Abstract
Background
Since the 1970s, incidence rates for esophageal and gastric cardia adenocarcinomas have risen substantially for reasons that are not well understood. We sought to determine the role of dietary factors in these tumor types.
Methods
This analysis on dietary factors included 206 esophageal adenocarcinoma, 257 gastric cardia, 366 distal gastric adenocarcinoma patients and, 1,308 control subjects from a population-based, case-control study conducted in Los Angeles County. Polytomous logistic regression was used to calculate odds ratios (ORs), as an estimate of the relative risk, and corresponding 95% confidence intervals (CIs) for the three tumor types.
Results
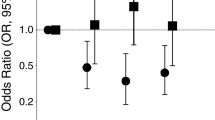
Intake of fiber had a significant impact on risk of esophageal and gastric cardia adenocarcinoma after adjustment for age, gender, race, birthplace, education, cigarette smoking, body size, history of reflux, and vitamin use. Compared to subjects in the lowest quartile of fiber intake, subjects in the highest quartile of intake showed odd ratios of 0.44 (95% CI = 0.26–0.76) for esophageal adenocarcinoma (P trend = 0.004) and 0.58 (95% CI = 0.38–0.88) for gastric cardia adenocarcinoma (P trend = 0.016); these inverse associations remained after further adjustment for intake of fat. Positive associations between dietary fat and the three tumor types weakened after adjustment for fiber intake and were no longer statistically significant. For distal gastric cancer, a significant inverse association with fiber was observed only after adjustment for fat intake. The significant inverse associations with fiber remained after further adjustment for H. pylori infection for all three tumor types.
Conclusions
High intake of fiber was associated with significant reduced risks of esophageal and gastric cardia adenocarcinoma even after adjustment for dietary fat, H. pylori infection and other covariates.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
High body weight [1, 2], history of gastroesophageal reflux disease [2–4], and tobacco use [2, 5] are significant risk factors for esophageal and gastric cardia adenocarcinoma in several population-based case-control studies conducted in the US and Sweden. While numerous studies have found that diet plays an important role in the etiology of esophageal squamous cell cancers [6] and distal gastric cancers [7, 8], fewer studies have specifically investigated the influence of dietary factors on risk of esophageal and gastric cardia adenocarcinoma. Three studies have investigated dietary associations and risk of gastric cardia and esophageal adenocarcinoma combined [9–11] and three studies have examined one of the tumor types [12–14]. However, only two population-based studies have investigated dietary factors separately for gastric cardia and esophageal adenocarcinoma in the same study [15, 16], allowing comparison within the same population of risk patterns for the two tumor types. In the US multicenter study [15], low intake of fiber was a significant risk factor for both esophageal and gastric cardia adenocarcinoma but high fat intake was associated with an increased risk of esophageal but not gastric cardia adenocarcinoma. In the nationwide Swedish case-control study, risk of esophageal adenocarcinoma was not significantly influenced by either fat or fiber intake whereas risk of gastric cardia cancer increased nonsignificantly with intake of dietary fat and decreased significantly with increasing fiber intake [16, 17]. Thus, a primary objective of this analysis is to investigate further the impact of dietary fat and fiber on risk of esophageal, gastric cardia, and distal gastric adenocarcinoma in a population-based case-control study in Los Angeles County [2], a study which shares many similarities with both the US multicenter and the Swedish nationwide studies.
Additionally, a recent analysis of the European Prospective Investigation in Cancer (EPIC) cohort found high intake of meat, particularly processed meat, to be associated with increased risk of esophageal adenocarcinoma and gastric noncardia cancer, but not gastric cardia cancer. The finding between processed meat and noncardia gastric cancer was particularly strong among persons who were positive for H. pylori infection [18]. Thus, a secondary objective of this analysis is to investigate further risk associations with intake of total meat, red meat, poultry, fish/shellfish, and processed meat in this population-based study of esophageal, gastric cardia, and distal gastric adenocarcinomas. We also investigated whether these dietary associations are modified by history of H. pylori infection in a subset of subjects for whom infection status was determined [19].
Material and methods
As described previously [2], patients included as cases in our case-control study were men and women, aged 30–74 years, with histologically confirmed esophageal, gastric cardia or distal gastric adenocarcinoma, who had no prior history of cancers of these sites. We included patients diagnosed between 1992 and 1997 with esophageal adenocarcinoma (C15.0–C15.9), and gastric cardia adenocarcinomas (C16.0) and patients diagnosed between 1992 and 1994 with distal gastric cancers (i.e., those diagnosed in the antrum/pylorus (C16.3, C16.4), fundus/body (C16.1, C16.2), and lesser and greater curvature (C16.5, C16.6), as well as mixed subsite (C16.8) and subsite not otherwise specified (C16.9)). All incident cancers were identified by the Los Angeles County Cancer Surveillance Program (CSP), a population-based tumor registry.
We conducted in-person interviews with patients or their next-of-kin (NOK) when patients were unable to be interviewed due to death or illness. Of the 1,716 eligible patients identified, interviews were completed with 947 case patients (i.e., 55% [947/1716] of those identified and 77% [947/(1716–310-174)] of those approached). We were unable to obtain interviews for 769 patients: 310 had died or were too ill to be interviewed and did not have a NOK available for interview; physicians denied permission to contact 174 patients; 146 patients could not be located; and 139 patients did not wish to participate. Information was incomplete for five patients and 942 patients were included in the analyses [2]. Age, gender, and race distributions did not differ between case patients we interviewed and those we did not interview.
Control subjects were individually matched to interviewed case patients on gender, race and date of birth (±5 years). Whenever possible, we sought two control subjects for cases diagnosed with esophageal or gastric cardia adenocarcinoma to increase the study’s statistical power. Control subjects must not have had a diagnosis of stomach or esophageal cancer. A neighborhood control subject was sought by use of a systematic algorithm based on the address of the case patient. If the first eligible matched control subject refused to participate, the second eligible one in the sequence was invited, and so on. Of the 947 case patients interviewed, 528 had one participating control subject, 382 had two or more participating control subjects, and 37 had no eligible control subject identified. For over half of the case patients (n = 521), the first eligible matched control subject was interviewed; the second or a later matched control was interviewed for the remaining cases.
The study was approved by the Institutional Review Board of the Keck School of Medicine of the University of Southern California, and written informed consent was obtained from each study participant before interview.
Interview method
Cases and their matching controls were interviewed by the same interviewer in almost all instances. Next of kin (NOK) interviews accounted for 271 of the 942 interviews with case patients (66 for esophageal adenocarcinoma, 85 for gastric cardia and 120 for distal gastric cancer patients). Although it was not feasible to blind the interviewers to case (or NOK) or control status, interviewers (and study participants) were not aware of the study hypotheses. A reference date was defined as one year before the date of diagnosis of the case subject; this same reference date was used for each patient’s matched control subject(s).
The diet history included 124 food items or food groups (9 beverages, 115 foods), encompassing the following categories: vegetables, fruits/juice, smoked/cured meats, noodles/pasta/legumes, rice/potatoes, meat/poultry/fish, cereal, bread/spreads/snacks/sweet and beverages. Food items were selected to include the most frequently consumed foods in each ethnic group to capture at least 85% of the intake of the major nutrients of interest for all participants. The diet history we used was modeled after one we had previously used in a case-control study of multiethnic populations in California and Hawaii that was developed by researchers at the University of Hawaii [20]. Subjects reported both frequency and portion size for each food item consumed at least 12 times per year that represented their usual diet one year before reference date (one year before diagnosis for cases and the same reference period for control subjects). Identification of serving size was facilitated by the use of serving cups for various portion sizes. An estimate of caloric intake (used as an adjustment variable in the analysis) was derived from the questionnaire data for each participant, using a food composition table available at the University of Hawaii. The nutrient database at the University of Hawaii is based on the United States Department of Agriculture [21], supplemented with data from a variety of other sources [22]. Risk associations in relation to total fat, dietary fiber, and other macronutrients and micronutrients were investigated.
Intake of total meat, red meat, poultry, fish/shellfish, and processed meat were estimated, in grams per day, from information reported in the questionnaires. Red meat included beef, hamburger, pork, veal, and lamb. Poultry included chicken, turkey, duck and goose. Fish included fresh/canned fish and shellfish. Processed meat included bacon, sausages, hot dogs, smoked or cured ham, luncheon meat, and canned meat.
In addition to the food frequency questionnaire, subjects were asked whether they used vitamin supplements, specifically intake of multivitamins, vitamin C, D, A, or E or beta-carotene, on a regular basis (at least once per week for 6 months or longer). Age first started to use and duration of use were asked for each specific vitamin. Since the content of multiple vitamin supplements varies substantially and since individuals may change brands and types of vitamins used, we created a composite index of vitamin use by considering the number of different vitamins (0 if did not take any vitamins, and 6 if took all 6 types of vitamins) taken by a participant, and whether the participant used multivitamin supplements or single supplements. We did not attempt to estimate the contribution of specific vitamins to our nutrient analysis.
Nutrient densities expressed as intake per 1,000 kcal were used in all data analyses as a means for adjusting for total reported energy intake. Nutrient density intakes and meat intakes were categorized into quartiles using sex-specific cutpoints based on daily intakes in all controls. Indicator variables were created to represent quartile membership. Quartile ORs were computed by defining the lowest quartile of intake as the referent category. Conditional and unconditional logistic regression methods were used to calculate ORs and 95% CI. These results were comparable to the ORs and 95% CIs that were estimated using polytomous logistic regression which modeled three outcome categories (esophageal adenocarcinoma, gastric cardia, and distal gastric cancers) and we report below results from the polytomous logistic regression analyses. The information on tumor subsite and histology were obtained from the pathology reports documenting each cancer. Variables included in the regression model (age: <39, 40–49, 50–59, 60–69, 70+, sex: male/female, race: White, African-, Latino-, Asian-American, birthplace: US born, non-US born, and education: less than high school, high school, some college, college graduate or more) were defined in the same manner as in our previous reports [2]. In addition, smoking history (never, former, current), body mass index (kg/m2) (in quartiles), and history of gastro-esophageal reflux diseases were included as covariates in the analyses.
Tests for trend based on Wald tests were computed by fitting a polytomous logistic regression model to ordinal values representing levels of exposure. In the analyses, the dietary factors of interest were adjusted for the covariates mentioned above but they were not adjusted for other nutrients except for total calories. All reported trend test significance levels (P values) are two-sided. All analyses were performed using the EPILOG Windows (version 1.01s) statistical software system (Pasadena, CA).
Results
Characteristics of the study participants and intake patterns of selected dietary factors are shown in Table 1. As we have previously reported, cigarette smoking, high body weight, and history of reflux (i.e., history of hiatal hernia, esophagitis, Barrett esophagus, excess acid, or gastro-esophageal reflux that was diagnosed by a physician at least 2 years before reference date) were significant risk factors for esophageal and gastric cardia adenocarcinoma [2, 3]. There was a deficit of vitamin users among case patients, particularly among distal gastric cancer patients compared to controls. Subsequent analyses on dietary factors included adjustment for vitamin use (number of vitamin supplements used) as well as other significant risk factors (smoking, body size, and gastroesophageal reflux diseases) in this study population [2, 3].
Risk patterns for the three tumor types in relation to total calories and macronutrients are shown in Table 2. Caloric intake did not differ significantly between esophageal adenocarcinoma or gastric cardia cancer patients and controls. However, caloric intake was significantly lower in distal gastric cancer patients than in controls (P trend = 0.016). After adjusting for caloric intake, the risk of esophageal (P trend = 0.035), gastric cardia (P trend = 0.046), and distal gastric adenocarcinoma (P trend = 0.063) increased with increasing intake of total fat. When specific components of fat were considered, risk of all three tumor types increased with increasing intake of saturated and monounsaturated fat but was unrelated to intake of polyunsaturated fat. Risk of esophageal adenocarcinoma was significantly associated with intake of both saturated and monounsaturated fats while gastric cardia adenocarcinoma risk was significantly associated with higher intake of saturated fat and distal gastric cancer risk was significantly associated with higher intake of monounsaturated fat. Cholesterol intake was associated with all three tumor types, but this finding was, statistically, significant only for distal gastric cancer (P trend <0.001). In contrast, risks of gastric cardia and of distal gastric cancer were statistically significantly and inversely associated with intake of carbohydrates. High intake of total fiber (highest (>15.0 gm/day) vs lowest (≤9.31 gm/day) quartile) was associated with a statistically significant 31–56% risk reduction of all three tumor types. A statistically significant trend of decreasing risk with increasing fiber intake was observed for esophageal and gastric cardia adenocarcinoma, but not for distal gastric cancer (Table 2).
We examined risk patterns in relation to intake of fat and fiber simultaneously (Table 3). For all three tumor types, the increased risk associated with total fat diminished and was no longer statistically significant after adjusting for total fiber intake whereas the effect of fiber remained statistically significant and the risk estimates were largely unchanged. The positive association between cholesterol intake and distal gastric cancer was unaltered with additional adjustment for fiber intake. Vegetables, fruits, and cereals each accounted for 35%, 21%, and 44% of the fiber intake among control subjects in this study. Our results point to inverse associations from all sources of fiber although vegetable and fruit fibers appeared to have stronger effects on risk of esophageal and gastric cardia adenocarcinomas, whereas cereal fibers had more apparent effects on distal cancers (Table 3, bottom).
Our finding on vegetable and fruit fiber is not merely a marker for fruit and vegetable intake or other plant-based micronutrients. Intake of fruits and vegetables was not significantly associated with risk of these tumor types. For example, for esophageal adenocarcinoma, the adjusted ORs associated with quartiles of vegetable intake were 1.00, 0.92, 0.95, 0.67 (P trend = 0.23) and the adjusted ORs associated with quartile of fruit intake were 1.00, 0.90, 0.77, 0.98 (P trend = 0.76). When we considered intake of other plant-related micronutrients (e.g., alpha carotene, beta carotene, lycopene, lutein, tocopherol, vitamin C, folate) and fiber intake in stepwise regression analyses, only fiber intake entered the model in analyses of esophageal and gastric cardia adenocarcinoma. In the stepwise regression analysis for distal gastric cancer, both intake of fiber and lycopene were selected but intake of lycopene was positively associated with risk (data not shown).
Risk patterns for the three tumor types by intake of all meats combined (including fish) and separately by type of meat are presented in Table 4. Risk of esophageal adenocarcinoma was not significantly influenced by total meat intake (P trend = 0.33) but the risk of gastric cardia (P trend = 0.027) and distal gastric cancers (P trend = 0.004) increased with increasing quartile of meat intake. Risk tended to increase with increasing intake of red meat for the three tumor types but this was statistically significant only for gastric cardia cancers (P trend = 0.031). Intake of processed meat was associated with a significant increase of distal gastric cancer but it had no significant impact on risk of esophageal or gastric cardia adenocarcinoma. Intake of poultry and fish/shellfish was unrelated to risk of these three tumor types. For all three tumor types, the decreased risk associated with total fiber remained statistically significant after adjustment for intake of red meat and processed meat (data not shown). The positive association between processed meat and distal gastric cancer risk also remained after adjustment for fiber intake (fiber adjusted OR for the quartiles of processed meat intake were 1.00, 1.49, 1.20, 1.64; P trend = 0.027), but the effect of red meat intake on gastric cardia cancer risk diminished (fiber adjusted OR for the quartiles of red meat intake were 1.00, 1.14, 1.58, and 1.38, P trend = 0.13).
Table 5 shows risk patterns for fiber intake after adjustment for intake of total fat by race/ethnicity, body size, smoking history, history of reflux, vitamin use, and respondent type. Formal tests for interaction were not statistically significant suggesting that none of these variables significantly modified the fiber-risk associations.
As we have previously reported, history of H. pylori infection was a significant risk factor for distal gastric cancer, but it was not significantly associated positively or negatively with risk of gastric cardia or esophageal adenoacarcinoma in this population [19]. Risk patterns associated with intake of fiber, fat, cholesterol, and meats after adjustment for H. pylori status were generally similar to the overall results when H. pylori status was not included in the models (Table 6). In particular, the inverse association between fiber intake and risk remained statistically significant for esophageal adenocarcinoma and gastric cardia cancers and was strengthened for distal gastric cancer despite the reduced samples sizes. Few esophageal and gastric cardia adenocarcinoma patients were H. pylori positive and thus most of the results restricted to persons who were positive for infection were not statistically significant but the patterns were similar (data not shown). Among distal gastric cancer patients, the increased risk associated with intake of fat and cholesterol and the decreased risk associated with intake of fiber remained statistically significant among H. pylori positive persons (data not shown).
Discussion
In this population-based case-control study of adenocarcinoma of the esophagus and stomach, low intake of fiber was a significant risk factor for all three tumor types. Although high intake of total fat was also significantly associated with risk for all three tumor types (in multivariate analyses that included adjustment for total caloric intake), the effect of total fat diminished and was no longer statistically significant after additionally adjusting for fiber intake (Table 3). The inverse association with fiber also remained after adjustment for H. pylori status. The inverse association with fiber intake appears strongest for esophageal adenocarcinoma, intermediate for gastric cardia, and weaker for distal gastric adenocarcinomas in this study population.
Our findings on fiber and risk of esophageal and gastric cardia adenocarcinoma are consistent with the findings from the US multicenter study [15] but they differ somewhat from the Swedish nationwide study [16]. In the Swedish study, fiber was not associated with risk of esophageal adenocarcinoma, and the strong inverse association between fiber and gastric cardia cancer was primarily due to cereal fiber [16]. In contrast, in this Los Angeles County study, persons with a low intake of fiber from vegetables and fruits were at elevated risk for esophageal and gastric cardia adenocarcinoma whereas the effect of cereal fiber intake was modest. Cereal fiber, particularly from wheat, represented a major source of fiber (>60%) in the Swedish study. Several constituents of wheat fiber may be anticarcinogenic (e.g., indoles, phenolic compounds) [23, 24], but intake of this type of cereal fiber may be relatively low in the US population. Although the US multicenter study [15] and several other US studies [10, 11, 25] have found strong inverse association with fiber, sources of fiber were not presented. Slightly stronger inverse association with fruit fiber than with vegetable fiber was reported in one US study which examined risk patterns for esophageal adenocarcinoma and esophagogastric junction cancers combined, but information on cereal fiber was not presented in this study [10]. Intake of fruits and vegetables was not significantly associated with risk of these tumor types in this study population.
Fiber intake (based on crude fiber) in the US may have declined as much as 30%, from 6.1 gm/day in the early 1900s to 4.3 gm/day in the mid 1970s [26]. The average dietary fiber intake in our study population (∼12 g/day) is similar to that reported for the US population in the 1980s [27] but considerably lower than the American Heart Association’s recommendation of 25–30 g/day [28]. The consistent inverse association with fiber in major subgroup analyses in the present study (Table 5) strengthens the overall finding. High fiber intake has been suggested to reduce the risk of hiatal hernia [29] and was recently found to be associated with a reduced risk of gastro-esophageal reflux symptoms and erosive esophagitis in a cross-sectional study [30]. Fiber may have beneficial effects via a mechanical action by helping to remove and/or limit the contact of carcinogens with the esophageal epithelium [28]. In addition, high-fiber foods generally tend to have a higher content of antioxidants and phytochemicals [23, 24]. Importantly, our findings on dietary fiber remained statistically significant after adjustment for dietary fat whereas the effects of dietary fat were substantially diminished after adjustment for dietary fiber (see below).
Our study results suggest that total fat is not independently associated with risk as the effects of fat were substantially diminished after adjusting for dietary fiber intake. Previous findings on the role of dietary fat and risk of esophageal and gastric cardia adenocarcinoma are mixed. In the US multicenter study, risk of esophageal adenocarcioma but not gastric cardia cancer increased in association with total fat intake [15]. High-fat intake was also associated with increased risk of esophageal adenocarcinoma in a small US study [25]. In contrast, in the Swedish nationwide study, total fat intake was lower among patients with esophageal adenocarcinoma and was nonsignificantly higher among patients with gastric cardia cancer [17]. Although a statistically significant dietary fat effect was found in two [9, 11] of the three US studies which included gastric cardia and esophageal adenocarcinomas combined in the analysis [9–11], it is unclear whether fiber intake was considered in the analyses.
Our results reveal no significant difference in meat intake between esophageal adenocarcinoma patients and controls. Although high intake of red meat was associated with a significant increased risk of gastric cardia cancer, this effect diminished after adjustment for fiber intake. High intake of processed meat was a significant risk factor for distal gastric cancer and this association remained after adjustment for fiber intake (Table 3). Thus, our findings on intake of meat and processed meat and risk of distal gastric cancer and gastric cardia cancer are generally concordant with the findings from the EPIC cohort studies [8, 18]. Processed meat was implicated as a risk factor for esophageal adenocarcinoma in the EPIC study, but this was based on a relatively small number of cases (n = 65). In this analysis which included larger numbers of esophageal adenocarcinoma (n = 206), processed meat was not associated with risk. It should be noted that hamburger was included in the category of processed meat in the EPIC study whereas hamburger was included as part of red meat in our analysis. When we re-analyzed our data and examined intake of hamburger separately (i.e., excluded from red meat and from processed meat), intake of hamburger was unrelated to risk of the three tumor types. Intake of processed meat (without hamburger) was significantly associated with risk of distal gastric cancer (P trend = 0.02) while it remained unrelated to risk of esophageal and gastric cardia adenocarcinoma. A few previous studies provided results for intake patterns of meat and processed meat and risk of these tumor types. No significant associations were found between intake of processed meat or red meat and risk of esophageal adenocarcinoma [12], gastric cardia [31], and esophageal adenocarcinoma and gastric cardia cancers combined [11].
Most previous studies have not considered H. pylori status in their investigations of diet and esophageal and gastric adenocarcinoma. While H. pylori status was determined on only about one-third to one-half of the control and case groups in this study [19], our dietary findings which adjusted for H. pylori status (Table 6) were largely concordant with our findings in all subjects combined when H. pylori status was not considered. In particular, the inverse associations between fiber intake and risk in all three tumor types remained statistically significant. However, unlike results from the EPIC study [18], our findings on processed meat and red meat showed that distal gastric cancer risk was not stronger when we restricted our analysis to participants who were H pylori positive.
Our study represents one of the few large population-based epidemiologic studies that have been designed specifically to investigate further the role of dietary factors in these three tumor types. The problem of measurement errors in dietary assessment, particularly in relation to using food frequency questionnaires in case-control studies is well recognized. Nondifferential misclassification would tend to weaken our findings toward the null suggesting that the inverse associations with fiber intake may be even stronger. In addition, the fiber finding remained after adjustment for other dietary (e.g., dietary fat) and non-dietary factors including H. pylori infection status, body size, smoking, and history of reflux. However, we cannot rule out the possibility of selective recall bias if eating patterns of the cases were affected by early disease symptoms which would lead to differential measurement error and bias away from the null.
The rising incidence of adenocarcinoma of the esophagus is well documented in many western countries and the causes of this increase are not well-understood. High intake of fiber was significantly inversely associated with risk of esophageal adenocarcinoma and gastric cardia adenocarcinoma. Better understanding of the mechanisms by which high fiber intake may reduce risk of these tumor types is warranted.
References
Chow WH, Blot WJ, Vaughan TL et al (1998) Body mass index and risk of adenocarcinomas of the esophagus and gastric cardia. J Natl Cancer Inst 90:150–155
Wu AH, Wan P, Bernstein L (2001) A multiethnic population-based study of smoking, alcohol and body size and risk of adenocarcinomas of the stomach and esophagus (United States). Cancer Causes Control 12:721–732
Wu AH, Tseng CC, Bernstein L (2003) Hiatal hernia, reflux symptoms, body size, and risk of esophageal and gastric adenocarcinoma. Cancer 98:940–948
Lagergren J, Bergstrom R, Lindgren A et al (1999) Symptomatic gastroesophageal reflux as a risk factor for esophageal adenocarcinoma. N Engl J Med 340:825–831
Gammon MD, Schoenberg JB, Ahsan H et al (1997) Tobacco, alcohol, and socioeconomic status and adenocarcinomas of the esophagus and gastric cardia. J Natl Cancer Inst 89:1277–1284
Chainani-Wu N (2002) Diet and oral, pharyngeal, and esophageal cancer. Nutr Cancer 44:104–126
Nomura AM, Hankin JH, Kolonel LN et al (2003) Case-control study of diet and other risk factors for gastric cancer in Hawaii (United States). Cancer Causes Control 14:547–558
Larsson SC, Bergkvist L, Wolk A (2006) Processed meat consumption, dietary nitrosamines and stomach cancer risk in a cohort of Swedish women. Int J Cancer 119:915–919
Kabat GC, Ng SK, Wynder EL (1993) Tobacco, alcohol intake, and diet in relation to adenocarcinoma of the esophagus and gastric cardia. Cancer Causes Control 4:123–132
Brown LM, Swanson CA, Gridley G, et al (1995) Adenocarcinoma of the esophagus: role of obesity and diet. J Natl Cancer Inst 87:104–109
Zhang ZF, Kurtz RC, Yu GP et al (1997) Adenocarcinomas of the esophagus and gastric cardia: the role of diet. Nutr Cancer 27:298–309
Chen H, Ward MH, Graubard BI, et al (2002) Dietary patterns and adenocarcinoma of the esophagus and distal stomach. Am J Clin Nutr 75:137–144
Cheng KK, Sharp L, McKinney PA et al (2000) A case-control study of oesophageal adenocarcinoma in women: a preventable disease. Br J Cancer 83:127–32
Ekstrom AM, Serafini M, Nyren O et al (2000) Dietary antioxidant intake and the risk of cardia cancer and noncardia cancer of the intestinal and diffuse types: a population-based case-control study in Sweden. Int J Cancer 87:133–140
Mayne ST, Risch HA, Dubrow R et al (2001) Nutrient intake and risk of subtypes of esophageal and gastric cancer. Cancer Epidemiol Biomarkers Prev 10:1055–1062
Terry P, Lagergren J, Ye W et al (2001) Inverse association between intake of cereal fiber and risk of gastric cardia cancer. Gastroenterology 120:387–391
Terry P, Lagergren J, Wolk A et al (2000) Reflux-inducing dietary factors and risk of adenocarcinoma of the esophagus and gastric cardia. Nutr Cancer 38:186–191
Gonzalez CA, Jakszyn P, Pera G et al (2006) Meat intake and risk of stomach and esophageal adenocarcinoma within the European Prospective Investigation Into Cancer and Nutrition (EPIC). J Natl Cancer Inst 98:345–354
Wu AH, Crabtree JE, Bernstein L et al (2003) Role of Helicobacter pylori CagA+ strains and risk of adenocarcinoma of the stomach and esophagus. Int J Cancer 103:815–821
Kolonel LN, Hankin JH, Whittemore A et al (2000) Vegetables, fruits, legumes and prostate cancer: A multiethnic case-control study. Cancer Epidemiol Biomarkers Prev 9:795–804
Department of Agriculture (1992) Nutrient Database for Standard Reference, Release 10 tape, Human Nutrition Information Service. Bethesda, MD: United States Department of Agriculture
Holland B, Welch AA, Unwin ID, Buss DH, Paul AA, Southgate DAT (1991) McCance and Widdowson’s the composition of foods, ED. 5. Royal Society of Chemistry, Cambridge, England
Lupton JR,Turner ND (1999) Potential protective mechanisms of wheat bran fiber. Am J Med 106:24S–27S
Marlett JA, McBurney MI, Slavin JL (2002) Position of the American Dietetic Association: health implications of dietary fiber. J Am Diet Assoc 102:993–1000
Chen H, Tucker KL, Graubard BI et al. (2002) Nutrient intakes and adenocarcinoma of the esophagus and distal stomach. Nutr Cancer 42:33–40
Gortner WA (1975). Nutrition in the United Status, 1900 to 1974. Cancer Research 35:3246–3253
Lanza E, Jones DY, Block G et al. (1987) Dietary fiber intake in the US population. Am J Clin Nutr 46:790–797
Van Horn L (1997) Fiber, lipids, and coronary heart disease. A statement for healthcare professionals from the Nutrition Committee, American Heart Association. Circulation 95:2701–2704
Burkitt DP (1981) Hiatus hernia: is it preventable? Am J Clin Nutr 34:428–431
El-Serag HB, Satia JA, Rabeneck L (2005) Dietary intake and the risk of gastro-oesophageal reflux disease: a cross sectional study in volunteers. Gut 54:11–17
Wu-Williams AH, Yu MC, Mack TM (1990) Life-style, workplace, and stomach cancer by subsite in young men of Los Angeles County. Cancer Res 50:2569–2576
Acknowledgments
We thank all the study participants for their contributions and Annie Fung, Isaura Rivera, Timothy Stirton, and June Yashiki for their help with data collection. The authors have contributed to the conception and design (A Wu, L Bernstein), obtaining funding (A Wu, L Bernstein), data collection (A Wu), data management (C Tseng), statistical analysis (A Wu, C Tseng, L Bernstein), interpretation of study results (A Wu, J Hankin, L Bernstein), and preparation (A Wu, C Tseng) and reviewing of the manuscript (A Wu, C Tseng, J Hankin, L Bernstein). The authors have no conflicts of interest, i.e., involvements that might raise the question of bias in the work reported or in the conclusions, implications or opinions stated in this report. Supported by grant no. 3RT-0122 and 10RT-0251 from the California Tobacco Related Research Program, and grant no. CA59636 from the National Cancer Institute, and NIEHS Grant # 5P30 ES07048. Incident cancer cases for this study were collected by the USC Cancer Surveillance Program (CSP), which is supported under subcontract by the California Department of Health. The CSP is also part of the National Cancer Institute’s Division of Cancer Prevention and Control Surveillance, Epidemiology, and End Results Program, under contract number N01CN25403.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, A.H., Tseng, CC., Hankin, J. et al. Fiber intake and risk of adenocarcinomas of the esophagus and stomach . Cancer Causes Control 18, 713–722 (2007). https://doi.org/10.1007/s10552-007-9014-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10552-007-9014-8