
On the other hand, the incidence of colorectal cancer increased among males and the incidence of pancreatic cancer increased among females. Likewise, the incidences of breast, ovarian, kidney, prostate, and brain cancers all increased. Thus, it appears that the incidences of cancer types frequently diagnosed in developed countries are increasing in Tianjin, and the incidences of cancers frequently diagnosed in developing countries are decreasing. It is apparent that, during the 20-year study period, the decrease exceeded the increase in the overall cancer incidence.
The changing pattern in the overall cancer incidence is consistent with our current understanding of cancer etiological factors [26–28]. For example, populations in developing countries are prone to cancers in which infectious agents and associated non-malignant diseases play a significant etiologic role. These include cancers of the stomach, uterus, cervix, liver, and possibly esophagus. The changes in the cancer distribution in Tianjin also parallel the economic and social metamorphosis occurring during the corresponding decades. For example, economic growth and increases in personal income have led to improvements in people’s living conditions. The number of kitchen ventilators per family increased considerably in Tianjin, which greatly improved the indoor environment. Increases in the use of refrigerators have led to substantial reductions in the consumption of smoked, salted, and pickled food and increases in the fresh food intake among Tianjin residents. These changes may account for the decreases in the incidence of esophageal, stomach, and liver cancers. Noticeable improvements in public sanitation and personal hygiene, paticularly regular gynecological examinations in females, have likely contributed to the dramatic decline in the incidence of uterine cancer. The universal vaccination for HBV has likely led to the decrease in the liver cancer incidence as well.
Recent rapid growth in the economy in China has resulted in dramatic changes in the dietary habits of Tianjin residents. Data from Tianjin Statistical Bureau [29] show that between 1985 and 2000, the average annual consumption of cereal and vegetables decreased among residents and the consumption of fruits, animal products, and plant oils increased. The proportion of carbohydrates in the total energy intake decreased from 69% to 53%, and the proportion of fat in the total intake increased from 19% to 33%. These changes in nutrient and energy intake may explain the changes in the incidences of cancers of certain sites [30–33]. For example, the increase in colorectal, breast, and pancreatic cancer may be attributable to the rise in the consumption of animal products and dietary fat. A decline in physical activities may also have contributed to the increase in the overall cancer incidence. Although the economic development liberated urban residents from labor-intensive work and made them less dependent on walking and bicycling for transportation, the decrease in physical activities coupled with an increase in nutrient and energy intake have resulted in overweight problems. This is shown by the increase in the proportions of overweight and obese adults among Chinese from 6% and 0.6%, respectively, in 1982 to 18.5% and 2.2%, respectively, in 1997 [34]. It is well known that overweight is a risk factor for several cancers, including kidney, breast, colorectal, and ovarian cancers [35–38].
Cigarette smoking is a major risk factor for many cancers, especially lung cancer. As we previously reported, the average number of cigarettes smoked in Tianjin has steadily decreased since 1986, a decline that might explain the decrease in the lung cancer incidence seen in late 1990s [39], although the latent period of the effect may be even longer.
We should also point out that there are potential limitations in our study. The accuracy of observed incidence rates may be influenced by several factors including the completeness of registration, changes in disease classification, and advances in diagnostic technology. Whereas it is difficult to document the completeness of the registration by the Tianjin Cancer Registry, missing registration should not be a major problem because there has been a legal reporting requirement for all cancer cases in Tianjin. Changes in the coding system from ICD to ICD-O may have resulted in loss of specificity in some reported cases, but the number of cases involved is very low. Advance in diagnostic technology is perhaps a single most important variable in the study because better diagnostic technology leads to more early detection of cancer thus may increase the number of cancer cases over the study period.
China has undergone and is continuing to undergo tremendous changes in living conditions and lifestyle, especially the large cities. The modernization and westernization of lifestyle among Chinese appear to have both positive and negative effects on individual health. Our analyses of the cancer incidence in Tianjin showed that the early stage of modernization in China has actually had beneficial effects in terms of reducing the overall age-adjusted cancer incidence. Because there is now an alarming increasing trend in the incidence of several cancer types, more noticeable in developed countries, it is likely that China will have an increase in the overall cancer incidence in the coming decades, when the early decrease in cancers such as uterine cancer reaches its limit. As a matter of fact, the incidence of uterine and esophageal cancers is already quite low, leaving very little room for a further decline. On the other hand, the incidences of breast, ovarian, kidney, and prostate cancers are still going up. In addition to an increase in the incidences in cancers that are more prevalent in developed countries, we also observed an increasing trend in cancers in the younger age group (0–44 years old) among both males and females. This is an alarming finding that suggests that a future increase in the overall cancer incidence in Tianjin may be more likely to occur soon. Therefore, a comprehensive and timely survey of the cancer incidence in Tianjin is necessary to further monitor such an increasing trend that may lead to the development and implementation of an effective primary prevention program.
References
Twombly R (2005) Cancer surpasses heart disease as leading cause of death for all but the very elderly. J Natl Cancer Inst 97:330–331
Parkin DM, Bray F, Ferlay J et al (2005) Global cancer statistics, 2002. CA Cancer J Clin 55:74–108
Parkin DM (2001) Global cancer statistics in the year 2000. Lancet Oncol 2:533–543
Kanavos P (2006) The rising burden of cancer in the developing world. Ann Oncol Suppl 8:15–23
Brennan P (2002) Gene–environment interaction and aetiology of cancer: what does it mean and how can we measure it? Carcinogenesis 23:381–387
Thun M, Henley S, Calle E (2001) Tobacco use and cancer: an epidemiologic perspective for geneticists. Oncogene 21:7307–7325
Notani PN (2001) Global variation in cancer incidence and mortality. Current Science 81:10
Parkin DM (2004) International variation. Oncogene 38:329–340
Ferlay J, Bray F, Parkin DM et al (2001) Cancer incidence and mortality Worldwide, Globocan 2000. Lyon: IARC
Lowenfels A, Maisonneuve P (2002) Epidemiologic and etiologic factors of pancreatic cancer. Hematol Oncol Clin N 16:1–16
Giovannucci E, Goldin B (1997) The role of fat. Fatty acids, and total energy intake in the etiology of human colon cancer. Am J Clin Nutr 66:S1564–S1571
Franceschi S, Bidoli E, Herrero R et al (2000) Comparison of cancers of the oral cavity and pharynx worldwide: etiological clues. Oral Oncol 36:106–115
Muir C, Waterhouse J, Mack T et al (1987) Cancer incidence in five continents vol. V. Lyon: IARC
Parkin DM, Whelan SL, Ferlay J et al (1990) Cancer incidence in five continents vol.VI. Lyon: IARC
Parkin DM, Whelan SL, Ferlay J et al (1997) Cancer incidence in five continents vol. VII. Lyon: IARC
Parkin DM, Whelan SL, Ferlay J et al (2002) Cancer incidence in five continents vol. VIII. Lyon: IARC
World Health Organisation (1992) International statistical classification of diseases and related health problems, tenth revision ICD-10. Geneva: WHO
Kim HJ, Fay MP, Feuer EJ et al (2000) Permutation tests for joinpoint regression with application to cancer rates. Stat Med 19:335–351
Joinpoint Regression Program, Version 3.0 (2005) In: Statistical research and applications branch national cancer institute. Bethesda, USA
Chen JG, Zhu J, Parkin DM et al (2006) Trends in the incidence of cancer in Qidong, China, 1978–2002. Int J Cancer 6:1447–1454
Jin F, Devesa SS, Chow WH et al (1999) Cancer incidence trends in urban Shanghai, 1972–1994: an update. Int J Cancer 4:435–440
Remontet L, Estève J, Bouvier AM et al (2003) Cancer incidence and mortality in France over the period 1978–2000. Rev Epidemiol Sante 51:3–30
Edwards BK, Brown ML, Wingo PA et al (2005) Annual report to the nation on the status of cancer, 1975–2002, featuring population-based trends in cancer treatment. J Natl Cancer I 19:1407–1427
Stracci F, Canosa A, Minelli L et al (2007) Cancer mortality trends in the Umbria region of Italy 1978–2004: a joinpoint regression analysis. BMC Cancer 7:10–18
Crocetti E, Capocaccia R, Casella C et al (2004) Population-based incidence and mortality cancer trends (1986–1997) from the network of Italian cancer registries. Eur J Cancer Prev 4:287–295
Firket J (1955) Frequency, etiological factors and geographical distribution of lung cancer. Rev Med Liege 1:3–12
Weiderpass E, Pukkala E (2006) Time trends in socioeconomic differences in incidence rates of cancers of gastro-intestinal tract in Finland. BMC Gastroenterol 6:41
Lu SN, Su WW, Yang SS et al (2006) Secular trends and geographic variations of hepatitis B virus and hepatitis C virus-associated hepatocellular carcinoma in Taiwan. Int J Cancer 8:1946–1952
Statistical Bureau of Tianjin (1986–2001) Yearbooks of Tianjin in 1985–2000. Beijing: China Statistical Press
Ji J, Couto E, Hemminki K (2005) Incidence differences for gallbladder cancer between occupational groups suggest an etiological role for alcohol. Int J Cancer 3:492–493
Tsugane S (2004) Dietary factor and cancer risk–evidence from epidemiological studies. Gan To Kagaku Ryoho 6:847–852
Palmer S (1985) Diet, nutrition, and cancer. Prog Food Nutr Sci 3–4:283–341
Gonzalez CA (2006) Nutrition and cancer: the current epidemiological evidence. Br J Nutr 96:42S–45S
Koo LC, Mang OW, Ho JH (1997) An ecological study of trends in cancer incidence and dietary changes in Hong Kong. Nutr Cancer 3:289–301
Wu Y, Zhou B (2002) The current status and future trend of adult overweight and obesity in China. Zhong Hua Liu Xing Bing Xue Za Zhi 1:11–15
Rodriguez C, Calle EE, Fakhrabadi-Shokoohi D et al (2002) Body mass index, height, and the risk of ovarian cancer mortality in a prospective cohort of postmenopausal women. Cancer Epidem Biomar 9:822–828
Franceschi S, Favero A, La Vecchia C et al (1996) Body size indices and breast cancer risk before and after menopause. Int J Cancer 2:181–186
Lukanova A, Bjor O, Kaaks R et al (2006) Body mass index and cancer: results from the Northern Sweden Health and Disease Cohort. Int J Cancer 2:458–466
Chen KX, Wang PP, Sun BC et al (2006) Twenty-year secular changes in sex specific lung cancer incidence rates in an urban Chinese population. Lung Cancer 51:13–19
Acknowledgements
This work was supported by the Department of Health of Tianjin. The authors thank all the colleagues in the Department of Epidemiology and Biostatistics of the Tianjin Medical University Cancer Hospital and Institute for their contribution to this study and the Tianjin Central Disease Control for the contributions in data collection. The authors also thank Ms. Beth Notzon of Department of Scientific Publications of The University of Texas M. D. Anderson for her editorial assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Song, F., He, M., Li, H. et al. A cancer incidence survey in Tianjin: the third largest city in China—between 1981 and 2000. Cancer Causes Control 19, 443–450 (2008). https://doi.org/10.1007/s10552-007-9105-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10552-007-9105-6