Abstract
Background
Given its high recurrence risk, guidelines recommend systemic therapy for most patients with early-stage triple-negative breast cancer (TNBC). While some clinicopathologic factors and tumor-infiltrating lymphocytes (TILs) are known to be prognostic in patients receiving chemotherapy, their prognostic implications in systemically untreated patients remain unknown.
Methods
From a cohort of 9982 women with surgically treated non-metastatic breast cancer, all patients with clinically reported ER-negative/borderline (≤10%) disease were selected for central assessment of ER/PR/HER2, histopathology, Ki-67, and TILs. The impact of these parameters on invasive disease-free survival (IDFS) and overall survival (OS) was assessed using Cox proportional hazards models.
Results
Six hundred five patients met the criteria for TNBC (ER/PR < 1% and HER2 negative). Most were T1–2 (95%), N0–1 (86%), grade 3 (88%), and had a Ki-67 >15% (75%). Histologically, 70% were invasive carcinoma of no special type, 16% medullary, 8% metaplastic, and 6% apocrine. The median stromal TIL content was 20%. Four hundred twenty-three (70%) patients received adjuvant chemotherapy. Median OS follow-up was 10.6 years. On multivariate analysis, only higher nodal stage, lower TILs, and the absence of adjuvant chemotherapy were associated with worse IDFS and OS. Among systemically untreated patients (n = 182), the 5-year IDFS was 69.9% (95% CI 60.7–80.5) [T1a: 82.5% (95% CI 62.8–100), T1b: 67.5% (95% CI 51.9–87.8) and T1c: 67.3% (95% CI 54.9–82.6)], compared to 77.8% (95% CI 68.3–83.6) for systemically treated T1N0. Nodal stage and TILs remained strongly associated with outcomes.
Conclusions
In early-stage TNBC, nodal involvement, TILs, and receipt of adjuvant chemotherapy were independently associated with IDFS and OS. In systemically untreated TNBC, TILs remained prognostic and the risk of recurrence or death was substantial, even for T1N0 disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Triple-negative breast cancer (TNBC) accounts for 15–20% of all cases of breast cancer (BC) [1] and is characterized by earlier relapse and worse survival compared to non-TNBC subtypes [2]. While endocrine and targeted therapies have transformed the landscape of hormone receptor- and HER2-positive BC, cytotoxic chemotherapy remains the mainstay of treatment for TNBC. Efforts to better understand the heterogeneity within TNBC have resulted in the identification of distinct molecular subtypes [3,4,5], but to date these data have not changed the clinical management of early-stage TNBC.
Beyond hormone receptor and HER2, clinicopathological features such as TNM stage, grade, Ki-67, and histological subtype are often used to risk-stratify newly diagnosed BC. However, in TNBC, it remains unknown if these features provide independent prognostic information when analyzed in the context of TIL density and the presence or absence of systemic chemotherapy. Given the high recurrence and mortality risk, guidelines recommend the consideration of adjuvant or neoadjuvant chemotherapy for nearly all patients with non-metastatic TNBC [6]. As such, contemporary studies have only evaluated clinicopathological features in patients exposed to cytotoxics. The presence of high TILs, a surrogate for adaptive immune activation, is prognostic in early-stage TNBC treated with chemotherapy, being associated with higher rates of pathologic complete response in the neoadjuvant setting [7,8,9,10,11] and improved disease-free (DFS) and overall survival (OS) [12,13,14,15] in the adjuvant setting. However, the prognostic effect of TILs independent of systemic chemotherapy is unknown.
In this study, we sought to better understand the prognostic value of classic clinicopathological features and TILs in early-stage TNBC in the presence and absence of chemotherapy. To this end, we assembled a large cohort of surgically treated Mayo Clinic TNBC patients with long-term follow-up. Given that the definition of TNBC has evolved over time [16,17,18,19], we performed central pathology review of ER, PR, and HER2 status, in an effort to ensure that the cohort was representative of TNBC by modern definitions (ER/PR < 1%, HER2-negative by current guidelines [19]). We subsequently assessed the histologic subtype, grade, Ki-67, and TILs and evaluated their impact on the outcomes of the cohort and in a subset of systemically untreated TNBC patients. To ensure reproducibility of TIL assessment, we followed the recommendations proposed by the 2014 International TILs Working Group [20].
Methods
Study population
We used the Mayo Clinic, Rochester, MN database to identify patients who underwent surgery for stage I–III BC between January 1, 1985 and December 31, 2012, and who were clinically HER2 negative or unknown and did not receive anti-HER2 therapy. We excluded patients with prior cancer, bilateral BC, metastatic disease within 60 days of surgery, non-invasive or benign breast disease only, receipt of adjuvant endocrine therapy, known ER > 10% or ER-negative or unknown who received any neoadjuvant therapy.
Pathologic assessment
For eligible patients, tissue sections from formalin-fixed, paraffin-embedded (FFPE) tumor blocks were obtained and centrally evaluated for ER, PR, and HER2 at the Pathology Research Core (Mayo Clinic, Rochester, MN). ER and PR immunoreactivity were assessed using clones 1D5 and PgR363, respectively (Dako, Carpinteria, CA), with staining ≥1% considered positive. HER2 immunoreactivity was assessed using the HercepTest Kit (Dako, Carpinteria, CA), reflexed to fluorescence in situ hybridization (FISH, for IHC 2+ cases only), and categorized according to current ASCO/CAP guidelines [19].
For centrally confirmed TNBC tumors, we assessed the Ki-67 labeling index (MIB-1 monoclonal antibody, Dako, Carpinteria, CA, 1:400) and abstracted clinical data. A dedicated breast pathologist (DWV) blinded to clinical information classified histology into four groups (Table 1) and quantified TILs on full-face hematoxylin and eosin (H&E) sections from the surgical specimen. Stromal TILs were evaluated following the TILs Working Group recommendations [20], by counting all mononuclear cells in the stromal compartment within the borders of the invasive tumor and reported as a percentage value. Intratumoral TILs were also quantified and reported as a percentage value. Lymphocyte-predominant breast cancer (LPBC) was defined as having >50% stromal or intratumoral TILs [12, 15].
Statistical analysis
IDFS and OS were defined as per the STEEP classification [21]. Patients who were event-free at the last date of disease evaluation were censored for IDFS. If death occurred >365 days after the last disease evaluation where the patient was found to be recurrence-free, IDFS was censored on the last date of disease evaluation. The Kaplan–Meier method was used to estimate the distributions of IDFS and OS. The log-rank test was used to compare survival distributions among groups. Univariate Cox proportional hazards models were fit to assess the association between each baseline variable and clinical outcomes. Multivariate Cox proportional hazards models were fit to assess the association of each baseline covariate with clinical outcomes while adjusting for patient and disease characteristics. Results are expressed as hazard ratios (HRs) and 95% confidence intervals (CIs). Wilcoxon rank-sum test was used to compare the distribution of continuous variables between groups. Fisher’s exact test was used to assess the association between categorical variables. All statistical tests were two-sided and p values less than 0.05 were considered statistically significant. The association between TILs and outcomes was evaluated as a continuous variable, with separate analyses for stromal and intratumoral TILs, and as a categorical variable (LPBC vs. non-LPBC, and low vs. high stromal TILs dichotomized at the median of the cohort: 20%). The statistical analyses were carried out using R version 3.2.3. This study was approved by the Mayo Clinic Institutional Review Board.
Results
Study population
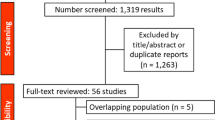
A total of 9982 women underwent surgery at Mayo Clinic, Rochester, MN for clinically HER2-negative or unknown BC between January 1, 1985 and December 31, 2012. Of these, 8826 were excluded after medical record review (Fig. 1). Of the remaining 1156 patients, FFPE tumor blocks were available in 1016. Tissue sections were centrally assessed for ER, PR, and HER2. After central review, 615 met the criteria for TNBC and were eligible for subsequent clinical data abstraction and evaluation of histology, Ki-67, and TILs. Ten patients with adenoid cystic carcinoma of the breast were excluded, leaving a total of 605 patients in the cohort.
Patient characteristics of the entire cohort
Characteristics of the study population are shown in Table 1. The median age was 56.3 years (range 26.9–93.7). Most were T1–2 (95%) and N0-1 stage (86%), grade 3 (88%), and had a Ki-67 >15% (75%). Histologically, most tumors were invasive carcinoma of NST (70%), but there were a substantial number of other subtypes as shown in Table 1.
The median stromal TIL content was 20% (range 0–90%), with the highest levels observed in carcinomas with medullary features (median 50%, range 0–90%). The median intratumoral TIL content was 3% (range 0–60%). Stromal and intratumoral TILs were moderately correlated (Spearman correlation coefficient = 0.6448). The breakdown of TIL content per 10% increments is shown in Table 1. Twenty-seven percent had TIL levels consistent with LPBC (>50% intratumoral or stromal TILs).
Surgery was evenly divided between lumpectomy and mastectomy. Patients with lumpectomy were more likely to receive adjuvant radiation compared to those who had a mastectomy (80% vs. 20%, p < 0.001). Patients with larger tumors and nodal involvement were more likely to receive adjuvant chemotherapy (T1: 52%, T2: 77%, T3/4: 72%, p < 0.001; N0: 55%, N1: 86%, N2: 88%, N3: 93%, p < 0.001). Rates of chemotherapy use increased in the later time periods: 1985–1990: 39%, 1991–1995: 61%, 1996–2000: 69%, 2001–2005: 76%, and 2006–2012: 76%, p < 0.001). While there was no difference in tumor size over time (p = 0.303), patients treated in the later time periods had a lower number of involved lymph nodes (p = 0.009). Distribution of chemotherapy regimens is shown in Table 1.
The median follow-up was 7.4 years (95% CI 6.1–8.5) for IDFS and 10.6 years (95% CI 9.7–11.7) for OS. The 5-year IDFS and OS were 66.8% (95% CI 62.5–71.4) and 77.3% (95% CI 73.8–80.9), respectively.
Prognostic factors in the entire cohort
Univariate analysis showed that postmenopausal status, tumor size (>5 cm), nodal stage, lower TILs, and the absence of adjuvant chemotherapy were significantly associated with worse IDFS (Table 2; Fig. 2) and OS (eTable 1; eFig. 1). In contrast, the histologic subtype carcinoma with medullary features was associated with improved IDFS (Table 2) and OS (eTable 1; eFig. 2). Regarding TILs, both stromal and intratumoral TILs were significantly associated with IDFS and OS. Furthermore, patients with LPBC had nearly half the risk of an IDFS (HR 0.55; 95% CI 0.39–0.78, p < 0.001, Table 2) or OS event (HR 0.60; 95% CI 0.43–0.83, p = 0.001, eTable 1) compared to non-LPBC. The 5-year IDFS and OS rates of patients with LPBC were 77.4 and 81.2%, compared to 61.6 and 74.8% in non-LPBC, respectively.
Carcinomas with apocrine differentiation were generally of lower grade (grade 3: 29.7%), had lower Ki-67 (Ki-67 ≤ 15%: 73%), and occurred in older patients (median age 69.1) compared to invasive carcinoma of NST. While IDFS and OS were similar when comparing the apocrine to the other histologic groups, bone metastases were numerically more frequent (54.6%, eTable 2) and IDFS events appeared to occur later in carcinomas with apocrine differentiation (eFig. 2).
In a multivariate analysis including stromal TILs as a continuous variable, nodal involvement, lower stromal TILs, and the absence of adjuvant chemotherapy remained associated with worse IDFS and OS (Table 2, eTable 1). In a separate multivariate model, substitution of intratumoral TILs (instead of stromal TILS) resulted in similar findings (eTable 3).
Patient characteristics of the systemically untreated cohort
One hundred eighty-two patients did not receive adjuvant chemotherapy. Baseline characteristics of this subset are listed in Table 1. These patients tended to be older (median age: 64), with smaller (T1: 68%) and node-negative (N0: 82%) tumors, while TIL levels and other clinicopathological factors were similar.
The median follow-up for IDFS and OS were 8.5 years (95% CI 6.0–10.9) and 15.0 years (95% CI 12.4–19.3), respectively. The 5- year IDFS and OS were 59.8% (95% CI 52.2–68.6) and 72.5% (95% CI 66.1–79.5), respectively.
Prognostic factors in the systemically untreated cohort
Univariate analysis showed that postmenopausal status, larger tumor size (≥T2 vs. T1), nodal involvement (≥N1 vs. N0), and lower stromal TILs were significantly associated with worse IDFS and OS, while lower intratumoral TILs were associated with worse IDFS but not with OS (Fig. 2; eTable 4; eFig. 3). Systemically untreated LPBC had half the risk of an IDFS event (HR 0.47; 95% CI 0.24–0.90, p = 0.020, eTable 4) compared to systemically untreated non-LPBC. Younger age and carcinoma with medullary features were associated with better OS, but not with IDFS.
Given that most systemically untreated patients were node negative, we assessed the effect of tumor size on 5-year IDFS rates. Among systemically untreated T1N0, the 5-year IDFS estimates by T1 sub-stage were T1a: 82.5% (95% CI 62.8–100), T1b: 67.5% (95% CI 51.9–87.8), and T1c: 67.3% (95% CI 54.9–82.6). Furthermore, the 5-year IDFS was 69.9% (95% CI 60.7–80.5) for systemically untreated T1N0 (n = 111) compared with 77.8% (95% CI 68.3–83.6) for systemically treated T1N0 (n = 81).
In an exploratory analysis (Fig. 3), we compared the IDFS of systemically treated and untreated patients according to TIL levels using the median TIL cut-off (low: <20% vs. high: ≥20%). While IDFS rates in systemically treated patients were higher regardless of TIL levels, the HR was smaller in systemically treated patients with high TILs (HR 0.57; 95% CI 0.38–0.87, p = 0.008) versus those with low TILs (HR 0.82; 95% CI 0.56–1.19, p = 0.3). For OS, receipt of adjuvant chemotherapy was associated with similar HR regardless of TIL levels [high TILs: HR 0.56 (95% CI 0.37–0.85, p = 0.006), low TILs: HR 0.6 (95% CI 0.4–0.89, p = 0.01)]. A similar analysis using LPBC (≥50% stromal or intratumoral TILs) is shown in eFig. 4).
Discussion
In this cohort study of over 600 patients with early-stage TNBC, we have thoroughly analyzed a series of readily accessible and centrally reviewed clinicopathological features and their relationship with long-term outcomes, both in the presence and absence of adjuvant chemotherapy. Our study is unique in that, as to our knowledge, it is the first study to evaluate the prognostic value of TILs in TNBC patients who did not receive adjuvant chemotherapy. Importantly, while this is a retrospective study, we generated these data by re-staining all archived specimens in a centralized pathology review setting, used current guidelines to assign ER, PR, and HER2 status [18, 19], and followed the current International Working Group recommendations for TIL assessment in BC [20].
When evaluating the entire cohort, we found that TILs, nodal stage, and administration of adjuvant chemotherapy were the only independent factors associated with prognosis on multivariate analysis. The median TIL content of our cohort and the magnitude of the prognostic effect with increasing TIL levels are consistent with other studies assessing TILs in early-stage TNBC [12,13,14,15].
It has been hypothesized that the association of high TILs with improved outcomes in the setting of chemotherapy may be related to the ability of chemotherapy to enhance immune responses. This putative effect may occur via the release of antigens due to tumor cell death, generation of neoantigens due to somatic mutations induced by chemotherapy, and favorable modification of the microenvironment through depletion of immunosuppressive cells, among other mechanisms [22]. However, when focusing on patients not exposed to adjuvant chemotherapy, we found that both stromal and intratumoral TILs were associated with IDFS, and stromal TILs were also associated with OS. While the literature and our exploratory analysis suggest that patients with higher TIL levels appear to derive a larger benefit from adjuvant chemotherapy, our data suggest that high TILs may represent the activation of an endogenous antitumor immune response that is present even in the absence of immune enhancements triggered by chemotherapy.
Histology did not independently influence IDFS or OS in our cohort. While carcinomas with medullary features (classically described to have a better prognosis [23]) were associated with better outcomes compared to invasive carcinomas of NST on univariate analysis, this association was lost once TILs were included in a multivariate model. Metaplastic carcinomas of NST (classically described to be associated with chemoresistance [24] and worse outcomes [25,26,27]) had similar IDFS and OS compared to invasive carcinomas of NST. Interestingly, and as noted in other series [28, 29], carcinomas with apocrine differentiation (known to nearly universally express the androgen receptor) had features that seemed to mirror luminal BC, including lower Ki-67, older age, higher rates of bone metastases, and later IDFS events.
Randomized clinical trials have demonstrated the benefit of polychemotherapy for TNBC [30]. In this cohort, receipt of adjuvant chemotherapy was associated with improved IDFS and OS, with the greatest benefit observed in patients receiving both anthracycline- and taxane-based chemotherapy. In contrast, the risk of recurrence in systemically untreated patients was substantial, even in those with T1N0 disease. These data support the modern-day practice of prioritizing biology over anatomy when making adjuvant therapy decisions.
Limitations and strengths
This study is limited by the constraints of a retrospective and single-center analysis. Patients in this cohort were treated across the span of nearly three decades, a period during which major changes in standard of care were introduced. Despite these limitations, our study represents the first thorough evaluation of the prognostic impact of TILs and other tumor features on the outcomes of TNBC patients in the presence and absence of chemotherapy. In the modern era, a prospective study of early-stage TNBC without the inclusion of chemotherapy would be unethical and not feasible. As such, exploration in existing patient cohorts is a necessary first step.
Conclusions
We have demonstrated that TILs and nodal burden are the only independent prognostic factors in early-stage TNBC treated with upfront surgery, and our data suggest that TILs may exert a prognostic effect regardless of receipt of adjuvant chemotherapy. Further dissection of the tumor microenvironment and the effect of TIL subpopulations on prognosis in both systemically treated and untreated patients will likely aid the generation of hypotheses for therapeutic opportunities in this highly aggressive subtype of BC. In addition, our data suggest that histology, grade, and Ki-67, while prognostic in other BC subtypes, do not modify prognosis and should not be used in treatment decision-making in TNBC. Finally, patients with T1 node-negative disease exhibit a substantial risk of recurrence and should be considered for systemic chemotherapy.
References
Perou CM, Sorlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA, Pollack JR, Ross DT, Johnsen H, Akslen LA, Fluge O, Pergamenschikov A, Williams C, Zhu SX, Lonning PE, Borresen-Dale AL, Brown PO, Botstein D (2000) Molecular portraits of human breast tumours. Nature 406(6797):747–752
Dent R, Trudeau M, Pritchard KI, Hanna WM, Kahn HK, Sawka CA, Lickley LA, Rawlinson E, Sun P, Narod SA (2007) Triple-negative breast cancer: clinical features and patterns of recurrence. Clin Cancer Res 13(15 Pt 1):4429–4434. doi:10.1158/1078-0432.CCR-06-3045
Lehmann BD, Bauer JA, Chen X, Sanders ME, Chakravarthy AB, Shyr Y, Pietenpol JA (2011) Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J Clin Invest 121(7):2750–2767. doi:10.1172/JCI45014
Purrington KS, Visscher DW, Wang C, Yannoukakos D, Hamann U, Nevanlinna H, Cox A, Giles GG, Eckel-Passow JE, Lakis S, Kotoula V, Fountzilas G, Kabisch M, Rudiger T, Heikkila P, Blomqvist C, Cross SS, Southey MC, Olson JE, Gilbert J, Deming-Halverson S, Kosma VM, Clarke C, Scott R, Jones JL, Zheng W, Mannermaa A, Jane Carpenter for AI, Eccles DM, Vachon CM, Couch FJ (2016) Genes associated with histopathologic features of triple negative breast tumors predict molecular subtypes. Breast Cancer Res Treat 157(1):117–131. doi:10.1007/s10549-016-3775-2
Prat A, Adamo B, Cheang MC, Anders CK, Carey LA, Perou CM (2013) Molecular characterization of basal-like and non-basal-like triple-negative breast cancer. Oncologist 18(2):123–133. doi:10.1634/theoncologist.2012-0397
Network NCC Breast Cancer (Version 2.2017). https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf
Denkert C, von Minckwitz G, Darb-Esfahani S, Ingold Heppner B, Klauschen F, Furlanetto J, Pfitzner B, Huober J, Schmitt W, Blohmer J-U, Kümmel S, Engels K, Lederer B, Schneeweiss A, Hartmann A, Jakisch C, Untch M, Hanusch C, Weber K, Loibl S (2017) Abstract S1-09: evaluation of tumor-infiltrating lymphocytes (TILs) as predictive and prognostic biomarker in different subtypes of breast cancer treated with neoadjuvant therapy—a metaanalysis of 3771 patients. Cancer Res 77(4 Supplement):S1–S09. doi:10.1158/1538-7445.sabcs16-s1-09
Denkert C, Loibl S, Noske A, Roller M, Muller BM, Komor M, Budczies J, Darb-Esfahani S, Kronenwett R, Hanusch C, von Torne C, Weichert W, Engels K, Solbach C, Schrader I, Dietel M, von Minckwitz G (2010) Tumor-associated lymphocytes as an independent predictor of response to neoadjuvant chemotherapy in breast cancer. J Clin Oncol 28(1):105–113. doi:10.1200/JCO.2009.23.7370
Yamaguchi R, Tanaka M, Yano A, Tse GM, Yamaguchi M, Koura K, Kanomata N, Kawaguchi A, Akiba J, Naito Y, Ohshima K, Yano H (2012) Tumor-infiltrating lymphocytes are important pathologic predictors for neoadjuvant chemotherapy in patients with breast cancer. Hum Pathol 43(10):1688–1694. doi:10.1016/j.humpath.2011.12.013
West NR, Milne K, Truong PT, Macpherson N, Nelson BH, Watson PH (2011) Tumor-infiltrating lymphocytes predict response to anthracycline-based chemotherapy in estrogen receptor-negative breast cancer. Breast Cancer Res 13(6):R126. doi:10.1186/bcr3072
Ono M, Tsuda H, Shimizu C, Yamamoto S, Shibata T, Yamamoto H, Hirata T, Yonemori K, Ando M, Tamura K, Katsumata N, Kinoshita T, Takiguchi Y, Tanzawa H, Fujiwara Y (2012) Tumor-infiltrating lymphocytes are correlated with response to neoadjuvant chemotherapy in triple-negative breast cancer. Breast Cancer Res Treat 132(3):793–805. doi:10.1007/s10549-011-1554-7
Loi S, Sirtaine N, Piette F, Salgado R, Viale G, Van Eenoo F, Rouas G, Francis P, Crown JP, Hitre E, de Azambuja E, Quinaux E, Di Leo A, Michiels S, Piccart MJ, Sotiriou C (2013) Prognostic and predictive value of tumor-infiltrating lymphocytes in a phase III randomized adjuvant breast cancer trial in node-positive breast cancer comparing the addition of docetaxel to doxorubicin with doxorubicin-based chemotherapy: BIG 02-98. J Clin Oncol 31(7):860–867. doi:10.1200/JCO.2011.41.0902
Loi S, Michiels S, Salgado R, Sirtaine N, Jose V, Fumagalli D, Kellokumpu-Lehtinen PL, Bono P, Kataja V, Desmedt C, Piccart MJ, Loibl S, Denkert C, Smyth MJ, Joensuu H, Sotiriou C (2014) Tumor infiltrating lymphocytes are prognostic in triple negative breast cancer and predictive for trastuzumab benefit in early breast cancer: results from the FinHER trial. Ann Oncol 25(8):1544–1550. doi:10.1093/annonc/mdu112
Adams S, Gray RJ, Demaria S, Goldstein L, Perez EA, Shulman LN, Martino S, Wang M, Jones VE, Saphner TJ, Wolff AC, Wood WC, Davidson NE, Sledge GW, Sparano JA, Badve SS (2014) Prognostic value of tumor-infiltrating lymphocytes in triple-negative breast cancers from two phase III randomized adjuvant breast cancer trials: ECOG 2197 and ECOG 1199. J Clin Oncol 32(27):2959–2966. doi:10.1200/JCO.2013.55.0491
Pruneri G, Vingiani A, Bagnardi V, Rotmensz N, De Rose A, Palazzo A, Colleoni AM, Goldhirsch A, Viale G (2016) Clinical validity of tumor-infiltrating lymphocytes analysis in patients with triple-negative breast cancer. Ann Oncol 27(2):249–256. doi:10.1093/annonc/mdv571
Harbeck N, Rody A (2012) Lost in translation? Estrogen receptor status and endocrine responsiveness in breast cancer. J Clin Oncol 30(7):686–689. doi:10.1200/JCO.2011.38.9619
Harvey JM, Clark GM, Osborne CK, Allred DC (1999) Estrogen receptor status by immunohistochemistry is superior to the ligand-binding assay for predicting response to adjuvant endocrine therapy in breast cancer. J Clin Oncol 17(5):1474–1481
Hammond ME, Hayes DF, Dowsett M, Allred DC, Hagerty KL, Badve S, Fitzgibbons PL, Francis G, Goldstein NS, Hayes M, Hicks DG, Lester S, Love R, Mangu PB, McShane L, Miller K, Osborne CK, Paik S, Perlmutter J, Rhodes A, Sasano H, Schwartz JN, Sweep FC, Taube S, Torlakovic EE, Valenstein P, Viale G, Visscher D, Wheeler T, Williams RB, Wittliff JL, Wolff AC (2010) American Society of Clinical Oncology/College Of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Clin Oncol 28(16):2784–2795. doi:10.1200/JCO.2009.25.6529
Wolff AC, Hammond ME, Hicks DG, Dowsett M, McShane LM, Allison KH, Allred DC, Bartlett JM, Bilous M, Fitzgibbons P, Hanna W, Jenkins RB, Mangu PB, Paik S, Perez EA, Press MF, Spears PA, Vance GH, Viale G, Hayes DF, American Society of Clinical O, College of American P (2013) Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. J Clin Oncol 31(31):3997–4013. doi:10.1200/JCO.2013.50.9984
Salgado R, Denkert C, Demaria S, Sirtaine N, Klauschen F, Pruneri G, Wienert S, Van den Eynden G, Baehner FL, Penault-Llorca F, Perez EA, Thompson EA, Symmans WF, Richardson AL, Brock J, Criscitiello C, Bailey H, Ignatiadis M, Floris G, Sparano J, Kos Z, Nielsen T, Rimm DL, Allison KH, Reis-Filho JS, Loibl S, Sotiriou C, Viale G, Badve S, Adams S, Willard-Gallo K, Loi S, International TWG (2015) The evaluation of tumor-infiltrating lymphocytes (TILs) in breast cancer: recommendations by an International TILs Working Group 2014. Ann Oncol 26(2):259–271. doi:10.1093/annonc/mdu450
Hudis CA, Barlow WE, Costantino JP, Gray RJ, Pritchard KI, Chapman J-AW, Sparano JA, Hunsberger S, Enos RA, Gelber RD, Zujewski JA (2007) Proposal for standardized definitions for efficacy end points in adjuvant breast cancer trials: the STEEP system. J Clin Oncol 25(15):2127–2132. doi:10.1200/JCO.2006.10.3523
Zitvogel L, Apetoh L, Ghiringhelli F, Kroemer G (2008) Immunological aspects of cancer chemotherapy. Nat Rev Immunol 8(1):59–73. doi:10.1038/nri2216
Huober J, Gelber S, Goldhirsch A, Coates AS, Viale G, Ohlschlegel C, Price KN, Gelber RD, Regan MM, Thurlimann B (2012) Prognosis of medullary breast cancer: analysis of 13 International Breast Cancer Study Group (IBCSG) trials. Ann Oncol 23(11):2843–2851. doi:10.1093/annonc/mds105
Chen IC, Lin CH, Huang CS, Lien HC, Hsu C, Kuo WH, Lu YS, Cheng AL (2011) Lack of efficacy to systemic chemotherapy for treatment of metaplastic carcinoma of the breast in the modern era. Breast Cancer Res Treat 130(1):345–351. doi:10.1007/s10549-011-1686-9
Yamaguchi R, Horii R, Maeda I, Suga S, Makita M, Iwase T, Oguchi M, Ito Y, Akiyama F (2010) Clinicopathologic study of 53 metaplastic breast carcinomas: their elements and prognostic implications. Hum Pathol 41(5):679–685. doi:10.1016/j.humpath.2009.10.009
Jung SY, Kim HY, Nam BH, Min SY, Lee SJ, Park C, Kwon Y, Kim EA, Ko KL, Shin KH, Lee KS, Park IH, Lee S, Kim SW, Kang HS, Ro J (2010) Worse prognosis of metaplastic breast cancer patients than other patients with triple-negative breast cancer. Breast Cancer Res Treat 120(3):627–637. doi:10.1007/s10549-010-0780-8
Lester TR, Hunt KK, Nayeemuddin KM, Bassett RL Jr, Gonzalez-Angulo AM, Feig BW, Huo L, Rourke LL, Davis WG, Valero V, Gilcrease MZ (2012) Metaplastic sarcomatoid carcinoma of the breast appears more aggressive than other triple receptor-negative breast cancers. Breast Cancer Res Treat 131(1):41–48. doi:10.1007/s10549-011-1393-6
Thike AA, Yong-Zheng Chong L, Cheok PY, Li HH, Wai-Cheong Yip G, Huat Bay B, Tse GM, Iqbal J, Tan PH (2014) Loss of androgen receptor expression predicts early recurrence in triple-negative and basal-like breast cancer. Mod Pathol 27(3):352–360. doi:10.1038/modpathol.2013.145
Masuda H, Baggerly KA, Wang Y, Zhang Y, Gonzalez-Angulo AM, Meric-Bernstam F, Valero V, Lehmann BD, Pietenpol JA, Hortobagyi GN, Symmans WF, Ueno NT (2013) Differential response to neoadjuvant chemotherapy among 7 triple-negative breast cancer molecular subtypes. Clin Cancer Res 19(19):5533–5540. doi:10.1158/1078-0432.CCR-13-0799
Group EBCTC (2012) Comparisons between different polychemotherapy regimens for early breast cancer: meta-analyses of long-term outcome among 100 000 women in 123 randomised trials. Lancet 379(9814):432–444
Funding
This work was supported by the Mayo Clinic Center for Individualized Medicine; the Mayo Clinic Cancer Center (Grant Number CA15083-40A2 to MPG, JNI); George M. Eisenberg Foundation for Charities; the Mayo Clinic Breast SPORE (Grant Number P50CA 116201-9 to MPG, FJC, JNI, DWV, MP, KRK); and NIH Grant R01CA192393.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Roberto A. Leon-Ferre, Mei-Yin Polley, Heshan Liu, Judith A. Gilbert, Victoria Cafourek, David W. Hillman, Ahmed Elkhanany, Margaret Akinhanmi, Jenna Lilyquist, Abigail Thomas, Vivian Negron, Judy C. Boughey, Minetta C. Liu, James N. Ingle, Krishna R. Kalari, Fergus J. Couch, Daniel W. Visscher, and Matthew P. Goetz declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
The need for obtaining informed consent was waived by the institutional review board, given that this study was retrospective and non-interventional.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Leon-Ferre, R.A., Polley, MY., Liu, H. et al. Impact of histopathology, tumor-infiltrating lymphocytes, and adjuvant chemotherapy on prognosis of triple-negative breast cancer. Breast Cancer Res Treat 167, 89–99 (2018). https://doi.org/10.1007/s10549-017-4499-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-017-4499-7